O Chem quiz II
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
What is an Arrhenius acid?
A substance that ionizes to yield protons (H+) when dissolved in water
What is an Arrhenius base?
A substance that ionizes to yield hydroxide ions (HO-) when dissolved in water
What is a Bronsted - Lowry acid?
A proton donor
What is a Bronsted - Lowry base?
A proton acceptor
What is a Lewis acid?
Electron pair acceptor
What is a Lewis base?
Electron pair donor
What is Amphoteric?
A molecule that’s capable of acting as an acid or base
What is Amphiprotic?
Capacity of a substance to either lose or gain a proton
What are the 6 strong acids we study?
HCl, HBr, HI, H2SO4, HNO3, HClO4
What are the 6 strong bases we study?
NaOH, LiOH, KOH, Ca(OH)2, Ba(OH)2, Sr(OH)2
Which 2 strong bases are only slightly soluble in water?
Ca(OH)2 and Sr(OH)2
What is Homolysis?
Bond cleavage in which both atoms contain one electron to produce free radicals
What are Free Radicals?
Neutral species containing an unpaired electron
What is Heterolysis?
Bond cleavage which leads to charged fragments (cation/anion)
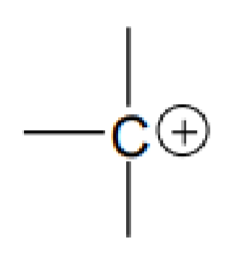
What molecule is this?
Carbocations
What are the 2 things that stabilize a charged system?
Induction and delocalization through resonance structures
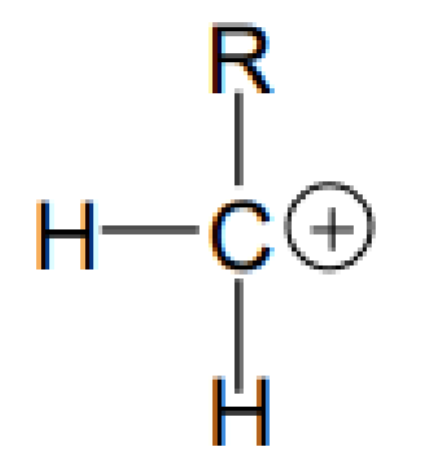
What type of carbon is this?
Primary carbon
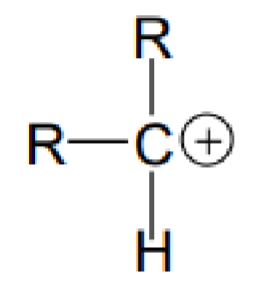
What type of carbon is this?
Secondary carbon
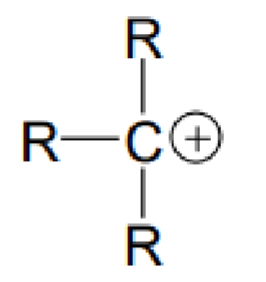
What type of carbon is this?
Tertiary carbon
Which Carbocation is most stable?
Tertiary carbon is the most stable
Which Carbocation is least stable?
Methyl is the least stable
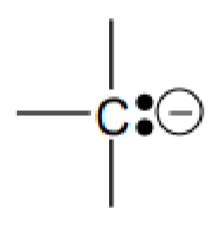
What molecule is this?
Carbanions
Which Carbanion is most stable?
Methyl is the most stable
Which Carbanion is least stable?
Tertiary carbon is the least stable
What is a Nucleophile?
A “nucleus loving species” includes any negative ion or neutral molecule that has an unshared electron pair
What is Nucleophilicity?
The tendency of a chemical species to donate an electron pair to an electrophile in order to form a covalent bond
What is a Electrophile?
“Electron loving species” Any electron deficient species
A negatively charged nucleophile is always …
A more reactive (better) nucleophile than its conjugate acid
If the nucleophilic atoms are different, compare electronegativities. The less electronegative …
The better the nucleophile
What makes a good leaving group?
A relatively stable molecule or ion that maintains its stability when the leaving group actually leaves
How does the strength of the base affect the favorability of the leaving group?
The stronger the base the worse the leaving group
What is polarizability?
The ability of an atom to distort its electron cloud
How does the size of an atom affect its polarizability?
The bigger the atom, the greater the polarizability
How does the polarizability of an atom affect the quality of its leaving group?
The greater the polarizability the better the leaving group
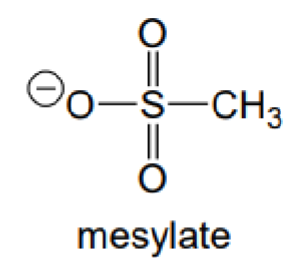
What is the abbreviation of this carboanion?
OMs
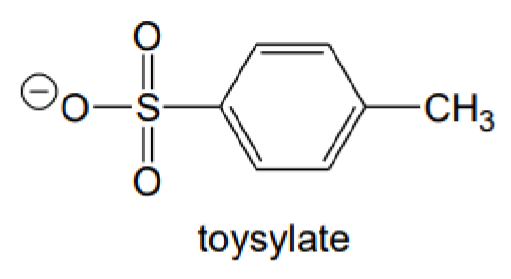
What is the abbreviation of this carboanion?
OTs
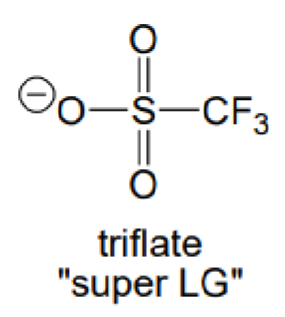
What is the abbreviation of this carboanion?
OTf
What are Protic solvents?
Solvents which contain a hydrogen attached to a highly electronegative element
What are polar aprotic solvents?
Solvent which does not contain a hydrogen attached to a highly electronegative element
Polar aprotic solvents can never …
Be used as a nucleophile
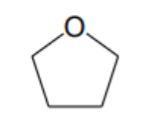
What is the abbreviation of this solvent?
THF
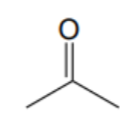
What is the name of this solvent?
Acetone
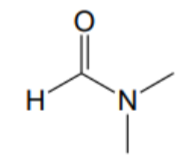
What is the name of this solvent?
DMF
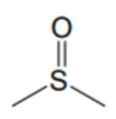
What is the name of this solvent?
DMSO
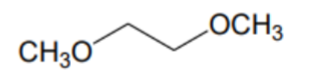
What is the name of this solvent?
DME
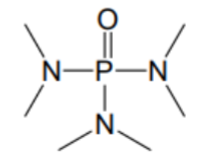
What is the name of this solvent?
HMPA
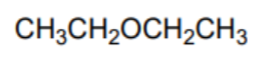
What is the name of this solvent?
Diethyl ether
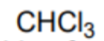
What is the name of this solvent?
Chloroform