Lab 9 - Acid Base Balance (new) (in progess)
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Balance of substances contributing to homeostasis
Acid-base balance
Found in the stomach, blood, extracellular fluids, cytosol, and urine
Acids and bases
The cells of which system release acids and bases?
Immune system
A hydrogen ion (H+) donor
Acid
A hydroxyl ion (OH-) donor
Base
Negatively charged ions that combine with hydrogen ions in a solution
Alkali (bases)
Measure of the acidity or alkalinity of a solution
pH
Equal to the negative logarithm of the hydrogen ion concentration
pH
A combination of two or more chemicals that minimize the pH change of a solution when a strong acid or base has been added
Buffer system
What two chemicals typically comprise a buffer system?
Weak acid and it’s salt
pH scale range
0-14
pH related to greater hydrogen ion concentration
Lower, acidic
pH related to lower hydrogen ion concentration
Higher, basic
pH indicating neutrality
7
When the number of hydrogen ions = hydroxyl ions
Neutrality (pH 7)
Neutral pH
7
A shift of how many pH units represents a tenfold increase or decrease in hydrogen ion concentration
One pH
A solution of pH 4 has how many times more hydrogen ions as a pH 5?
10x
What is the pH of water?
~7
What macromolecule primarily makes up albumin (egg white)?
Protein
What do proteins change when exposed to different pH or temperature?
Shape
Why does the body strictly regulate pH and temperature?
To prevent protein and enzyme denaturing
Can all proteins refold after denaturing? Can any?
Not all, only some can
Required by almost all biochemical reactions
Enzymes
Work optimally within relatively narrow temperature and pH ranges
Enzymes
How does acid affect the protein in egg white?
Denatures the protein, leading to unfolding and coagulation (white and solid clumps)
How does acid affect protein structure
Acid denatures protein
What would an acidic environment do to the body
It would disrupt necessary enzyme dependent biochemical reactions
Compound resisting pH change
Buffer
What does the buffer do with hydrogen ions (H+) when pH is falling (acidic)
Takes up hydrogen ions (H+) from the solution
What does the buffer do with hydrogen ions (H+) when pH is rising (basic)
Donates hydrogen ions (H+) to the solution
Protein function, and therefore cellular function is dependent on what levels
pH
Category of proteins that catalyze chemical reactions and are required for cell survival
Enzymes
What part of a protein is disrupted due to low or high pH
Three-dimensional structure
What conditions aside from pH impact three dimensional enzyme structure?
Heat and high salt concentration
Loss of the three dimensional structure of a protein
Denaturation
H2CO3 < > H++ HCO3-
Bicarbonate buffer system
H2PO4- < > H++ HPO42-
Phosphate buffering system
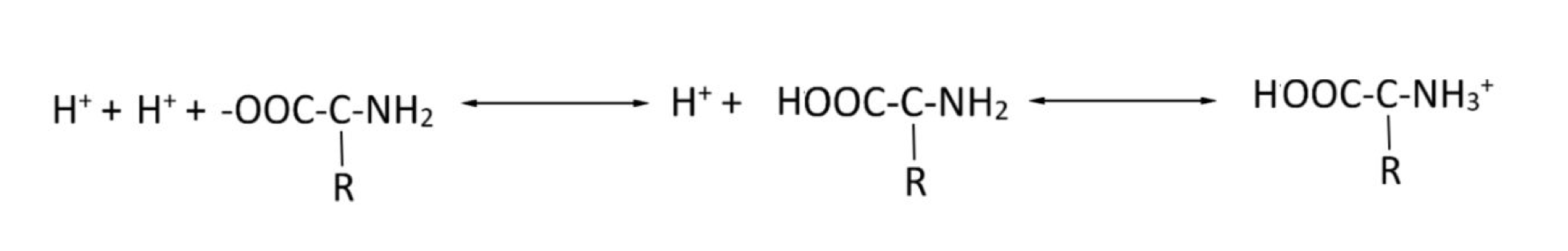
Which buffer system is this
Protein buffering system
If a solution is rising in pH, does it have more or less protein buffering molecules
Less, because it has already released hydrogen molecules to buffer the rise in pH
If a solution is rising in pH, will it need to produce hydrogen ions or take them up
Take them up
Consider the bicarbonate buffer system. If pH is rising, will carbonic acid (H2CO3) form or dissociate
Dissociate
Consider the bicarbonate buffer system. If pH is falling, will carbonic acid (H2CO3) form or dissociate
Form
Consider the phosphate buffering system. What will favour the formation of dihydrogen phosphate (H2PO4-)
Falling pH
Consider the phosphate buffering system. What will favour the dissociation of dihydrogen phosphate (H2PO4-)
Rise in pH
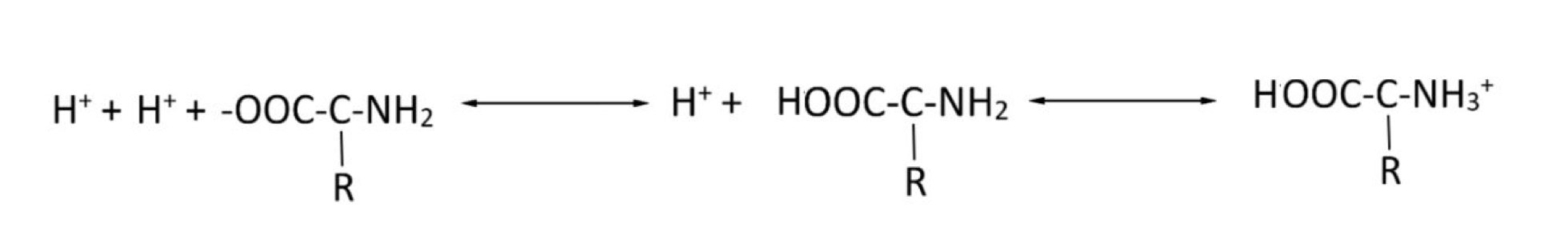
What part of the protein buffering system is the most acidic?
The carboxyl group (-COOH)
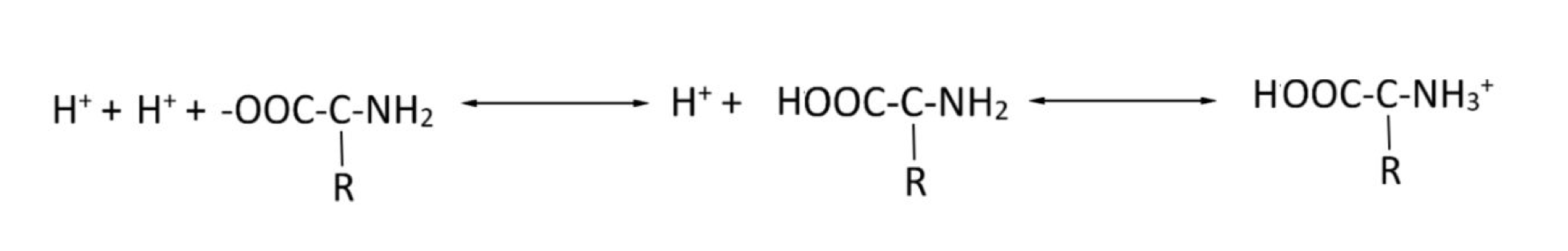
Consider the protein buffering system. Will a rise in pH favour the equation moving to the right or the left?
Shift to the right, favoring the dissociation of H+ ions
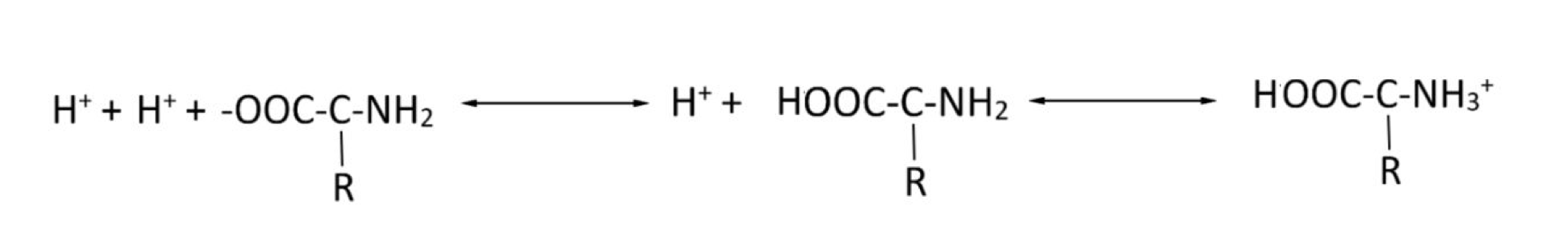
Consider the protein buffering system. Will a drop in pH favour the equation moving to the right or the left?
Left
What is produced when carbon dioxide and water combine
Carbonic acid
Why does carbonic acid lower blood pH
It is acidic and therefore a source of hydrogen ions
Is it possible to lower the amount of water in the blood sufficiently to prevent the formation of carbonic acid? Why or why not?
No, it would lead to fatal dehydration
What does the body have to eliminate to prevent carbonic acid formation
C02
What organ does the body use to eliminate CO2
The lungs
The removal of excess base in the blood left over from stomach acid production
Alkaline
What fluid becomes alkaline a base is removed from the blood
Plasma/serum
What organ helps the lungs with blood pH
Kidneys
Carbonic acid, dihydrogen phosphate, and proteins are always present in the blood. Based on this, would you expect the chemical buffering systems to act in seconds, minutes, or hours
Seconds
Why do chemical buffering systems act in seconds
The molecules they rely on are already in the blood
Which physiological buffering system is faster, respiratory or renal? Why?
Respiratory system, greater surface area
Which two ions absorbed and secreted by the kidneys work to regulate pH
Bicarbonate and hydrogen ions
Does bicarbonate reabsorption via kidney tubules increase or decrease blood pH
Increases (alkaline)
Does bicarbonate excretion via kidney tubules increase or decrease blood pH
Decreases (acidic)
Which two body systems work together to maintain blood pH
Respiratory and renal