AP BIO UNIT 1 REVIEW
1/196
Earn XP
Description and Tags
(QUIZLET IMPORTED)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
197 Terms
active site
a region on an enzyme that binds to a substrate during a reaction
allosteric site
The place on an enzyme where a molecule that is not a substrate may bind, thus changing the shape of the enzyme and influencing its ability to be active
amino acid
monomer of protein
amphipathic
having both a hydrophilic region and a hydrophobic region
carbohydrate
compound made up of carbon, hydrogen, and oxygen atoms; major source of energy for living organisms
carbon
element that makes up living things; forms many bonds
catalyst
substance that speeds up the rate of a chemical reaction
denaturation
A process in which a protein unravels, losing its specific structure and hence function; can be caused by changes in pH or salt concentration or by high temperature. Also refers to the separation of the two strands of the DNA double helix, caused by similar factors.
dissacharide
A molecule made of two monosaccharides.
hydrogen bond
Attraction between a slightly positive hydrogen atom and a slightly negative atom.
hydrophilic
Attracted to water
hydrophobic
Having an aversion to water; tending to coalesce and form droplets in water.
ion
A charged atom
lipid
macromolecule made mostly from carbon and hydrogen atoms; includes fats, oils, and waxes
macromolecule
A very large organic molecule composed of many smaller molecules
monomer
A simple compound whose molecules can join together to form polymers
monosaccharide
A single sugar molecule such as glucose or fructose, the simplest type of sugar.
nonpolar molecule
molecule that shares electrons equally and does not have oppositely charged ends
nucleic acid
macromolecule containing hydrogen, oxygen, nitrogen, carbon, and phosphorus
nucleotide
monomer of nucleic acids made up of a 5-carbon sugar, a phosphate group, and a nitrogenous base
organic molecule
A molecule containing carbon that is a part of or produced by living systems.
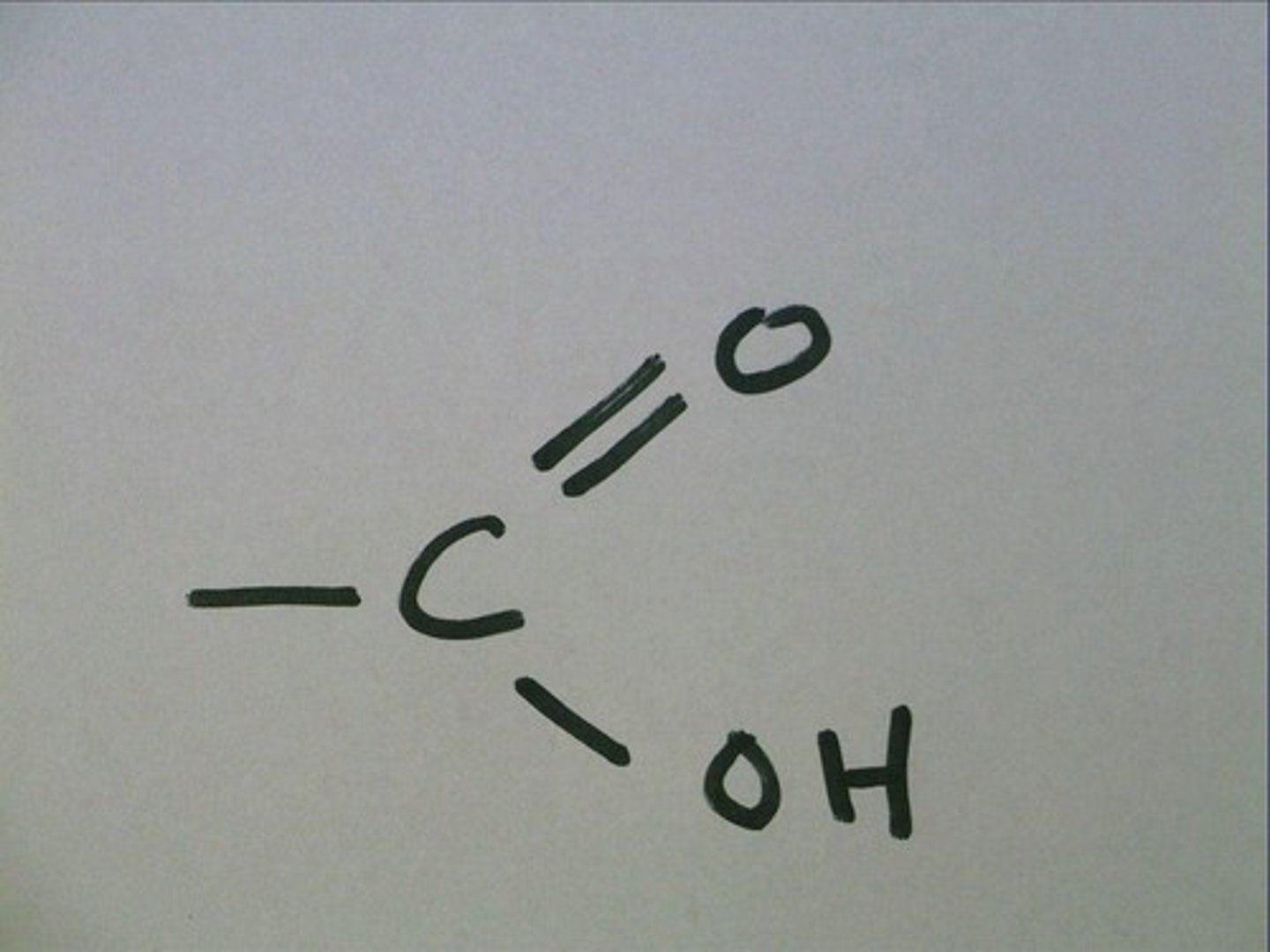
peptide bond
The chemical bond that forms between the carboxyl group of one amino acid and the amino group of another amino acid
polar molecule
molecule with an unequal distribution of charge, resulting in the molecule having a positive end and a negative end
polymer
A long molecule consisting of many similar or identical monomers linked together.
protein
A three dimensional polymer made of monomers of amino acids.
substrate
A specific reactant acted upon by an enzyme
cohesion
Attraction between molecules of the same substance
adhesion
An attraction between molecules of different substances
surface tension
the cohesive force at the surface of a body of liquid that prevents the molecules from separating
specific heat
The amount of energy required to raise the temperature of 1 gram of a substance by 1 degree Celcius
hydrophobic
Having an aversion to water; tending to coalesce and form droplets in water.
hydrophilic
Attracted to water
dehydration synthesis
A chemical reaction in which two molecules are bonded together with the removal of a water molecule.
hydrolysis
A chemical process that splits a molecule by adding water.
polysaccharide
Carbohydrates that are made up of more than two monosaccharides
monosaccharide
A single sugar molecule such as glucose or fructose, the simplest type of sugar.
disaccharide
A double sugar, consisting of two monosaccharides joined by dehydration synthesis.
elements in carbohydrates
CHO in a 1:2:1 ratio
elements in lipids
CHO
elements in proteins
CHON
elements in nucleic acids
CHONP
saturated fatty acids
have the maximum number of hydrogen atoms possible and no double bonds
unsaturated fatty acids
A fatty acid possessing one or more double bonds between the carbons in the hydrocarbon tail. Such bonding reduces the number of hydrogen atoms attached to the carbon skeleton.
R group
a functional group that defines a particular amino acid and gives it special properties.
protein primary structure
amino acid sequence
protein secondary structure
alpha helix and beta pleated sheet formed by hydrogen bonds between atoms of the polypeptide backbone
pyrimidines
Cytosine and Thymine; nitrogen bases containing one ring
purines
Adenine and Guanine; nitrogen bases containing a double-ring structure
noncompetitive inhibitor
A substance that reduces the activity of an enzyme by binding to a location remote from the active site, changing its conformation so that it no longer binds to the substrate.
competitive inhibitor
A substance that reduces the activity of an enzyme by entering the active site in place of the substrate whose structure it mimics.
Matter
Anything that takes up space and has mass.
Element
Substance that cannot be broken down to other substances through chemical reactions. Each has a symbol. 92 occurring in nature.

Compound
Substance consisting of two or more different elements combined in a fixed ratio.
*Characteristics different than those of its elements
CHON
4 elements essential to life
Trace Elements
Required by an organism only in minute quantities.
Atom
Smallest unit of matter that retains the properties of an element. Composed of subatomic particles. Mostly empty space.
Subatomic particles
Neutrons, protons, electrons. Make up atoms.
Proton
One unit of positive charge. Approximately one dalton. Located in nucleus.
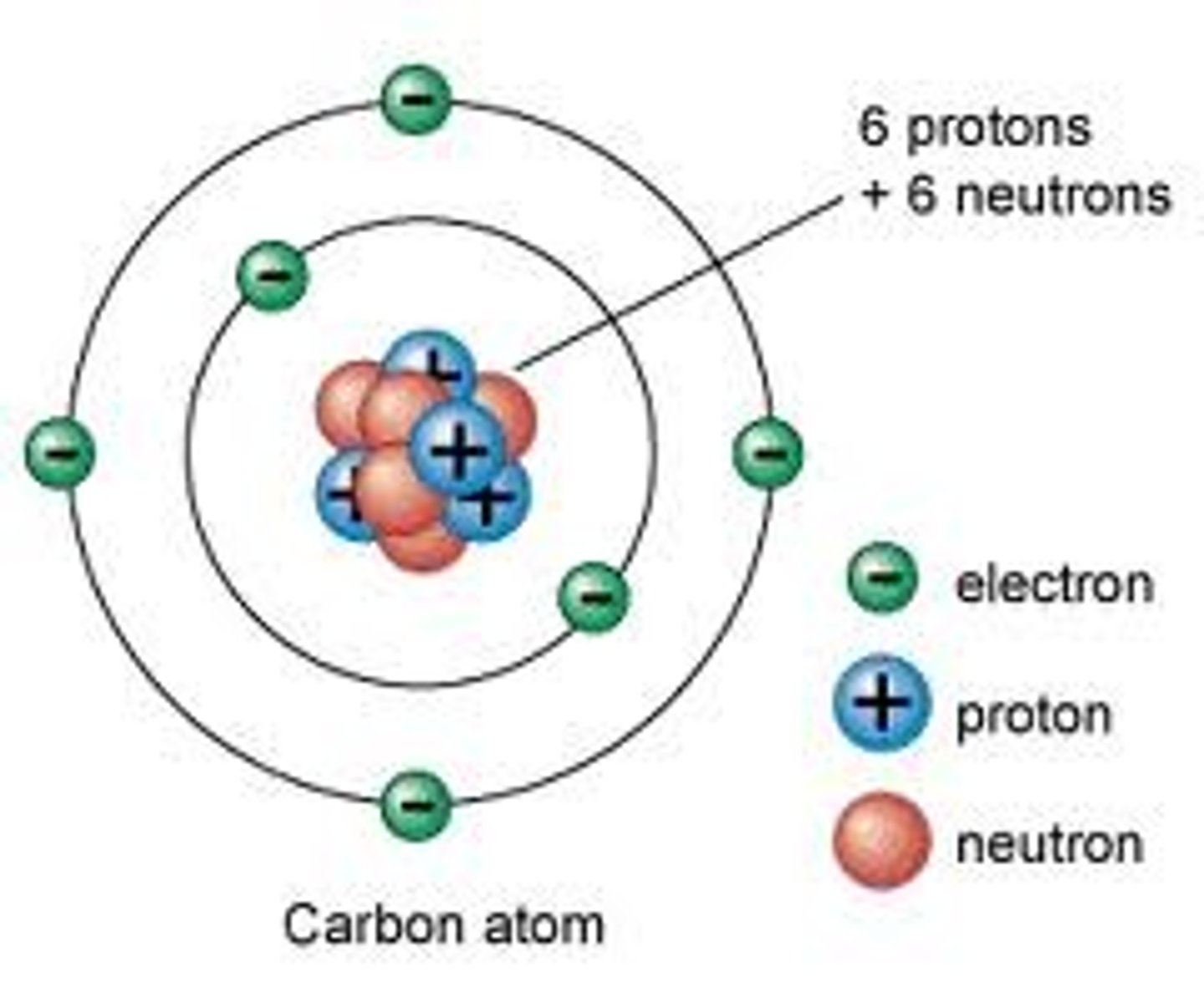
Electron
One unit of negative charge. Located outside of nucleus.
Atomic Nucleus
Dense core packed with protons and neutrons.
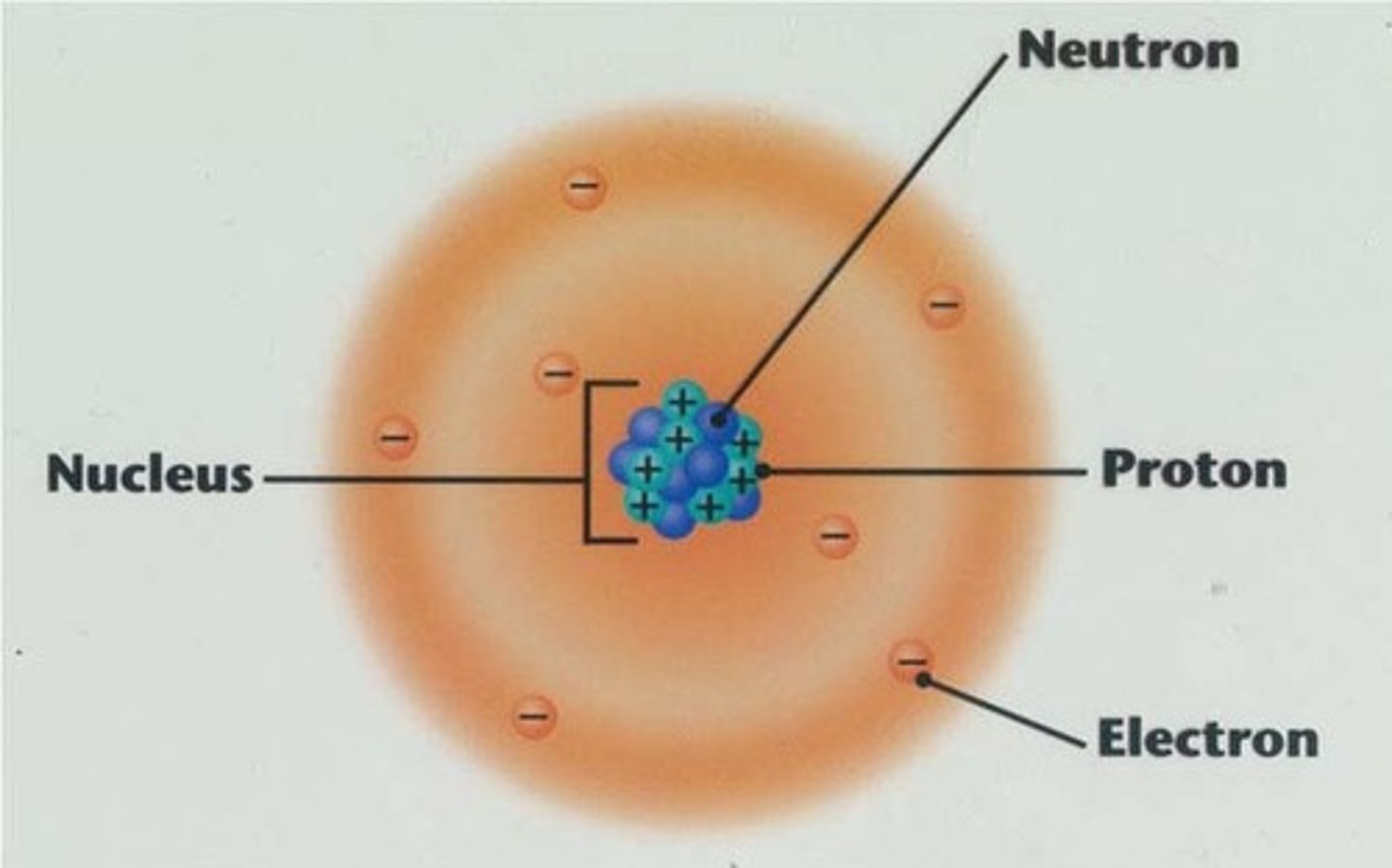
Neutrons
Electrically neutral. Approximately one dalton.

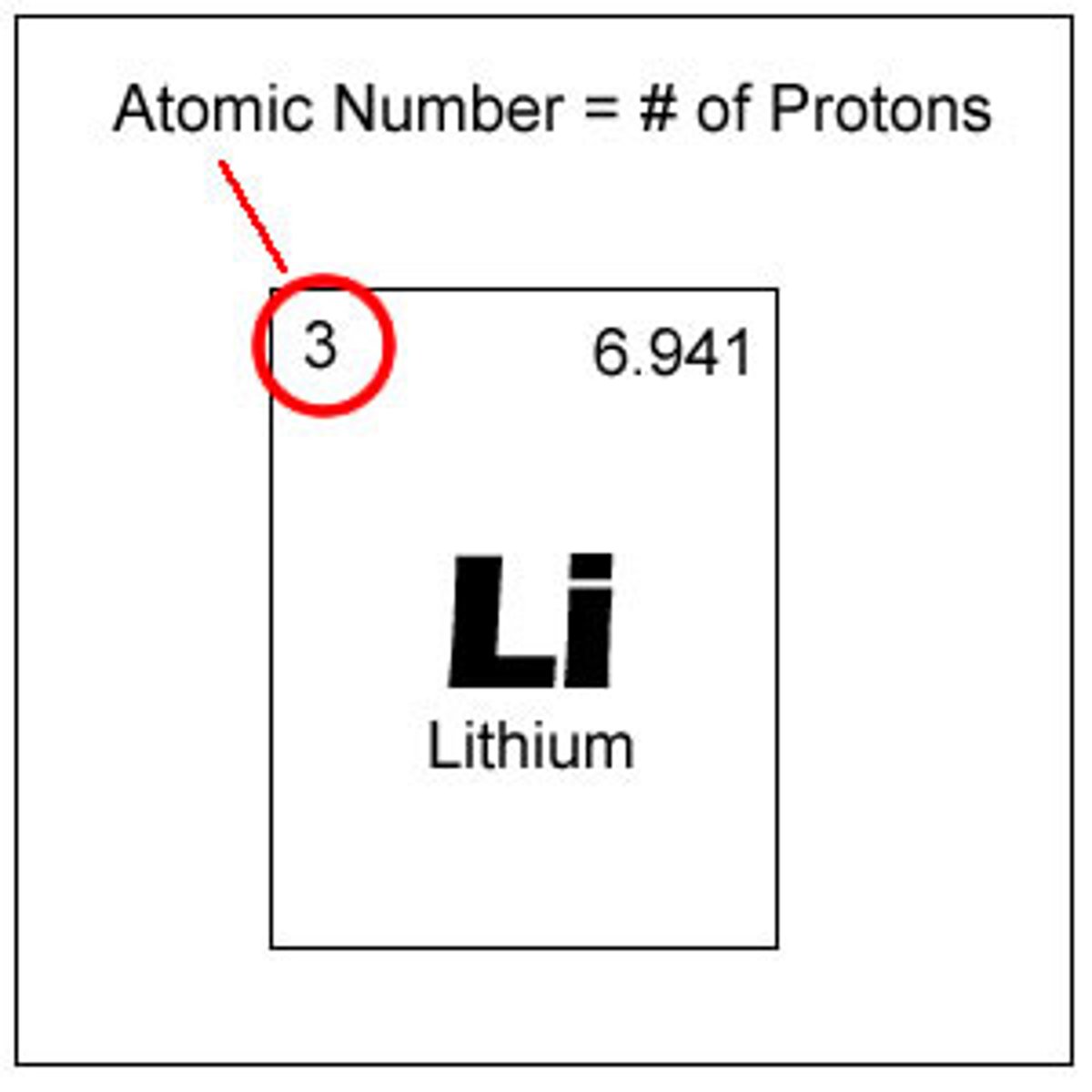
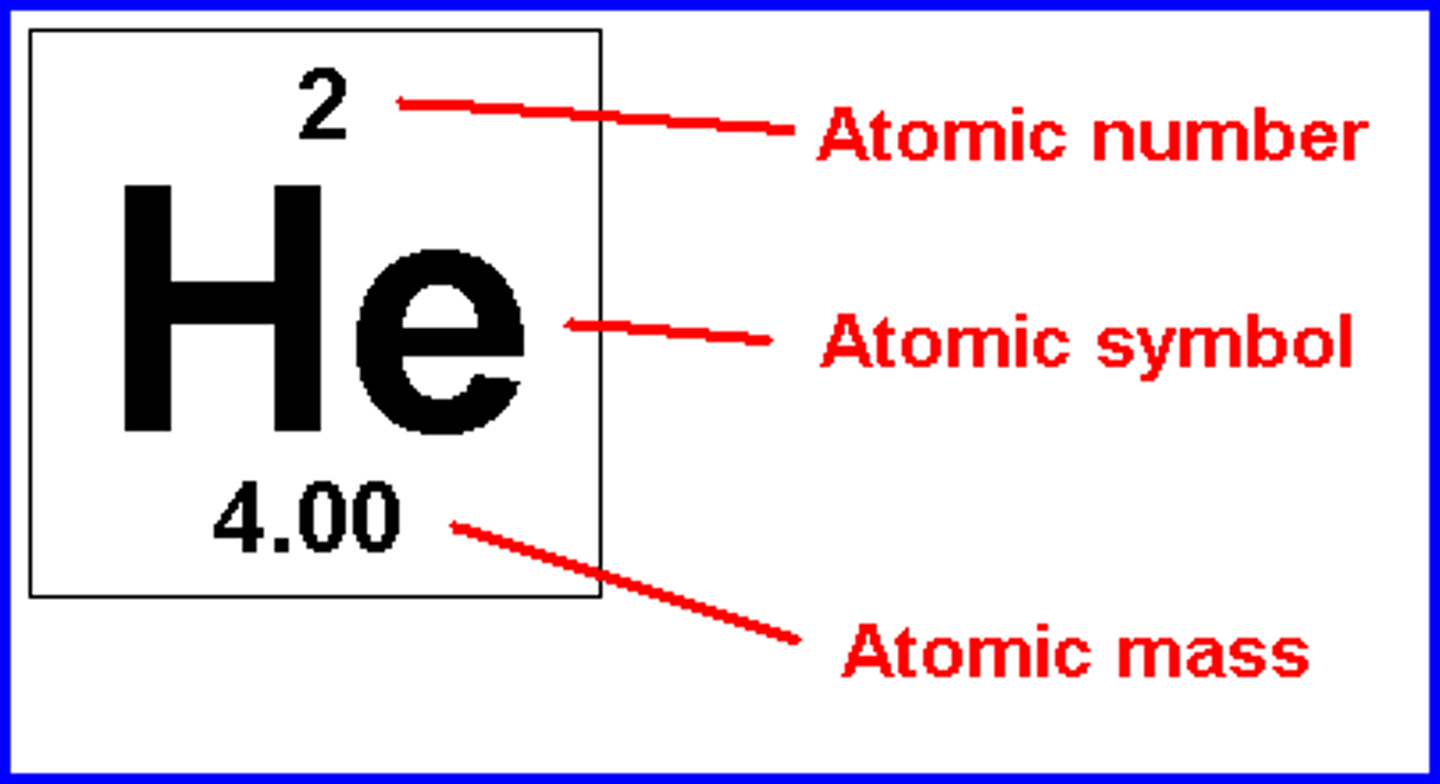
Atomic Number
The number of protons (which is unique to that element) and written as a subscript to left of element symbol. Also tells us number of electrons since P=E if atom is neutral
Mass Number
Sum of the protons and the neutrons. An approximation of the atomic mass.
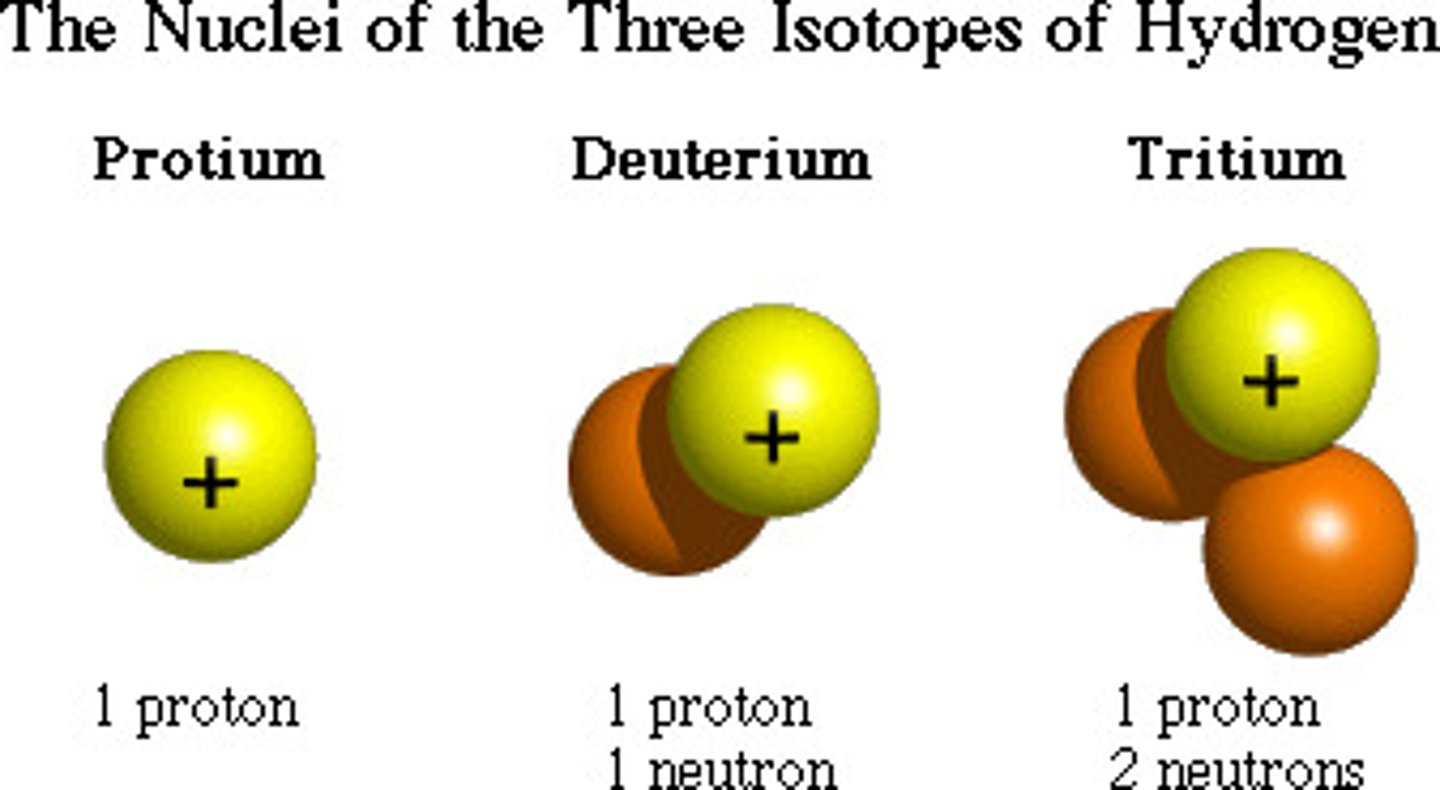
Isotopes
Same number of protons, but different number of neutrons and therefore different mass. Same element.
Radioactive Isotope
One in which the nucleus decays spontaneously, giving off particles and energy. Can decay to a different element. Have useful applications.
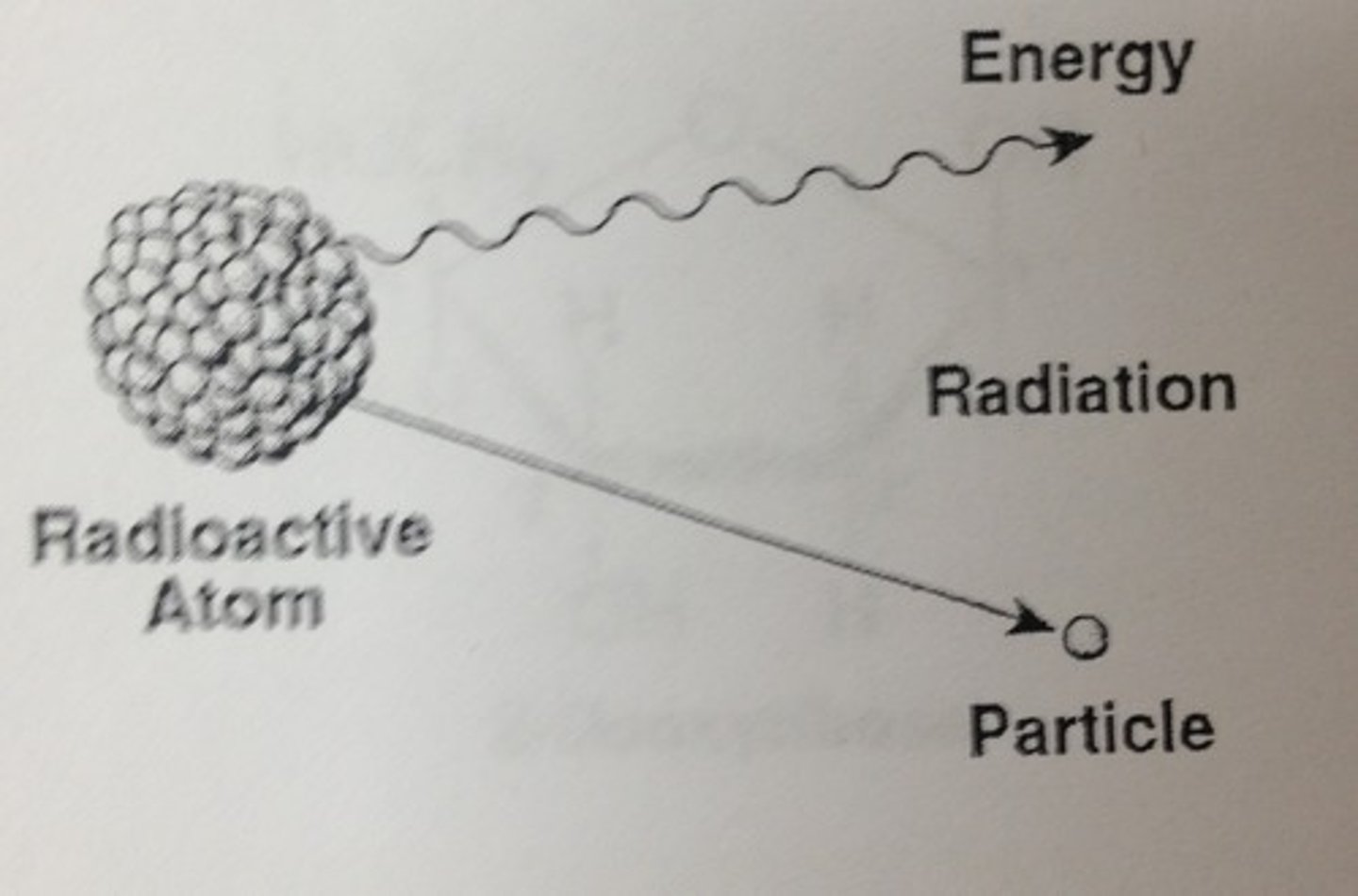
Energy
Capacity to cause change
Potential Energy
Energy matter possesses because of its location or structure. Electrons have this based on distance from nucleus.

Electron Shells
Where electrons are found, each with characteristic average distance and energy level. Electrons can change these by absorbing or losing energy.
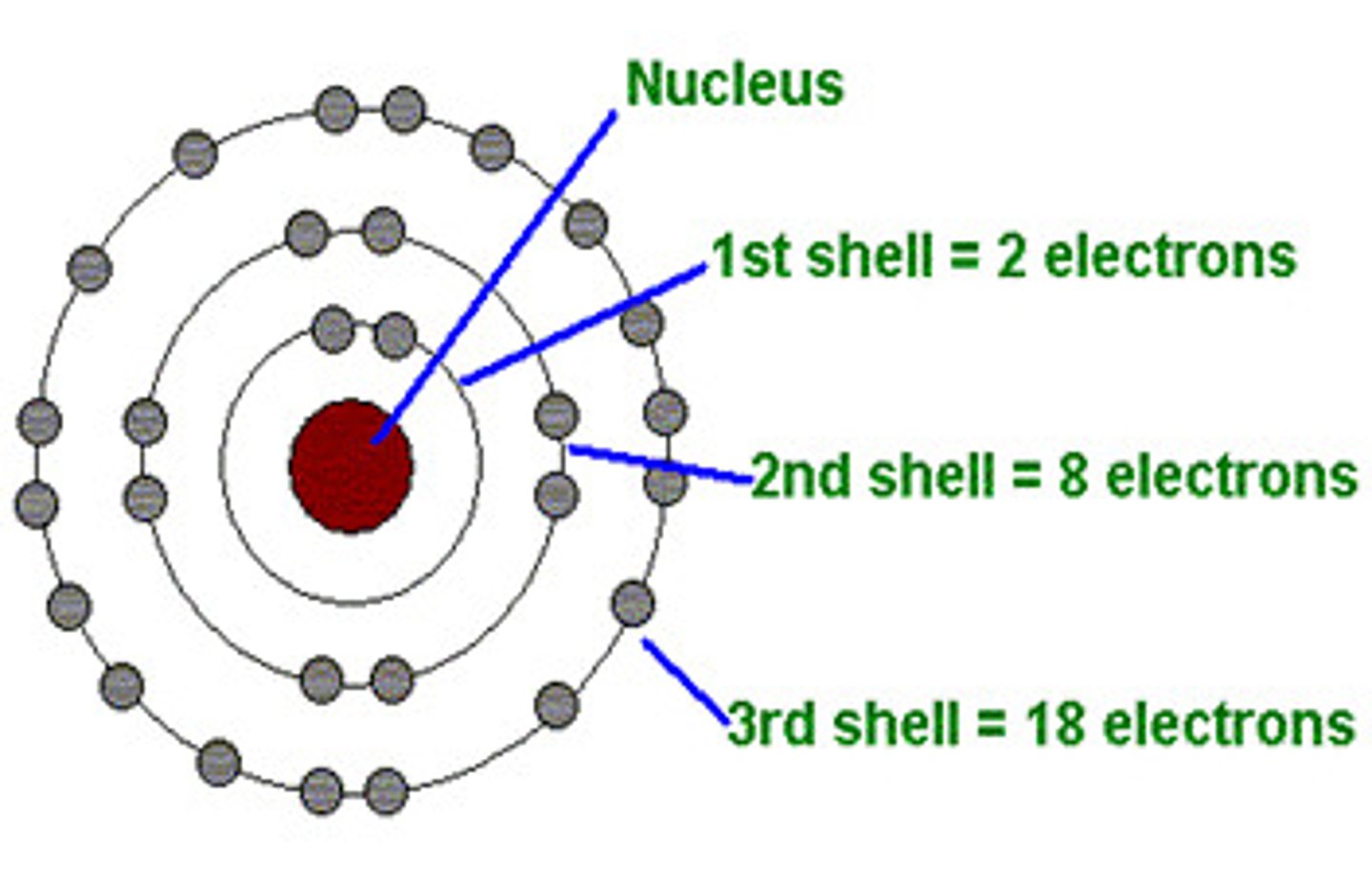
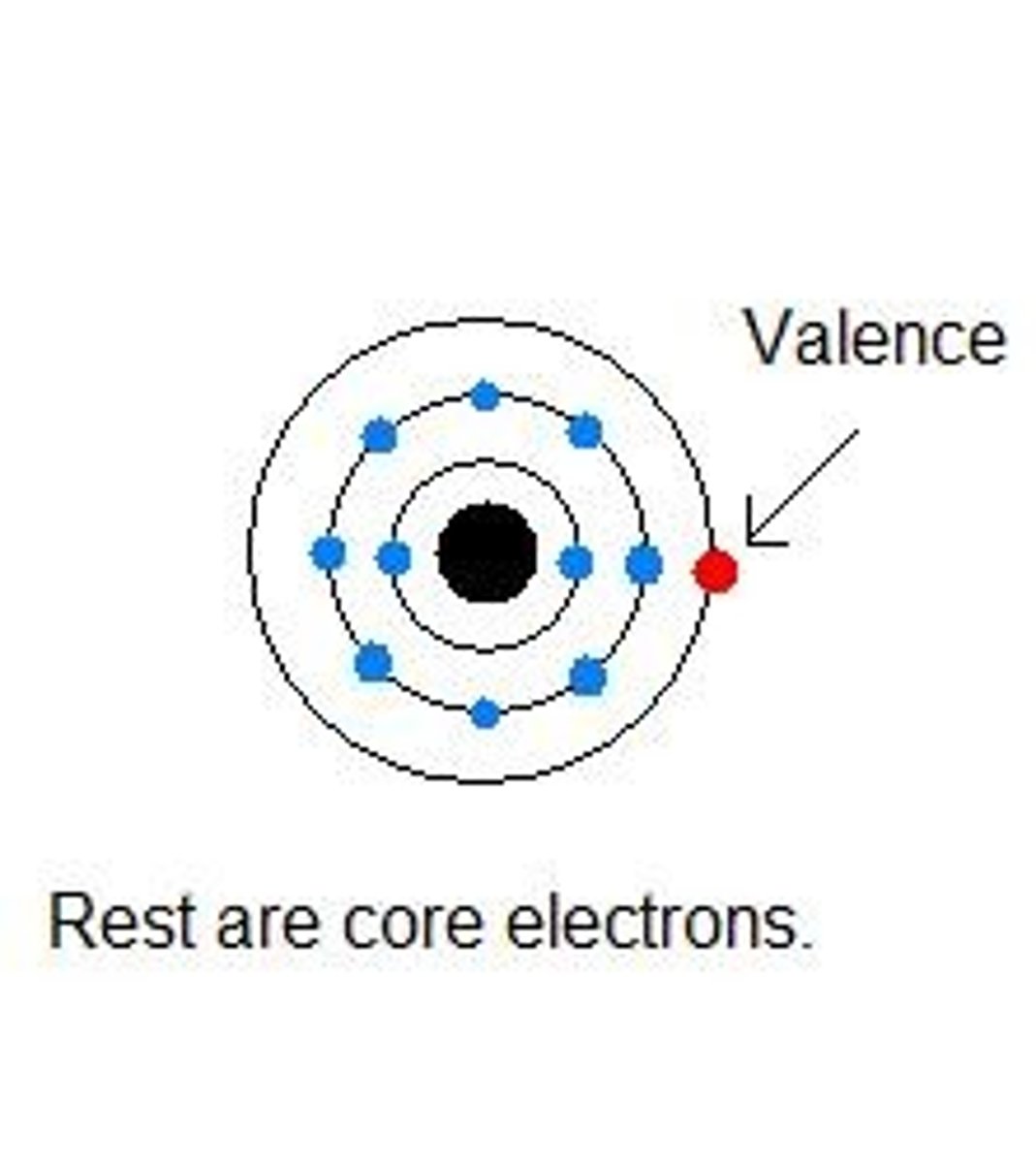
Valence Electrons
Electrons in the outermost shell.
Chemical Bonds
The attraction that holds atoms together during an interaction.
Covalent Bond
Sharing of a pair of valence electrons by two atoms.
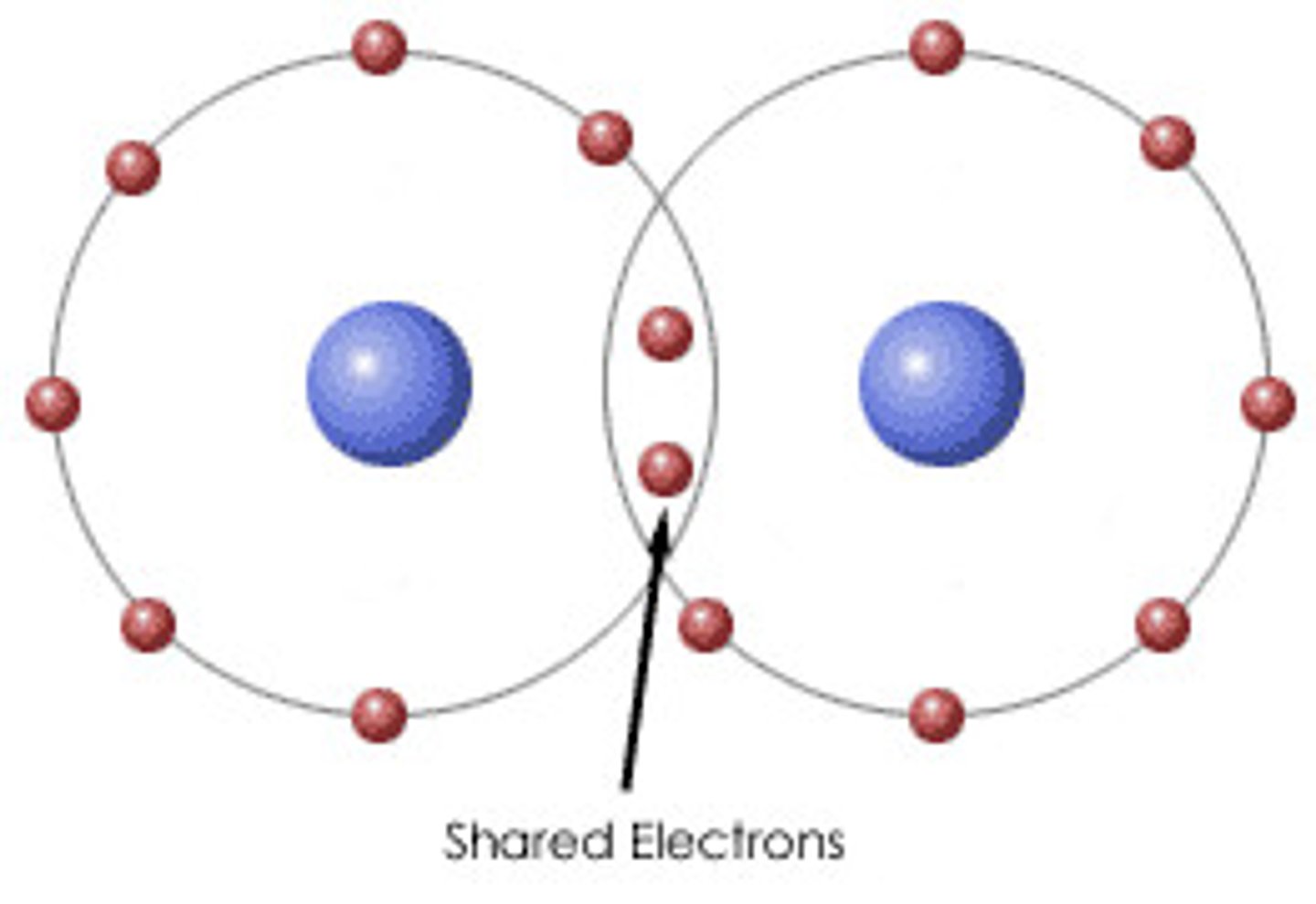
Molecule
Two or more atoms held together by covalent bonds.
Single Bond
One pair of shared electrons.
Structural Formula
H--H. Notation which represents both atoms and bonding.
Double Bond
Sharing two pairs of valence electrons.
Valence
Bonding capacity. Equal to the number of unpaired electrons required to complete the outermost (valence) shell.
Electronegativity
The attraction of a particular kind of atom for the electrons of a covalent bond.
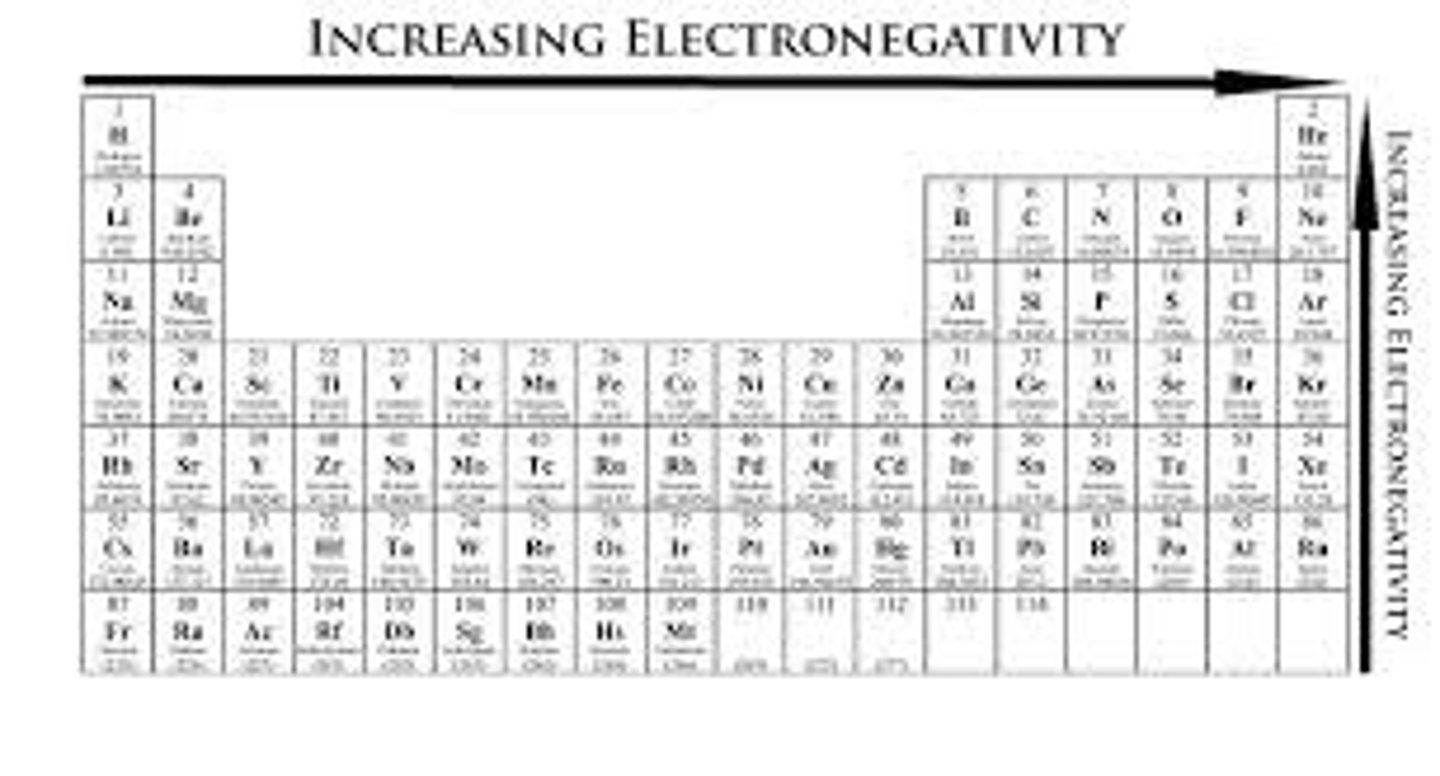
Nonpolar Covalent Bond
Covalent bond in which electrons are shared equally.
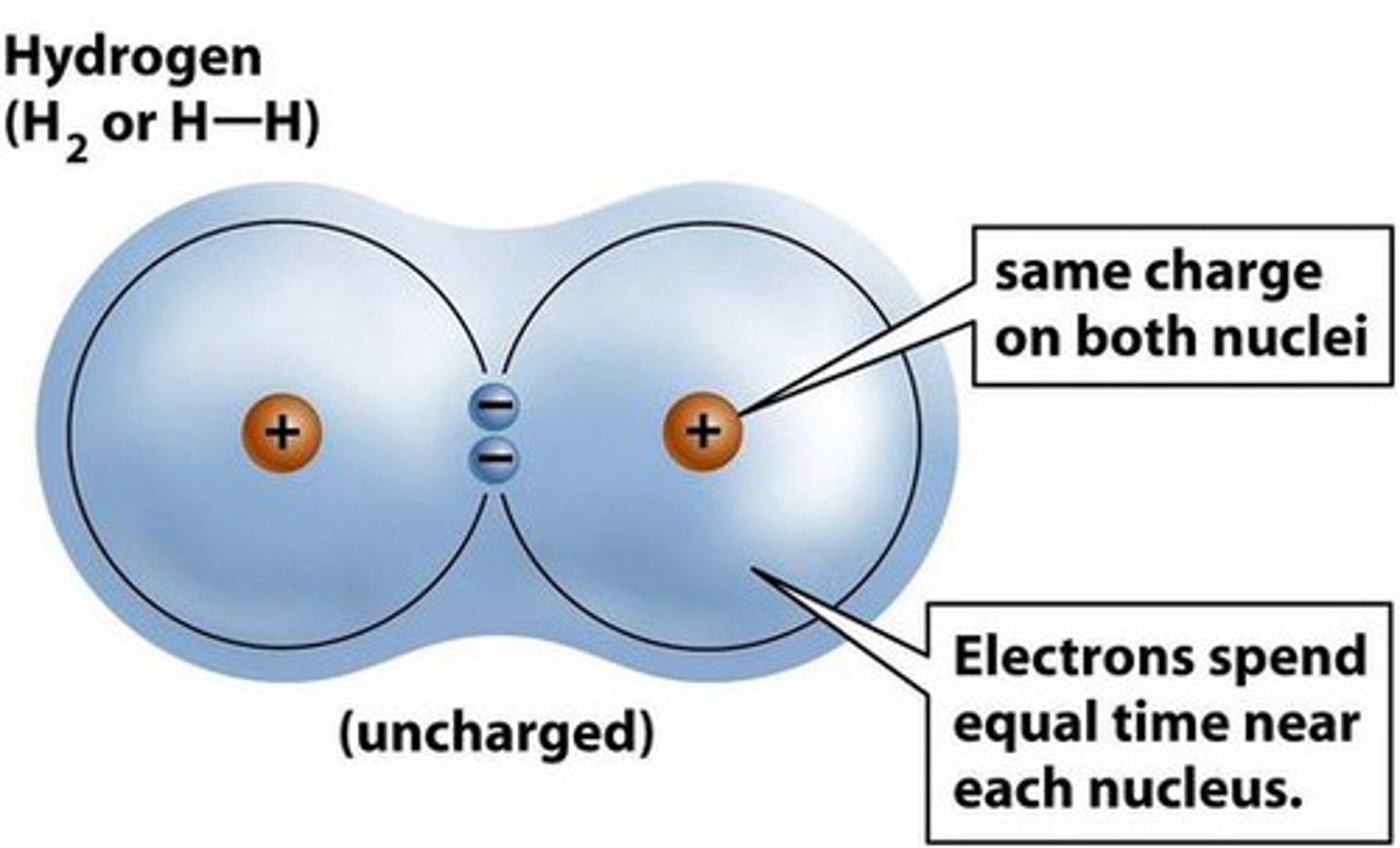
Polar Covalent Bond
Where one atom is bonded to a more electronegative atom and the electrons of the bond are not shared equally.
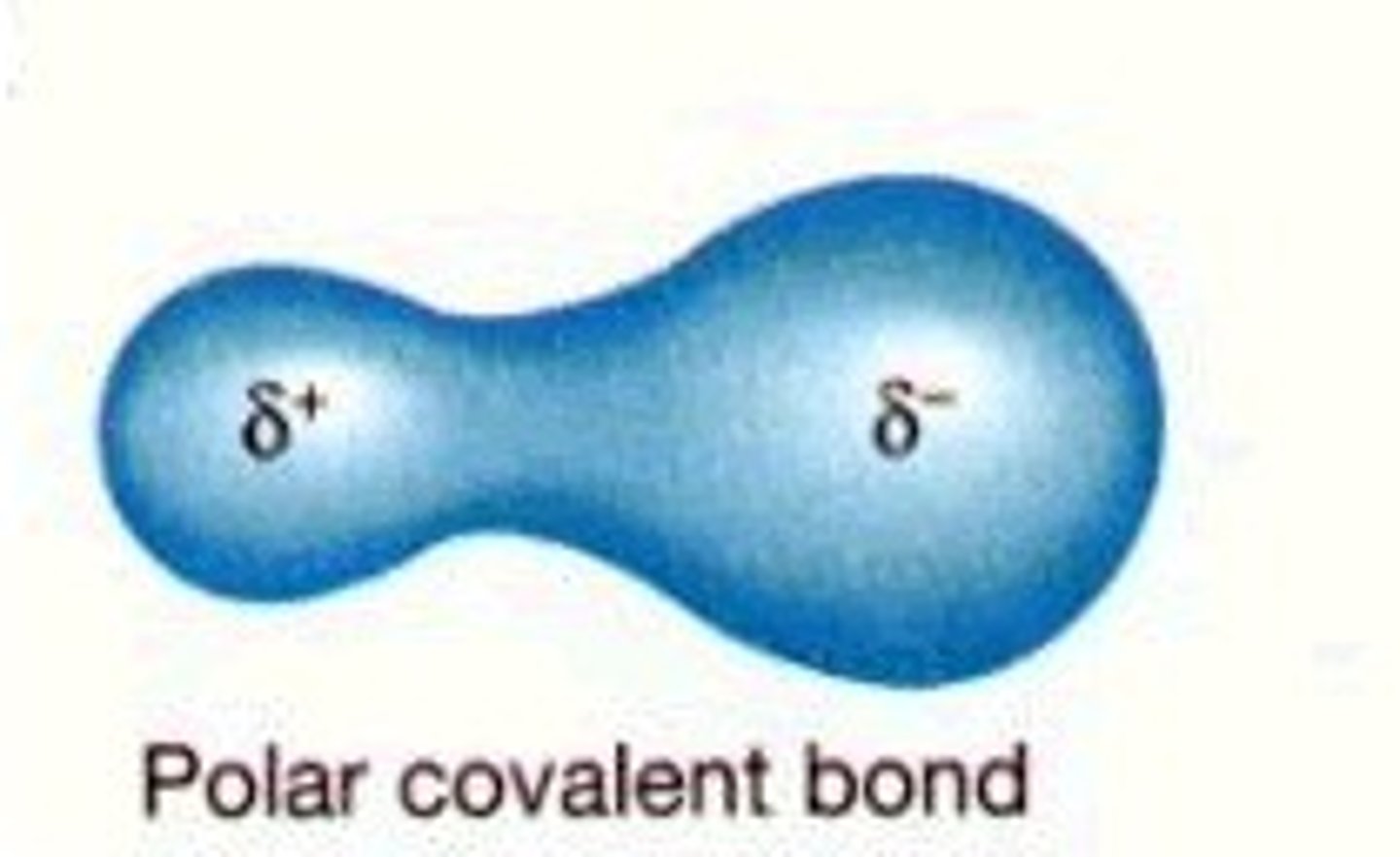
Ionic Bonds
The attraction of cations and anions. The transfer of electrons creates ions which form an ionic bond.
Ion
A charged atom.
Cation
A positively charged atom.

Anion
A negatively charged atom.
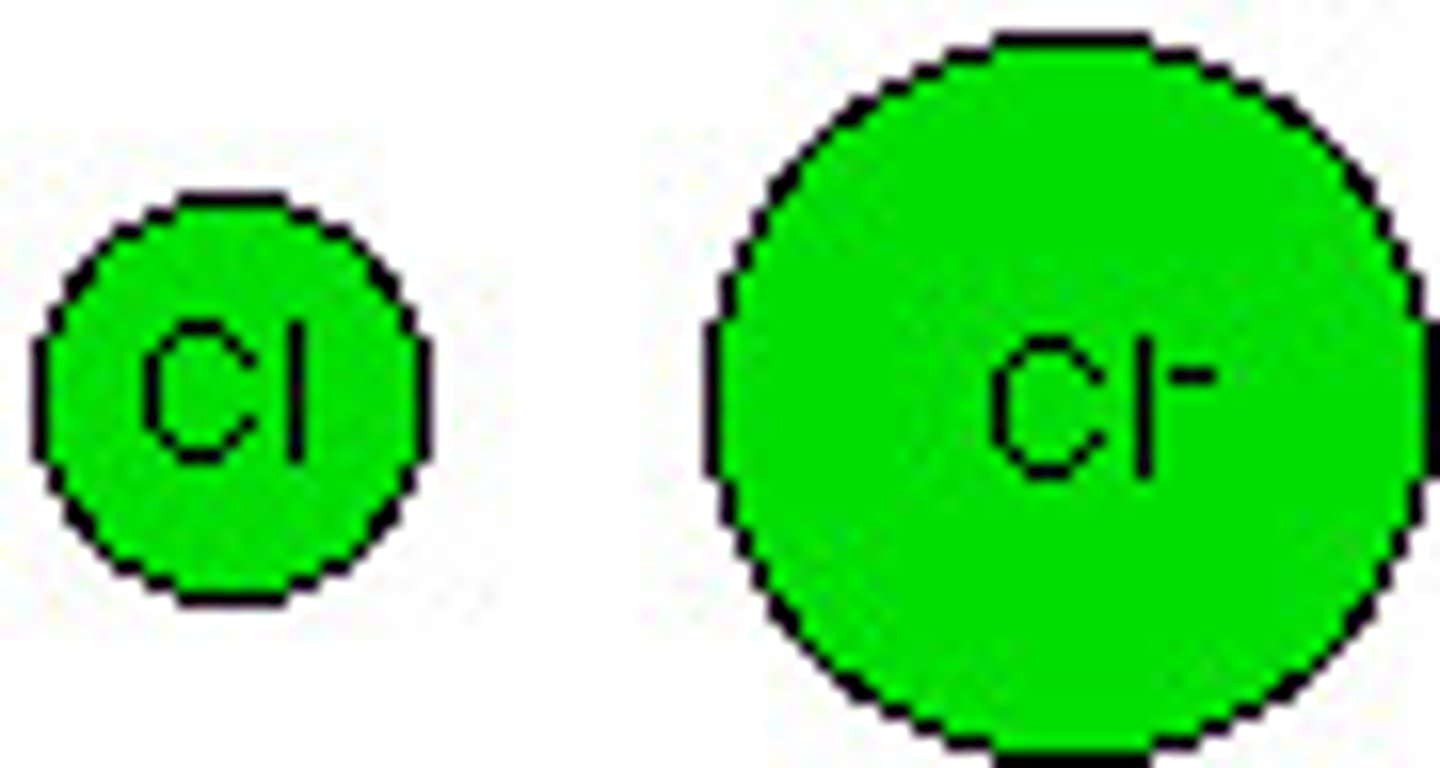
Ionic Compounds
Or Salts. Compounds formed by ionic bonds.
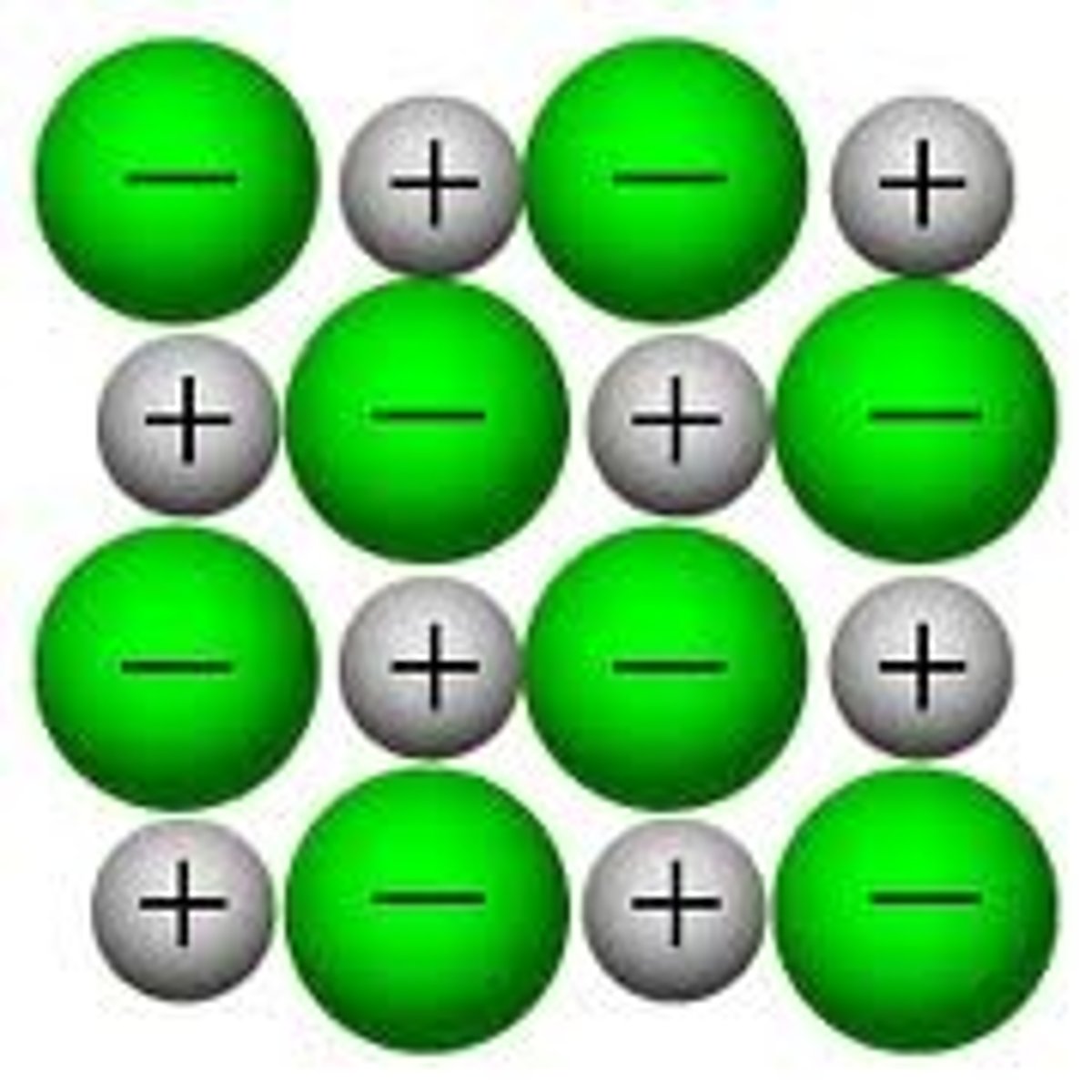
Hydrogen Bond
When a hydrogen atom covalently bonded to an electronegative atom is also attracted to another electronegative atom.
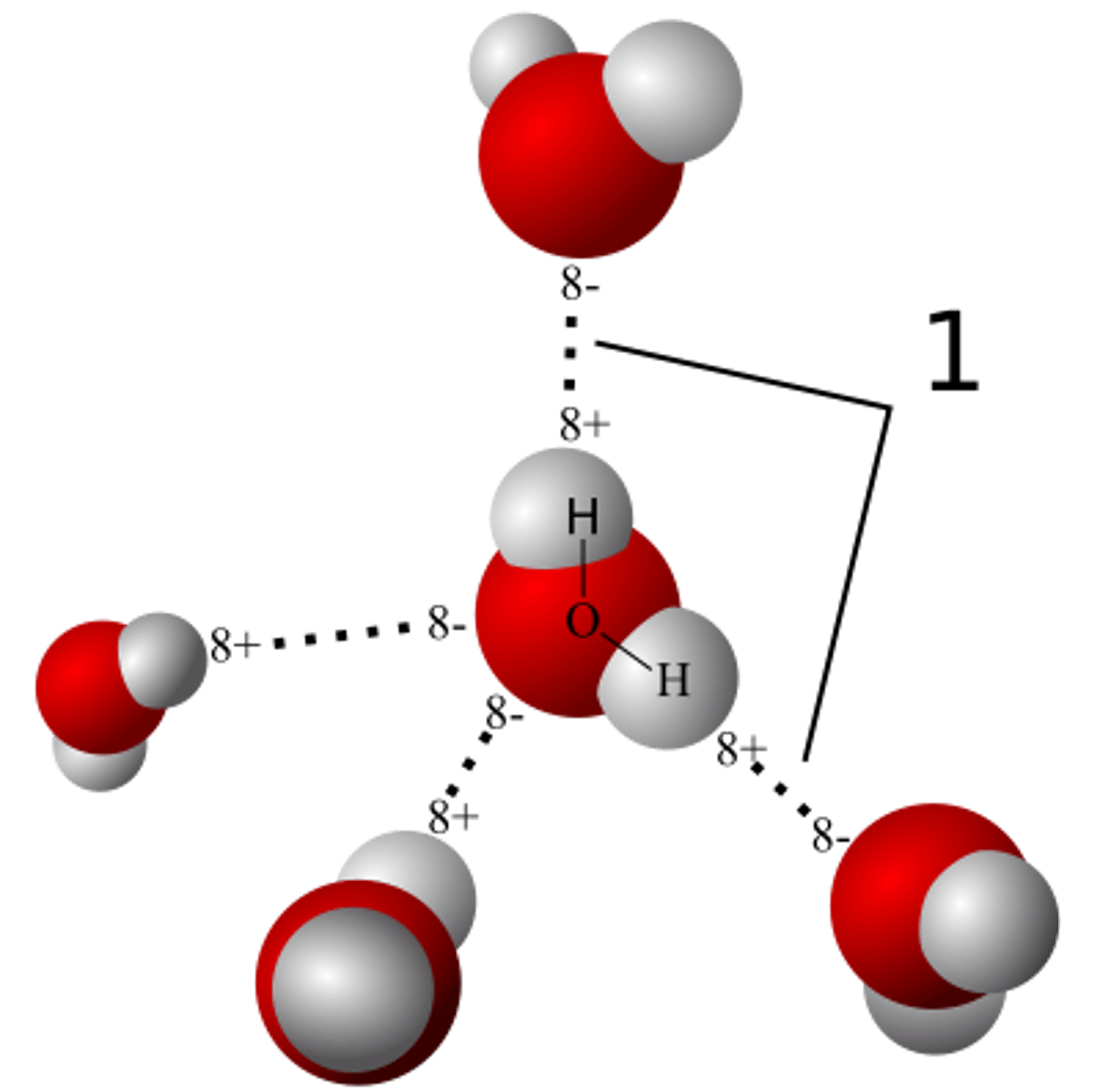
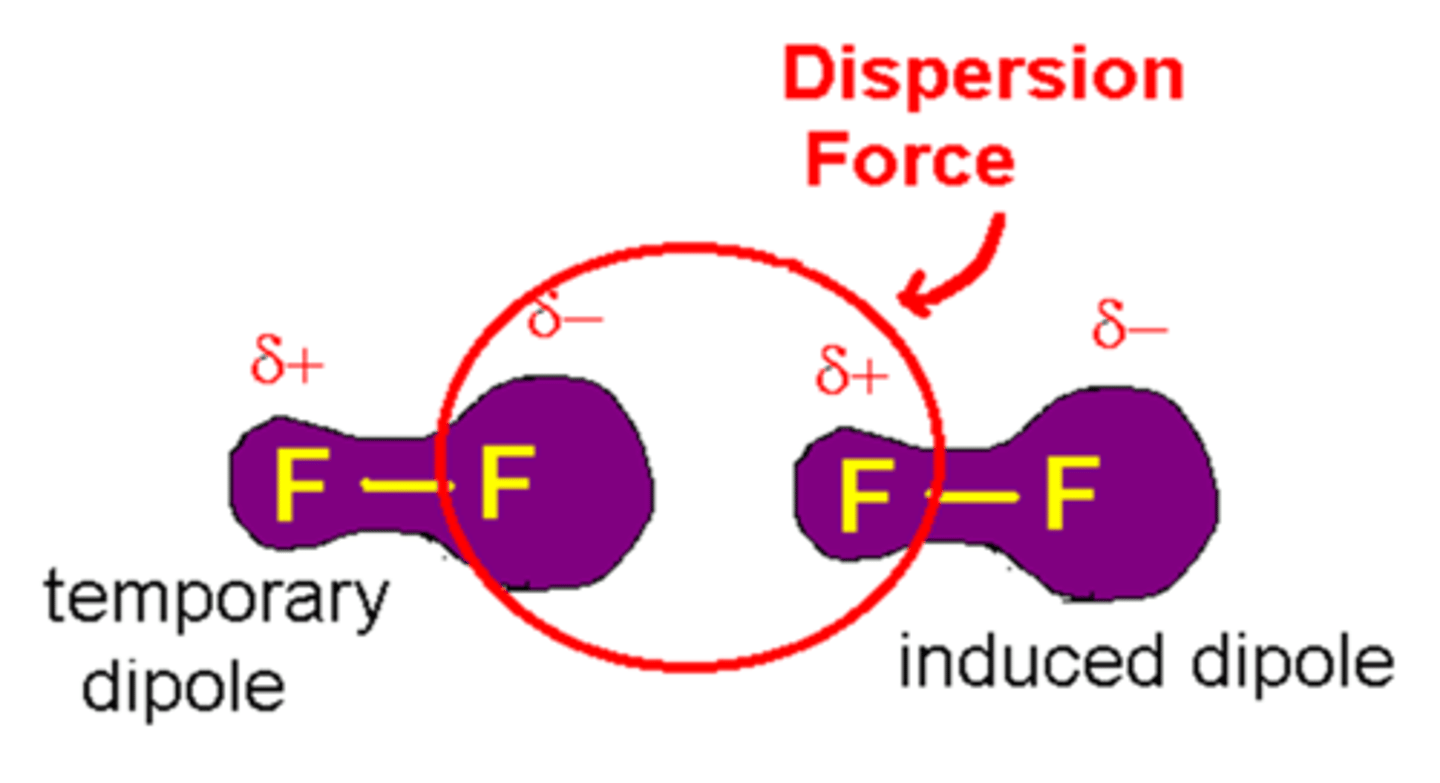
van der Waals Interactions
Ever changing regions of positive and negative charge that enable all atoms and molecules to stick to one another.
Molecular Shape
Determines how biological molecules recognize and respond to one another.
Reactants
The starting materials of a chemical reaction.
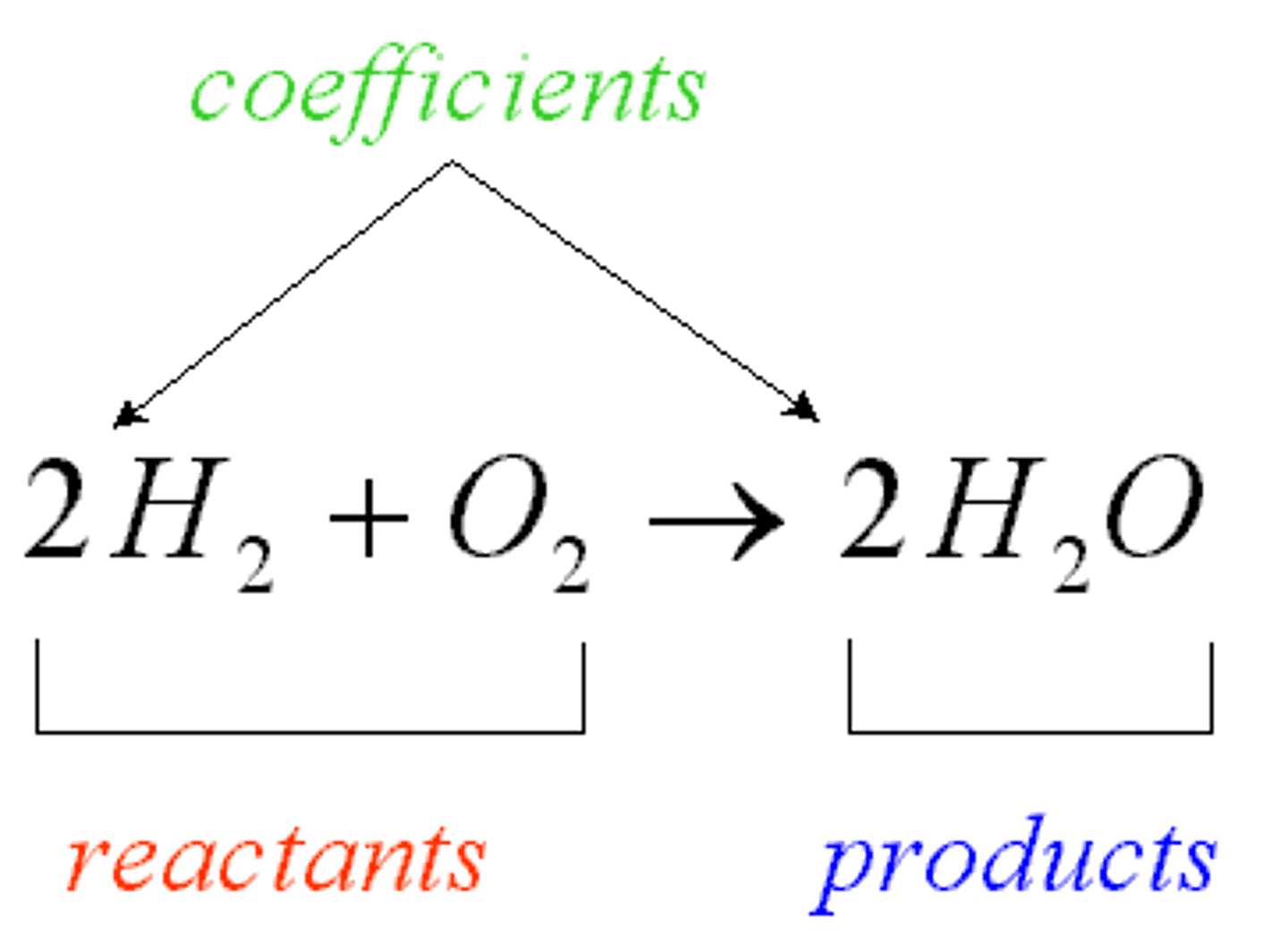
Products
The resulting material of a chemical reaction.
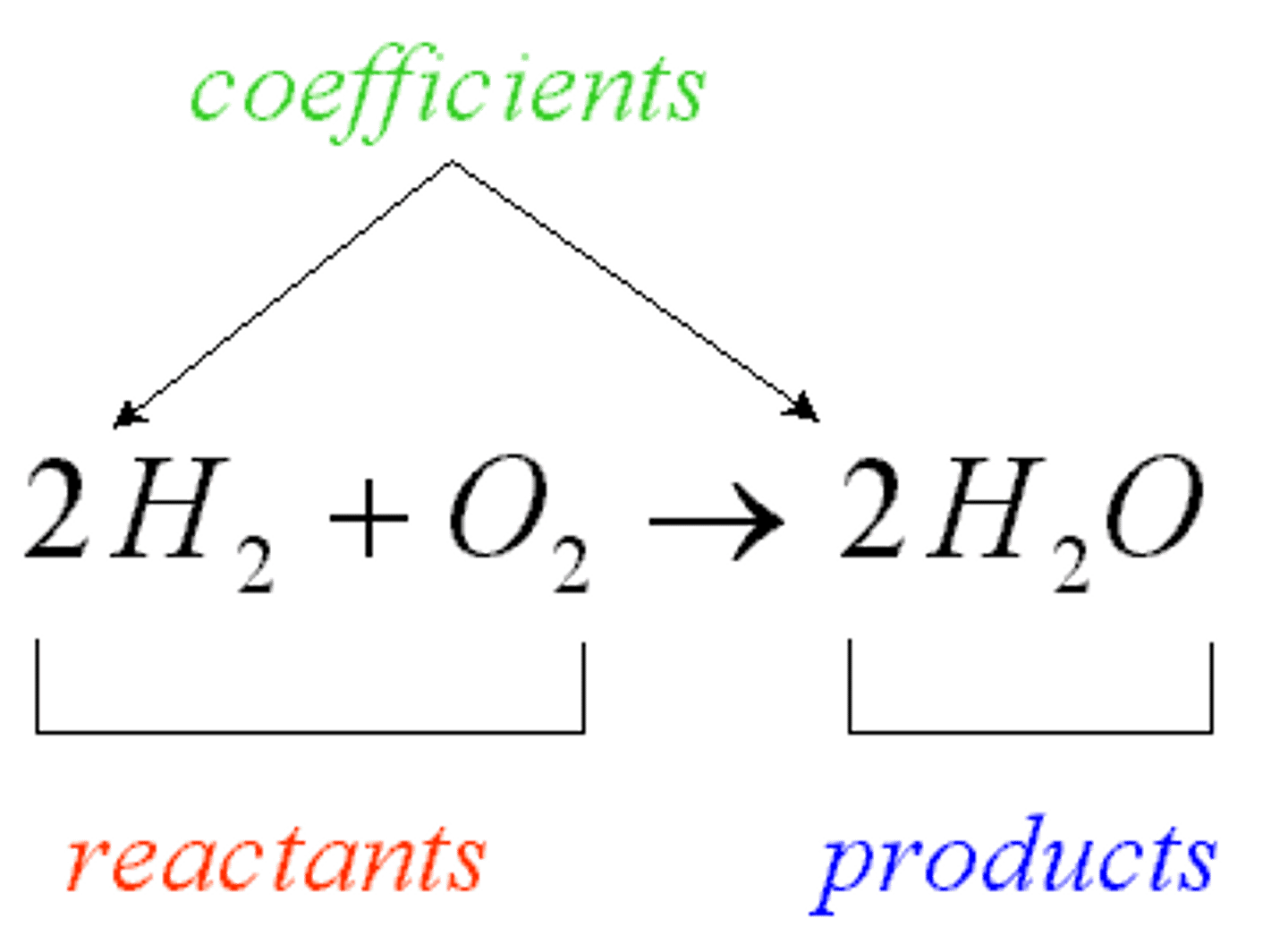
Chemical Equilibrium
The point at which the relative concentrations of the products and the reactants stop changing and offset one another exactly.

organic chemistry
The study of carbon & hydrogen compounds (organic compounds).
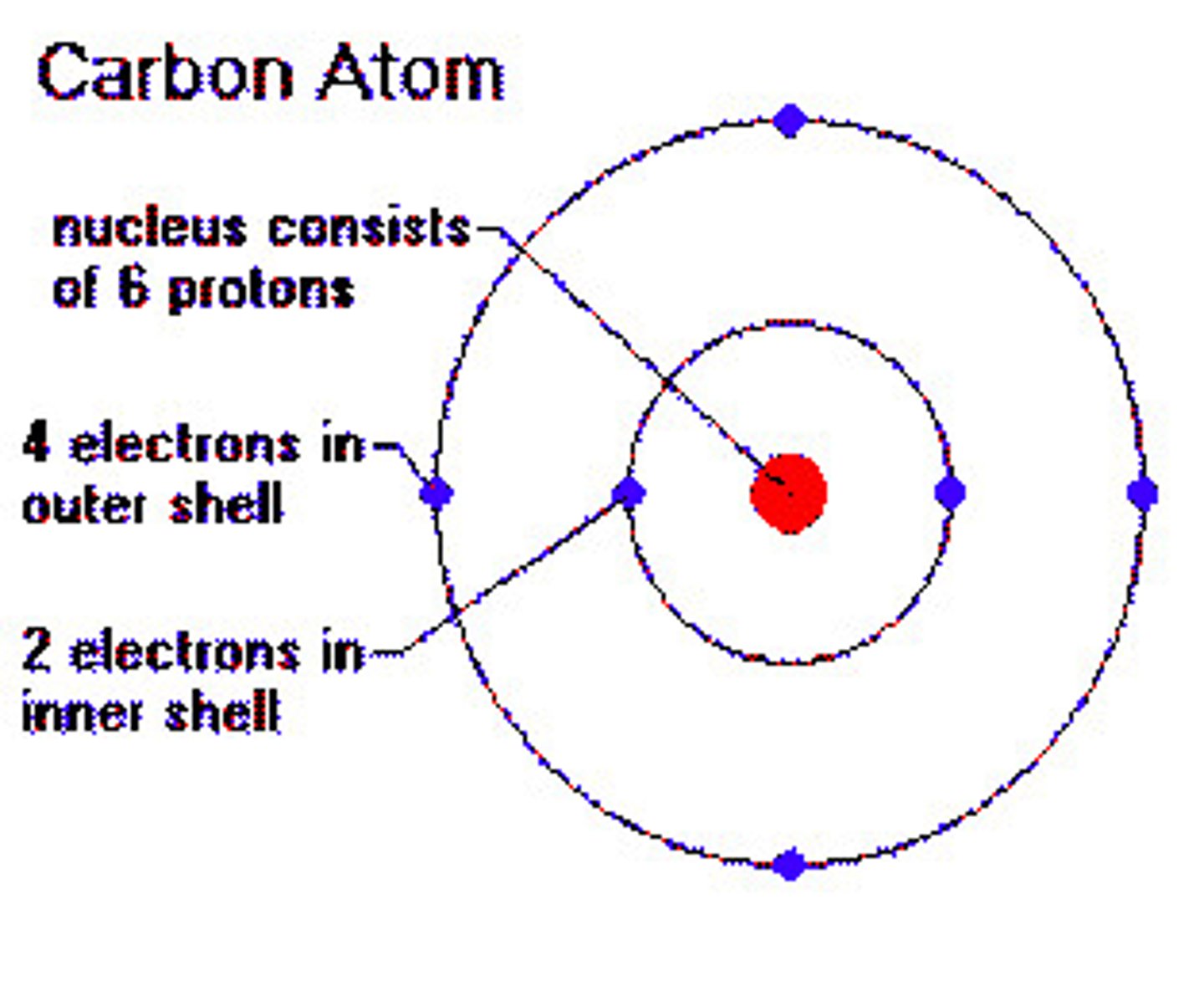
carbon
this atom has 4 electrons in valence shell and can form 4 covalent bonds with other atoms
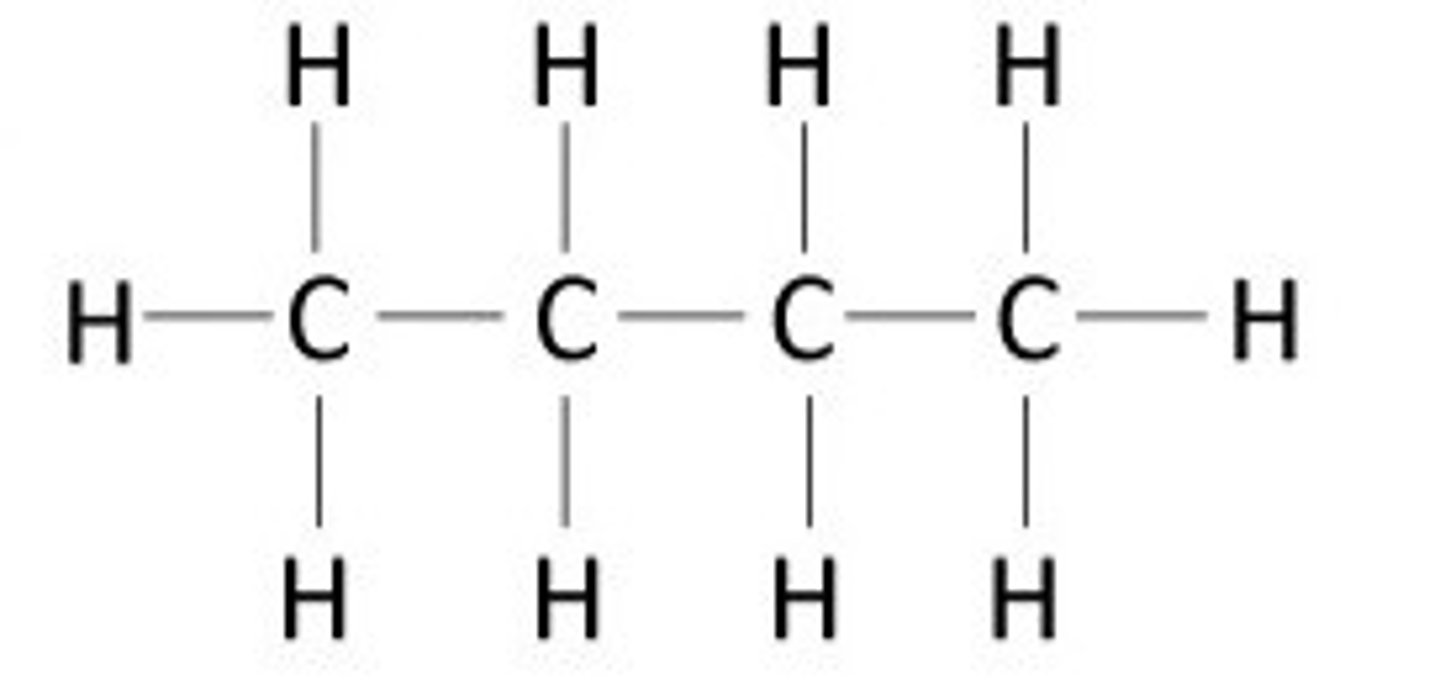
hydrocarbon
An organic molecule consisting only of carbon and hydrogen.
isomer
One of several compounds with the same molecular formula but different structures and therefore different properties. The three types are structural, geometric, and enantiomers.
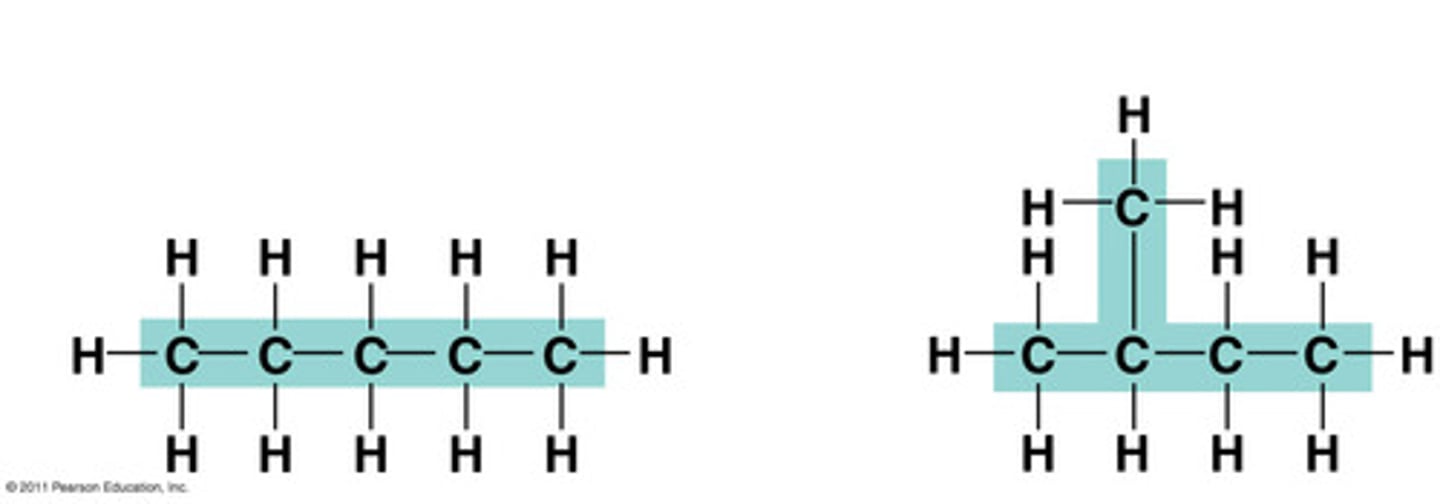
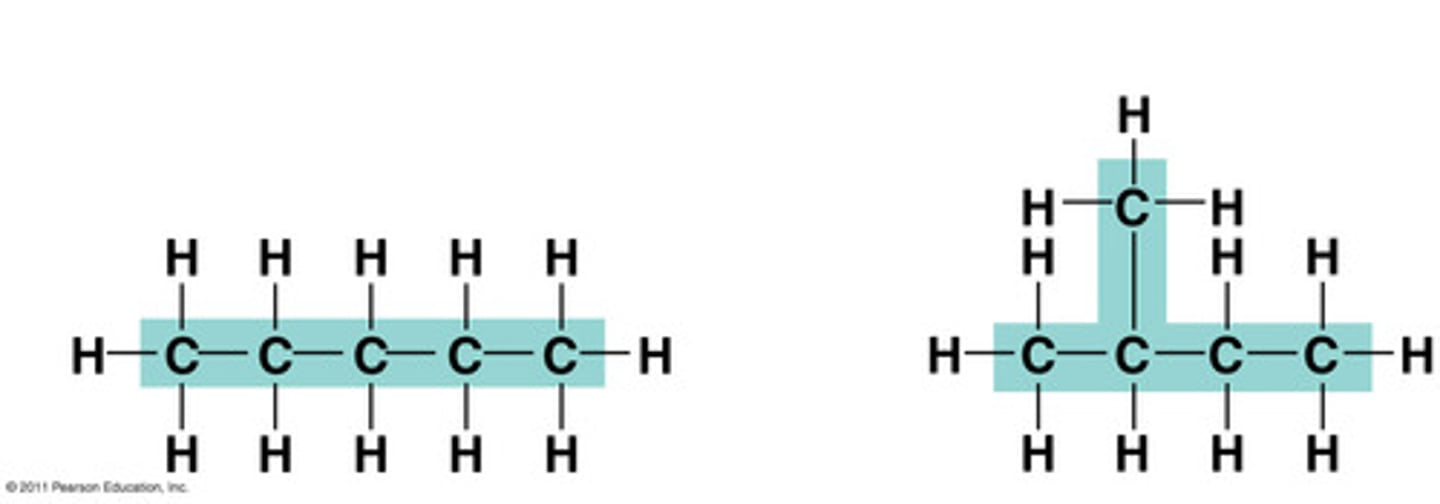
structural isomer
One or several compounds that have the same molecular formula but differ in the covalent arrangement of their atoms. (DIFFERENT SHAPE)
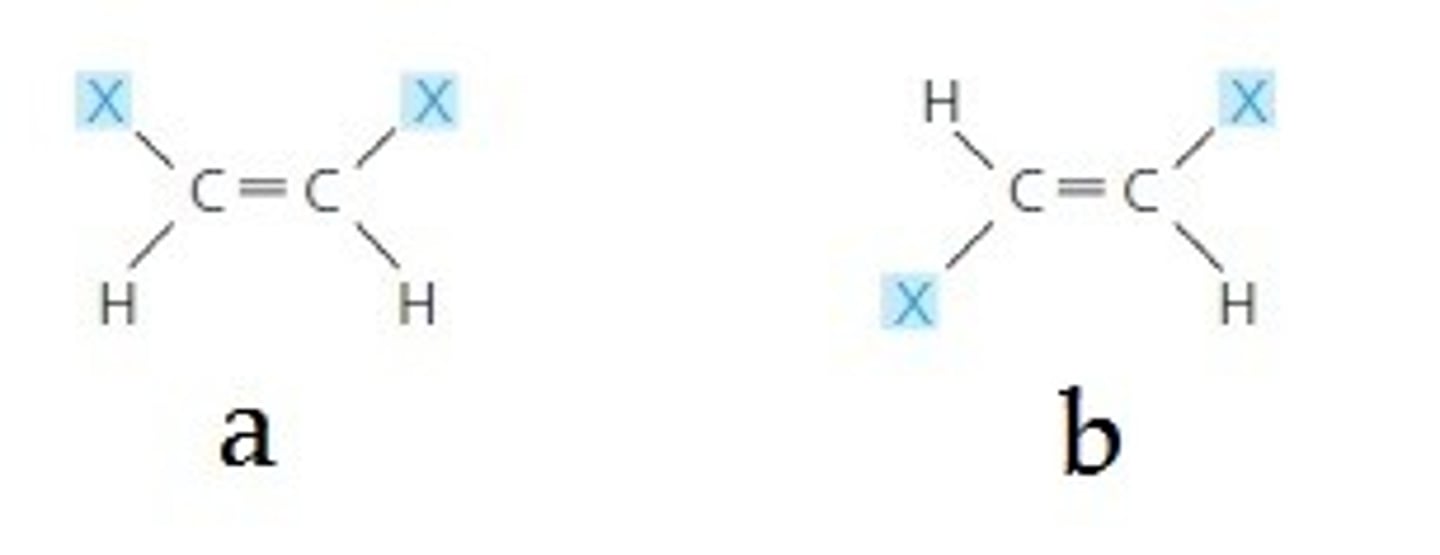
geometric isomer
One of several compounds that have the same molecular formula and covalent bonds between atoms but differ in the spatial arrangements of their atoms owing to the inflexibility of double bonds (DIFFERENT ARRANGEMENT)
enantiomer
One or two compounds that are mirror images of each other and that differ in shape due to the presence of an asymmetric carbon. (MIRROR IMAGE)

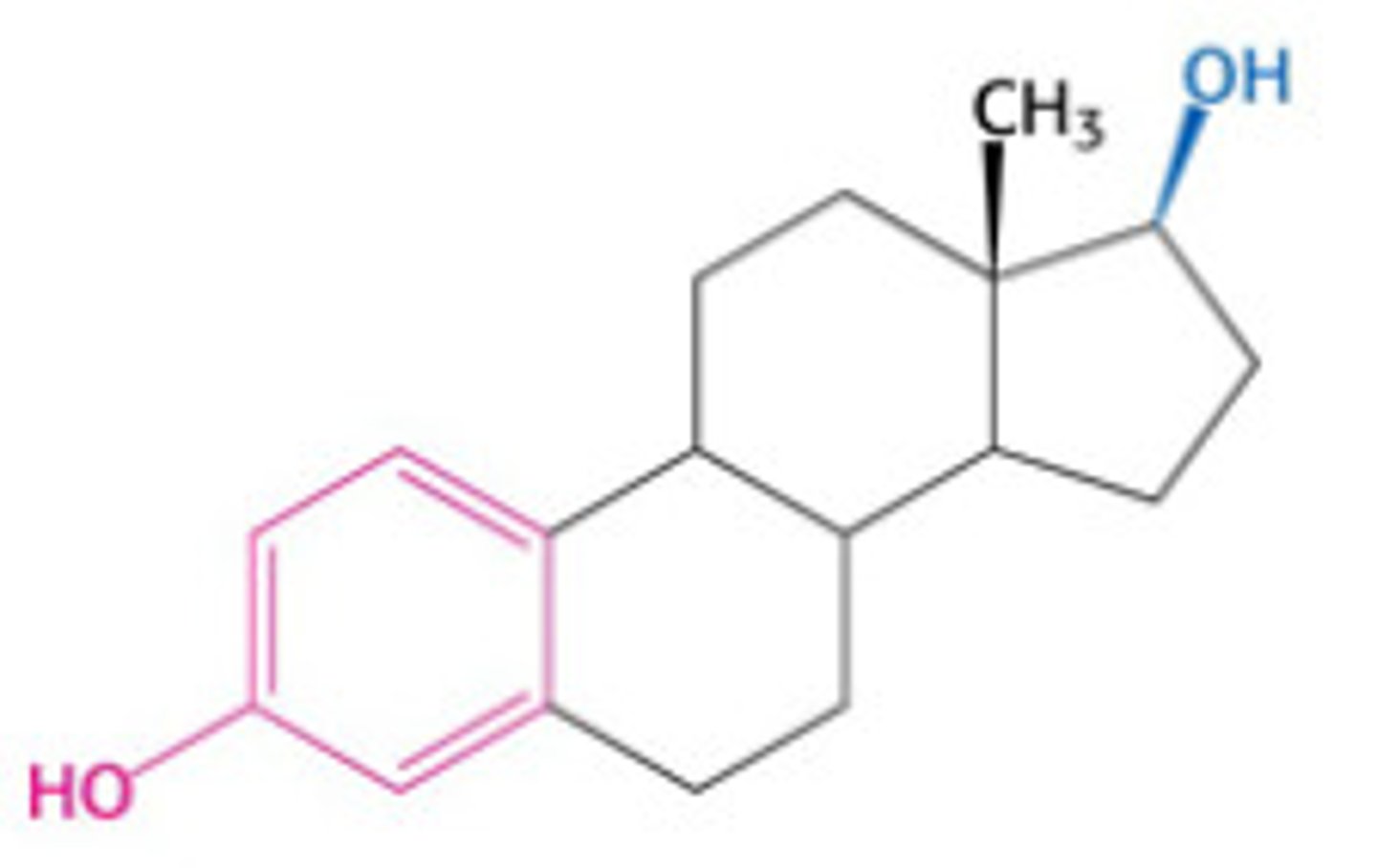
estradiol
A steroid hormone that stimulates the development and maintenance of the female reproductive system and secondary sex characteristics; the major estrogen in mammals.
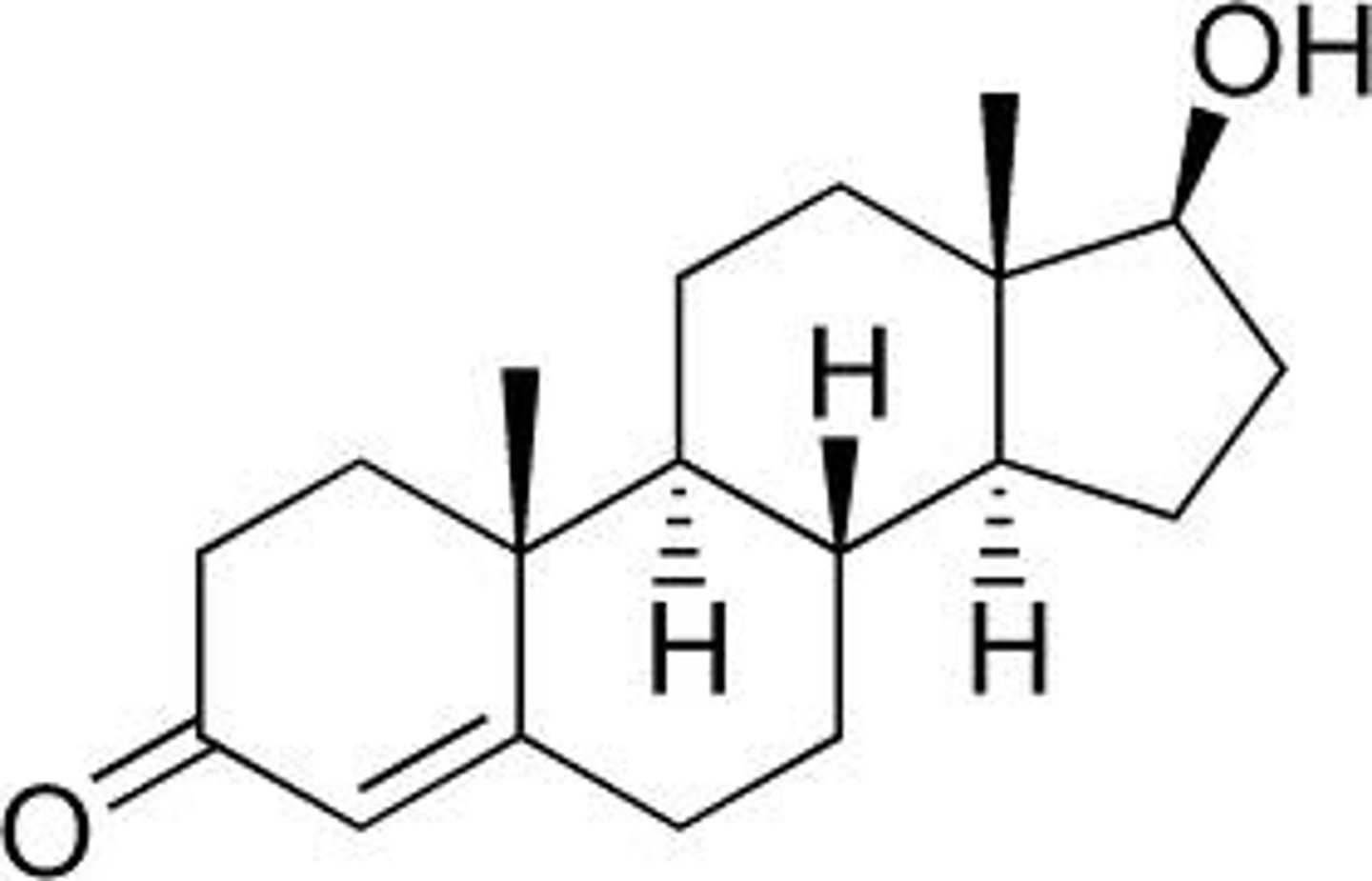
testosterone
Asteroid hormone required for development of the male reproductive system, spermatogenesis, and male secondary sex characteristics; the major androgen in mammals.
functional groups
A specific configuration of atoms commonly attached to the carbon skeletons of organic molecules and involved in chemical reactions.