Endothermic and exothermic
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
21 Terms
endothermic and exothermic reactions are
changes in heat
endothermic
heat moves from the surroundings into the system
exothermic
heat moves out of a system into the surroundings
endothermic
absorb heat energy
exothermic
release heat energy
endothermic
negative ΔH
exothermic
positive ΔH
H
Enthalpy
What is enthalpy?
The amount of heat energy in a system
ΔH
Σbonds broken - Σbonds formed
Q
Heat
Endothermic
energy in products are smaller than the energy in reactants
Exothermic
energy in products are larger than the energy in the reactants
Endothermic
ice melting
Exothermic
Gas condensing
Endothermic
bonds break
Exothermic
Bonds form
Endothermic
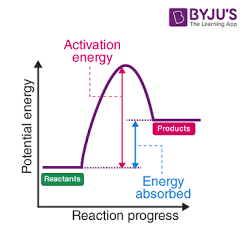
Exothermic
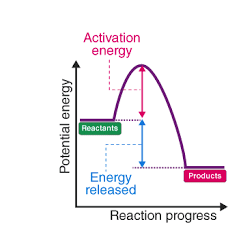
Activation energy
energy needed for a chemical reaction to occur
Bond enthalpy
energy required to break covalent bonds in a gaseous molecule