CHEM 2131 | E2 Mechanism
1/13
Earn XP
Description and Tags
E2
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
the E2 mechanism uses ____ instead of nucleophiles
bases
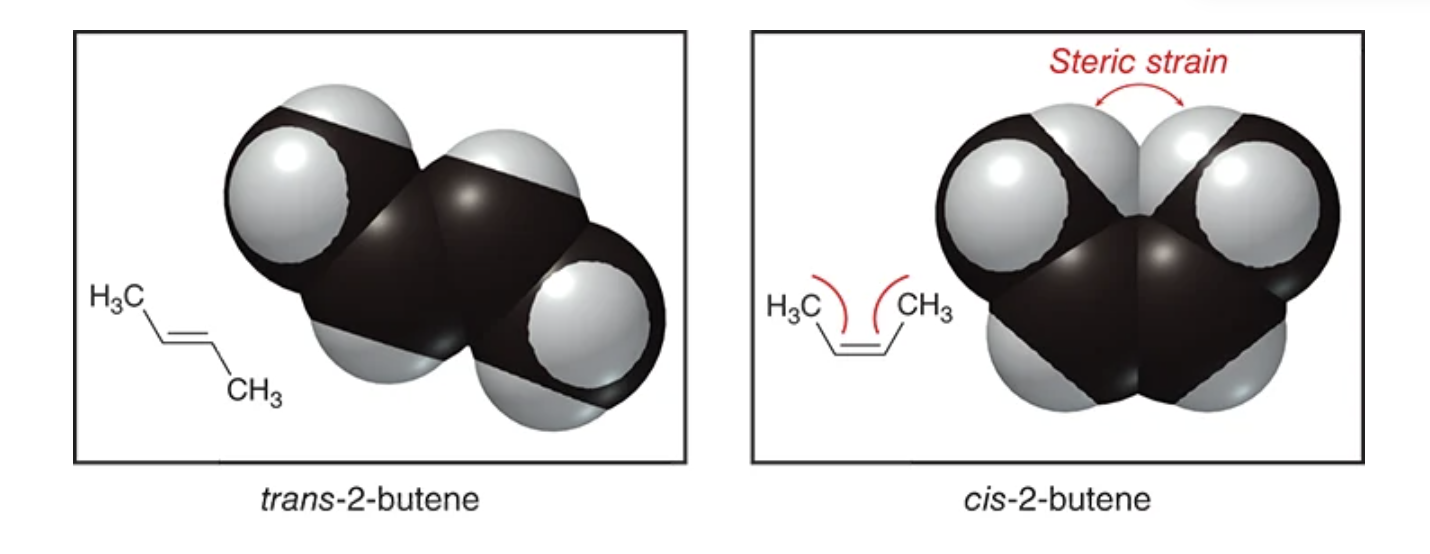
Alkenes are more stable when they are ______ ______
highly substituted
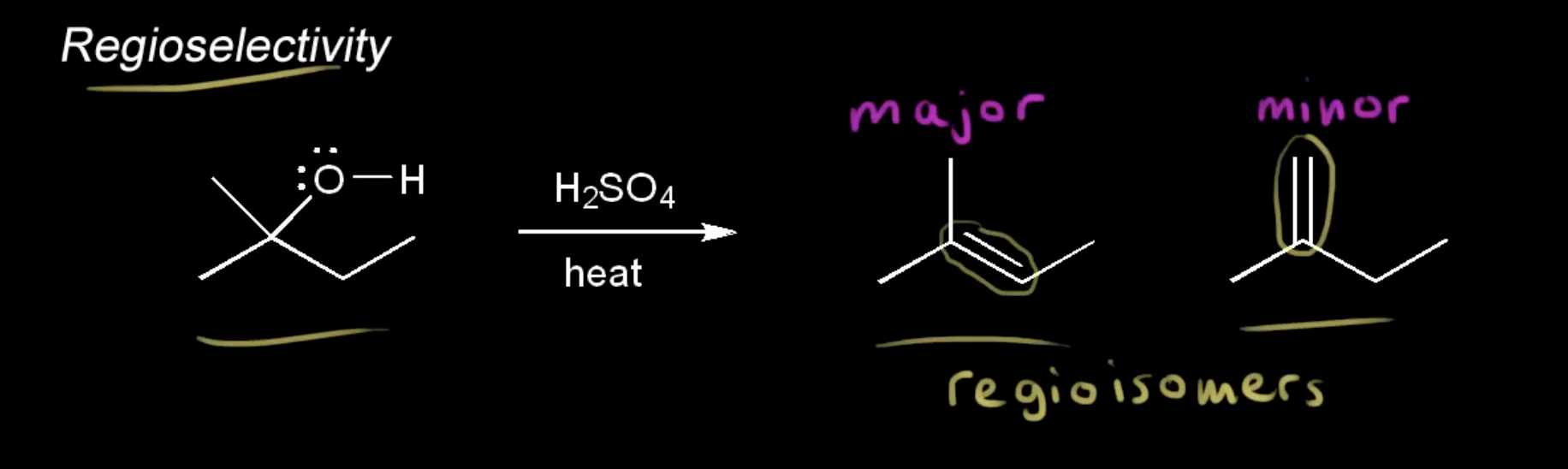
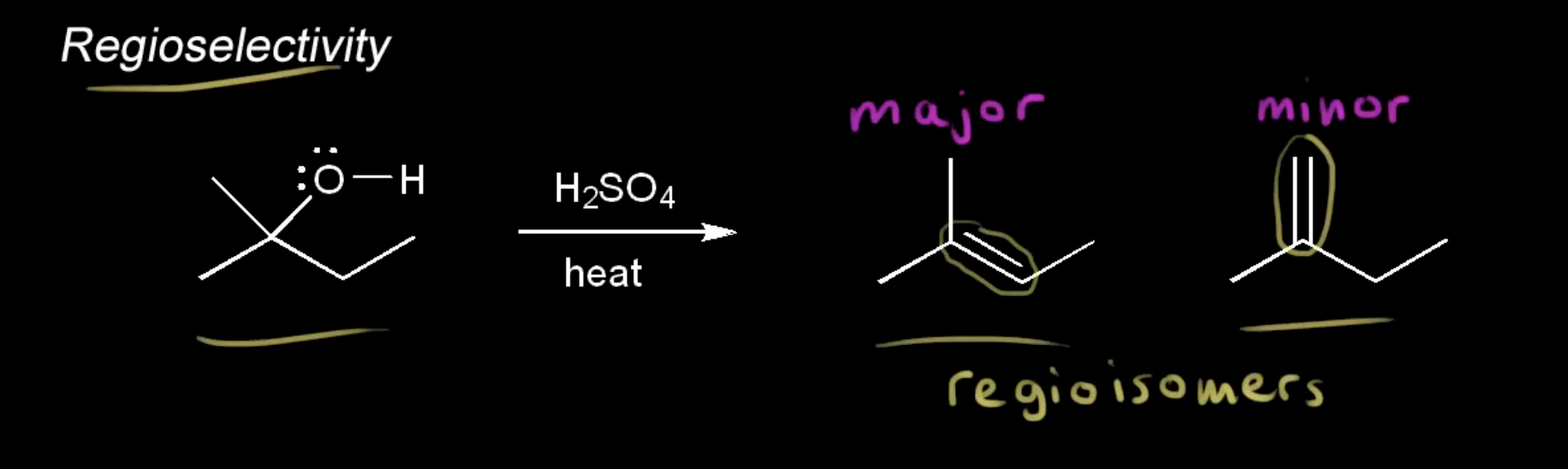
regioselectivity
an E2 reaction that can form more than 1 product (differs in placement of double bond), with a major and minor product
stereoselectivity
a type of chemical reaction that preferentially forms one stereoisomer over another when multiple options are possible (trans)
the Zaitsev product refers to the most ___ product (or the most substituted)
stable
the Hoffman product refers to the most ____ proton
accessible (less steric hindrance)
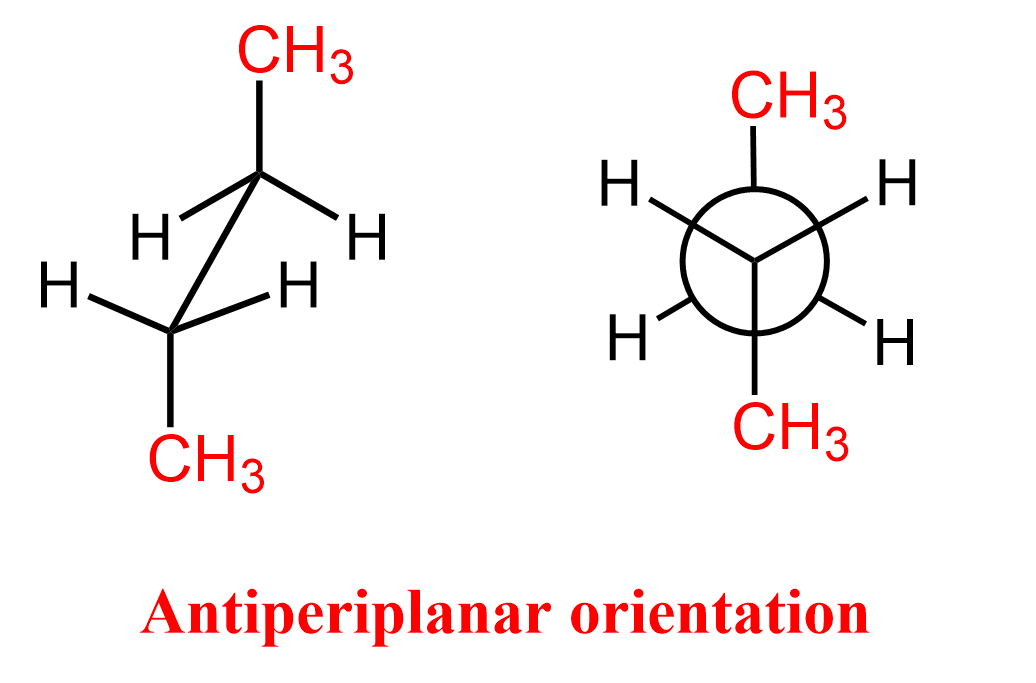
antiperiplanar
Anti-periplanar in E2 reactions means the -hydrogen and the leaving group are on opposite sides of the molecule ( dihedral angle) in the same plane; allows rxn to occur efficiently
on a cyclohexane ring, an E2 reaction only occurs from the chair conformation in which the leaving group is ____; the H and Cl must be ___ to each other (on opposite sides)
axial, trans
when a fat base is used, the major product is the ___ product
Hoffman
when a skinny base is used, the major product is the ______ product
Zaitsev
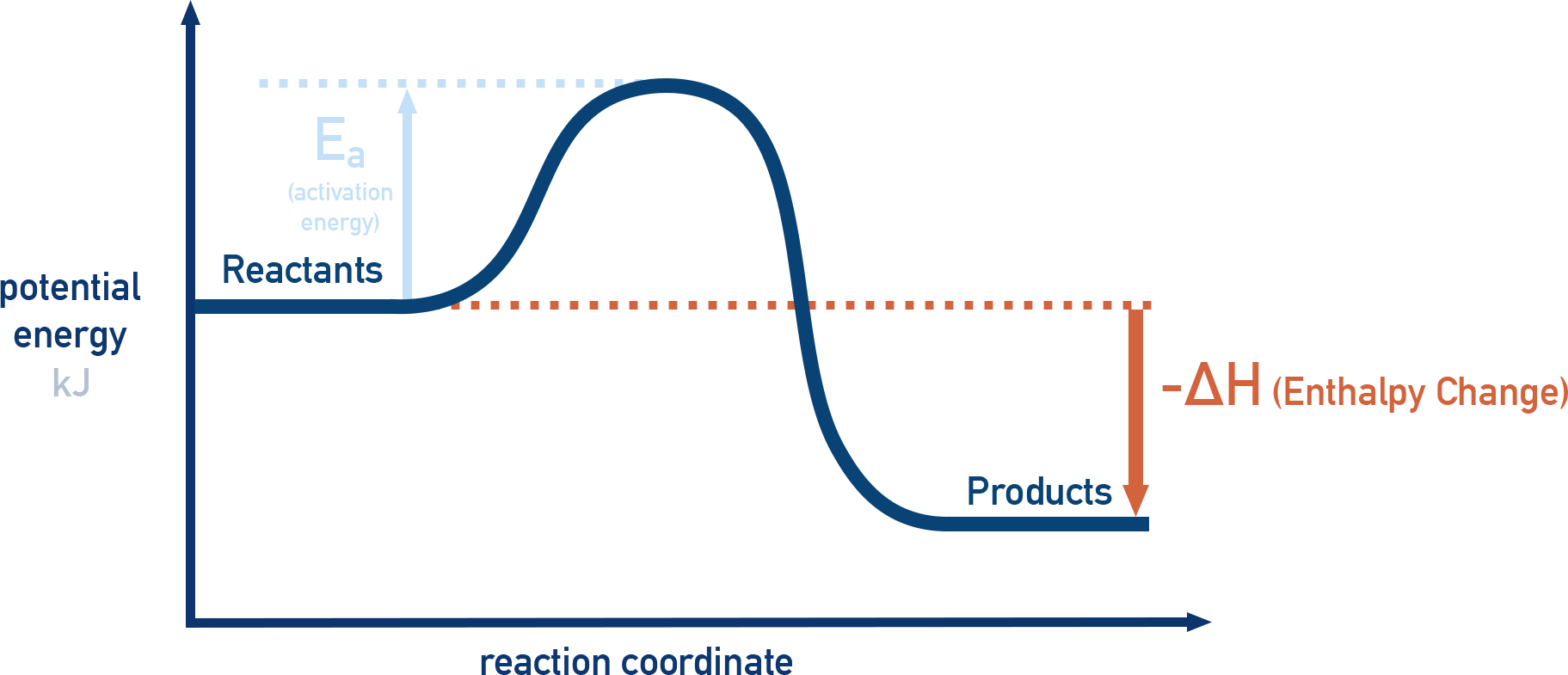
an E2 reaction is _____ overall
exothermic
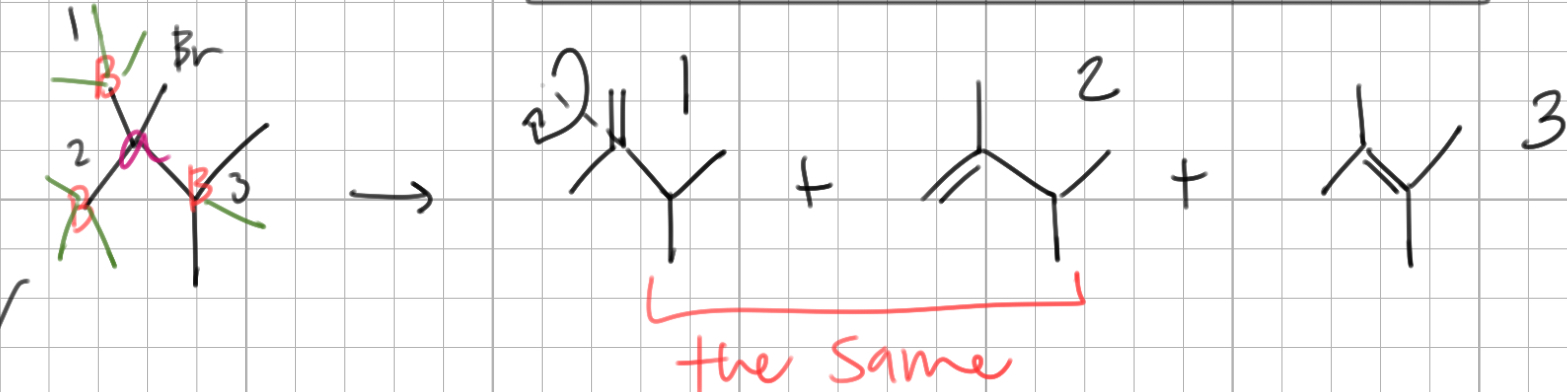
when determining if two products are the same, assess if the alpha carbon is a _____ _____, if it is not, determine which of the two beta carbons are the ___
chiral center, same
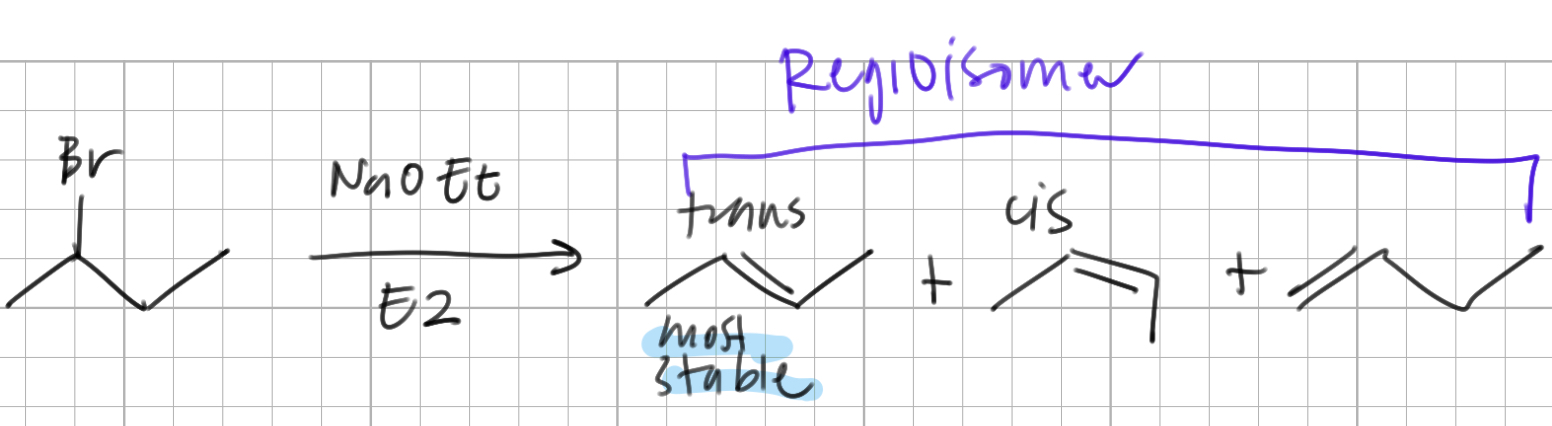
if there are two beta protons attached to a beta carbon (with sigma bond), there will be free ____ and two _____ will be produced, ___ and ___
rotation, stereoisomers, cis, trans
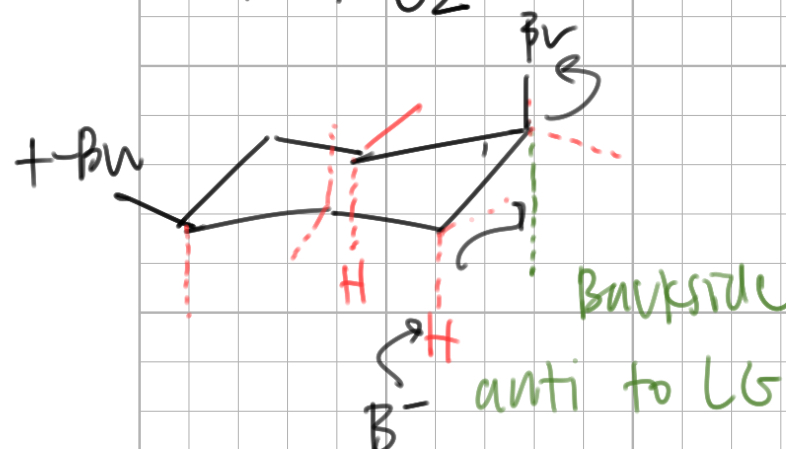
a fast E2 reaction occurs when the leaving group and beta H are both ___
axial