Topic B - The Particulate Nature of Matter
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Density
Mass per unit volume
Differences between solids, liquids and gases in terms of arrangement and energy
Solids:
Most dense
Strongest intermolecular bonds
Least KE and PE
Vibrate around a fixed point in a fixed arrangement
Liquids:
Irregular arrangement
Flow around each other
Stronger intermolecular bonds than gases
Gases:
Least dense
Weakest intermolecular bonds
Most KE and PE
Can collide with each other
Temperature
Average random kinetic energy of the particles in a substance.
Heat
Transfer of thermal energy from hot to cold.
Thermal equilibrium
When two bodies in contact reach the same temperature and energy transfers stop.
Internal Energy
The sum of the total random kinetic energy and the total intermolecular potential energy of all the molecules. U = EK + EP
SHC
The amount of thermal energy required to raise 1 kg of a substances temperature by 1K.

When calculating the temperature change in mixtures what’s the process?
Equate the two substances
Energy lost by steel = Energy gained by water
Sub in the values making sure that the temperatures are in different positions (Tf - x and y - Tf)
(4)(450)(Tf - 85) = (10)(4200)(52-Tf)
Solve
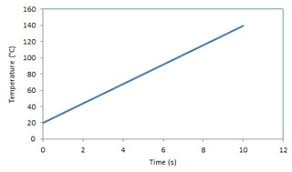
What is the gradient on a Temperature Time graph
P/mc
SLH
Amount of energy required to change the state of 1kg of substance from solid to liquid (fusion) or liquid to gas (vapourisation) without any change in temperature.
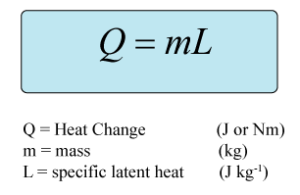
SLH Equation
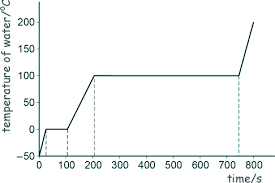
Describe the features of a temperature time graph
During a change of state the temperature stays constant ( so KE stays constant) and energy goes into increasing PE only ( weakening/forming intermolecular bonds)
A steeper gradients means a smaller SHC as there will be a greater change in temperature.
Longer time means that there will be more energy and therefore a bigger SLH
Convection and convection currents
Thermal energy transfer throw fluids.
Convection currents form as the fluids heat and up and rise it cools and loses KE and become more dense and sinks which starts a convection current.
Thermal Radiation
The transfer of energy by electromagnetic waves. It doesn’t require a medium and can travel through a vacuum.
All objects with a temp above 0K emit thermal radiation. The higher the temperature the higher intensity of radiation.
Conduction
In a sold if one side has a higher temperature then, the particles on that side are moving faster on average.
This kinetic energy is transferred to the neighbouring particles through collisions.
Metals are good conductors as they have free electrons which transfer even more energy.
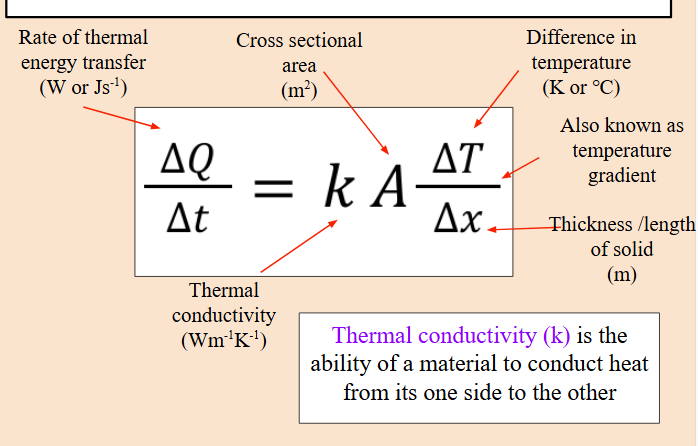
Rate of thermal energy transfer formula
Which state of matter absorbs the most wavelengths
Solids