Lab D: Reduction of Vanillin to Vanillyl Alcohol
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
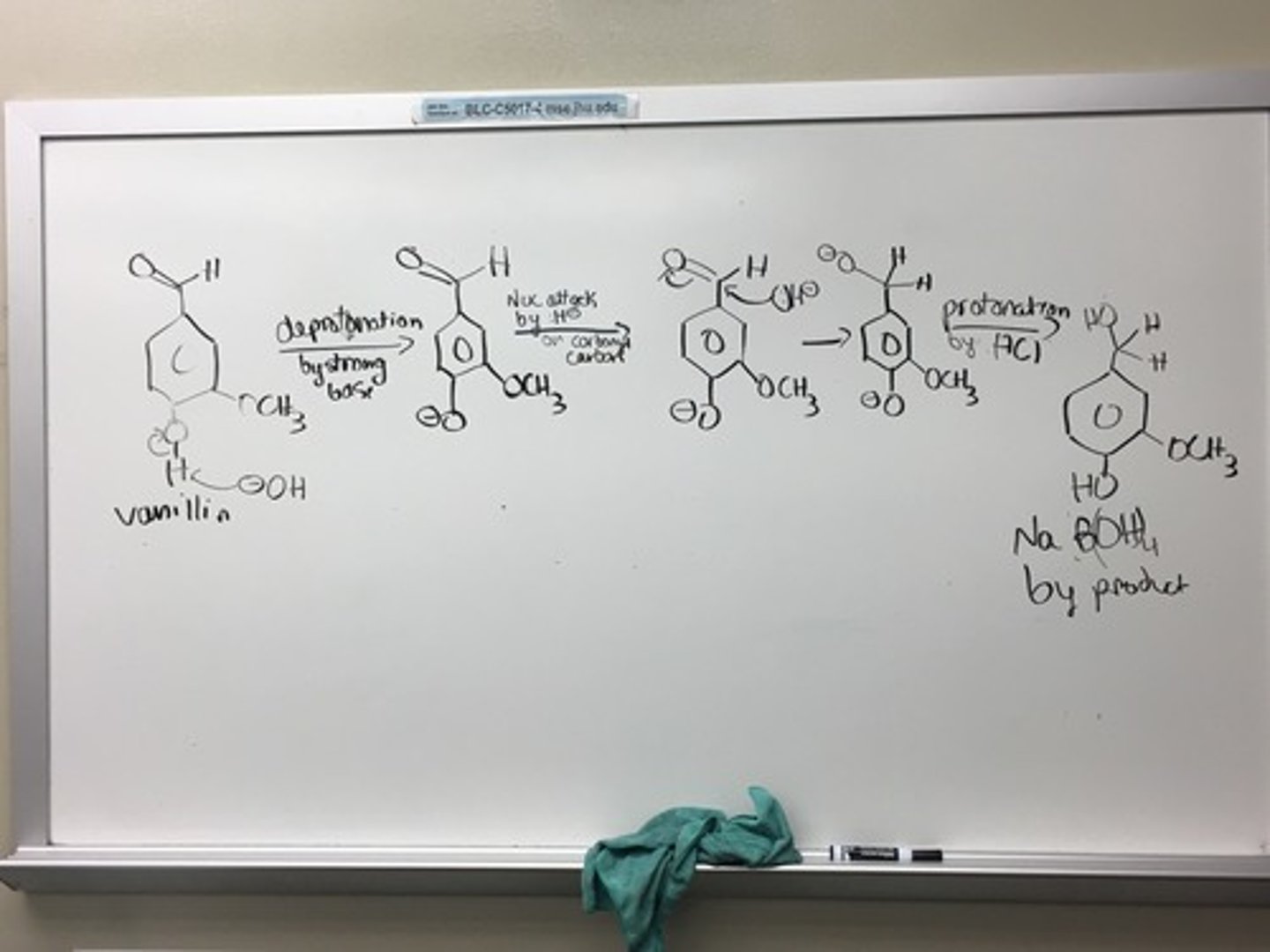
Reaction Mechanism of Vanillyl Alcohol
reducing aldehyde of vanillin to vanillyl alcohol
Calculating Limiting Reactant
every molecule of NaBH₄ is 4 H ions; have to multiply the mass of NaBH₄ by 4 to get mass of H ions; the limiting reactant is vanillin
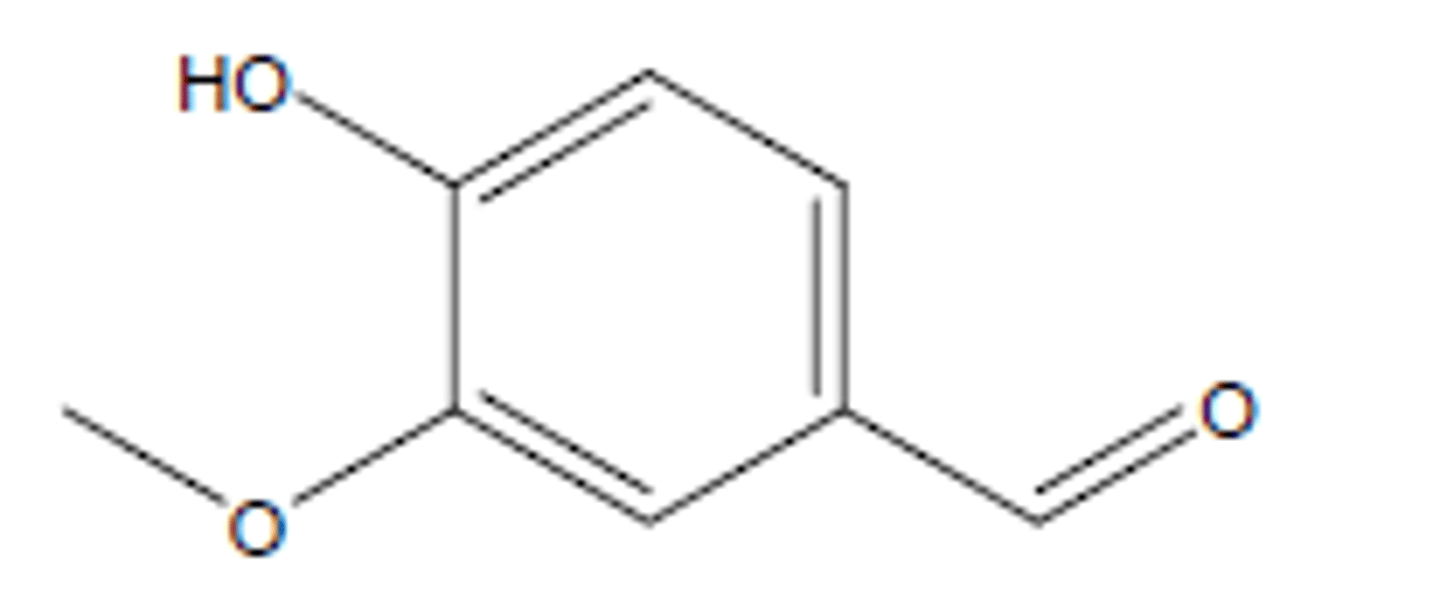
vanillin
aromatic compound found in vanilla extract; commonly used for flavoring, baking, and perfume industry; starting material for L-dopa to treat Parkinson's disease; 152.15 g/mol; 81-83°C; d 1.056 g/mL
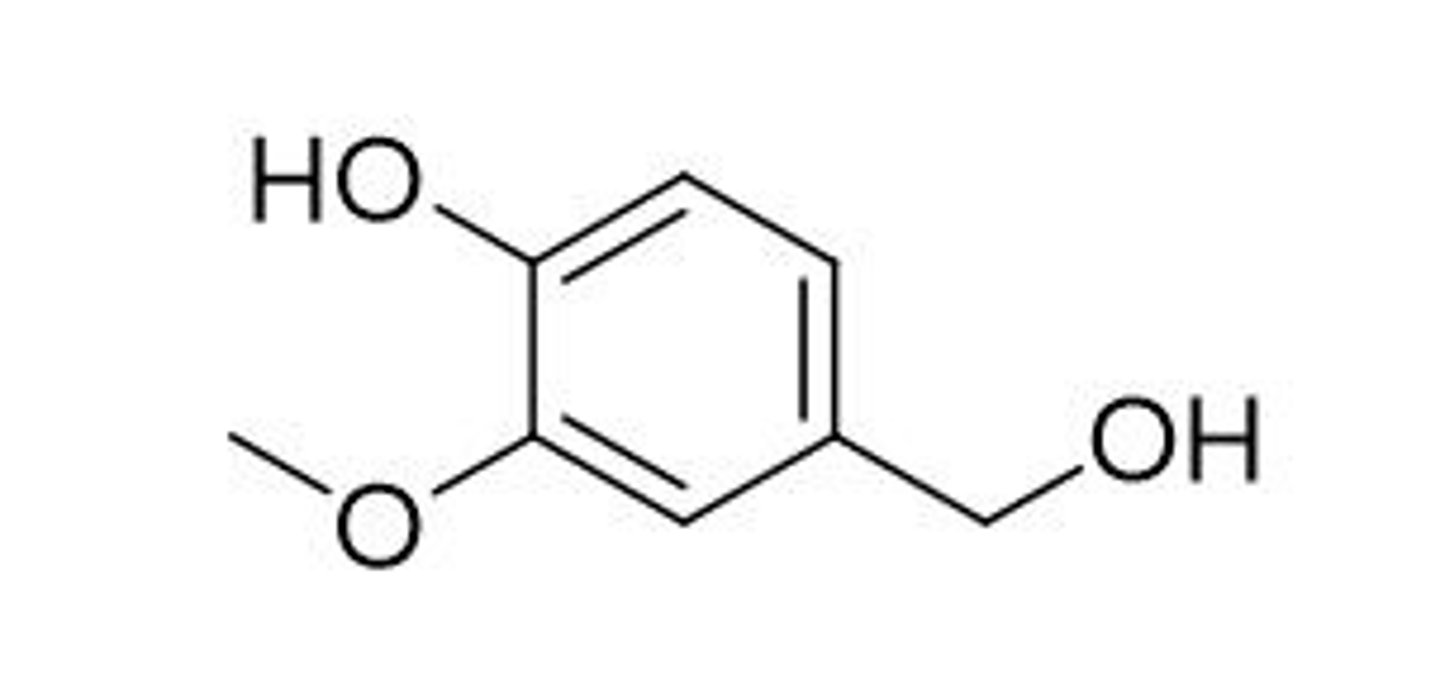
vanillyl alcohol
renewable source towards synthesizing biologically active molecules and flavoring ingredients; the African sugarcane moth and Leaffooted pine seed bug use this for chemical communication channels.
sodium borohydride (NaBH₄)
reducing agent; good source of hydride ions; hydride ions is a good nucleophile toward the carbonyl group; LiAlH₄ is extremely reactive with water, but NaBH₄ reacts slowly with water; count moles as H's provided, so 4 moles. MW: 154.16g/mol; d: 1.07 g/mL
HCl
reacts with excess NaBH₄ and produces hydrogen gas and boric acid; vanillyl alcohol then precipitate from solution
IR Spectra
Vanillin: OH on aromatic ring stretch
Aldehyde C=O stretch
Aromatic ring stretch
C=C ring stretch
Vanillyl Alcohol: OH on aromatic ring stretch
Absence of aldehyde C=O stretch
New stretch from CH2-OH group
Aromatic ring stretch
C=C ring stretch
ratio of eluent
4 ethyl acetate: 6 hexane