Chapter 15 Ideal gases
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
chapter 15.1 Ideal gases

15.2 intro gas laws
weather balloons are launched into the upper atmosphere to measure changes in temperature and pressure, and air currents and atmospheric pollutants
as the balloon rises, the atmospheric pressure around it drops, causing it to expand
the relationships between the temperature, pressure, and volume of an ideal gas can be described by a few simple gas laws

what happens when the temperature and mass of gas remains constant?
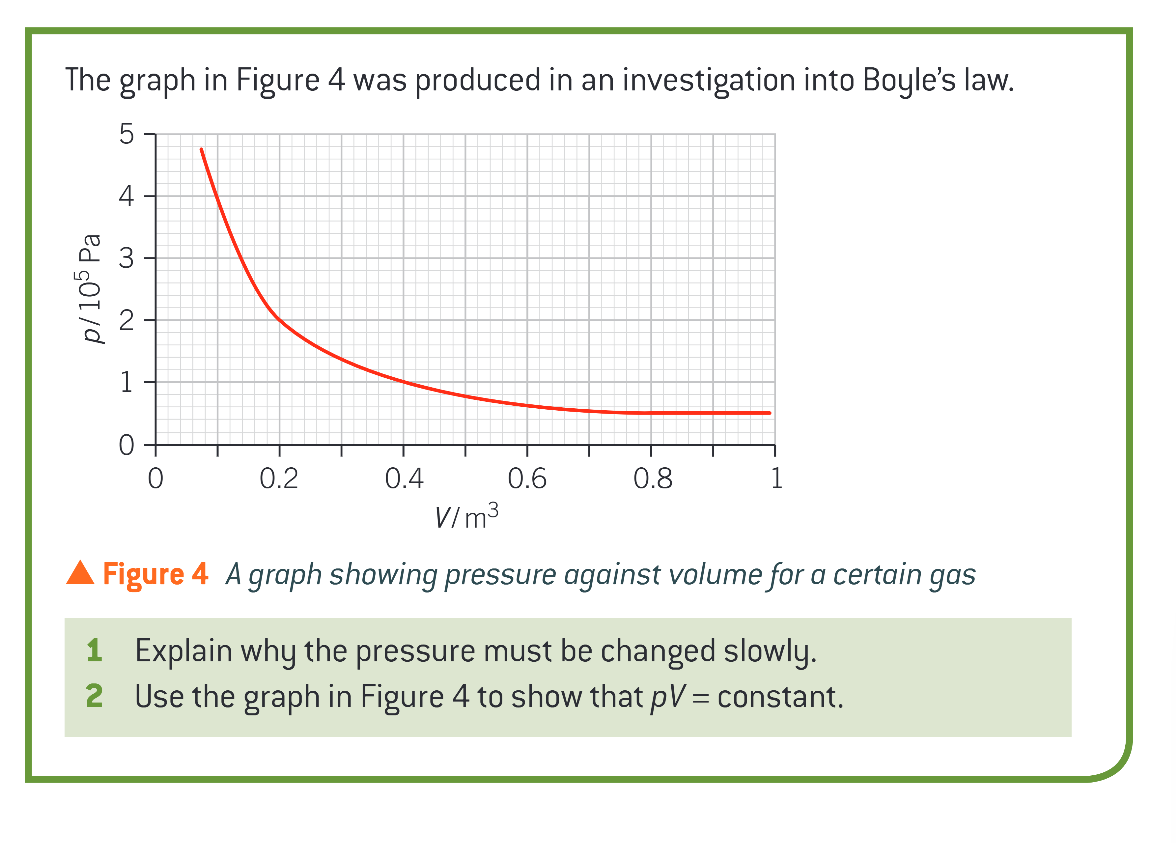
if a fixed mass of gas is kept in a sealed box, halving the volume of the box (slowly, to ensure the temperature remains constant) would do what to the gas and the box?
it would compress the gas and double the pressure it exerts on the box
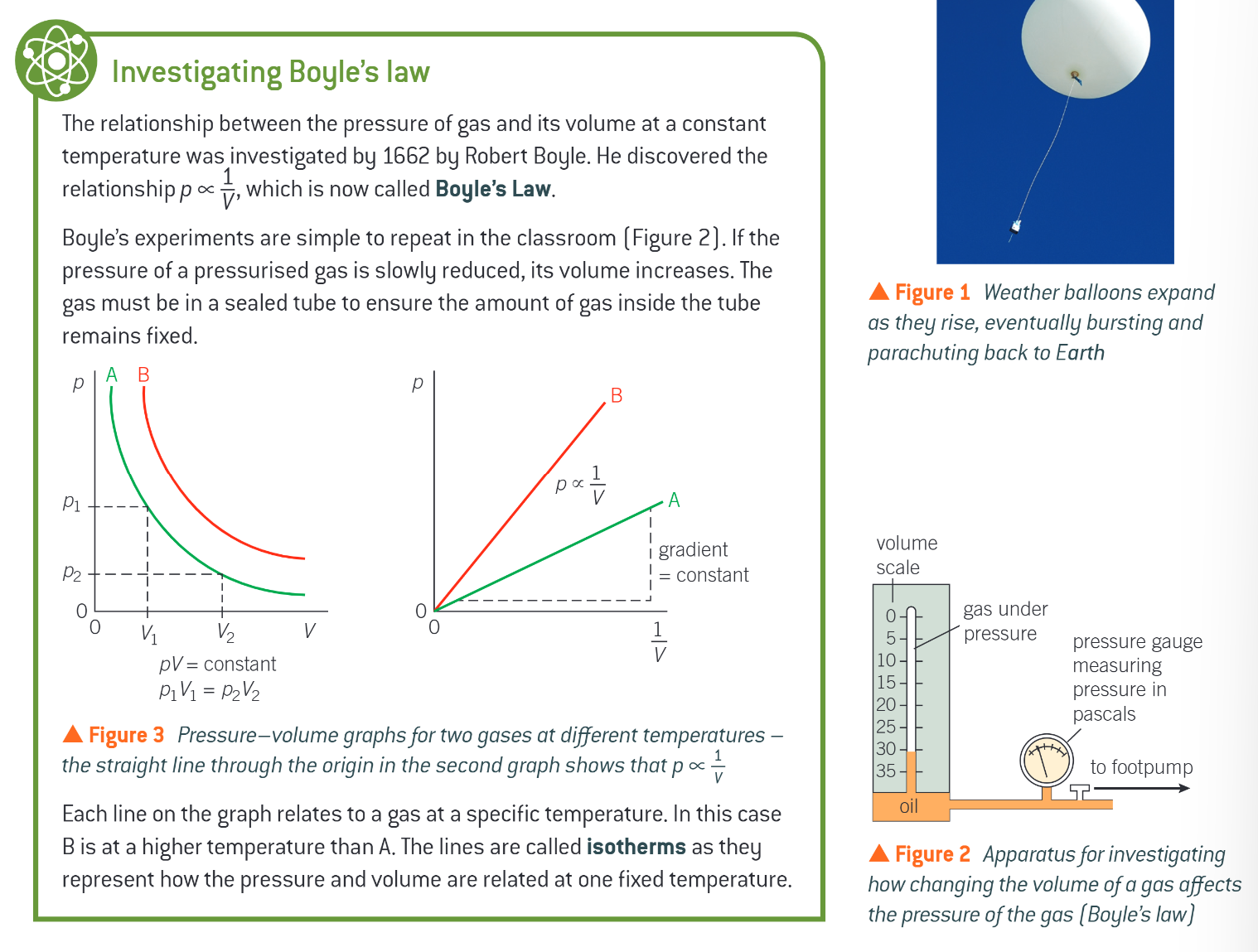
what are isotherms
the lines on the p against V graphs
they represent how the pressure and volume are related at one fixed temperature
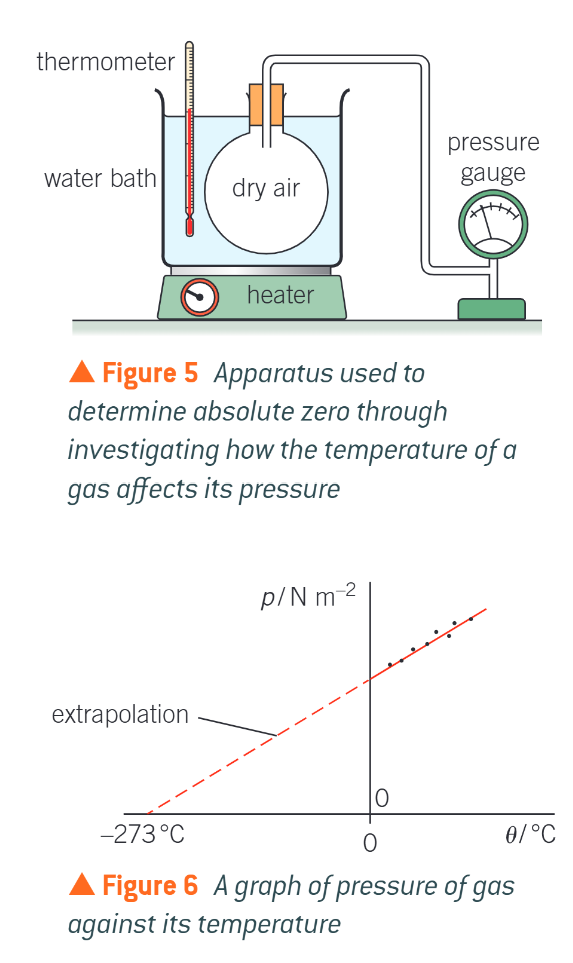
what is the relationship between p and T of a gas if the volume and mass of the gas remain constant

estimating absolute 0 - how do you do this
because the expression into the relationship between the pressure of a fixed volume and mass of gas and its temperature can provide an approximate value for absolute 0
with the set up shown in figure 5, the temperature of the water baths can be increased and the resulting increase in pressure of the gas inside the sealed vessel recorded
at absolute 0, the particles are not moving (the internal energy is at its minimum) so the pressure of the gas must be 0
plotting a graph of pressure against temperature theta in celsius from the experimental results gives a line that can be extrapolated back to a point where the pressure is 0
combining gas laws
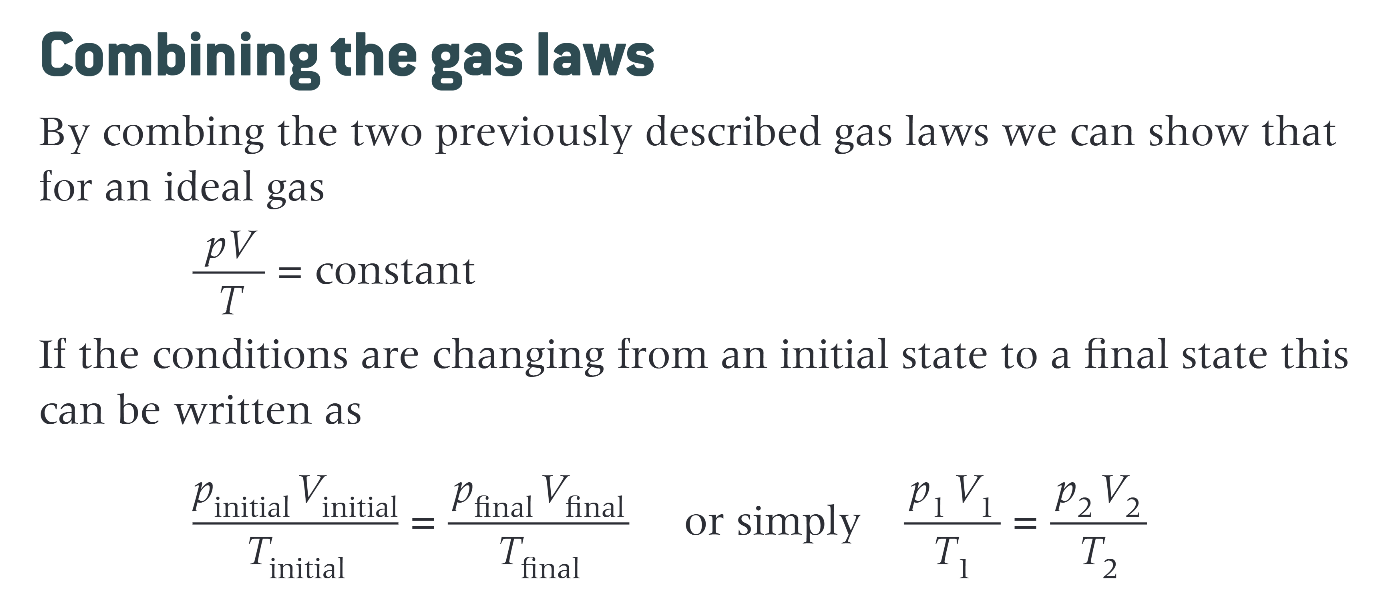





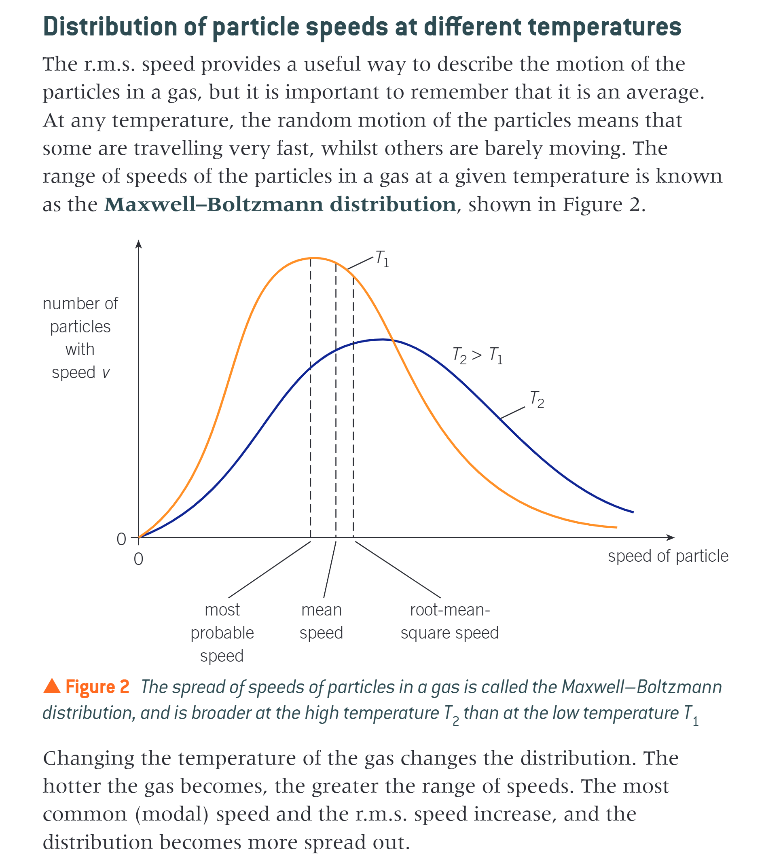
15.3 intro - root mean square speed - what happens when average velocity = 0
we have already seen how the particles (atoms or molecules) in a gas move in random directions at different speeds
if we calculated the average velocity of the particles in a gas, because velocity is a vector the average would be 0ms-1
all the velocities of such a large number of particles would simply cancel out
so in order to describe the typical motion of particles inside the gas, we use a different measure, the root mean square speed (r.m.s speed)




15.4 intro - the boltzmann constant



how do you derive the mean kinetic energy equation?


at a given temp what do all atoms or molecules of different gases have that are the same
they have the same mean KE
particles will have different ___ speeds as?
rms speeds as they have different masses

what is the internal energy of a gas
the sum of the kinetic and potential energies of the particles inside the gas
one of the assumptions of an ideal gas states that the electrostatic forces between particles in the gas are negligible except during collisions. this means that there is no what type of energy?
no electrical potential energy
all internal energy is in the form of the kinetic energy of the particles
doubling the temp of an ideal gas doubles the average KE of the particles inside the gas, which also doubles its?
internal energy