2.10 Equilibrium
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Reversible reaction
A reversible reaction is a reaction which can go in both the forward and backward directions.
E.g changes in state ice
What is closed system
no substances and no energy exchanged with the surroundings.
Nitrogen + hydrogen equilibrium ammonia formula
N2(g) + 3H2(g) |=| 2NH3(g)
Dynamic equilibrium
in a dynamic equilibrium the amounts of each reactant and product remain constant and the rate of the forward reaction is equal to the rate of the reverse reaction
Homogeneous reaction
a reaction in which the reactants and products are in the same physical state.
CATALYST MUST BE SAME STATE TOO
Heterogenous reaction
reaction in which the reactants and products are in a different physical state.
Position of equilibrium on products
equilibrium left, little product is formed.
equilibrium right, lots of product has formed.
Equation for equilibrium constant and units
Kc = [Products] over [reactants]
moldm-3
High Kc value indicates
> 1 position of equilibrium lies to the right-hand side.
<1 position of equilibrium lies to left
Le chateliers principle
if a change is made to the conditions of a system at equilibrium, then the position of the eguilibrium moves to oppose that change in conditions.
Adding catalyst to equilibrium
Does not impact position fo equilibrium as it speeds up rate of forward and reverse reactions equally

Increase Concentration of reactant on equilibrium
Reactant Forward reaction - faster (more particles more collision, more successful collisions) -
Equilibrium moved to right


Changing pressure
Increase pressure- equilibrium shifts to reduce it by moving to side with least number of gas molecules lower pressure

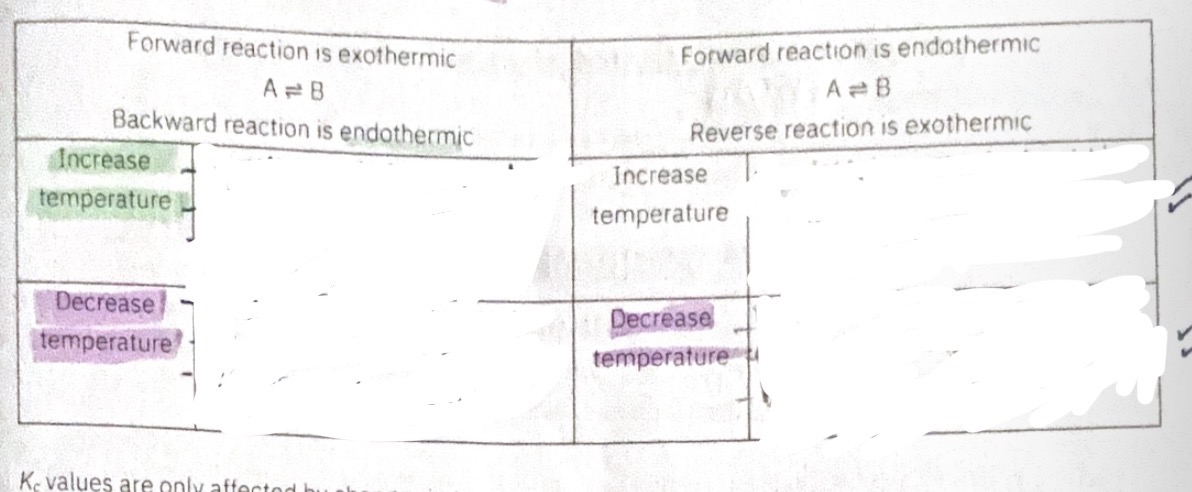
Changing temperature on equilibrium rules
Increase temp = favours endothermic
Decrease temp= favours Exothermic

Haber process formula
N2 (g) + 3H2 (g) {} 2NH3 (g) Delta H = -92kjmol-1 Exothermic
Temperature in haber process
Decrease temp - move position of equilibrium to right, to increase temp, gives high yield of ammonia
Why not use low temp in haber process
Low temp give very slow reaction so compromise temp of 450oC used give reasonable rate and yield
Pressure haber process
High pressure- equilibrium shifts right to decrease pressure (fewer gas molecules) , good yield of a,,o is
Why not very high pressure
Large amount of energy needed to compress gad and thick pipes required expensive, so compromise pressure of 200atm
Conditions for haber process
Temp : 450oC
Pressure: 200atm
Iron catalyst
Costs reduced in haber process
Recycling unused nitrogen and hydrogen.
Using the energy given off in the reaction to heat up the incoming gases
Contact process formula
Sulphuric acid produced
2SO2 (g) + O2(g) |=| 2SO3 (g) delta H -197
Temp on contact process
Increase temp - equilibrium right favour endothermic reaction - lower yield of product , compromise temp 450oC
Pressure on contact process
Increase pressure- equilibrium move right - fewer gas moles higher yield - compromise pressure of 2atm
Catalyst in contact process
vanadium (V) oxide as a heterogeneous catalvst allows equilibrium to be reached more quickly.
Conditions contact process
Temperature: 450°C
Pressure: 2 atm
Vanadium(V) oxide (V2O5) catalyst