BIOS 312 Exam 3 Review
1/229
Earn XP
Description and Tags
Unit 3 - Covers all lectures and microbes
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
230 Terms
What are viruses?
Genetic elements that cannot replicate independently of a living cell
Obligate intracellular parasites
What are the extracellular forms of viruses?
Virus particles or virions
Virions are the complete, infectious, and physical particle of a virus
These exist outside of the host
Facilitate transmission
Only replicate within a host
T/F: Most viruses are larger than prokaryotic cells
False; they range from 0.02 to 0.03 um
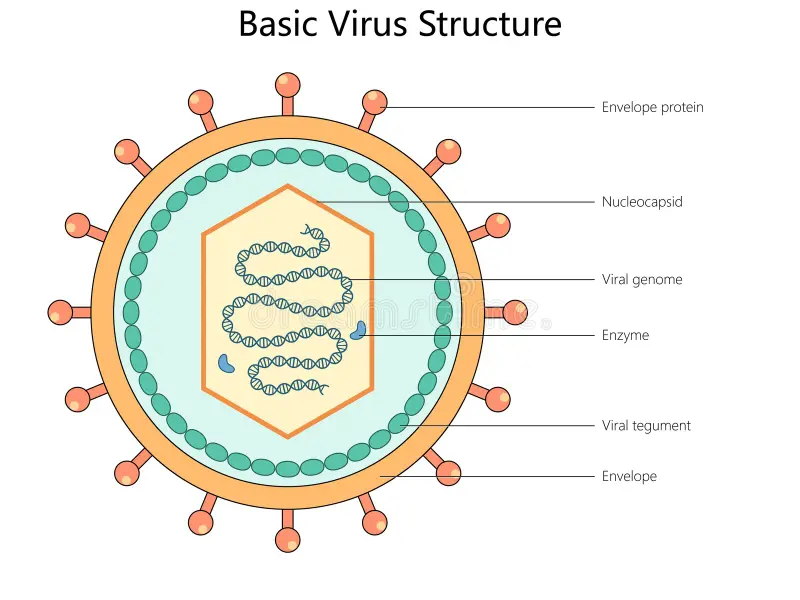
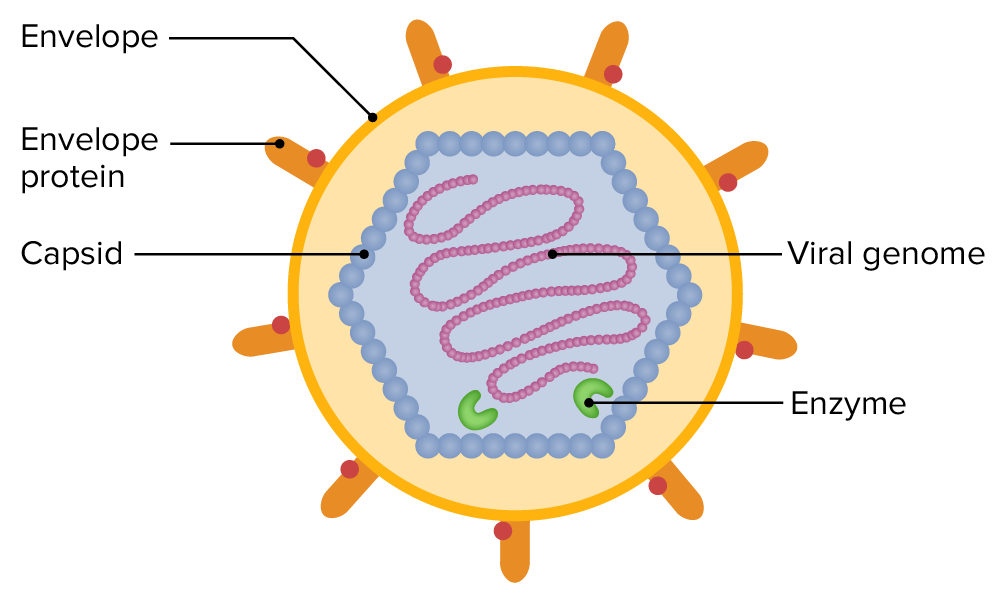
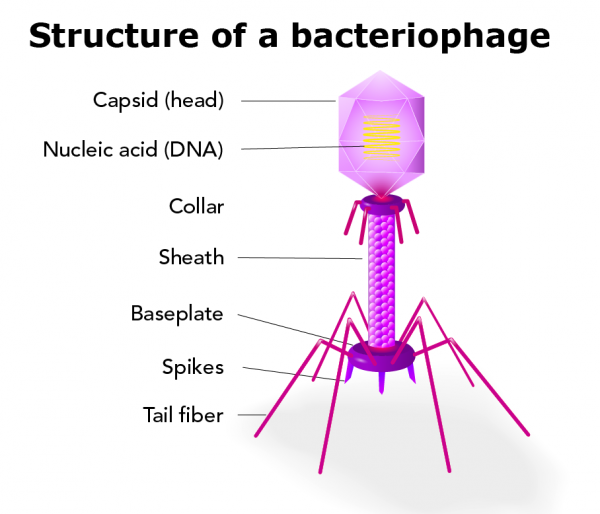
Describe the structure and components of a virion
Includes
Genome
Capsid: a protective shell formed by protein subunits called capsomeres
Nucleocapsid: combination of the nucleic acid and capsid
Viral tegument: a cluster of proteins that lines the space between the envelope and nucleocapsid
Envelope (optional): a lipid bilayer containing viral glycoproteins (spikes) for attachment
Enzymes (optional): specific proteins required for replication, sometimes packaged inside the capsid
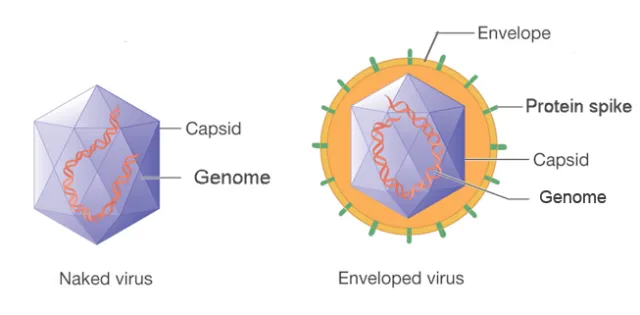
Compare naked viruses vs. enveloped viruses
Naked viruses (most bacterial viruses) have no other layers
Enveloped viruses (many animal viruses) have an outer layer consisting of a phospholipid bilayer (from host cell membrane) and viral proteins
Describe the size of a viral genome
Ranges from a 1.75 kilobase single strand within a circovirus
to a 1.25 megabase pair genome within a megavirus
Describe viral genomes
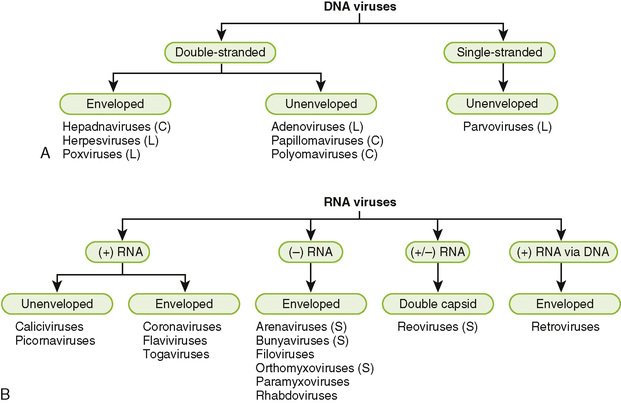
Can be either DNA or RNA; so they can be either single or double-stranded
Single-stranded genomes can be in either plus sense (same as mRNA) or minus sense (complimentary to mRNA)
Generally the genomes are smaller than those of cells
Are viral genomes circular or linear?
Most are linear, but some can be circular
How are viruses classified?
They are classified on the basis of the hosts they infect and their genomes
Bacterial viruses (bacteriophages; model systems)
Archaeal viruses
Animal viruses (extensively studied)
Plant viruses (less well-studied)
Other viruses
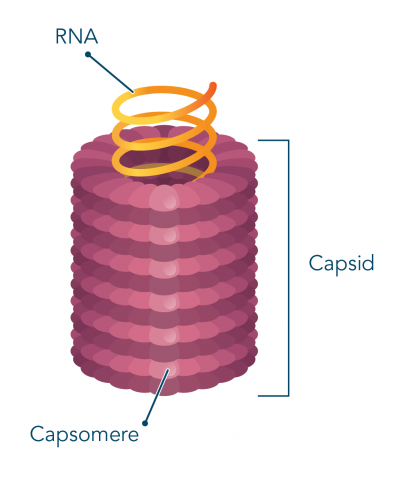
How does the capsid of a virus form?
It is made of individual proteins called capsomeres, that are arranged in precise, highly repetitive patterns around the nucleic acid
They can be assembled spontaneously (self-assembly) or with assistance from host-cell folding proteins
Helical virions are ___-shaped viruses
Rod-shaped (ex: tobacco mosaic virus)
Length of virus determined by length of nucleic acid
Width of virus determined by size and packaging of capsomeres
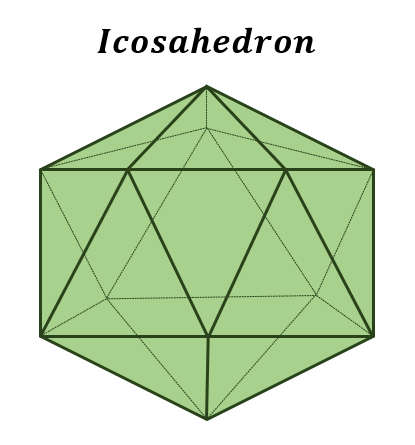
Icosahedral Virions
Viruses that are roughly spherical
Most efficient arrangement of subunits in a closed shell
Requires fewest capsomeres
Describe enveloped viruses
Have a lipoprotein membrane surrounding their nucleocapsid
Can be either RNA or DNA viruses
Can attach to and infect animal host cells
A few can infect plants/bacteria because of the cell walls surrounding the cell membrane
During infection, the entire virion enters animal cell
Can exit more easily
What kinds of enzymes can exist in a virion?
These are pre-formed enzymes
Lysozymes: makes hole in cell well to allow nucleic acid entry; lyses bacterial cell to release new virions
Neuraminidases: destroys glycoproteins and glycolipids; allows liberation of viruses from cell
Nucleic acid polymerases
RNA replicases: RNA-dependent RNA polymerases
Reverse transcriptase: RNA-dependent DNA polymerase in retroviruses
What is the major difference between prokaryotic and eukaryotic viruses?
Nucleic acid entry in prokaryotes
Virion entry in eukaryotes
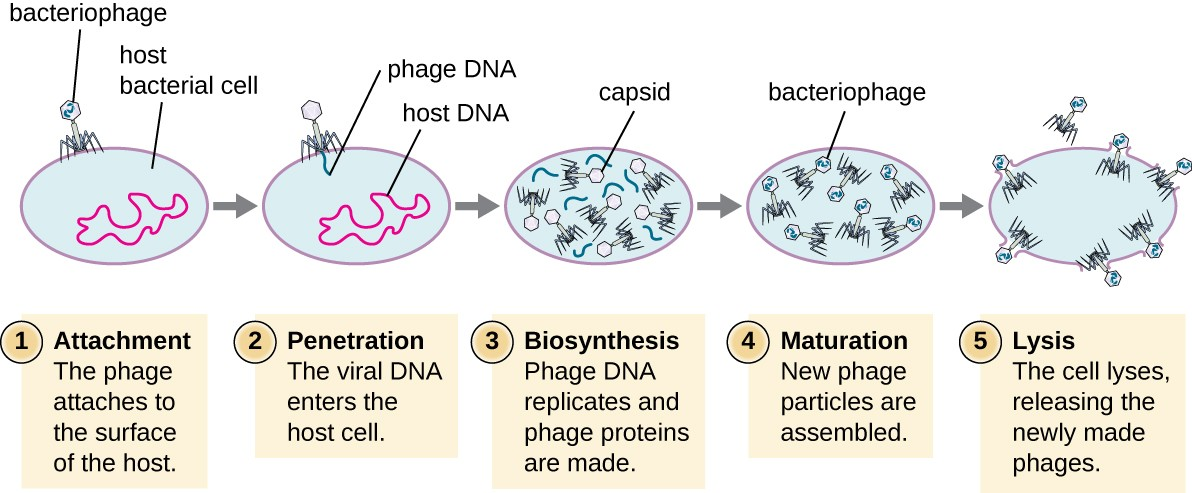
What are the phases of viral replication in a permissive (supportive) host?
Attachment (adsorption) of virion
Penetration (entry, injection) of the virion nucleic acid
Synthesis of viral nucleic acid and proteins by cell host metabolism as redirected by virus
Assembly of capsids and packing of viral genomes into new virions
Release (lysis) of mature virions from host cell
This is a prokaryotic virus
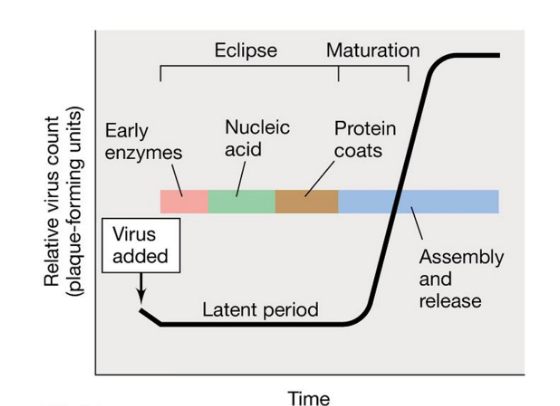
Describe the viral growth curve
Typically characterized by a one-step growth curve
Viral numbers increase once cell lyses
Phases of the curve
Eclipse: genome replicated and proteins translated
Maturation: packaging of nucleic acids in capsids
Latent period: eclipse + maturation
Release: cell lysis, budding, or excretion
Burst size: number of virions released
Time of life cycle
Bacterial viruses: 20-60 minutes
Animals: 8-40 hours
Compare virulent (lytic) viruses vs. lysogenic viruses
Virulent viruses: infect their host, replicate in the host, and then destroy the host to escape
Lysogenic viruses: viral DNA integrates into the host cell’s DNA, which alters the host as the viral DNA becomes part of its genome
Describe bacterial viruses (bacteriophages)
Very diverse group
The best studied are those that infect enteric bacteria (E. coli, Salmonella enterica)
Usually naked, although some can have lipid envelopes
Structurally complex and contain heads, tails, and other components
Most phages have dsDNA genomes
T/F: Attachment of a virion to a host is highly specific
True
Attachment requires complementary receptors on the surface of a susceptible host and its infecting virus
Receptors on host cell carry out normal functions for cell
Receptors include: proteins, carbohydrates, glycoproteins, lipids, lipoproteins, or complexes
T/F: The attachment of a virus to its host can result in changes to both virus and cell surface that facilitate penetration
True
What is a permissive cell?
A host cell that allows the complete replication cycle of a virus to occur
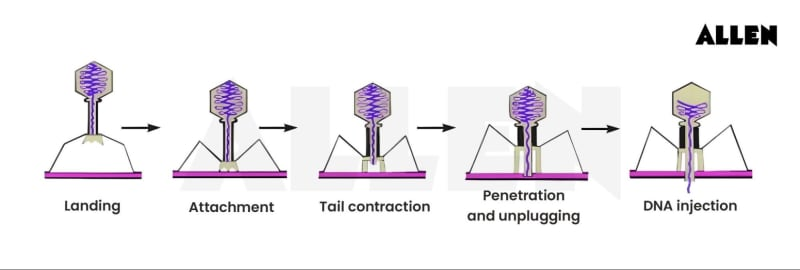
Describe Bacteriophage T4
A virus of E. coli
One of the most complex penetration mechanisms known
Virions attach to the cell via tail fibers that interact specifically with polysaccharides on the E. coli cell envelope
The tail fibers retract, and the tail core makes contact with the E. coli cell wall
Lysozyme-like enzymes forms a small pore in the peptidoglycan
The tail sheath contracts and viral DNA passes into the cytoplasm
Describe the synthesis step of the viral life cycle
The virus hijacks the host cell’s metabolism to synthesize proteins (including sigma factors) and replicate nucleic acids
RNA polymerase is also modified to recognize virus promoters
Describe the assembly and packaging step of the viral life cycle
Structural proteins for virion self-assembly and packaging of the nucleic acid in the capsid
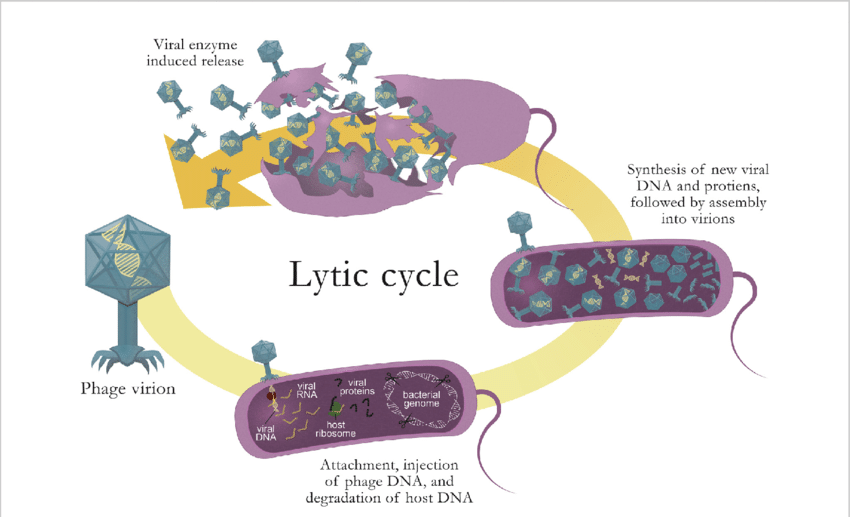
Describe the lytic cycle
Lytic infection cycle of virulent phages
A phage virion attaches and injects its phage DNA into a host cell; degrades host DNA
New viral DNA and proteins are synthesized followed by assembly into virions
Viral enzymes (lysozymes) induce the release of progeny phages
T/F: All viruses are lytic
False
T/F: Lysogenic viruses are ecologically significant
True; many environmental bacteria are infected with lysogens
T/F: The virulence of many human pathogens is controlled by lytic bacteriophages
False; it is controlled by lysogenic bacteriophages
What is the Bacteriophage Lambda?
A temperate phage that infects E. coli used for studies
Has a linear, dsDNA genome
Complementary, single-stranded regions 12 nucleotides long at the 5’-terminus of each strand
Upon penetration, DNA ends base-pair forming the cos site; DNA ligates and forms double-stranded cricle
When lysogenic, integrates into E. coli chromosomes at the lambda attachment site
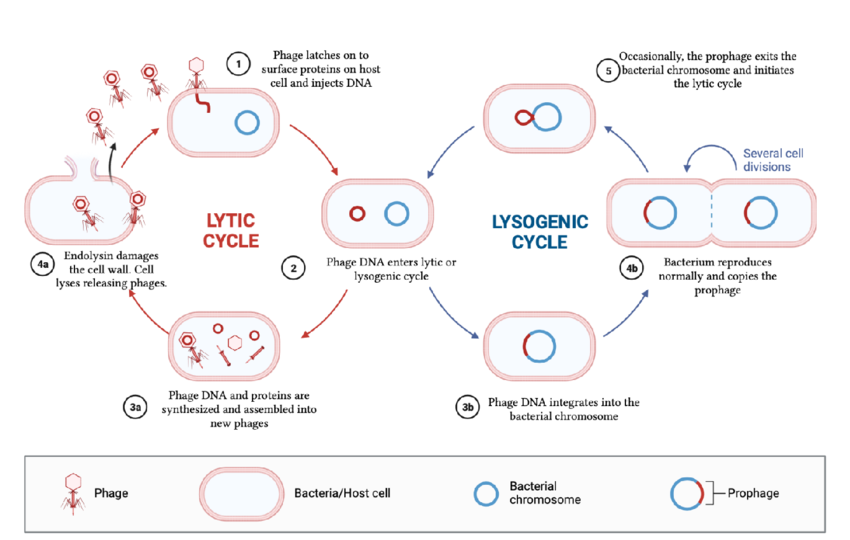
Temperate bacteriophages carry out two types of life cycles. What are they?
The lytic and lysogenic cycle
The lytic cycle for temperate bacteriophages is similar to the lytic cycle for virulent phages
Compare the lytic vs lysogenic cycle for viruses
Lytic cycle: immediate, rapid replication of viruses, and the destruction (lysis) of the host cell.
Lysogenic cycle: allows virus to remain dormant, integrating its genome into the host's DNA to be replicated passively during cell division without destroying the host
What determines if a virus becomes lytic or not?
Gene promoters and regulatory proteins
Describe what happens when Bacteriophage Lambda enters the lytic cycle
It synthesizes long, linear concatemers of DNA by rolling circle replication
Genome-sized lengths cut as cos sites; genomes packaged into phage heads
After the tails are added, lysis occurs
Transduction: packaging of host chromosomal genes and transfer to a new host can also occur
When it is lysogenic, its DNA integrates into E. coli chromosome at the lambda attachment site using lambda integrase
What are the two key repressor proteins involved in temperate phage replication?
Cl protein (lambda repressor): causes repression of lambda lytic events
Cro repressor: represses lambda cII and cIII proteins required for lysogeny, thus controls activation of lytic events
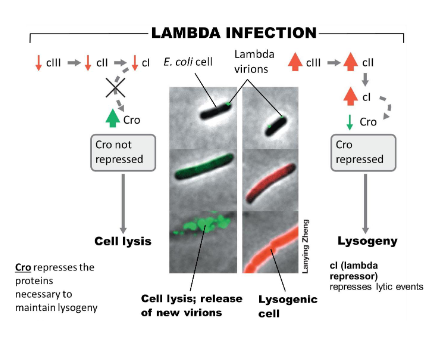
What are the outcomes of lysogeny?
Via binary fission, host cell and the provirus DNA are both replicated and passed on to the progeny
Upon induction of some event (ex: stress), the provirus DNA excises out of the host chromosomal DNA and initiates the lytic cycle
Genetic information in the provirus can provide new characteristics for the host cell
What components do animal viruses share with other viruses?
Capsid
RNA/DNA genome
Many pathogenic viruses have RNA genomes
What differentiates animal viruses from other viruses?
The entire virion enters the animal cell
Eukaryotic cells contain a nucleus, which is the site of replication for many animal viruses
Describe the viral infection of animal cells
Animal viruses bind specifically to host cell receptors, which are typically used for cell-cell contract or immune function
Different tissues and organs express different cell surface proteins
Viruses often only infect certain tissues
Entry usually occurs by fusion with the cytoplasmic membrane or endocytosis
Describe the process of animal virus infection
Uncoating occurs in the cytoplasm
DNA genomes enter the nucleus; RNA genomes are converted into DNA within the capsid
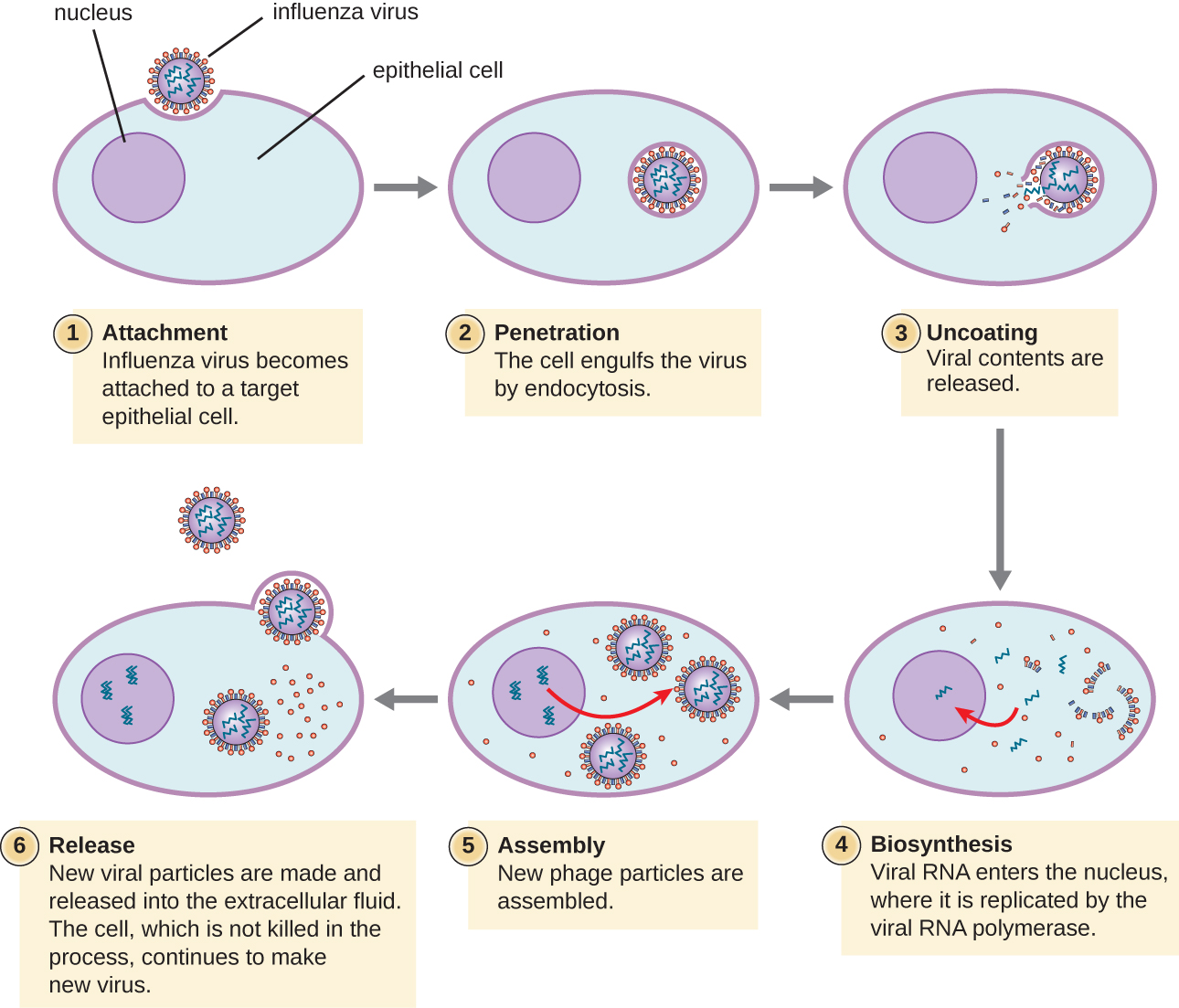
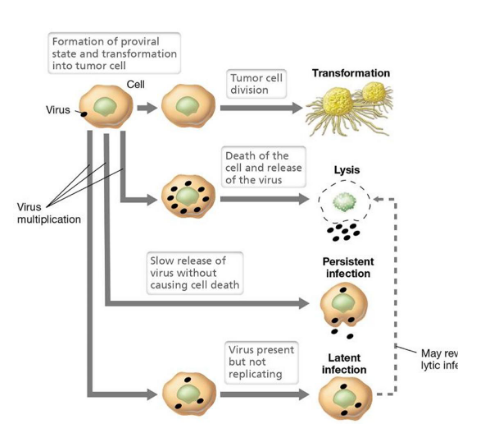
What are the outcomes of animal viral infection?
If the virus is virulent: host cell is lysed (most common outcome!)
If latent infection: viral DNA exists in host cell chromosomes and virions are not produced; cell is unharmed
If persistent infection: virions are released by budding and the cell does not lyse
The cell can also undergo transformation and become cancerous
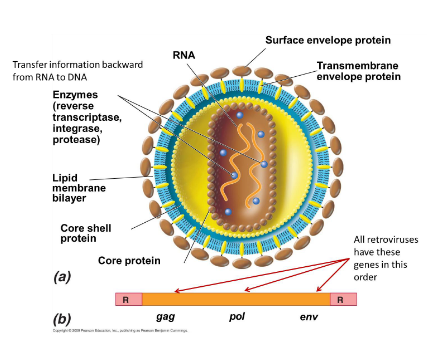
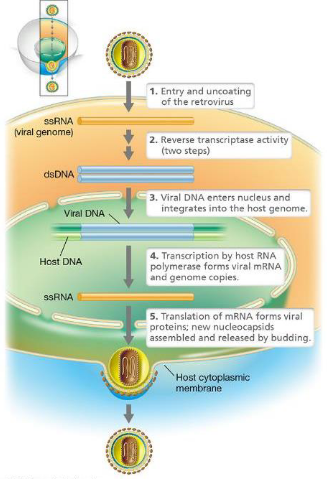
What are retroviruses?
RNA viruses
Enveloped virions that contain two copies of the RNA genome
Virion contains several enzymes, which include reverse transcriptase to make DNA copies of the genome
Genes included: gag, pol, env
Some have a fourth gene downstream of env that is active in cellular transformation
What are hepadnaviruses?
DNA viruses
How does retroviral replication work?
Virus enters the cell; it fuses to the cytoplasmic membrane at receptor sites
Envelope is removed. Genome and enzymes remain in the core
Reverse transcription leads to the creation of a linear double-stranded DNA that enters the nucleus
Retroviral DNA integrates into the host genome similar to a temperate bacteriophage
Transcription leads to the formation of mRNAs and viral RNA genome
Genomic RNA and nucleocapsids assemble
Enveloped virions are released from the cell by budding
Enumeration of viruses
Viruses replicate only in certain types of cells or in whole organisms
Bacterial viruses are easiest to grow = model systems
Animal viruses (and some plant viruses) can be cultivated in tissue or cell cultures
Plant viruses are typically most difficult because study often requires growth of the whole plant
Titer
The number of infectious agents per volume of fluid
Plaque Assay
One way to measure virus infectivity
Analogous to the bacterial colony
Plaques are clear zones that develop on lawns of host cells
Lawn can be bacterial or tissue culture
Each plaque results from infection by a single virus particle
Satellite Virus
A subviral agent composed of nucleic acid
The success of the virus depends on coinfection with another virus for their replication
Virioid
Infectious RNA that lacks a protein coat
(Smallest known pathogens (246-399 nucleotides) - only known to infect plants)
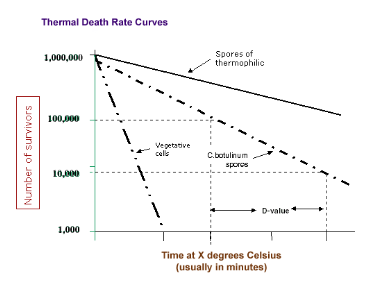
T/F: The death rate of bacteria is constant
True
What factors influence how well a disinfectant works?
The number of microbes (more microbes = more time needed to kill them)
Environmental factors (temperature; presence of organic material, etc)
Time of exposure: antimicrobials often require an extended time to affect resistant microbes or endospores
Microbial characteristics can also affect choice of chemical/physical growth control methods
What kind of environment does a disinfectant work better in?
In warm solutions or in the presence of organic matter (blood, vomit, feces)
Can also inhibit the action of chemical microbials
Why are bacteria encased in a biofilm more difficult to treat with disinfectant?
Because the microbes are suspended in media that is rich in proteins and fats
Many disinfecting agents target what of a microbe?
The cell membrane because damage to it can cause cellular contents to leak out of the cell and interfere with cell growth
Can also alter shape of enzymes (through heat or pH) leave cells susceptible to death
Alterations to DNA or RNA (ex: UV treatment) leaves cell unable to divide
Sterilization
The destruction of all living microorganisms
Most common method: heating
Commercial Sterilization
Destroying all pathogenic and spoilage microorganisms as well as heat-resistant spores, especially from C. botulinum, from shelf-stable foods
This means that a very small number of resistant bacterial spores may remain, but will not be dangerous
Complete sterilization is not required for many areas (like food or surgical wounds)
Disinfection
Microbial control aimed at destroying harmful bacteria
Eliminates only vegetative bacteria (not spores)
Methods: UV light, chemical disinfectants
Antisepsis: when used on living tissue; thus the agent becomes an antiseptic
Degerming
Mechanically eliminating germs instead of killing them directly
Ex: getting skin swabbed with alcohol for a flu shot
Sanitization
Reducing the number of microbes to prevent disease
What is the suffix for treatments that eliminate/kill microorganisms?
-cide
What is the suffix for treatments that inhibit the growth of microorganisims?
-stasis
What are the physical methods used to control microbial growth?
Heat
Filtration
Low temperatures
High pressure
Desiccation
Osmotic Pressure
Radiation
How does heat affect microbial growth?
Heat denatures enzymes
Thermal Death Point (TDP): lowest temperature at which all cells in a liquid culture are killed in 10 minutes
Thermal Death Time (TDT): minimal time for all bacteria in a liquid culture to be killed at a particular temperature
Decimal Reduction Time (DRT): Minutes to kill 90% of a population at a given temperature
Autoclave
Provides steam under pressure at 121C at 15 psi for 15 minutes
Kills all organisms and endospores
Steam must contact the item’s surface
Large containers require longer sterilization times
Test strips used to indicate sterility
Pasteurization
Uses heat to reduce spoilage organisms and pathogens
Equivalent treatments
63C for 30 min
High-temperature short-time (HTST): 72C for 15 sec
Ultra-high-temperature (UHT): 140C for 4 sec
Thermoduric organisms survive
Dry Heat Sterilization
Kills by oxidation
Flaming
Incineration
Hot-air sterilization
Filtration
Passage of substance through screen-like material
Used for heat-sensitive materials
High-efficiency particulate air (HEPA) filters remove microbes > 0.3um
Membrane filters remove microbes >0.22 um
T/F: Low temperature has a bacteriostatic effect
True; slows the growth
Refrigeration
Deep-freezing
Lyophilization (freeze drying)
Desiccation
Absence of water prevents metabolism
Osmotic pressure uses salts and sugars to create a hypertonic environment, causing plasmolysis
What are the different types of radiation that can be applied to controlling microbial growth?
Ionizing radiation (x-rays, gamma, electron beams)
ionizes water to create reactive hydroxyl radicals
damages DNA causing lethal mutations
Nonionizing radiation (UV, 260nm)
damages DNA by creating thymine dimers
Microwaves
kill by heat; not especially antimicrobial
What should be known about chemical methods for controlling microbial growth?
Can be used to control growth of microbes on living/non-living tissue
Few agents achieve sterility - most reduce microorganism numbers to safe levels
Not all agents work on all microorganisms
What is the Use-Dilution Test?
To measure the effectiveness of liquid disinfectants on hard, non-porous surfaces
A metal/glass cylinder is dipped into a culture and then submerged into a disinfectant
The cylinders are placed into a sterile broth medium to check for growth - turbidity determines effectiveness
Disk Diffusion Test
Filter paper disks are impregnated with a disinfectant and placed onto a lawn of bacteria
Zone of clearing around disk = the agent is effective at disinfecting the bacteria

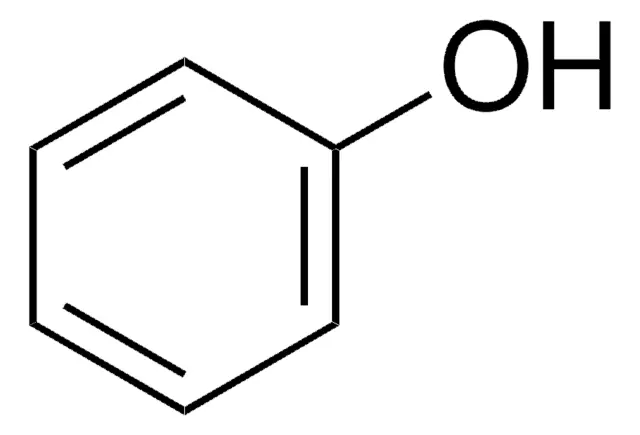
What are phenols?
A type of disinfectant
Not frequently used on living tissue (is an irritant, does not smell good)
Significant antimicrobial effects at 1% solution
Phenolics (derivatives of phenols) reduce the irritant qualities of phenol
Works well with soap and detergents
Persist for a long time after application
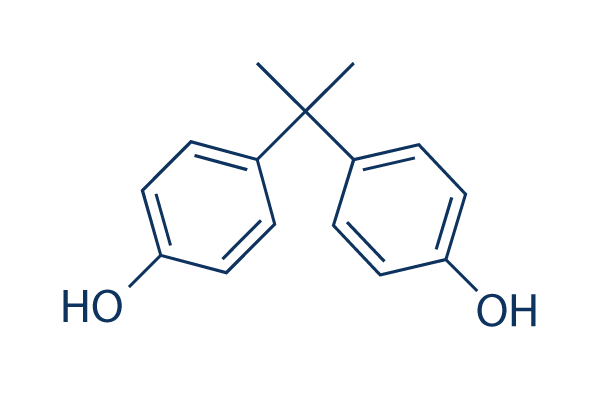
What are bisphenols?
A type of disinfectant derived from phenols
Part of the prescribed lotion pHisoHex
Triclosan (another version) is found in antimicrobial soups and in cutting boards/plastic knife handles to prevent foodborne illness
Prevents synthesis of fatty acids
What are biguanides?
Broad spectrum disinfectants
Attacks the bacterial membrane
Works well against gram-positive bacteria
Can work on gram-negative, except for pseudomonads
Generally not sporacidal
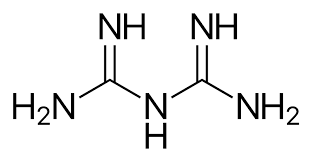
What are halogens?
Iodine and chlorine → effective alone and as constituents of inorganic/organic compounds
Iodine: one of the oldest disinfectants and is available in tinctures
A liquid form of compressed chlorine gas can be used to disinfect municipal water supplies and swimming pools
What do alcohols do?
Kill bacteria and fungi, not endospores and non-enveloped viruses
Generally denature proteins and dissolve lipids (disrupts membranes)
Evaporate rapidly and leave no residue
Common forms: ethanol and isopropanol
Can be used to enhance the activity of other disinfectants
What do heavy metals and their compounds do?
Several metals are biocidal and antiseptic (mercury, copper)
Silver and copper can prevent growth (oligodynamic action)
What are surfactants?
Surface active agents
Can decrease surface tension and dislodge bacteria and other microorganisms from surfaces
What do soap and detergent do?
Allows for the mechanical removal of microbes when added to scrubbing
Soap is an emulsifier and breaks things up into small pieces
Not antiseptic
What are acid-anionic sanitizers?
Important for cleaning food-processing facilities
Combinations of phosphoric acid and surface-active agents
Have negatively charged particles that disrupt plasma membranes
What are quaternary ammonium compounds?
The most widely used surfactants
Positively-charged
Bactericidal against gram-positive bacteria
Fungicidal
What is used as chemical food preservatives?
Sulfur dioxide = prevents food spoilage
Organic acids = inhibit metabolism
Sorbic acid, benzoic acid, calcium propionate prevent molds in acidic foods
Nitrates and nitrates prevent endospore germination
What are bacteriocins?
Proteins produced by one bacterium that inhibits another
What type of antibiotic prevents the spoilage of cheese
Nisin and natamycin
What do aldehydes do?
Inactivate proteins by cross-linking with functional groups
-NH2, -OH, -COOH, -SH
Used for preserving specimens and in medical equipment
Formaldehyde and ortho-phthalaldehyde
Glutaraldehyde: one of the few liquid chemical sterilizing agents
Describe chemical sterilization
Gaseous sterilants cause alkylation (replacing hydrogen atoms of a chemical group with a free radical)
Cross-links nucleic acids and proteins
Used for heat-sensitive material
Ethylene oxide
What is plasma?
Fourth state of matter, consisting of electrically excited gas
Free radicals destroy microbes
Used for tubular instruments
What are supercritical fluids?
CO2 with gaseous and liquid properties
Used for medical implants
What are peroxygens and other forms of oxygen used for?
Oxidizing agents
Used for contaminated surfaces and food packaging
O3, H2O2, and peracetic acid
What did Paul Ehrlich do?
Introduced concept of a “magic bullet” that selectively destroys pathogens and leaves the surrounding tissue unaffected
Basis for chemotherapy and selective toxicity
What did Alexander Fleming do?
Discovered the first true antibiotic penicillin
Accidentally inoculated a plate of S. aureus with a mold from the air and observed clearance around the mold
What is an antibiotic?
A substance produced by a microorganism that inhibits the growth of other microorganisms
Easy to discover
Very few of them useful clinically
More than half of our antibiotics are produced from Streptomyces spp
Filamentous bacteria that lives in the soil
A few are produced by endospore-producing bacteria like Bacillus
Others produced by mold
Either bactericidal: kills cells
or bacteriostatic: prevents microbes from gorowing
T/F: Synthetic drugs like sulfa are antibiotics
False, they are antimicrobial drugs
Antibiotics naturally occur from microorganisms
How were many of the antibiotics used today discovered?
By screening colonies of antibiotic-producing organisms or by screening soil samples
What is selective toxicity?
Generated through attacking structures unique to prokaryotes (ex: lipopolysaccharides, porins)
To pass through the channels made by porins, drugs must be small and hydrophilic
Lipophilic drugs do not pass through Gram-negative cell walls
Compare broad-spectrum and narrow-spectrum agents
Narrow-spectrum antibiotics: act against a limited group of bacteria
Broad-spectrum antibiotics: act against a larger group of bacteria
T/F: During treatment, narrow-spectrum antibiotics are used first, then broad spectrum antibiotics
False
Giving broad-spectrum antibiotics first saves time, but it can also act on normal microbiota
The harvesting of energy is mostly done through what kind of reactions?
Redox reactions
The most electronegative element is the electron acceptor