Biology 173 - Intro Bio Lab Unit 1; Enzyme Action, Gut Microbiome, & Basic PCR
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
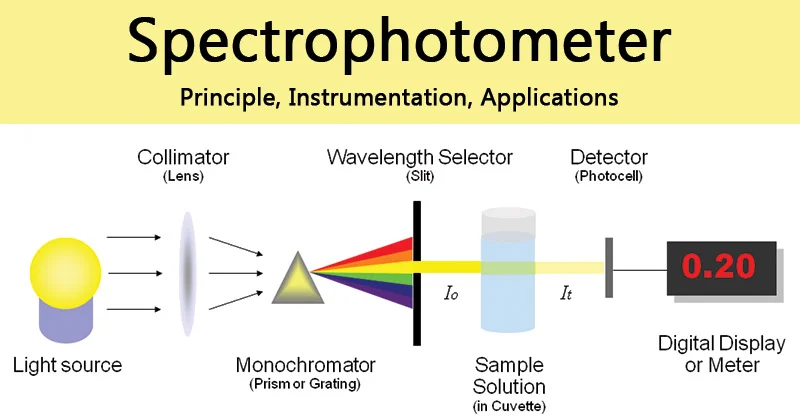
How does a spectrophotometer work?
Measures the amount of light that passes through or reflects off a sample to determine its color or the amount of chemicals in a solution
Uses a diffraction grating to separate light into different wavelengths, which are each allowed to exit one at a time
The light passes through the sample
A detector measures the amount of light that passed through the sample (transmittance) and the amount absorbed (absorbance)
What is metabolism?
The totality of an organism’s chemical reactions
What is a catalyst?
Chemical agent that speeds up a reaction without being consumed in the reaction
What is an enzyme?
A catalytic protein that speeds up chemical reactions in living organisms without being consumed or permanently altered in the process
How do enzymes and other catalysts speed up specific reactions?
By lowering the Ea (activation energy) barrier
Enzymes hasten spontaneous reactions that would occur eventually
How do enzymes affect Gibbs free energy (ΔG) of a chemical reaction?
They don’t! Enzymes have nothing to do with energetic favorability. They only lower the activation energy (ΔG‡) of the reaction, so they speed up the reaction rate by stabilizing the transition state. No change in free energy of the reactants or products.
HOW does the enzyme lower the Ea activation energy barrier?
It takes energy to break bonds, the enzyme gives you the push to break the bonds and form new ones to get to a lower energy state.
More specifically, the enzyme:
Orients the substrates correctly
Strains the bonds
Provides a favorable environment
What is the difference between exergonic and endergonic reactions?
Exergonic reaction releases energy:
products have less energy than reactions
Endergonic reaction absorbs energy
Requires inputting energy to proceed

In the Lucy and the Chocolate Factory example from class, describe how the video represents the substrate, active site, enzyme, and product.
Substrate: chocolate + wrapper
Enzyme: Lucy & Ethel
[Enzyme] is constant (2 units)
Active site: their hands
Product: wrapped chocolate
[Substrate] is the speed of the conveyor belt delivering chocolates
What are enzyme kinetics?
Study of enzyme mechanisms through reaction rates under various conditions
What are the two factors that affect the speed of an enzymatic reaction?
[substrate] & [enzyme]
The concentrations of both are important, though each have different effects
How is time or rate important for an enzyme? What are the two “time requirements” that affect the rate of enzyme catalysis?
Think of enzyme catalysis in two parts
find time - time to bind to the next substrate
processing time - time to convert substrate into product
What does a Vmax graph, where the rate of the reaction is graphed against substrate concentration, show/tell you?
All of the rates of the reaction at increasing substrate concentrations for an enzyme
It’s important to note that this graph does not have time anywhere on it; it’s not about a single situation over time but about all of the specific reaction rates that occur at each specific substrate concentration for a specific enzyme
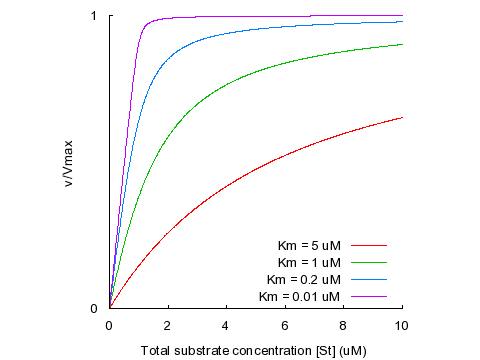
Where is Vmax located on this graph?
It occurs for each color line at the highest point, where each line plateaus.
What is Vmax?
The maximum rate of the reaction
This occurs when all of the active sites on each enzyme are full (active saturation)
What is KM?
The substrate concentration at which the reaction rate is half-maximal, or half of its Vmax.
What does KM tell you about an enzyme?
The affinity of the enzyme for the substrate
The lower the KM, the greater the affinity
Many cases, you will need to consider Km the “apparent” affinity for an enzyme. if there are things that reduce how well the enzyme binds to the substrate, it will appear that the affinity is affected, though we know the enzyme’s true Km is the same as it’s the same enzyme.
Why is it that a lower KM indicates a greater affinity for the substrate?
The KM is the substrate concentration at which the reaction rate is half of the maximum velocity. Therefore, a lower KM value indicates that a lower substrate concentration was needed to already get the reaction to reach half of its maximum speed, showing that the enzyme is efficient, and doesn’t have to use a lot of substrate to work well, meaning it has a high affinity for the substrate.
Why does an enzymatic reaction reach a Vmax despite increasing substrate concentrations?
At a certain point, no matter how much substrate is added, the reaction cannot proceed faster because all the enzymes are saturated.
When an enzyme is saturated, it can’t handle any more substrate
High [substrate] decreases “find” time for an enzyme to find a substrate but does not affect “process” time, where the enzyme has to convert substrate into a product
How does a competitive inhibitor work?
Binds to the active site
Physically competes with substrate
Can a competitive inhibitor be influenced by the amount of substrate?
Yes; since a competitive inhibitor physically fights with substrate to bind to the active site, adding more substrate relative to the amount of inhibitor will make it less likely the inhibitor will bind to an enzyme before the substrate is able to.
How does a noncompetitive inhibitor work?
Binds to another part of the enzyme (allosteric site) and changes its shape
Shuts down the enzyme
Can a noncompetitive inhibitor be influenced by the amount of substrate?
No; because the noncompetitive inhibitor shuts down the enzyme/prevents substrates from binding through a different site, it is not affect by substrate concentration
Are things like temperature and pH noncompetitive inhibitors?
No, since these are not tangible things that bind to the enzyme in any way, they are not exactly inhibitors. However, they can affect the rate of the reaction.
What is a non-specific inhibitor?
Affects ALL enzymes in the system
Something like a poison, completely denatures all enzymes
What effect does a competitive inhibitor have on KM, and why?
KM will be higher.
The substrate now has to compete with an inhibitor to get to the active site, so it takes a higher [substrate] to get the same rate of product formation (or same proportion of active sites filled by substrate).
![<p>K<sub>M </sub>will be higher.</p><p>The substrate now has to compete with an inhibitor to get to the active site, so it takes a higher [substrate] to get the same rate of product formation (or same proportion of active sites filled by substrate).</p><p></p>](https://knowt-user-attachments.s3.amazonaws.com/b0336eff-2867-4e52-b24d-53eed394e2b7.png)
What effect does a noncompetitive inhibitor have on KM, and why?
There will be little to no effect on Km, depending on whether the substrate binding and processing is affected.
This is because Km is a measure of AFFINITY, or how well the enzyme appears to be bonding to the substrate. The inhibitor lowers the amount of overall active enzyme molecules available, which doesn’t affect the Km since enzyme concentration does not change how well the enzyme bonds to the substrate. Therefore, the enzyme will still appear to be bonding to the substrate with the same affinity and Km will be the same.
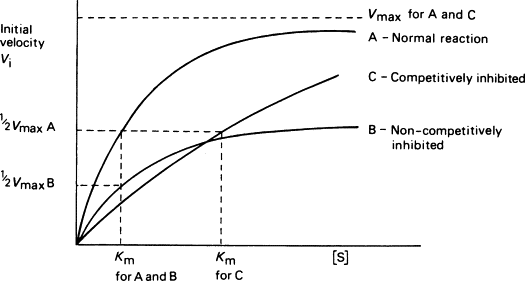
What effect does a competitive inhibitor have on Vmax, and why?
No effect; it stays the same.
Vmax measures the maximum reaction rate, and competitively inhibiting an enzyme doesn’t change highest possible reaction speed. It may take a greater substrate concentration to get there as increasing substrate concentration relative to inhibitor helps raise reaction rate, but ultimately the MAXIMUM rate will be the same.
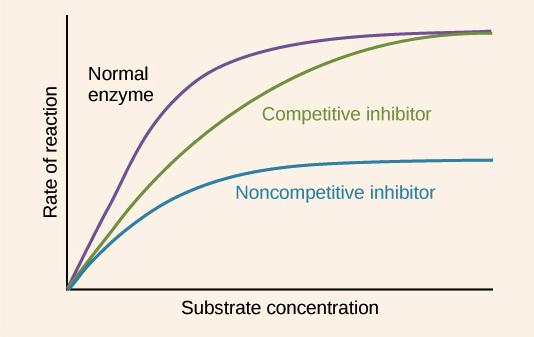
What effect does a noncompetitive inhibitor have on Vmax, and why?
Vmax is lowered.
Another way you can think about Vmax is when enzymes are totally saturated. Because the inhibitor changes the shape of the enzyme, preventing it from reacting, it reduces the amount of enzyme molecules that can catalyze the reaction. This means a lower rate because there are less enzymes that can be saturated.
With a competitive inhibitor, what effect does increasing substrate concentration have on percent inhibition?
Increasing substrate reduces % inhibition. This is because more substrate (relatively to the inhibitors) means less competition with inhibitors as the ratio between the two is much lower.
With a noncompetitive inhibitor, what effect does increasing substrate concentration have on percent inhibition?
Increasing substrate has no effect on % inhibition. This is because the active site’s shape is physically changed, crippling the enzyme, so no matter how much substrate concentration is increased, there is no change in how well the enzyme is working or not.
Give the percent inhibition formula
(rate without inhibitor) - (rate with inhibitor)/(rate without inhibitor)
x 100
How does the enzyme polyphenoloxidase work? How can its reaction rate be measured?
Oxidizes polyphenolics to quinones
Orthoquinone breaks down into other compounds
Some of those are colored (absorb strongly at 525nm)
Increase in absorbance over time tells us rate (= slope)
What is a hypothesis?
A possible explanation that leads to a testable prediction and motivates experiments
More broad
Based on observations and assumptions
Must be falsifiable, making specific predictions that can be critically tested
Not an “educated guess,” or an “if-then” statement
Give an example of a hypothesis you could make after observing why a desk lamp isn’t working
The light isn’t coming on because the bulb is burnt out
The light isn’t coming on because the bulb is loose and not plugged in
What is the difference between a hypothesis and a prediction?
A hypothesis is more of an explanation of an observation, an idea of how something works, while a prediction is a specific prediction of what will happen in the future based on the hypothesis.
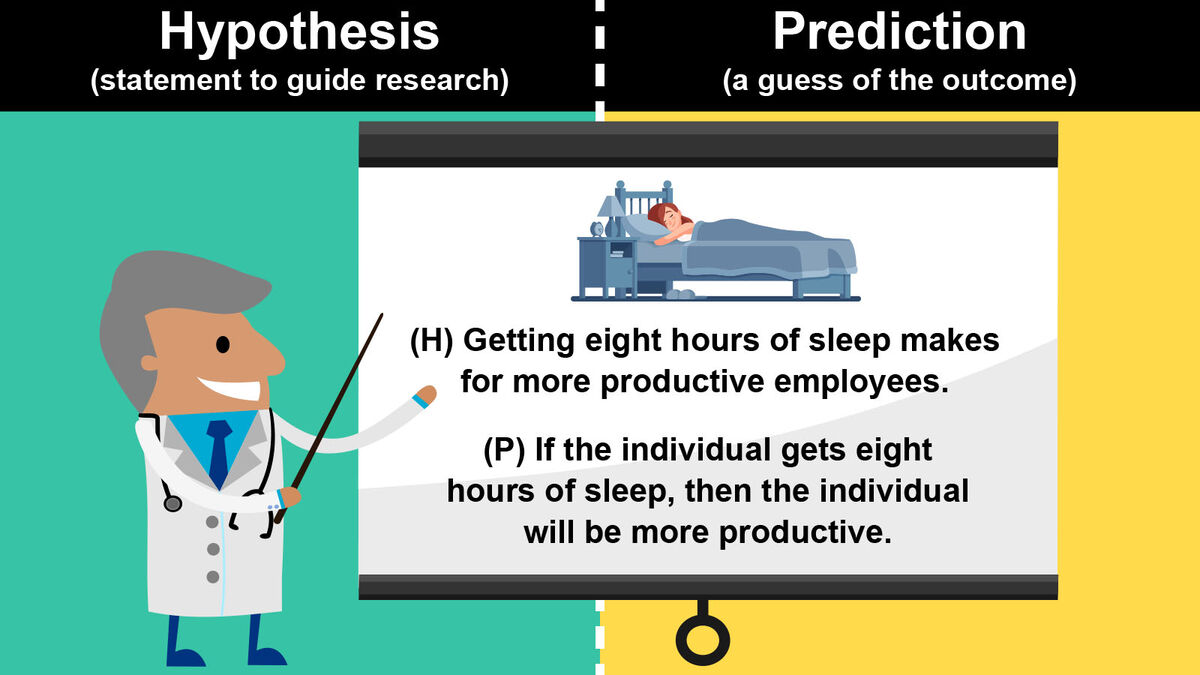
Is the following a hypothesis? “If a plant receives fertilizer, then it will grow to be bigger than a plant that doesn’t receive fertilizer.”
No, this is a prediction.
An example hypothesis for this situation could be something like “Plant growth is affected by fertilizer.”
What are reasons you might use a control?
Provides a baseline value for comparison
Makes sure everything is working properly
What possible issue regarding the experiment does the use of the control help eliminate?
Using a control helps make sure there aren’t other variables, such as confounding variables that were the real cause of the results.
The control helps you narrow down the results of the experiment to the manipulated variable.
What is a negative control?
A control in which a variable that you expect to have no effect is applied.
If the experiment does show a result for this negative control, you know something else is influencing results.
Ex. placebos, saline solution, etc.
What is a positive control?
A control in which a variable that you expect will have a specific effect is applied.
If the experiment doesn’t show results for this positive control, you know that something else is influencing results, and it could have stopped the effect of the independent variable you changed, too.
Ex. a drug that will have a specific effect, a medication that you know will work
To make a dilution that is 1/3 it’s original concentration, how many parts solution to how many parts water should you add?
1 part substance + 2 parts water
To make a serial or stepwise dilution that is 1/9 its original concentration, how many steps/dilutions should you include if each dilution is a 1/3 dilution?
Twice
1/3 × 1/3
What are quantitative observations?
Measurements that record numerical values, things you can organize into tables and graphs
What are qualitative observations?
Measurements that record descriptions or thoughts; not a distinct “measurable” quantity
List the steps of the Scientific Method
Explore/Observe → Ask a Question → Conduct Research → For a Hypothesis → Experiment → Analyze data and Form Conclusions
→ Do results support hypothesis?
Yes → Report Results
No → Go back to hypothesis, retesting
What were the results of running the reaction at 0C, 20C (room temp), 45C, and 65C?
In order from fastest to slowest, the temperature conditions were 45, 20, 0, 65.
Warmth helps the reaction run a little faster; high heats denature the enzyme
What were the results of freezing and thawing the enzyme vs boiling it and letting it cool?
Thee reaction rate was for the control ≈ frozen, which was much greater than the boiled enzyme.
Boiled enzyme was denatured
The boiled enzyme had a cloudiness meant that the protein unfurled, which made the vial cloudy and increased absorbance
This is why absorbance doesn’t matter → look at reaction rates
What were the results of testing adding 100µl of arsenite to the enzyme with varying substrate concentration?
Percent inhibition should be close at each concentration
There is no inhibition happening; arsenite is not an inhibitor for this enzyme because the Vmax doesn’t change
Learned that there are no - SH groups in PPO (enzyme). So while arsenite might be a potential inhibitor for other enzymes, it is not in this case because this enzyme doesn’t have sulfhydryl group in its tertiary structure to disrupt.
What were the results of checking whether para-hydroxybenzoic acid is an inhibitor?
PHBA has a chemical structure similar to catechol
PHBA show partial inhibition
% inhibition at high substrate concentration is less than % inhibition at low substrate concentration
PHBA is a competitive inhibitor
Results of using polyphenoloxidase have a metal ion cofactor (copper or iron)?
The enzyme has a copper cofactor for sure
You don’t know if the enzyme for sure has an iron cofactor
Need to test a chelator that only draws out iron to improve this experiment
The effect of increasing salt on the enzyme
% inhibition is not truly appropriate, as it isn’t actually an inhibitor
Inhibitor has to bind to the enzyme
Salt denatures the enzyme
Should have pretreated your enzyme to allow for denaturation
As the concentration of salt increases, reaction rate decreases (to a certain point)