Chemistry A level 16/03/26
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
What is the enthalpy change of formation?
What is the enthalpy change of combustion?
Standard enthalpy change of formation of a compound is the energy transferred when 1 mole of the compound is formed from its elements under standard conditions (298 K and 100 kpa) and states.
Enthalpy change of combustion is the enthalpy change when one mole of a substance is burned completely in oxygen in standards conditions and states.
Enthalpy change of atomisation
Bond dissociation enthalpy
Enthalpy change where one mole of a gaseous atoms is formed from its element in standard states.
Enthalpy change when one mole of a covalent bond is broken into two gaseous atoms/free radicals
First electron affinity
Second electron affinity
Enthalpy change when one mole of gaseous atoms gains one mole of electrons forming one mole of gaseous 1- ions. (exothermic- electrostatic attraction with nucleus)
Enthalpy change when one mole of gaseous 1- ions gains one mole of electrons forming one mole of gaseous 2- ions. (endothermic- repulsion between negative ion and electron)
First ionisation enthalpy
Second ionisation enthalpy
Enthalpy change required to remove 1 mole of electrons from 1 mole of gaseous atoms to form 1 mole of gaseous ions with a +1 charge
Enthalpy change to remove 1 mole of electrons from one mole of gaseous 1+ ions to produces one mole of gaseous 2+ ions
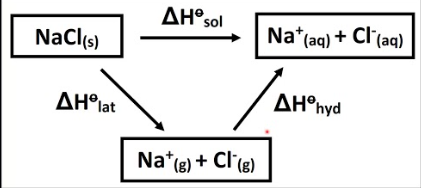
Enthalpy of lattice formation
Enthalpy of lattice dissociation
Enthalpy change when 1 mole of an ionic crystal lattice is formed from its constituent ions in gaseous form. (Na+ (g) + Cl− (g) → NaCl (s))
Enthalpy change when 1 mole of an ionic crystal lattice form is separated into its constituent ions in gaseous form
Enthalpy of hydration
Enthalpy of solution
Enthalpy change when one mole of gaseous ions become aqueous ions (exothermic)
Enthalpy change when one mole of an ionic solid dissolves in water to form an infinitely dilute solution.
What are the trends for lattice enthalpy?
What are the assumptions for lattice enthalpy
Trends: larger ion=larger enthalpy, greater charge= larger enthalpy
Assumptions: perfect ionic model, 100% ionic and spherical
Perfect ionic= no difference between theoretical and experimental (born haber) values.
What is the formula for entropy?
Entropy change?
ΔS = ΣS products - ΣS reactants (J K^-1 mol^-1)
Change of state, change of moles
Gibbs free energy
∆G = ∆H − T∆S
Negative= reaction is feasible as a spontaneous reaction
Rate equation
The Arrhenius equation
r = k[A]^m[B]^n, mol dm^3 s^-1
k = Ae^-EA/RT, A= Arrhenius constant, R= gas constant
Can be mapped out as ln k = ln A – EA/r x 1/T, with +x and -y, 1/T against ln k
Kp equation
How to work out partial pressure
Kp = pp(products)^m/pp(reactants)^n, only temp has effect on Kp (not pressure or catalyst)
mole fraction = moles of gas/ total moles of all gases
partial pressure = mole fraction x total pressure of all gases
How to work out emf
Cathode vs anode
EMF = rhs - lhs
Cathode is the negative electrode where reduction takes place
Anode is the positive electrode where oxidation takes place
Hydrogen fuel cell
Requires continuous supply of oxygen and hydrogen
Only has water as by-product
Danger = hydrogen flammable
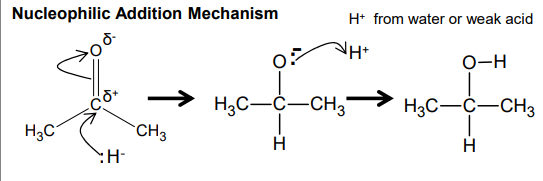
How are carbonyls reduced?
Nucleophilic addition: Uses reducing agent (NaBH4- source of nucleophilic hydride ions that attack the positive C=O carbon), aqueous ethanol
Catalytic hydrogenation: Nickel catalyst, H2, high pressure
Carbonyl addition with hydrogen cyanide
Nucleophilic addition with potassium cyanide (forms :CN- nucleophile) and sulfuric acid (supplies extra hydrogen to form alcohol group from C=O)
Forms racemate: planar carbonyl group has equal chance of being attacked from both sides
Carboxylic acid properties
Small chains up to C4 are soluble, forming hydrogen bonds with the water molecules, larger chains have hydrophobic tails that get in the way of the hydrogen bonds
Larger chain = weaker acid (electron density pushed towards COO- of carboxylate making it less stable, and therefore less likely for the acid to dissociate into)
Electronegative chlorine withdraws electron density from COO- making it more stable.
What is esterification?
Carboxylic acid + alcohol ←→ ester + water
Requires strong acid catalyst, heat under reflux, low yield, reversible
What can esters be used for?
Flavourings and perfumes- sweet smelling, non-toxic, unreactive with water
Use as plasticizer for polymers
Use as a solvent (for polar organic substances)
What is ester hydrolysis?
With acid: ester ←→ carboxylic acid + alcohol (dilute HCl, heat under reflux)
With base ester → sodium carboxylate salt + alcohol (dilute NaOH, heat under reflux)
Acid vs base hydrolysis of esters
Acid hydrolysis is reversible, doesn’t give good yield.
Base hydrolysis is not reversible, carboxylate salt is resistant to attack by weak nucleophiles, therefore reaction goes to completion.
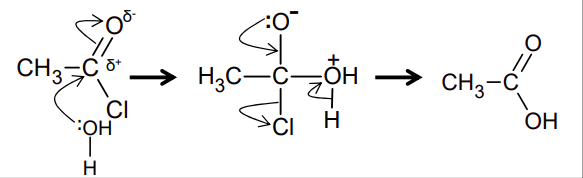
What is the reaction of Acyl chloride/acid anhydride with water?
Acyl chloride + water → carboxylic acid
RCOCl + H2O → RCO2H + HCl
Acid anhydride + water → carboxylic acid
(RCO)2O + H2O → 2RCO2H
Nucleophilic addition elimination
What is the reaction of Acyl chloride/acid anhydride with alcohol?
Acyl chloride + alcohol → ester
RCOCl (l) + CH3CH2OH → RCO2CH2CH3 + HCl (g)
Acid anhydride + alcohol → ester
(RCO)2O + CH3CH2OH → RCO2CH2CH3 +RCO2H
Nucleophilic addition elimination
What is the reaction of Acyl chloride/acid anhydride with ammonia?
What is the reaction of Acyl chloride/acid anhydride with primary amines?
Acyl chloride + ammonia → primary amide
Acid anhydride + ammonia → primary amide
Acyl chloride + primary amine → secondary amide
Acid anhydride + primary amine → secondary amide
Nucleophilic addition elimination
Why are acid anhydrides used instead of acyl chlorides?
Safer and less violent reaction
Doesn’t produce dangerous corrosive HCl fumes as a byproduct
Cheaper to produce and use
More stable and easier to store (less hydrolyzed by moisture)
What is a transition metal?
What is a ligand?
A metal with its atoms or ions containing incomplete d subshells
A molecule that can donate an electron pair, forming a coordinate bond
What is the chelate effect?
The substitution of monodentate ligand with a bidentate or a multidentate ligand leads to a more stable complex
Increase of entropy - more moles of products
Enthalpy change close to zero- same number of dative coordinate bonds, same energy required to break them
Therefore, Gibbs free energy is negative, the complex is stable
TM: What shapes do the complexes form?
Depends on coordinate number:
6 = octahedral, 4= tetrahedral (larger ligands), square planar, 2= linear