Chapter 17 Thermodynamics
1/23
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
what is the trend in lattice enthalpies for ionic compounds
as the ions get smaller the lattice enthalpies increase
because the ionic radius is smaller so the oppositely charged ions are closer together
resulting in stronger ionic bonding
how to calculate enthalpy of solution
enthalpy of solution = enthalpy of lattice dissociation + enthalpy of hydration
reason for discrepancy between experimental and theoretical values of lattice enthalpy
the ionic compound has covalent character
factors which increase polarisation for anions
large size
high negative charge
factors which increase polarisation for cations
small size
high positive charge
define entropy
the measure of disorder in a chemical system
how to work out change in entropy for a reaction
ΔS = ΣSproducts - ΣSreactants
units for entropy
JK-1mol-1
what 2 factors determine the feasibility of a chemical reaction
enthalpy change and entropy change
equation for Gibb’s free energy
ΔG = ΔH - TΔS
ΔG = Gibb’s free energy change
ΔH = enthalpy change
T = temperature in kelvin
ΔS = entropy change
what value of ΔG is required for a reaction to be feasible
ΔG negative or equal to 0
why does water have a higher entropy of vapourisation compared to other liquids
water has hydrogen bonds
so in the liquid state it is more ordered than other liquids without hydrogen bonds
how to work out temperature at which a reaction is feasible
when feasible ΔG = 0
T = ΔH/ΔS
explain in terms of molecules why entropy is 0 at 0K (2)
molecules have 0 kinetic energy so they don’t move
there is no disorder
why does entropy increase with temperature (2)
particles gain kinetic energy so start to move more
disorder increases
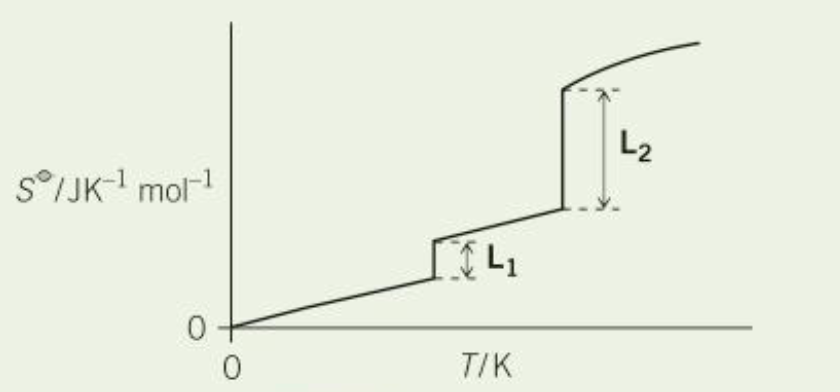
why is L2 longer than L1 (2)
L2 corresponds to boiling, L1 corresponds to melting
there is a greater change in disorder in boiling compared to melting
in terms of forces acting on particles why is the first electron affinity of oxygen exothermic (1)
there is a force of attraction between the nucleus of the oxygen atom and an external electron
standard free energy change for formation of MgO, ΔG = -570kJ mol-1. Suggest one reason why a sample of MgO appears to be stable in air at room temperature despite negative value for ΔG (1)
a protective layer of MgO forms