Chemistry CCEA AS Level 1.2 (Atomic Structure)
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is the atomic number?
The number of protons in the nucleus of an atom
What is the mass number?
The total number of protons and neutrons in the nucleus of an atom
What is an ion?
A charged atom, formed by the loss or gain of electrons
What is the relative atomic mass?
The average mass of one atom of an element relative to 1/12 of the mass of an atom of carbon-12
What is an isotope?
Atoms which have the same atomic number but a different mass number
What is an orbital?
A region of space within an atom that can hold up to 2 electrons with opposite spins
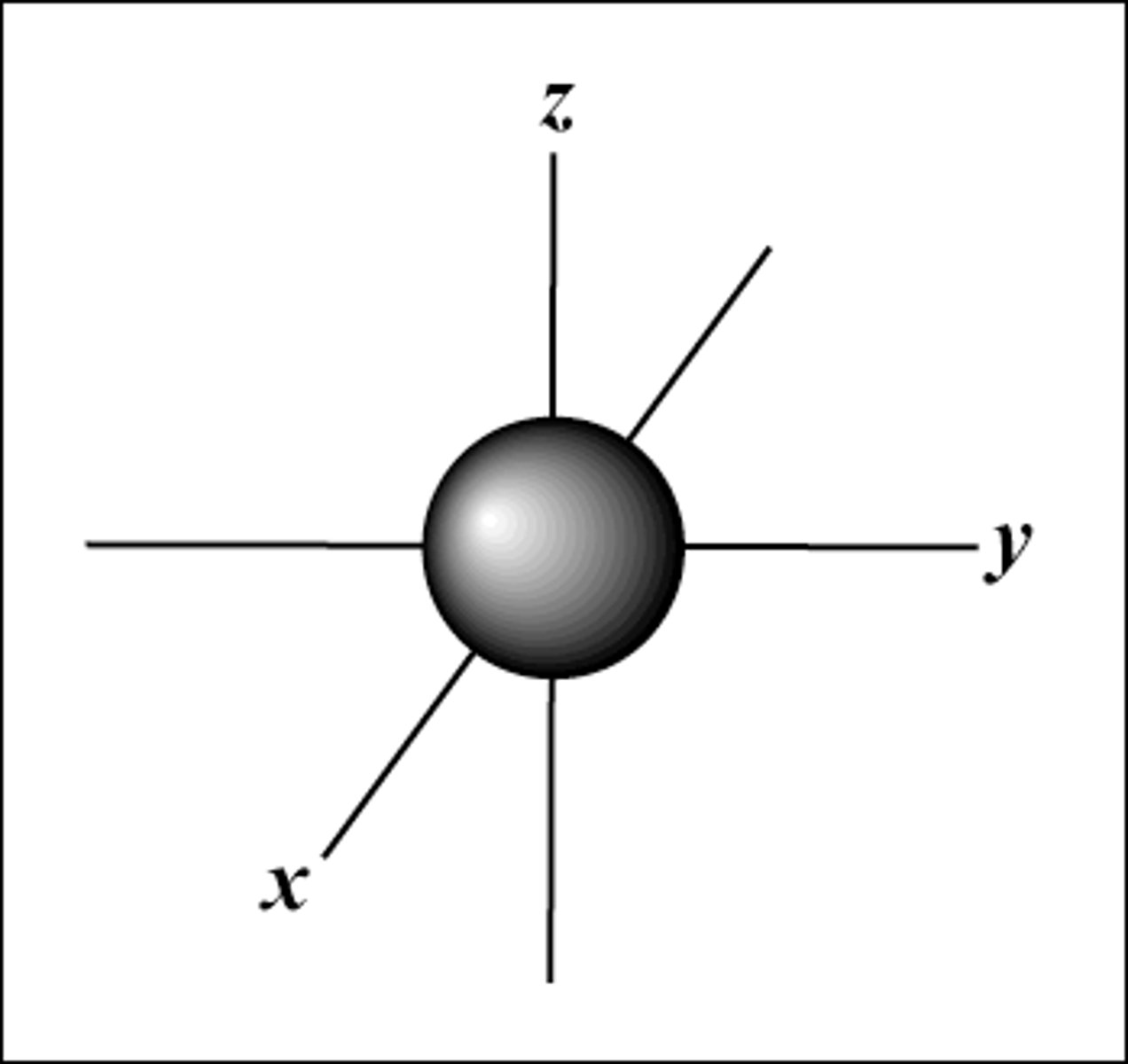
What does an s orbital look like?
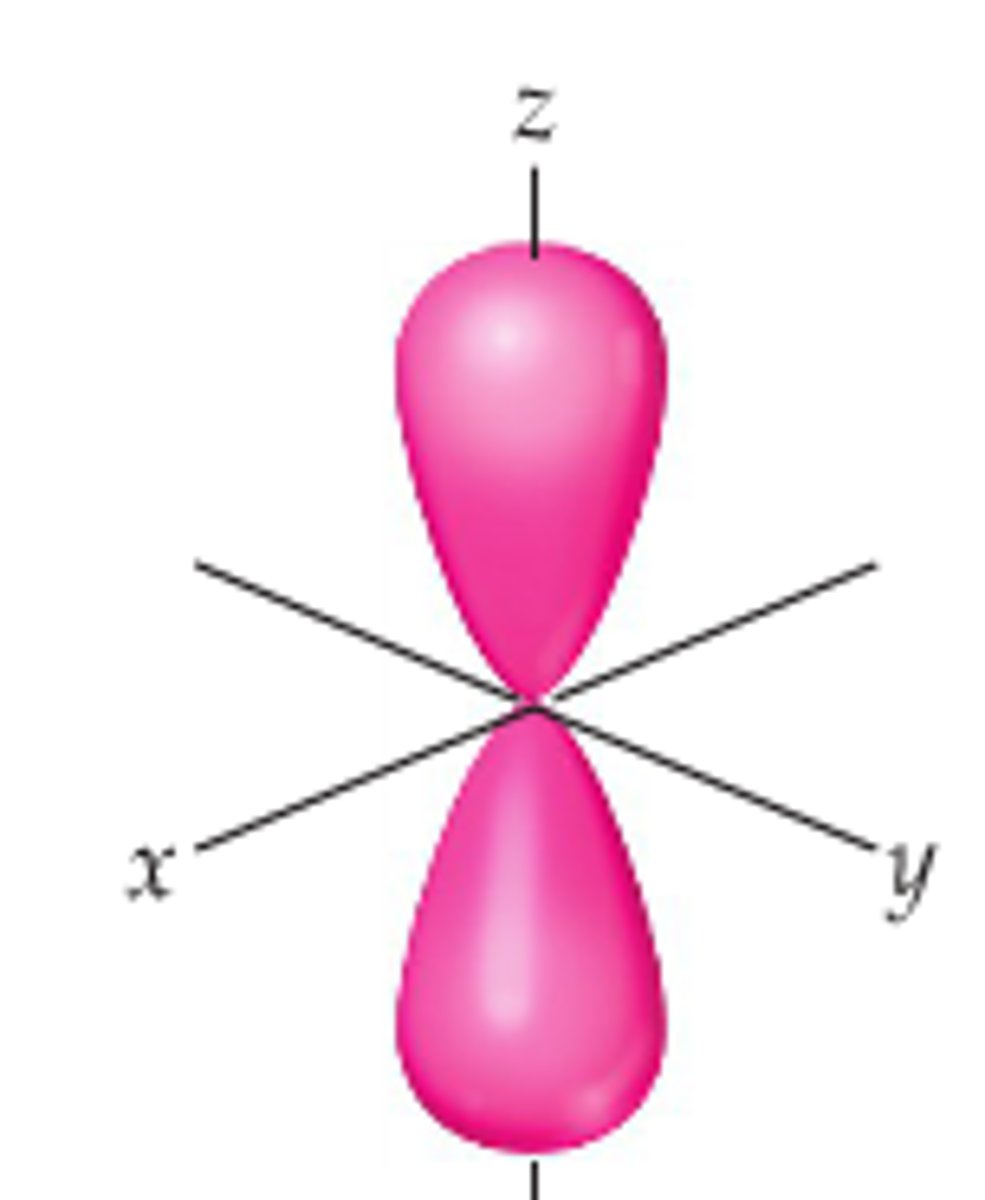
What does a p orbital look like?
How many electrons than a s sub-shell hold?
2
How many electrons can a p sub-shell hold?
6
How many electrons can a d sub-shell hold?
10
What is first ionisation energy?
The energy required to convert one mole of gaseous atoms in gaseous ions with a single positive charge
What is ionisation energy influenced by?
- Nuclear charge
- Electron shielding
- Atomic radius
- Filled/half-filled shells
How does nuclear charge affect ionisation energy?
The greater the charge the stronger the attraction for the outer electrons, meaning the ionisation energy is higher
How does electron shielding affect ionisation energy?
Inner electron shells repel the outer-shell electrons and also shield the outer electrons from the effects of the nuclear charge reducing the net attractive force on the outer electrons. This means the outer electrons are less strongly held and ionisation value is lower
How does atomic radius affect ionisation energy?
The larger the atomic radius the further the electron is from the nucleus and the less energy is need to remove them as the nuclear attraction experienced by the electron is small
How do filled/half-filled shells affect ionisation energy?
Extra stability is achieved when subshells are filled or half-filled, meaning the ionisation energy is greater