EARTH 232 - MODULE 1 & 2
1/121
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
122 Terms
formula for ductile solids or fluids
P = pgh. (p = density of overlying rock, g = acceleration due to gravity (9.8), h = thickness of rocks above.
pressure gradient in mantle
increases with depth, as rock density increases.
pressure gradient in core
pressure increasing with depth slows down, as acceleration due to gravity is low in the core
two primary heat sources in Earth
primordial heat: heat stored in earth from formation (10-25), and heat released from from the decay of radioactive isotopes in the continental crust (75%-90%)
main mechanisms of heat transfer
conduction and convection
conduction
transfer of energy from hot to cold. important from rigid/brittle materials. It is efficient but slow in Silicate Rocks.
Convection
density driven heat transfer, and is good for ductile materials capable of flow.
advection
passive/physical transfer of heat by rocks in motion. ex: tectonic uplift.
solidus
temperature over which rock melting begins at various pressures
liquidus
when 100% of the rock has melted
three principal ways of partial melting in crust and upper mantle
1) Raise temp, 2) lower pressure, 3) add fluids
Partial Melting: Raise the Temp
add heat will result in rock to reach their solidus. the lower mantle plumes rise into upper mantle causing oceanic/continental hotspots.
Partial Melting: Decrease the Pressure
lowers melting temp of rocks. rising ductile mantle material go through this.
Partial Melting: Add Fluids
H2O. lowers melting temp of rocks. fluid inclusions and hydrous minerals.
factors affecting newly formed melts
chemistry of the rock mass being melted
percent of rock mass melted
presence/absence of fluids, and type/amount of fluid
lithostatic pressure
How does the type of rock influence magma composition?
The type of rocks being melted influences the magma composition, as different minerals have varying melting temperatures and silica contents.
What are partial melts?
Partial melts are melts that are richer in silica than the original rock mass due to the melting of silica-rich minerals.
Which minerals are typically lower in melting temperature?
Silica-rich minerals like quartz and alkali feldspar have lower melting temperatures compared to silica-poor minerals such as olivine.
What do partial melts of the upper mantle commonly produce?
Partial melting of the upper mantle, which is ultramafic (olivine + pyroxene), commonly produces mafic melts.
What is produced from partial melting of lower continental crust?
Partial melting of lower continental crust, which is mafic (pyroxene + plagioclase), commonly produces intermediate-silica melts.
What does partial melting of middle-to-upper continental crust produce?
Partial melting of the middle-to-upper continental crust, which is intermediate-felsic (plagioclase, alkali feldspar, quartz), commonly produces felsic melts.
What is the rheological critical melt percentage (RCMP)?
The RCMP is the amount of melt (as a percentage of the rock melted) that needs to accumulate before it can migrate away.
How does mineral shape affect the RCMP?
The RCMP is lower if all minerals have spherical shapes but higher for a rock mass containing minerals with different crystal habits.
What role does viscosity play in partial melt migration?
Viscosity determines the percentage of partial melt needed for migration; more viscous melts require a larger percentage of partial melt to migrate.
How does the viscosity of felsic and mafic melts differ in terms of RCMP?
A larger percentage of partial melt is needed for viscous silica-rich (felsic) melts than for less viscous silica-poor (mafic) melts.
What effect does the addition of fluid have on melt viscosity and RCMP?
The addition of fluid lowers melt viscosity, which in turn decreases the RCMP.
What promotes melt separation?
Melt separation is promoted by:
a) Buoyancy: melt is less dense than unmelted rock and rises.
b) Compaction/filter-pressing: melt is squeezed upwards under lithostatic pressure.
c) Tectonic compression: melt is squeezed upwards.
d) Tectonic extension: creates fractures for melt to rise.
What is the typical RCMP range for mafic and felsic melts?
The RCMP typically ranges from 1-7% for mafic melts and 15-30% for felsic melts
What happens to the magma chamber roof when magma reaches shallow depths in the crust?
Magma can physically push up the roof rocks via folding or faulting due to magma buoyancy and/or overpressure when it is close to the surface.
What limits wall rock assimilation adjacent to a magma chamber?
Wall rock assimilation is limited because it consumes heat (heat of fusion) and magmas are not far above their solidus temperatures, leaving little excess heat.
What is stoping in relation to magma chambers?
Stoping is the process where blocks of brittle roof rock are broken off by rising magma. These blocks must be large and dense enough to sink through the magma, creating space for further magma rise.
What evidence supports the occurrence of stoping?
Petrographic evidence for stoping includes the presence of xenoliths, which are rocks not genetically related to the magma, surrounded by igneous rock crystallized from the magma.
How does ductile wall rock deformation occur?
If the surrounding rocks are ductile and denser than the magma, they flow downward, allowing magma to rise upward, which is efficient at deeper crustal and mantle depths.
What happens to wall rocks when magma is added to a magma chamber?
Adding magma increases pressure against the wall rocks, potentially causing faulting of brittle rocks or folding of more ductile rocks to accommodate radial expansion of the magma chamber.
How does tectonic extension facilitate magma movement?
Tectonic extension opens fractures to create space for magma but is constrained by the rate of plate tectonic extension, typically a few to several cm/year.
What is diapirism?
Diapirism is the process by which less dense magma rises through more dense surrounding rock, causing the rock to flow downward, typically in a ductile manner.
How does diapirism contribute to the formation of magma chambers?
Diapirism allows large magma bodies to form by enabling magma to rise and displace surrounding rock, leading to the development of magma chambers.
In what type of rock does diapirism occur effectively?
Diapirism occurs effectively in ductile rocks where the surrounding material can flow downward; it is less effective in the brittle upper crust.
What is the result of reduced density of magma during diapirism?
The reduced density of magma during diapirism enables it to rise through surrounding rock until it reaches a point of similar density, promoting lateral spreading.
What mechanisms may assist diapirism in magma chamber formation in the brittle upper crust?
In the brittle upper crust, other mechanisms such as faulting and folding help create space for magma as diapirism is limited.
How is heat transferred in molten magma?
Heat is transferred via convection in less viscous, largely molten magma.
How does heat transfer occur in highly viscous magma?
In highly viscous, largely crystallized magma, heat is transferred via conduction.
What is the main mechanism of heat loss from lava flows once a solid crust forms?
Once a solid crust forms on lava flows, conduction becomes the main mechanism of heat loss.
What does Fourier's Law describe in the context of heat transfer?
Fourier's Law describes the relationship between heat flux, thermal conductivity, and thermal gradient: Heat Flux = (Thermal conductivity)(Thermal Gradient).
What is the thermal conductivity range for silicate rocks?
Thermal conductivities for silicate rocks are low, about 2 to 3 W / [m . °C].
What happens to the temperature of surrounding rock after magma intrusion?
Upon magma intrusion, the temperature of adjacent country rocks rises to about half of the initial magma temperature (T / T0 = 0.5).
What happens to the temperature gradient between magma and rock over time?
Initially steep, the temperature gradient flattens over time as heat conducts further into the country rock and the magma cools.
How does the rate of magma cooling change over time?
The rate of magma cooling slows at a rate proportional to the square root of time.
How does latent heat of crystallization affect magma cooling?
Latent heat of crystallization causes magma crystallization to take longer than expected, around three times longer than predicted by Fourier’s Law.
What are the distances reached by the solidification front after 1, 10, 100, and 1000 years?
The solidification front reaches 7.66 m after 1 year, 24.3 m after 10 years, 76.6 m after 100 years, and 243 m after 1000 years.
How long can large magma chambers take to cool and crystallize?
Large magma chambers within the Earth’s crust can take tens to hundreds of thousands of years to completely cool and crystallize.
What is the effect of fast-cooling lavas on magmatic differentiation?
Fast-cooling lavas on Earth’s surface allow considerably less time for magmatic differentiation, leading to less diversity of magma and igneous rock types.
What did Bowen's reaction series help to illustrate?
Bowen's reaction series illustrates the crystallization behavior of minerals from a melt, highlighting continuous and discontinuous series of mineral formation as temperature decreases.
What are the two types of reactions observed in Bowen’s reaction series?
The two types of reactions are a continuous series involving plagioclase (more Ca-rich crystallizing first, followed by more Na-rich) and a discontinuous series involving mafic minerals reacting with the remaining melt.
What does Bowen's reaction series assume about the melt and crystallized minerals?
Bowen's reaction series assumes chemical equilibrium between the remaining melt and the minerals that have crystallized.
Why is Bowen's reaction series considered an oversimplification?
Bowen's series is considered an oversimplification because it does not describe the crystallization sequence for all magmas, as it varies based on starting melt composition, lithostatic pressure, and other differentiation processes.
What factors affect the sequence of mineral crystallization from magma?
Factors affecting the sequence of mineral crystallization from magma include starting melt composition, lithostatic pressure, and various magmatic differentiation processes.
What is Stoke's Law used for in geology?
Stoke's Law is used to determine the settling velocity of crystals through magma.
What is the formula for Stoke's Law?
The formula for Stoke's Law is V = (2/9) * (r² * (ps - pl) * g) / η, where V is the settling velocity.
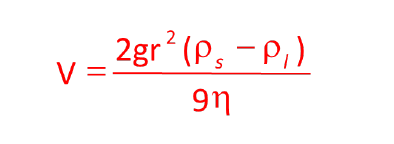
What does 'V' represent in Stoke's Law?
In Stoke's Law, 'V' represents the settling velocity of the crystal (cm/s).
What does the variable 'g' represent in Stoke's Law?
In Stoke's Law, 'g' represents the acceleration due to gravity, which is approximately 980 cm/s².
What does 'r' stand for in the context of Stoke's Law?
In Stoke's Law, 'r' stands for the radius of a spherical particle (cm).
What do 'ps' and 'pl' represent in Stoke's Law?
In Stoke's Law, 'ps' is the density of the solid spherical particle (g/cm³) and 'pl' is the density of the liquid melt (g/cm³).
What does 'η' represent in Stoke's Law?
In Stoke's Law, 'η' represents the viscosity of the liquid melt, measured in poise (1 g/[cm sec] = 1 poise).
What are solid solutions in the context of minerals?
Solid solutions are minerals that can have a range of compositions, characterized by high-temperature and low-temperature endmembers (e.g., Mg-rich and Fe-rich for mafic minerals; Ca-rich and Na-rich for plagioclase).
What is crystal zoning in minerals?
Crystal zoning is a variation in composition within a mineral crystal that occurs when equilibrium is not maintained during cooling of the magma.
What influences the development of crystal zoning?
Crystal zoning develops favorably at intermediate cooling rates; slow cooling allows for equilibrium exchange, while rapid cooling freezes the melt quickly, preventing zoning.
How can crystal zoning be observed?
Crystal zoning can be observed under a petrographic microscope as changes in color, extinction angle, or birefringence across the crystal.
What is an example of a mineral that exhibits crystal zoning?
Plagioclase is a common mineral that exhibits crystal zoning, observable as concentric bands of varying brightness in cross-polarized light.
What is normal zoning in plagioclase?
Normal zoning in plagioclase is characterized by a trend from a Ca-rich core to a Na-rich rim due to coupled substitution requiring charge balance during crystallization.
Why is coupled substitution in plagioclase difficult?
Coupled substitution in plagioclase is difficult because it involves breaking strong Si–O and Al–O bonds and the slow diffusion of highly charged Al³⁺ and Si⁴⁺ cations in the melt.
What occurs during equilibrium chemical exchange as magma cools?
If equilibrium chemical exchange is maintained as magma cools, the solid solution mineral will have a homogeneous composition that shifts towards the low-temperature endmember over time.
What does liquid immiscibility refer to in magmatic systems?
Liquid immiscibility refers to the phenomenon where melts of different compositions do not mix and separate based on their densities, similar to oil and water.
What is an example of liquid immiscibility involving ultramafic and felsic melts?
In an Fe-rich mafic magma, a less dense felsic melt (>75% SiO2) may separate from a denser ultramafic melt (~40% SiO2), though this separation may be impeded by high viscosity and density of previously formed crystals.
What is required for sulfide saturation in a mafic or ultramafic melt?
Less than 0.1 wt% sulfur is required to achieve sulfide saturation in a mafic or ultramafic melt, leading to the separation of a denser sulfide melt.
What happens to the sulfide melt in a magma chamber?
The denser sulfide melt sinks to the bottom of the magma chamber, undergoing fractional crystallization to produce economically important sulfide minerals.
What types of elements are concentrated by sulfide melts?
Sulfide melts can produce economically significant concentrations of chalcophile elements, such as copper, nickel, and platinum group elements (PGEs).
What occurs in an alkali-rich and CO2-rich magmatic system regarding liquid immiscibility?
In this system, a denser alkaline silicate melt can separate from a less dense carbonate-rich melt, leading to the crystallization of alkaline igneous rocks and carbonatite rocks.
How does the density of melts affect their behavior in magma?
The behavior of melts in magma is influenced by their density; denser melts tend to sink, while less dense melts rise, leading to separation and compositional diversity in magmatic systems.
What does fluid saturation in magma refer to?
Fluid saturation in magma occurs when it holds the maximum amount of dissolved fluid, resulting in the exsolution of a free fluid phase.
How can magmatic heat lead to fluid saturation?
Magmatic heat can cause melting or dehydration of hydrous minerals in surrounding rocks, allowing water to dissolve into the magma until it becomes fluid-saturated.