Card 141 Lecture 23: Acid-Base Regulation
1/203
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
204 Terms
What is the normal physiological pH range of body fluids?
7.2–7.4
What is the normal plasma [H⁺] concentration?
3–5 × 10⁻⁸ mol/L
Which buffer system is the most important in the ECF?
Bicarbonate buffer system
Which organ eliminates CO₂ to regulate acid–base balance?
Lungs
Which organ eliminates non‑volatile acids
Kidneys
Increased [H⁺] stimulates ______ ventilation.
Increased (hyperventilation)
(↑[H⁺] → ↑ ventilation → ↑ CO₂ loss)
The kidneys regulate acid–base balance by secreting ______ and reabsorbing ______.
H+; HCO3-
Ammonia buffering occurs primarily in the ______.
Renal tubules
T/F: Proteins are the most important extracellular buffer.
False: bicarbonate is the most important ECF buffer; proteins are mainly intracellular.
T/F: Phosphate is the major buffer in the renal tubule.
True
What’s the reaction for hemoglobin buffering?
H⁺ + Hb ⇌ H‑Hb
What’s the reaction for ammonia buffering?
NH₃ + H⁺ ⇌ NH₄⁺
What is the pK of the bicarbonate buffer system?
6.1
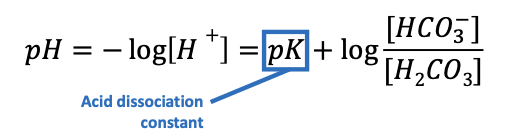
What is the Henderson–Hasselbalch equation for bicarbonate?
pH = 6.1 + log([HCO₃⁻] / (0.03 × pCO₂))
What happens to pH when CO₂ increases?
pH decreases (acidosis)
What happens to pH when bicarbonate increases?
pH (alkalosis)
CO₂ + H₂O ⇌ H₂CO₃ ⇌ ______ + HCO₃⁻
H+
The CO₂ solubility factor used in the Henderson–Hasselbalch equation is
0.3
T/F: The bicarbonate buffer system has a pK close to physiological pH.
False: pK for bicarb is 6.1, far from 7.4.
T/F: Adding acid shifts the bicarbonate buffer curve toward more H₂CO₃.
True
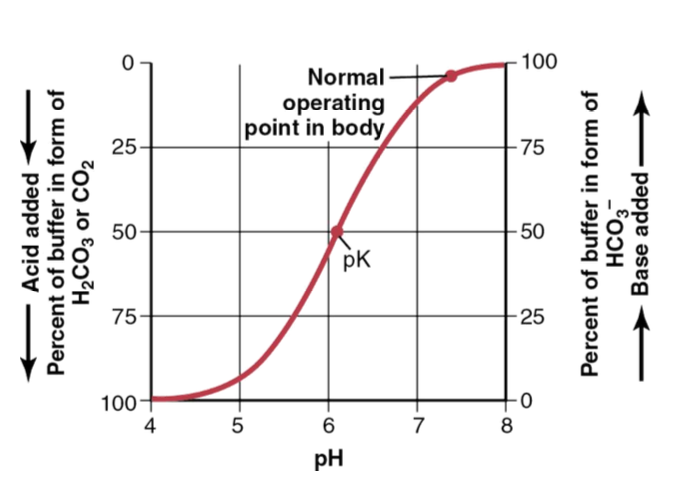
At the normal operating point, what percentage of the buffer is in the HCO₃⁻ form?
75&
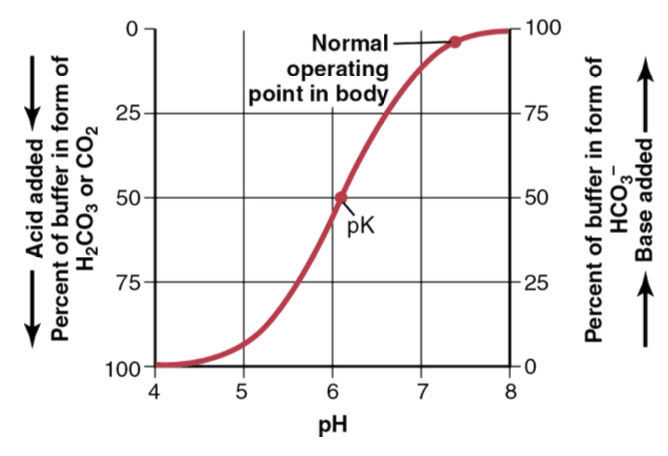
What happens to the buffer ratio when base is added?
HCO₃⁻ increases relative to H₂CO₃
Why is the bicarbonate buffer system effective despite a low pK?
Because CO₂ and HCO₃⁻ are tightly regulated by lungs and kidneys
What happens to ventilation when [H⁺] increases?
Ventilation increases (hyperventilation)
What percentage of metabolic acid disturbances can respiratory compensation correct?
50–75%
A fall in pH from 7.4 to 7.0 indicates a(n) ______ in [H⁺].
increase
Hyperventilation causes a ______ in pCO₂.
decrease
T/F: Respiratory compensation can fully normalize pH in metabolic acidosis.
False: it only partially corrects pH
T/F: CO₂ retention leads to alkalosis.
False: CO₂ retention leads to respiratory ACIDosis
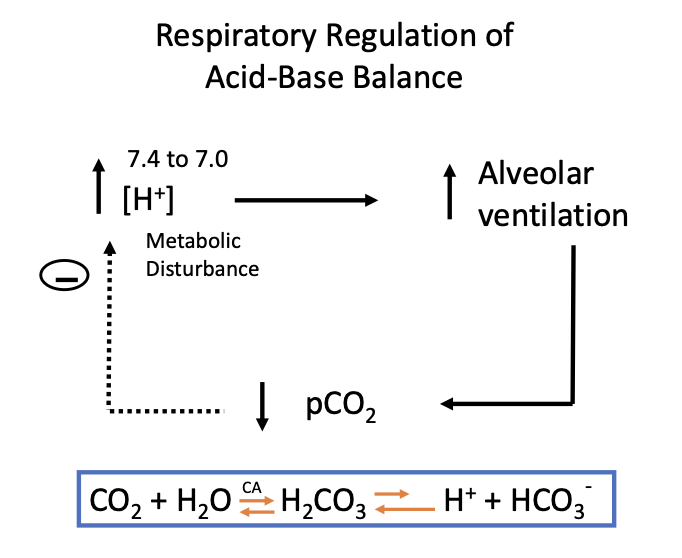
In the diagram, what metabolic disturbance causes increased ventilation?
Metabolic acidosis (↑H⁺)
What is the primary disturbance in metabolic acidosis?
Decreased HCO₃⁻
What is the primary disturbance in respiratory acidosis?
Increased pCO₂ (hypoventilation)
What causes respiratory alkalosis?
Hyperventilation (↓CO₂)
What happens to pH during metabolic alkalosis?
pH increases
Respiratory acidosis is caused by ______ ventilation.
Hypoventilation
Metabolic alkalosis is caused by increased ______ or loss of ______.
HCO₃⁻; H⁺
T/F: Metabolic acidosis is caused by increased hydrogen ions (H+)
True
T/F: Respiratory alkalosis results from decreased CO₂ elimination.
False: it results from increased CO₂ elimination
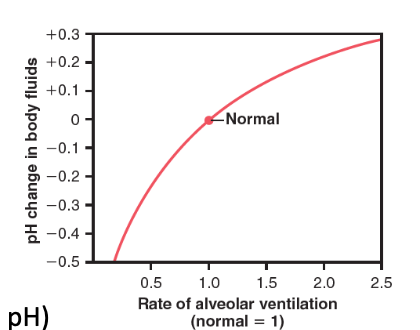
As alveolar ventilation increases above normal, what happens to pH?
pH increases (alkalosis)
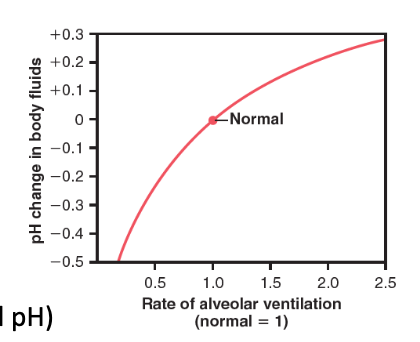
At half‑normal ventilation, what happens to pH?
pH decreases significantly (acidosis)
Which direction does the reaction shift during respiratory acidosis?
Right (toward H⁺ + HCO₃⁻)
Which direction does the reaction shift during respiratory alkalosis?
Left (toward CO₂ + H₂O)
Hypoventilation causes ______ pCO₂ and ______ pH.
Increased; decreased
Hyperventilation causes ______ pCO₂ and ______ pH.
Decreased; increased
T/F: Respiratory disturbances change pCO₂ first.
True
What is the rapid compensatory mechanism for metabolic acidosis?
Respiratory hyperventilation
(Acidosis – stimulate chemoreceptors – hyperventilation – decreased PaCO₂)
What is the rapid compensatory mechanism for metabolic alkalosis?
Respiratory hypoventilation
(Alkalosis – inhibits chemoreceptors – hypoventilation – increased PaCO₂)
What is the slow compensatory mechanism for respiratory acidosis?
Increased renal H⁺ secretion and increased HCO₃⁻ reabsorption
What is the slow compensatory mechanism for respiratory alkalosis?
Decreased renal H⁺ secretion and decreased HCO₃⁻ reabsorption
Respiratory compensation for metabolic acidosis occurs within ______.
minutes
Renal compensation for respiratory disturbances takes ______ to ______.
hours to days
T/F: Respiratory compensation can fully normalize pH in metabolic disorders.
False: respiratory compensation is rapid but incomplete
T/F: Renal compensation for respiratory alkalosis increases HCO₃⁻ reabsorption.
False: it decreases HCO₃⁻ reabsorption
In respiratory compensation for metabolic acidosis, what happens to PaCO₂?
It decreases
In renal compensation for respiratory acidosis, what happens to plasma HCO₃⁻?
It increases
What are the three general mechanisms for regulating plasma bicarbonate?
Filtration, reabsorption, and addition of new HCO₃⁻
What happens to HCO₃⁻ in alkalosis?
It is excreted
("Alkalosis – excrete HCO₃⁻)
What happens to HCO₃⁻ in acidosis?
Plasma HCO₃⁻ increases (via reabsorption + new generation)
What determines whether new HCO₃⁻ is added to plasma?
The amount of H⁺ secretion relative to filtered HCO₃⁻
If H⁺ secretion equals filtered HCO₃⁻, ______ HCO₃⁻ is excreted.
No
If H⁺ secretion is less than filtered HCO₃⁻, ______ is excreted.
HCO₃⁻
If H⁺ secretion exceeds filtered HCO₃⁻, ______ HCO₃⁻ is added to plasma.
new
T/F: New bicarbonate is added to plasma when H⁺ secretion is less than filtered HCO₃⁻.
False: new HCO₃⁻ is added only when H⁺ secretion exceeds filtered HCO₃⁻.
T/F: All filtered HCO₃⁻ is reabsorbed in alkalosis.
False: in alkalosis, HCO₃⁻ is excreted.
What happens to plasma HCO₃⁻ when H⁺ secretion is high?
Plasma HCO₃⁻ increases
What happens when H⁺ secretion is less than filtered HCO₃⁻?
HCO₃⁻ appears in urine
How much HCO₃⁻ is filtered per day?
4320 mmol/day
How much H⁺ is secreted per day?
4400 mmol/day
How much HCO₃⁻ is normally excreted?
1 mmol/day
How much new HCO₃⁻ is generated daily?
80 mmol/day
The kidneys eliminate ______ mmol/day of non‑volatile acids.
~80
Approximately ______% of filtered HCO₃⁻ is reabsorbed.
99%
(4319/4320) x 100%
Most HCO₃⁻ reabsorption occurs in the ______.
Proximal tubule (85%)
T/F: The kidneys excrete large amounts of bicarbonate under normal conditions.
False: only 1 mmol/day is excreted
T/F: New bicarbonate production equals the amount of non‑volatile acid excreted.
True: it’s around 80 mmol/day
What percentage of HCO₃⁻ reabsorption occurs in the proximal tubule?
85%
Which segment accounts for ~10% of HCO₃⁻ reabsorption?
Thick ascending limb (TAL)
Which segment contributes ~5% of HCO₃⁻ reabsorption?
Distal nephron/collecting duct
Which transporter secretes H⁺ into the tubular lumen in the PCT?
Na⁺–H⁺ exchanger (NHE) and H⁺‑ATPase
Which enzyme converts H₂CO₃ into CO₂ and H₂O in the lumen?
Carbonic anhydrase
What happens to CO₂ after it is formed in the lumen?
It diffuses into tubular cells
What percentage of HCO₃⁻ reabsorption occurs via this mechanism?
90-95%
For every H⁺ secreted, one ______ is reabsorbed.
HCO3-
Carbonic anhydrase catalyzes the reaction: H₂CO₃ → ______ + ______.
CO₂ + H₂O
T/F: The descending limb of the loop of Henle reabsorbs bicarbonate.
False: it does not reabsorb HCO₃⁻
T/F: HCO₃⁻ is directly transported across the apical membrane.
False: CO₂ diffuses in, then HCO₃⁻ is formed intracellularly
Which membrane contains carbonic anhydrase?
Luminal (brush border) and intracellular surfaces
Which ions are cotransported with HCO₃⁻ across the basolateral membrane?
Na+
What happens to secreted H⁺ in the lumen?
It combines with filtered HCO₃⁻ to form H₂CO₃
What is the primary function of Type A intercalated cells?
Secrete H⁺ and reabsorb HCO₃⁻ (correct acidosis)
What is the primary function of Type B intercalated cells?
Secrete HCO₃⁻ and reabsorb H⁺ (correct alkalosis)
Which transporter secretes H⁺ in Type A cells?
H⁺‑ATPase
Which exchanger secretes HCO₃⁻ in Type B cells?
Pendrin (Cl⁻/HCO₃⁻ exchanger)
Type A cells correct ______.
acidosis
A for acid
Type B cells correct ______.
alkalosis
B for base
Which nephron segments reabsorb bicarbonate?
All segments EXCEPT the descending limb of the loop of Henle