Precision medicine
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
How old is precision medicine?
Very old and has been since (460-370 BC)
Hippocrates was known as “father of medicine” and practitioner of precision medicine
Whats precision medicine?
Prescribed treatments and prognosis informed by the patient’s own measurements
What are some ancient and modern techniques of precision medicine?
Ancient one is the phlegm and bile
Modern is temp, blood pressure, hormone level, MRI scan, cognitive test, genetic markers
Why is modern better than ancient medicine?
More data = greater precision
LOOK AT AND UNDERSTAND INTUITION CHECK
Whats family history?
An important data point in precision medicine
Family history serves as indirect genetic information (indirect cuz doesnt measure genotype, as its inferred risk is based on someones relatives
Can be used to assess disease risk over 3 sectors (high, moderate, average)
Why is family history underused?
Time pressure model in healthcare, no time
People might not be informed of their history
Doctors might not be trained to take family history
Why is using family history alone imprecise?
Because its an indirect means of assessing genetic contribution
2 people may have different risks even if they have same allele or not (de novo mutations would be labelled average risk even though they have disease variant)
Why is precision med better than just family history?
It directly assesses genetic variation
It was one of the major goals of the human genome project
What did the human genome project give us?
A reference genome (normal baseline)
Sequencing tools and methods
Opportunity for extensive associations
STUDY THE FOLLWOING SUMMARY OF FAMILY HISTORY DIRECT GENETIC TESTING
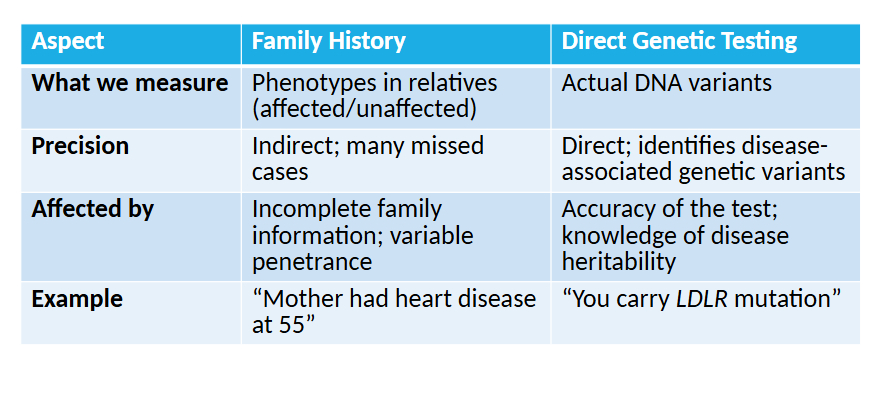
Explain early onset familial alzheimers disease (EOFAD) and its characteristics
Autosomal dominant
Numerous atypical phenotypes such as Visual-spatial difficulties, behavioural changes, Parkinsonism
Diagnosed before 65 years-old
Near complete penetrance = An individual with mutation nearly always will get EOFAD
Pathologies are indistinguishable from late onset alzheimers disease so people with this cant get a quick diagnosis and experience a DIAGNOSTIC ODYSSEY
What can geentic testing do?
Identify the cause of disease
Enable early diagnosis
Brings peace of mind
Gives access to condition-specific resources
Helps prepare for the future
Assists with career planning
Via carrier testing and newborn screening
Whatre some considerations for rare diseases?
Your right to know
Who gets access?
What traits should be screened?
Explain late onset alzheimers disease (LOAD)
Progressive, incurable, neurodegenerative disorder
Strong disease heterogeneity which means every dementia patient is unique
Complex exposome which is cumulative of all the exposures in a persons life from birth
Is highly polygenic → has far more key genes than EOFAD
How can risk for late onset alzheimers disease (LOAD) be quantified?
Via a polygenic score
Whatre the steps of creating a polygenic score?
Choose or create GWAS data for generating a PGS
Select variants for inclusion in PGS by applying a significance threshold (leniant, strict, middle → shows importance of p-value)
Identify number of effect alleles that each individual carrier
Use effect sizes to weight each variant (highest ODDS-Ratio = strongest bearing on trait)
Multiply # of effect alleles by effect sizes (odds ratio) to get polygenic score
NEED TO TEST DATA TO SEE IF POLYGENIC SCORE/ DATA IS USEFUL
Whatre polygenic scores?
Combination of multiple genetic variants which act together to increase and decrease risk of disease
They have a comparable risk to rare monographic mutations. Greater population impact
Explain the GWAS diversity gap
Reduced PGS accuracy in those non-european ancestry (4 in 5 people)
79% of GWAS participants were european individuals
Whatre some applications of polygenic score?
1. Identify high-risk individuals for early disease screening
2. Personalized medicine based on genetic risk
3. Assess disease risk in prospective parents
4. Control for genetic confounding in research
Whatre the 2 factors used to evaluate whether a genetic test is appropriate given clinical context?
Clinical validity = Extent to which a test is predictive of disease
Clinical utility = Degree to which a test will result in changes to medical care
Whatre the 4 aspects of clinical validity?
Sensitivity: Fraction of individuals with the disease who carry the risk variant = a/(a+c)
Specificity: Fraction of individuals without the disease who do not carry the risk variant = d/(b+d)
Positive predictive value: Proportion of individuals who carry the risk variant and develop the disease = a/(a+b)
Negative predictive value: Proportion of individuals who do not carry the risk variant and do not develop the disease = d/(c+d)
Whats clinical utility?
Degree to which a test will result in changes to medical care
Testing of variants may alter medical managements through the identification of improved treatments and preventative measures
Understand case example
Study this summary side
