chem ch.16 questions from book
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
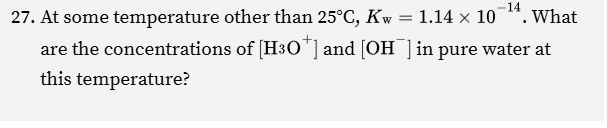
Remember that [H] x [OH] = kw
kw = 1.14e-14
So if we only have kw here , and we see [H]+ as X, and (OH) as X
That’s the same as
X² = 1.14e-14
we can solve for x
![<p>In this equation, you’ll set up an ice table and find that it ends with</p><p></p><p>Ka = (2.5e-5)² / X-0.1<br><br>Because the value for [H]+ was so small, JUST LIKE WE DO FOR Ka,</p><ul><li><p>You can remove the X to find Ka.</p></li></ul><p></p><p></p>](https://assets.knowt.com/user-attachments/669d8785-e5ec-42f6-a87d-a07a2b41aeb0.png)
In this equation, you’ll set up an ice table and find that it ends with
Ka = (2.5e-5)² / X-0.1
Because the value for [H]+ was so small, JUST LIKE WE DO FOR Ka,
You can remove the X to find Ka.
x

If both the components are a strong base and strong acid component the pH will be…
7

Count the O’s, more O’s attatched = higher strength
See other atoms
F > Cl > Br> I
O > N > C> H
For strength!
Important Count oxygen groups first, this is the biggest dealbreaker for strength.
a
Remember these
As the strength of the bond to H increases, the strength of the acid → decreases
Strong H–X bond → harder to lose H⁺ → weaker acid
As the polarity of the bond to H increases, the strength of the acid → increases
More polar bond → H⁺ more easily released → stronger acid
As the size of the anion bonded to H increases, the strength of the acid → increases
Larger anion → negative charge better stabilized → H⁺ easier to lose → stronger acid
a
When solving a polyprotic equation, what’s the point of using K1 vs K2?
You write out the full expression of lets say
H3C6O2 acid
for k1
H3C6O2 + H2O —> H2C6O2 + H3O
For k2
H2C6O2 + H2O —> HC6O2 + H3O
For k3
HC6O2 + H2O —> C6O2 + H3O
So if you’re asked to find the concentration of C6O2, this is where we’d have to use K3 to find it, we’d need to make 3 seperate ice tables to find the concentration of C6O2.
For pH, you can usually just use the value of H3O+ at k1.
stopped at 16.9, finish 16.9 and all of 17.0 except the portions she said not to tomorrow. Then just review.
What are 2 traits to look for in a Lewis base?
negative charge
lone pair
What traits to look for in a lewis acid?
positive charge
incomplete octet -
Check for a periodic table if we’re gonna get one on the exam, because the image explains it pretty clearly based on valence electrons, but you need a periodic table to know that.


memorise these i guess
d

Make ice table, it’s unique for this one because when k is small for this example
we can do ka (given just didnt write it) = (20 + x) (x) / (30 - x)
and it becomes
Ka = (20)(x) / 30
so we eliminate the x that’s combined with a value in both the numerator and denominator.


a
A buffer forms on weak acids and bases between a conjugate acid (1 more H+) and its conjugate base (has 1 less H+)
only B is forming a buffer because there’s a 1H+ difference between them
Note, any time there’s a strong acid or base present, a buffer can’t form.
So HI and I, even though they’re conjugate base + acid, they can’t form a buffer because HI is strong

x
Adding products shifts to left
(remember adding reactants right, decrease reactant left, increase products left, decrease products right
So PCl5 goes up, PCl3 goes down, Cl goes down
Ammonia =
Ammonium ion =
Ammonia = NH3
Ammonium = NH4
Should be NH3 + h2O —> NH4 + OH

list the steps for solving and the setup
% ionization (0.01, which is 1% as decimal) = H3O+ eq / acid initial (0.1). don’t include 100.
we solve for H3O, then go back to the balanced equation to see the concentrations for HZ and Z so we can plug into Ka

Find the concentration of OH ions in each of the following, sometimes it may be necessary to use Kw to solve.
A. Find H3O with pH —> Use Kw to solve for OH once you have H+
B. Strong acid disassociates completely, so H+ = HNO3 conc, then use Kw to solve for OH
C. Convert pOH to OH-
D. Water : 1e-14 = [H][OH) is the same as 1e-14 = X² , so we solve for x to get the conc of OH

In the HH equation
What’s unique about the values that can be used for log(base/acid)
moles can be used instead of just M
x

Find pKA
Plug given pH in
simplify
do 10(-)value to get the ratio of ion to acid.



For a. just calculate the pH by doing
pka = -log(ka)
Then do log (base/acid)
For b. write out a reaction
Weak acid + strong base (OH) —> conjugate acid + h2o
make BAA chart


solve
M1V1 = M2V2
(0.05L) (0.5M KOH) = (0.5L) (M2)
Solve for M2, this is the new concentration of OH after adding, because KOH disassociates completely.
Then use that to solve for pH, because you have conc of OH- ions.

d
Liam for titration
If starting with weak acid + strong base
do weak acid + OH —> Weak base conjugate(minus 1H) + H2O
if starting with weak base + strong acid
do weak base + H3O —> weak acid conjugate (+1H) + H2O
If it’s Strong acid + Strong base
Do H+ + OH- —> H2O
General rule of thumb, never include the full equation for a strong base or acid, just simplify to H+ or OH
Convert initial molality of Acid and Base in buffer to moles, i.e do 0.250M of acid x 0.500L to get moles
Calculate moles of new substance’s OH or H+ by doing the ml added x the M it contained
Write out in ice table, subtracting the new substance value found in step 2 from the new initial substance found in step 1
Do the new EQ concentration / Total volume (volume used in step 1 + volume in step 2)
plug into the henderson pH equation.

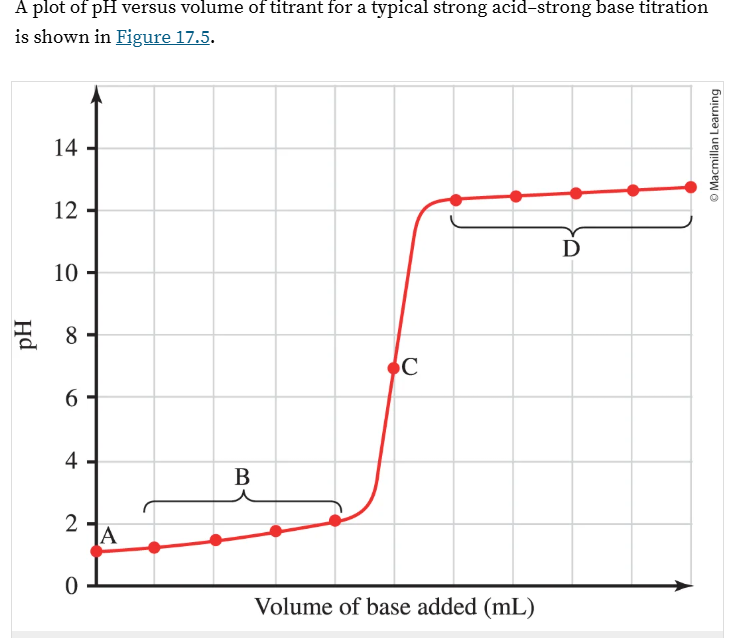
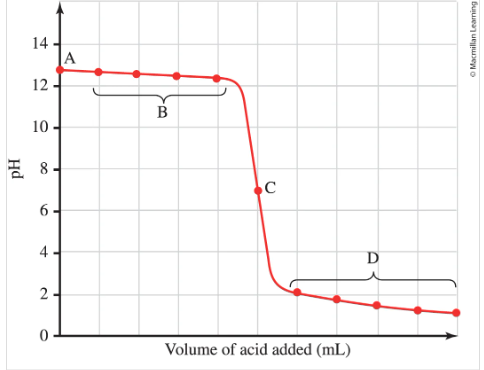
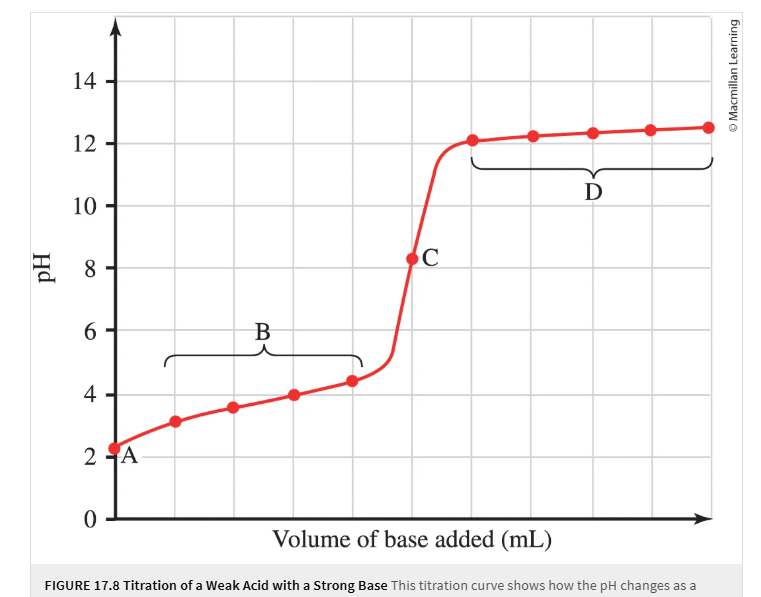
What does the X and Y axis represent?
What do regions A, B, C, and D represent?
Strong Acid-Strong base titration
X axis = volume of base added in ml
Y axis = pH
A = initial pH of acid alone. it can be found by doing -log[H3O]
B = after base has been added but before the EQ point. This pH can be calculated from the conc of H+ that hasn’t been neutralized
C = EQ point at pH = 7.0
D = after EQ point where excess base determines the pH. First calc the pOH from the conc of excess OH-, then calc the pH.

SA - SB titration
calc pH by doing -log(0.300)
For b.
First, convert the molality to moles of HCl, and moles of NaOH
this is done by doing Molality x Liters of solution
After you do this, plug them into Ice Table, Leave the initial value of the acid and the inital value of the base, and subtract do Initial H+ - Initial OH, this will give you an EQ value in moles. Divide that EQ value by the total amount of volume in the solution, to get it into molality. From here, you can calculate pH
c. pH of a titration of a Strong Acid and Strong base will always be 7
D. First, multiply the moles of the base by the volume you’re now adding
Then, the value you just obtained in part D, you are going to subtract the initial concentration found in part A from it.
After this, you will divide by the total liters added of the base
This will give you an answer in OH-, —> convert to pOH —> convert to pH
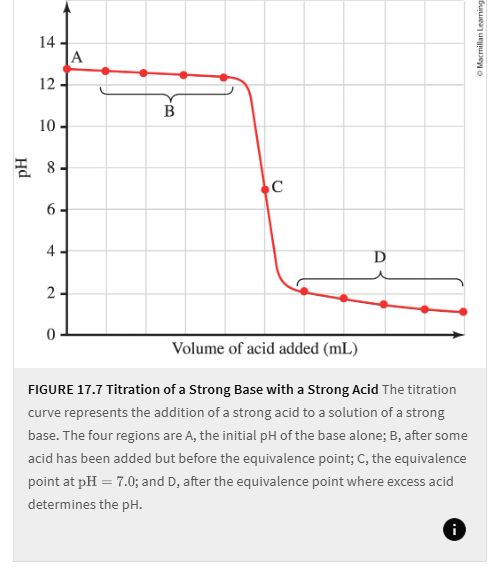
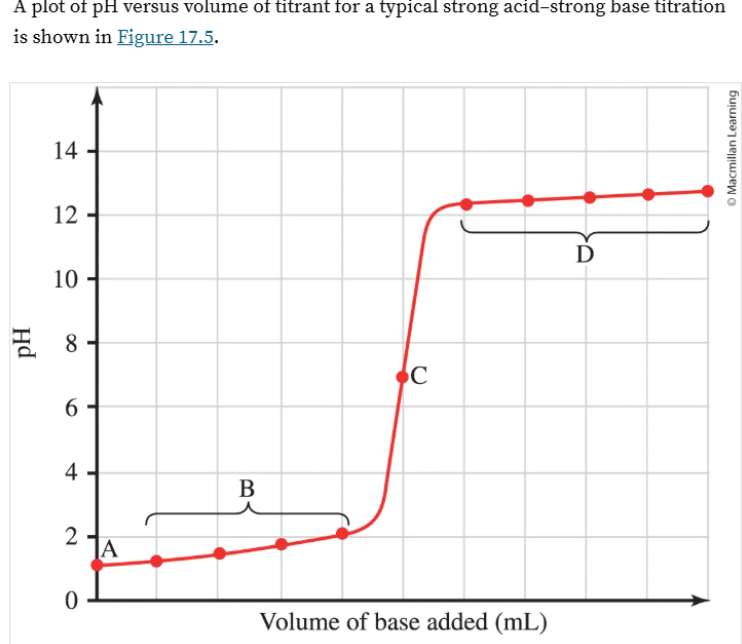
The following shows
Titration of a strong base with a an acid
The following shows
Titration of a strong Acid with a strong base

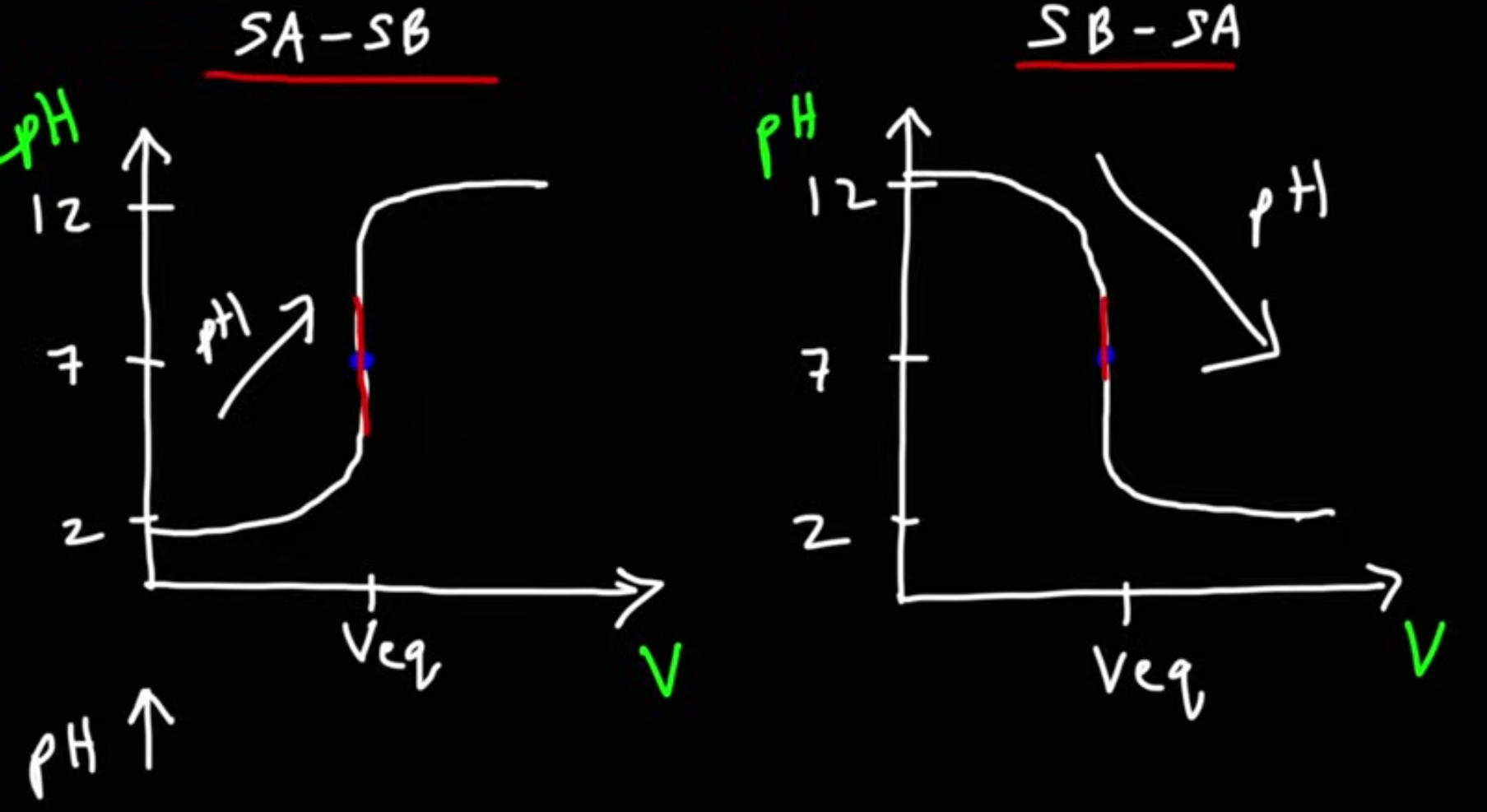
SA-SB
Strong acid, with stong base added
SB-SA
Strong base, with strong acid added.
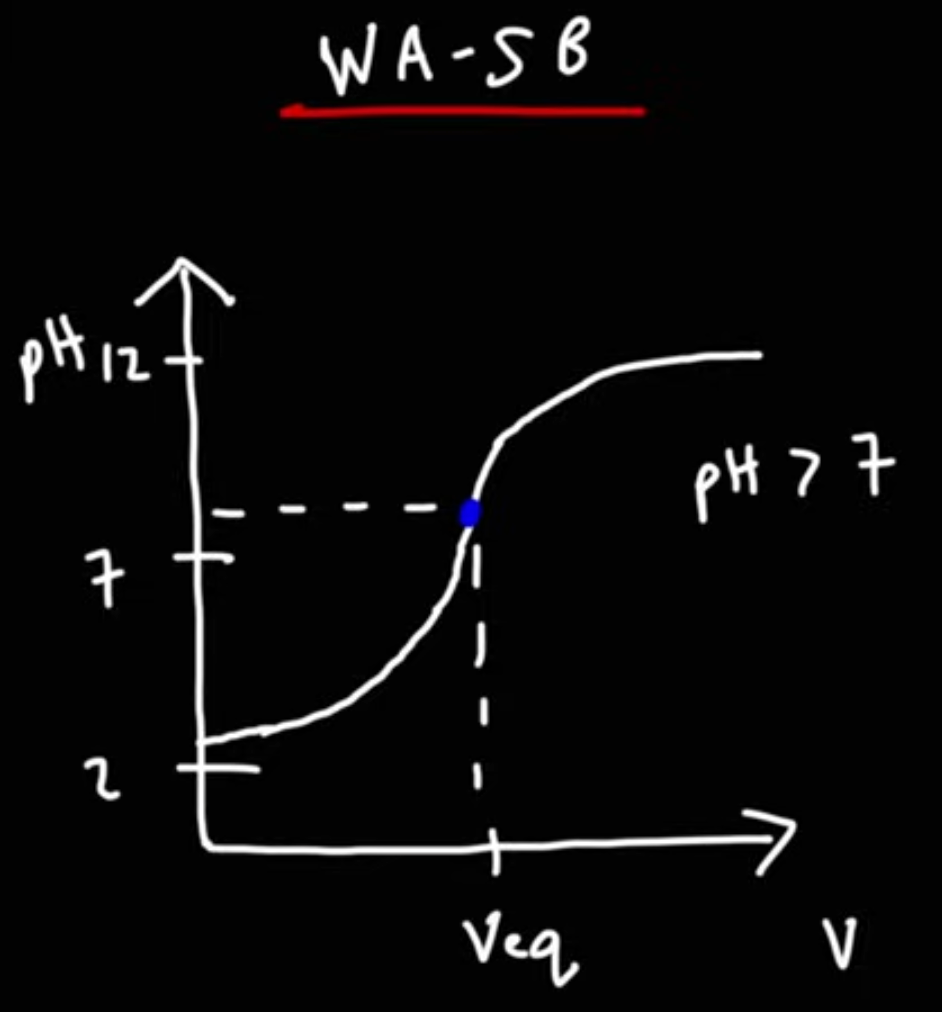
What’s the following show?
WA-SB
a. Inital pH
B: Buffer range where some conjugate base has been produced but not all WA has been consumed
C: Equilavence point where pH is more than 7
D after equilavence point where excess base determines pH

Determine which will have highest pH at EQ point of titration given pKA between several acids
The higher the pKA, the weaker the acid, and the higher the pH.

a
phenol, highest pKA, highest pH, weakest acid.
ammonium ion, pkA, middle pH,
hypobromous acid, lowest pKA, strongest acid.

a
Step 1: Before NaOH added pH
Write out HA ←→ H + A
Make an Ice table to calculate pH using the given Ka
Half of the equilavence point
Rewrite equation as
HA + OH —> A + H2O
Show me the exact math yo
value (i.e -2) = log (base/acid)
How do you solve for the base and acid?
10-2 gives you ratio of base to acid, as it will cancel the log
-2 = log(base/0.750)
How do you solve for moles of base?
10-2 will cancel the base, and become 0.01
0.01 = X/.750
solve for X

A. 0.0500mol NaOH in 1L of water
Find [OH-] M
0.0500mol NaOH/ 1L = 0.0500 M
From here, you can find pOH —> PH
![<p>A. 0.0500mol NaOH in 1L of water</p><ol><li><p>Find [OH-] M </p></li></ol><p>0.0500mol NaOH/ 1L = 0.0500 M</p><p>From here, you can find pOH —> PH</p><p></p>](https://assets.knowt.com/user-attachments/052023ae-55d0-4b89-bdd1-e7fa9e184635.png)
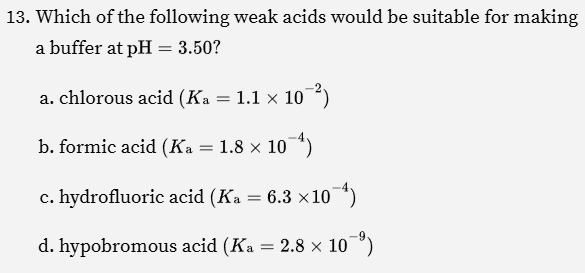
For solving this problem, what’s the trick that we want to look for?
Find a value where pKA = pH
Buffer works best when pH = pKa

What do you do to solve this?
Find a pKA that matches 7.4
Step 1, titration
What’s the first step for all titrations
Step 2, calculating pH before adding any acid or base, what do you write out?
Step 3, calculating pH after adding base. What is it for
SA + SB
vs WA + SB?
Step 4: Calculating pH at EQ for SA + SB
Step 5 Calculating pH at halfway point for WA + SB
Step 6: Calculating pH at Eq for WA + SB
step 1: MaVa = MbVb , this gives you the volume at the Equilavence point.
Step 2: Calculate pH using an Ice table, simply write the disassociation of the SA or WA and calculate with an ice table (if it’s a WA it will form a conjugate base + H+). If it’s a SA, it will match the [M] of the H+.
Step 3. Any pH before EQ point
Rewrite expression for SA + SB as
OH + H —> H2O
B
A
A
Rewrite expression for WA + SB as
WA + OH —> H2O + Conjugate base of WA
B
A
A
pH at EQ for SA + SB = 7
pH at halfway point for WA + SB = pKa
pH at Eq point for WA + SB
WA + OH —> H2O + Conjugate base of WA
you’ll eliminate all OH + WA and be left over with conjugate base of WA
Take the moles of Conjugate base of WA produced, and divide by total volume of WA + OH added to get it in (M)
Create an Ice table showing how the Conjugate base reacts with water
Conjugate Base WA + H2O —> OH + Conjugate Acid of WA
Use Kb instead of Ka, as you’re solving for a base
Solve for X
Convert X to OH, and solve for pH
Any amount past the EQ point for SA-SB or WA-WB
take the leftover from the
B
A
A
and convert it into (M) then into pH
A buffer consists always of a
conjugate acid and its base