Octet Expansion
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is octet expansion?
Octet expansion is the ability of elements to use available d-orbitals so that there are more than 8 electrons in the outer shell.
The molecules formed by octet expansion are sometimes called what?
Hypervalent.
Give an example of an element which can form a compound with more than 8 electrons in the outer shell.
Phosphorus- can form PCl5 with 10 electrons in the outer shell of the phosphorus atom.
Phosphorus can show a valency of 3 and 5. This means that both PCl3 and PCl5 can exist. Explain why.
Phosphorous is able to achieve a valency of 5 by promoting an electron from the 3s orbital to the 3d orbital; this allows it to have 5 unpaired electrons available for bonding.
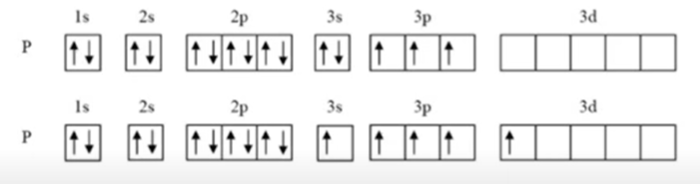
Why can octet expansion not occur with elements in Periods 1 and 2?
Elements in these periods do not have access to d-orbitals.