Acid-Base Balance Taylors Ch 41 Brunners Ch 10
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
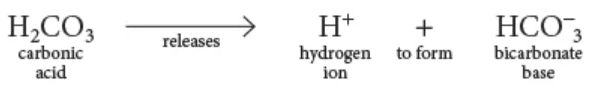
an acid
_______is a substance containing H+ that can be released, such as carbonic acid
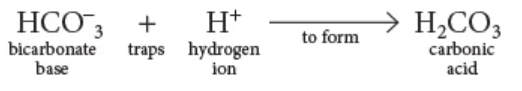
base
An alkali, or ________, is a substance that can accept or trap H+ ions, such as the bicarbonate ion
pH
The unit of measure used to describe acid–base balance is ___ which is an expression of H+ ion concentration and the resulting acidity or alkalinity of a substance
alkaline, 7.35 - 7.45
normal blood plasma is slightly ________ and has a normal pH range of _________ with 7.4 being the most ideal pH
6.80; 7.80
death due to blood plasma pH being too alkaline on average occurs at ______ and death due to pH being too acidic on average occurs at ______
(HCO3–) bicarbonate : carbonic acid (H2CO3)
in order to maintain pH balance, the blood plasma requires a 20:1 ratio of __________
kidneys
______regulate the production of bicarbonate
lungs
_______regulate carbonic acid (H2O + CO2) and is also the first organ to compensate before others 90% of the time
accepts H+ ions easily; accepts H+ ions less readily
A base that ___________ is considered a strong base, whereas one that __________ is considered a weak base.
separates completely; only a small number
A strong acid ___________ in solution and releases all of its H+ ions, whereas a weak acid releases ____________
acidosis
an acid–base imbalance characterized by an increase in H+ concentration (decrease blood pH)
alkalosis
an acid–base imbalance characterized by a reduction in H+ concentration or increase in bicarbonate concentration (increase blood pH)