macronutrients exam 2
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
insulin summary
-released by pancreatic b cells
-fed state
-lower blood glucose
-insulin signals GLUT4 in skeletal muscle and adipose tissue
glucagon summary
-released by pancreatic alpha cells
-fasting state
-increases blood glucose
-binds to glucagon receptors (G-protein coupled)
epinephrine
-secreted from adrenal glands
-catecholamine
-released in "flight or fight" conditions
-released in response to vigorous exercise (considered a stress)
role in glucose metabolism
-stimulates breakdown of glycogen (liver and skeletal muscle)
-increases glycolysis (skeletal muscle)
-binds to adrenergic receptors- G protein coupled receptors
GLUT4 and exercise
contraction of muscle contraction acts as another trigger for GLUT4 to get to membrane
-why diabetes should exercise
-naturally "lowers" blood sugar
cortisol
-secreted from adrenal glands
-glucocorticoid (from cholesterol)
-stress hormone
role in glucose metabolism
-glycogen breakdown (liver and skeletal muscle)
-gluconeogenesis (liver)
-also stimulates protein breakdown in skeletal muscle
-secreted during prolonged fasting, vigorous exercise, illness, injury
-binds to intracellular receptors- need protein in blood to get into cell
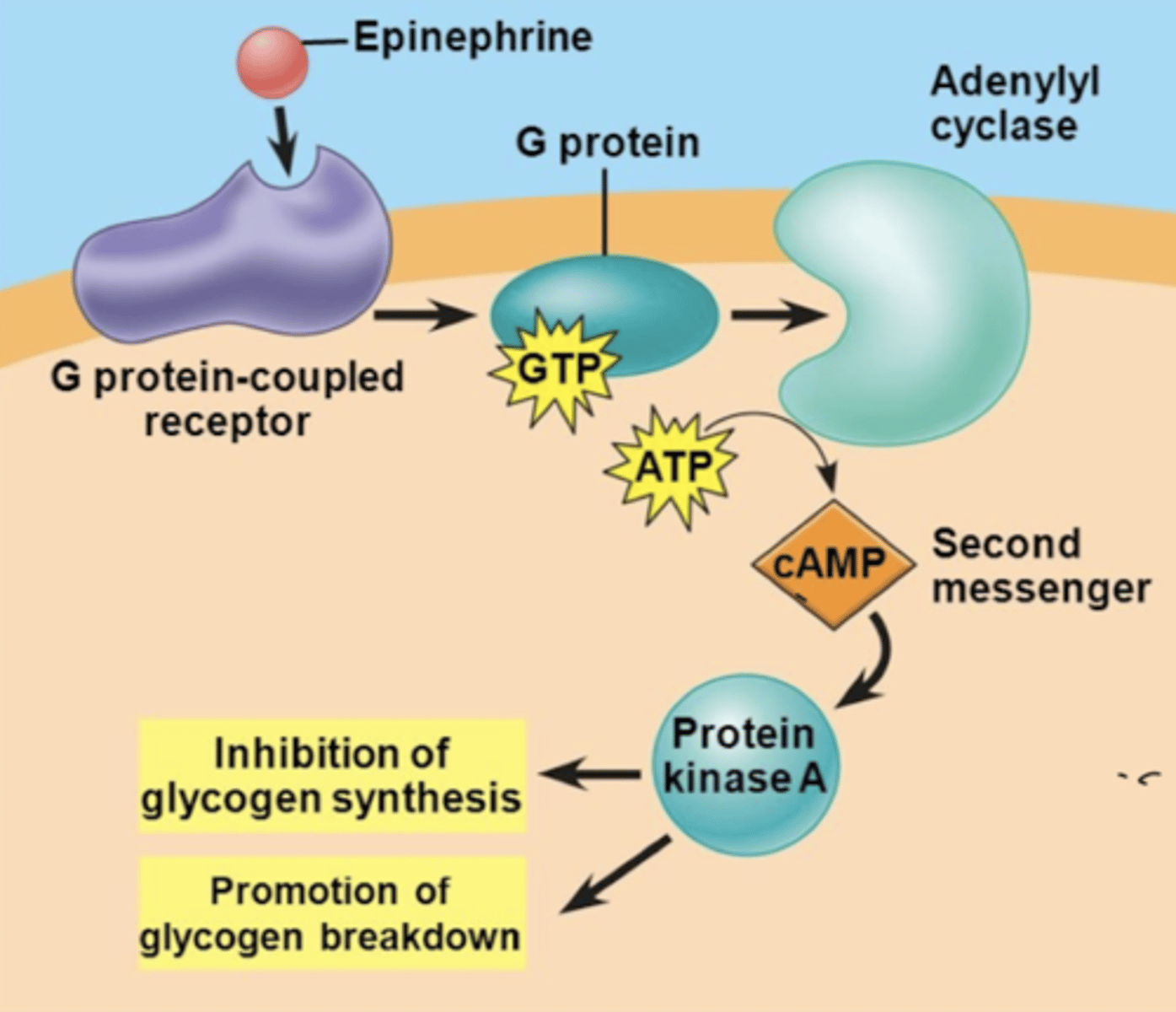
G-protein reaction
cascade reaction used by glucagon and epinephrine
-hormone binds to G protein
-G protein binds to adenylyl cyclase (triggers conversion of ATP into cAMP)
-cAMP acts as a SECOND messenger and activates protein kinase A
-protein kinase A is directly involved in regulating enzymes in metabolic pathway
*inhibits glycogen synthesis
*promotes glycogen breakdown
kinase- phosphorylates
-glycogen phosphorylase- breaks down glycogen
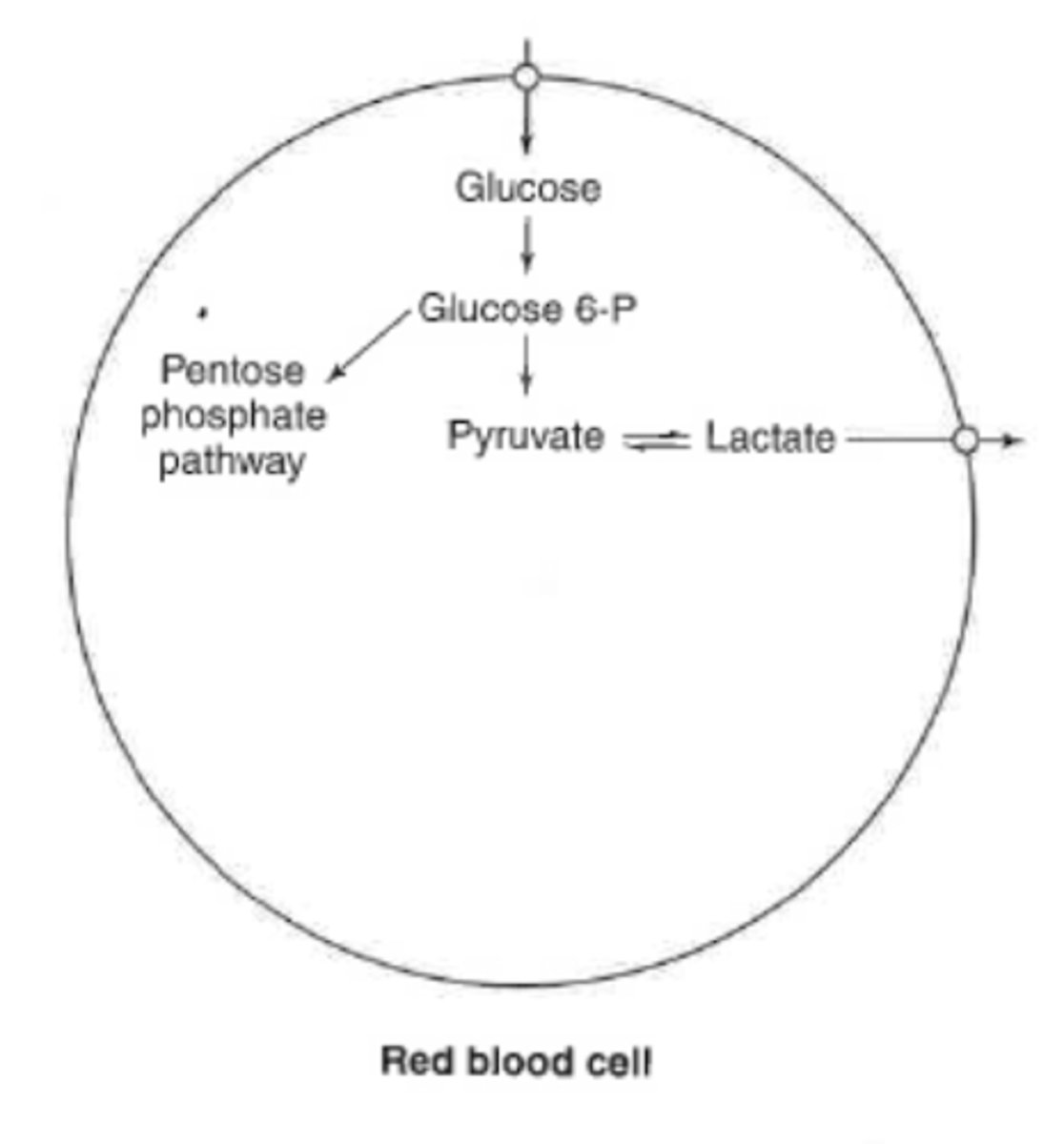
glucose metabolism in RBC
limited capacity
-anaerobic metabolism (pyruvate to lactic acid)
-lactic acid leaves RBC and goes to liver
lack mitochondria
-can not do aerobic
oxidative branch of PPP
-protection of RBC
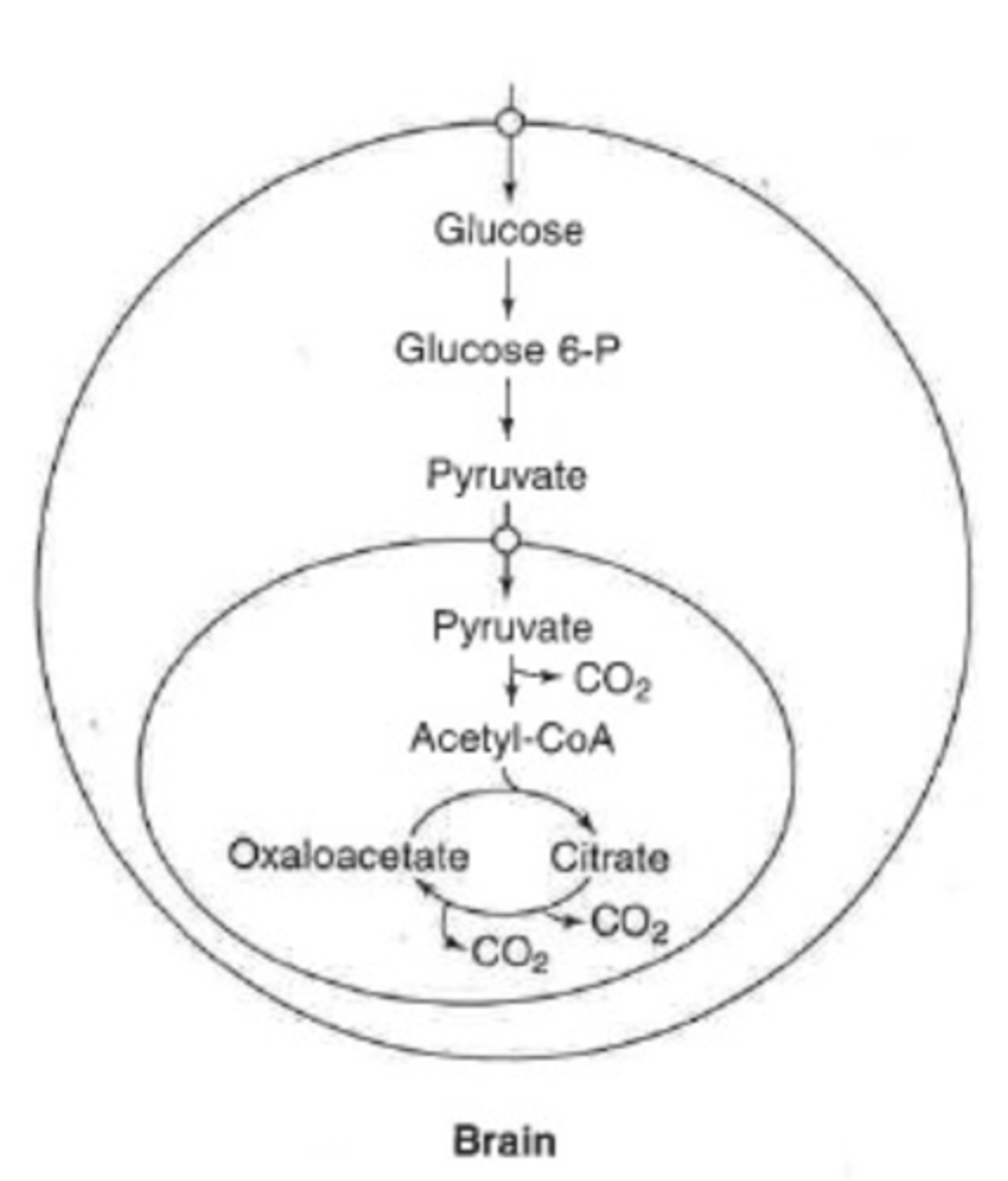
glucose metabolism in brain
can completely oxidize glucose
-aerobic metabolism
-TCA and ETS (greater than 90% of time)
completely dependent on glucose for energy
-even under times of starvation still requiring glucose (50%)
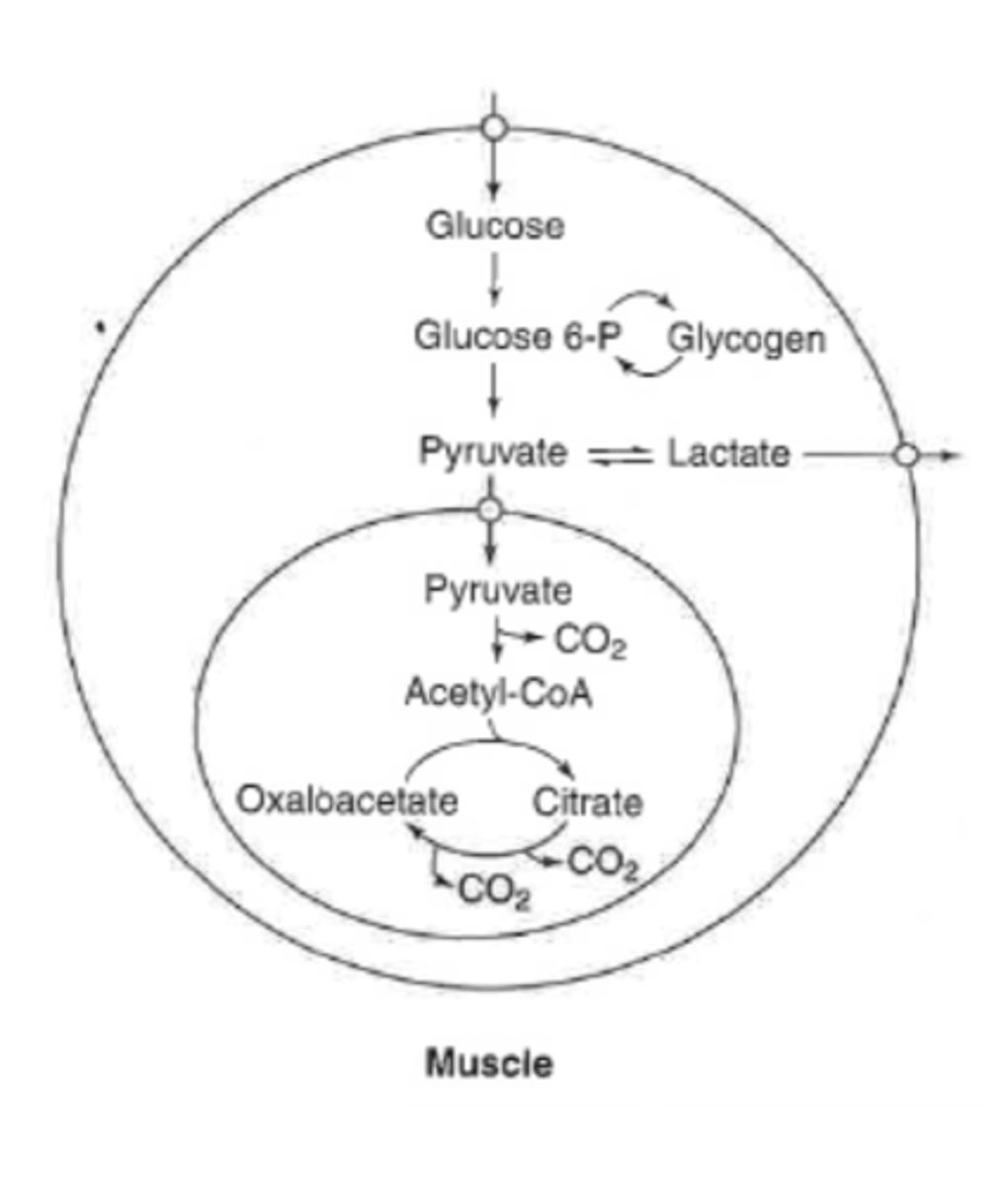
glucose metabolism in muscle
can completely of incompletely oxidize glucose
-anaerobic in times of high demand exercise (faster)
can store glucose as glycogen
-one of primary stores of glycogen
can also do PPP but does not do a lot
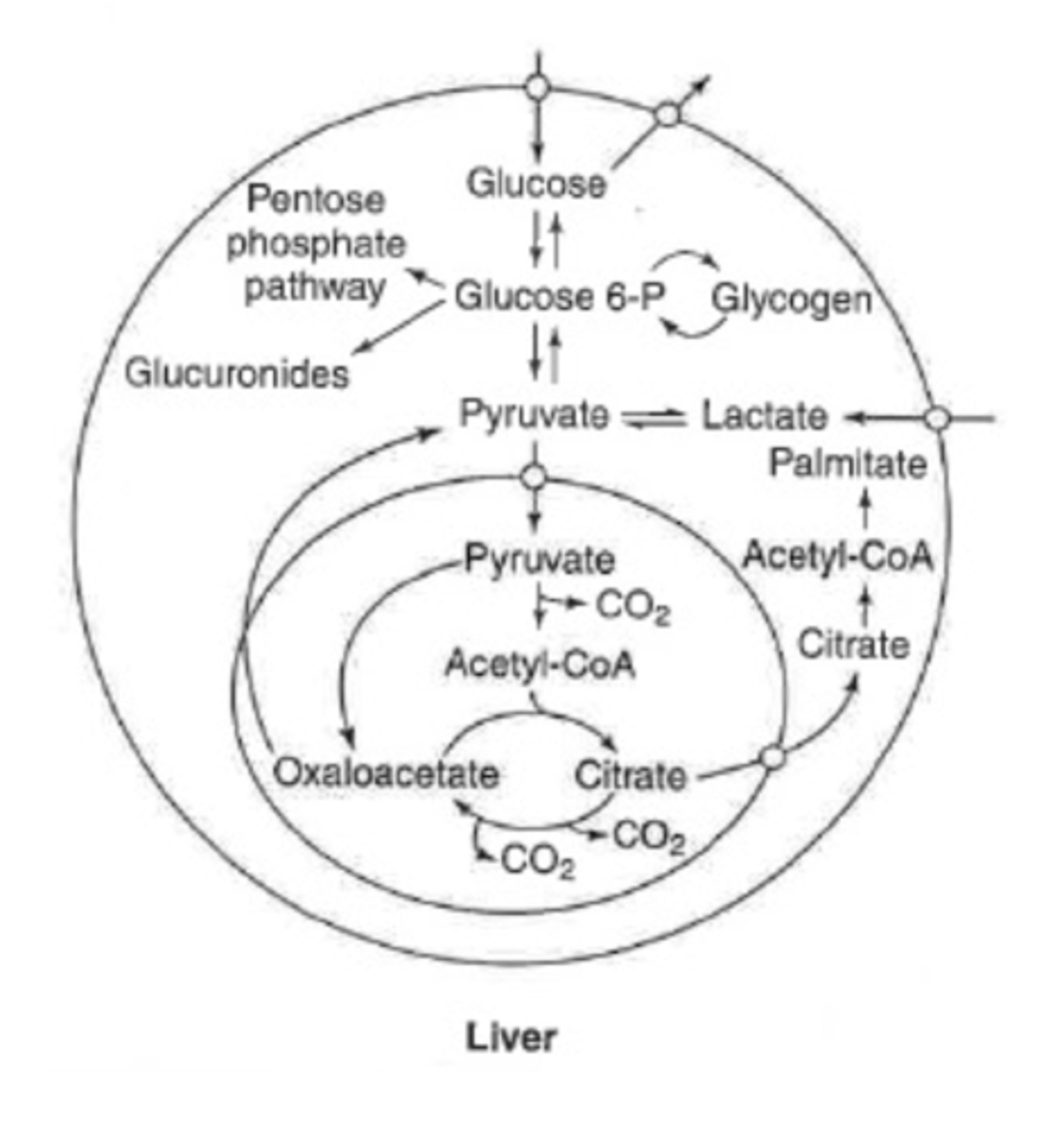
glucose metabolism in liver
can be completely oxidized
can store as glycogen
can convert to fatty acids or amino acids
can make glucose
(glycolysis or gluconeogenesis)
oxidative branch of PPP
glucose metabolism in adipocyte
glucose partially metabolized for fat synthesis
-get glucose from GLUT4 after meal
glycolysis
PPP
-lipid formation
*main reason adipocytes wants glucose is to STORE FAT
glycolysis
metabolism of glucose to pyruvate
-occurs in cytosol
-can proceed via anaerobic or aerobic metabolism
-generates ATP and NADH
-anaerobic glycolysis produces less ATP
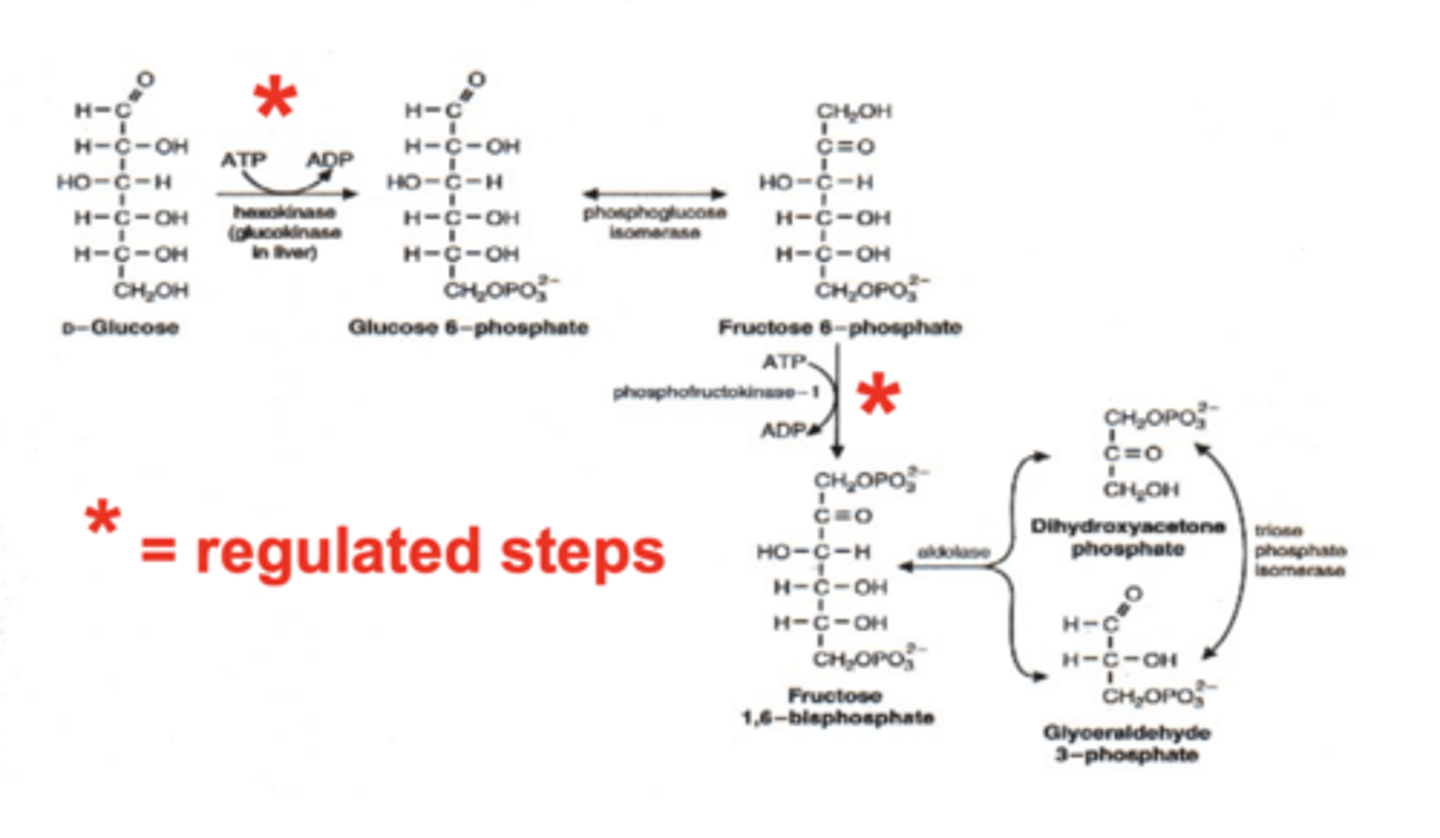
phase 1 glycolysis
priming of glucose
-requires ATP
regulated steps:
-hexokinase or glucokinase
-phosphofructokinase-1
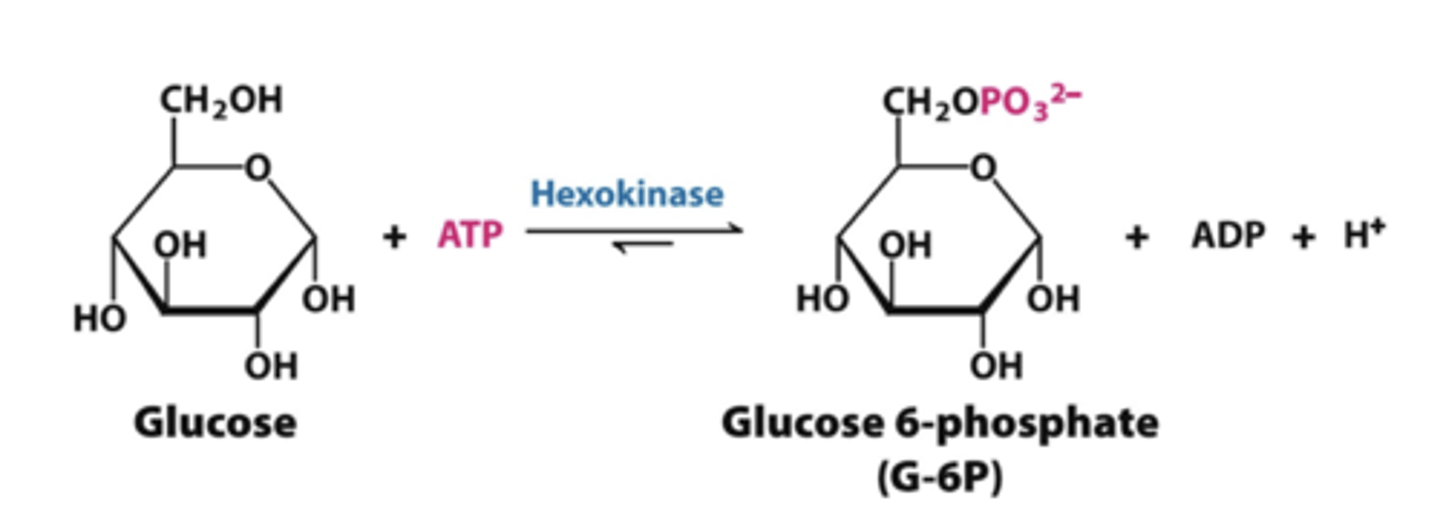
step 1 glycolysis
hexokinase or glucokinase
-glucose to glucose 6 phosphate
*have to put in ATP
REGULATED
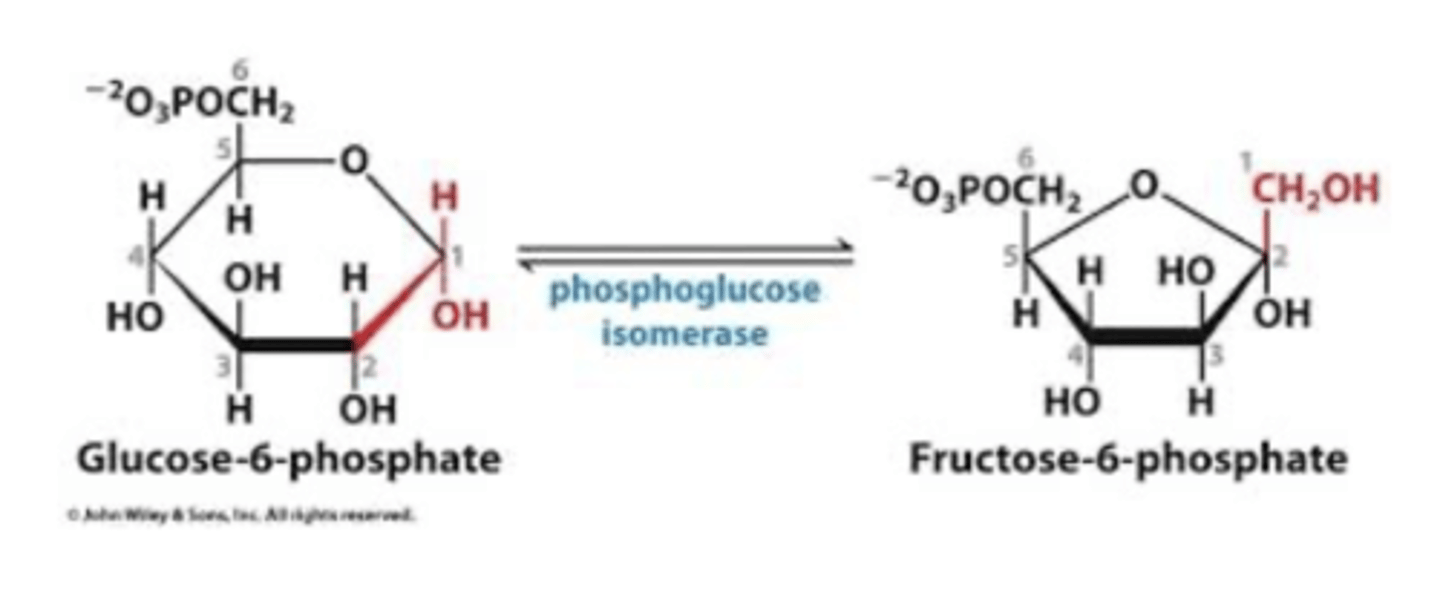
step 2 glycolysis
phosphoglucose isomerase
-glucose 6 phosphate to fructose 6 phosphate
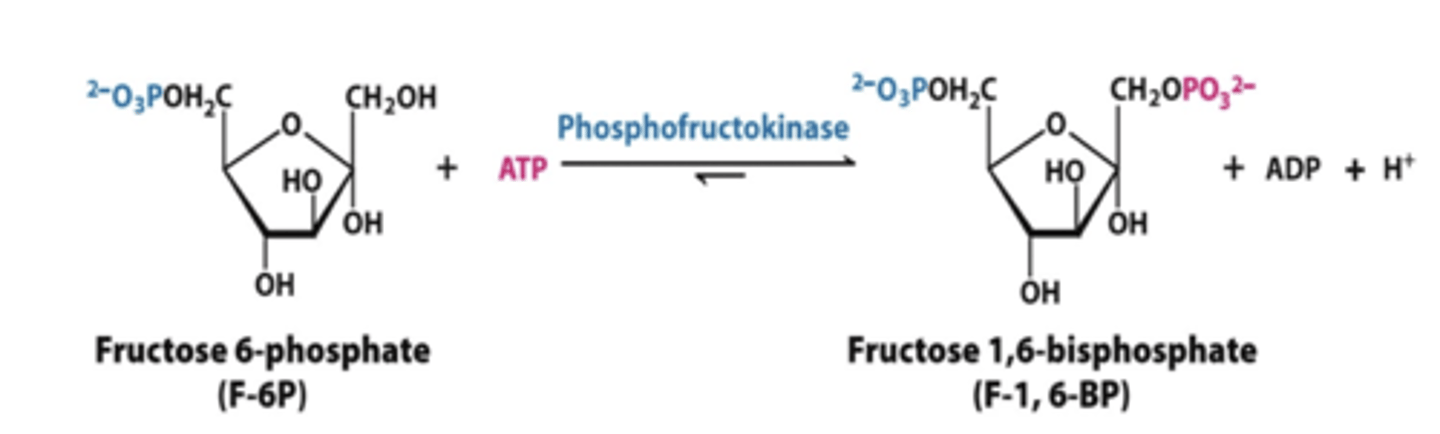
step 3 glycolysis
phosphofructokinase-1
-fructose 6 phosphate to fructose 1,6 bisphosphate
*have to put in ATP
REGULATED
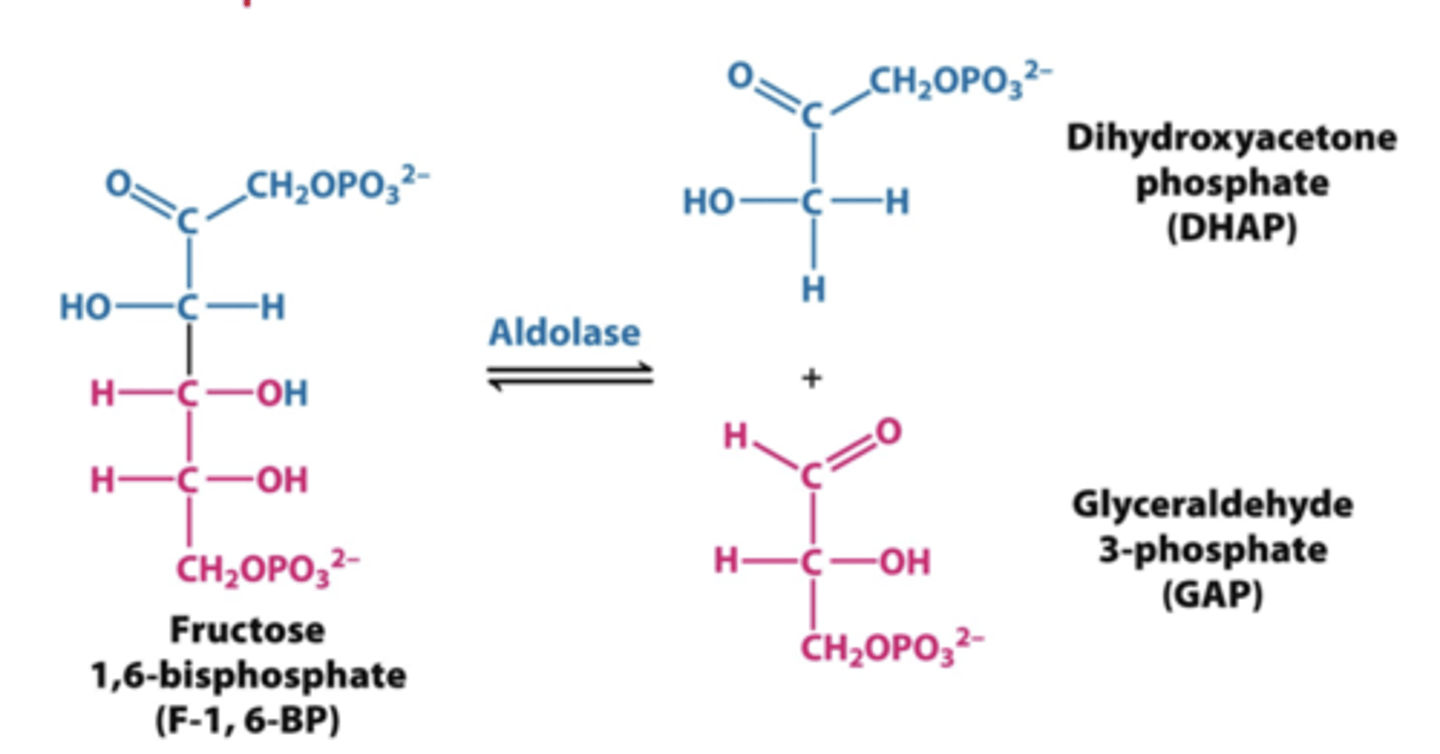
step 4 glycolysis
aldolase
-splits fructose 1,6 bisphosphate into two molecules
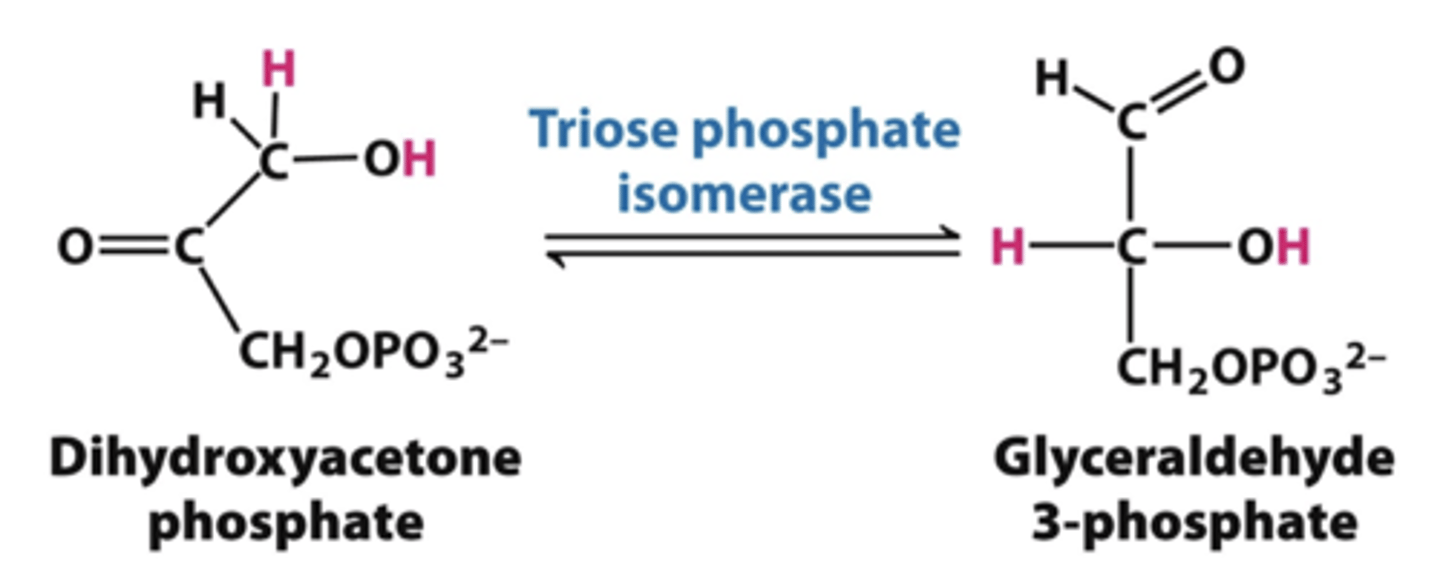
step 5 glycolysis
triose phosphate isomerase
-splits dihydroxyacetone phosphate into glyceraldehyde 3 phosphate
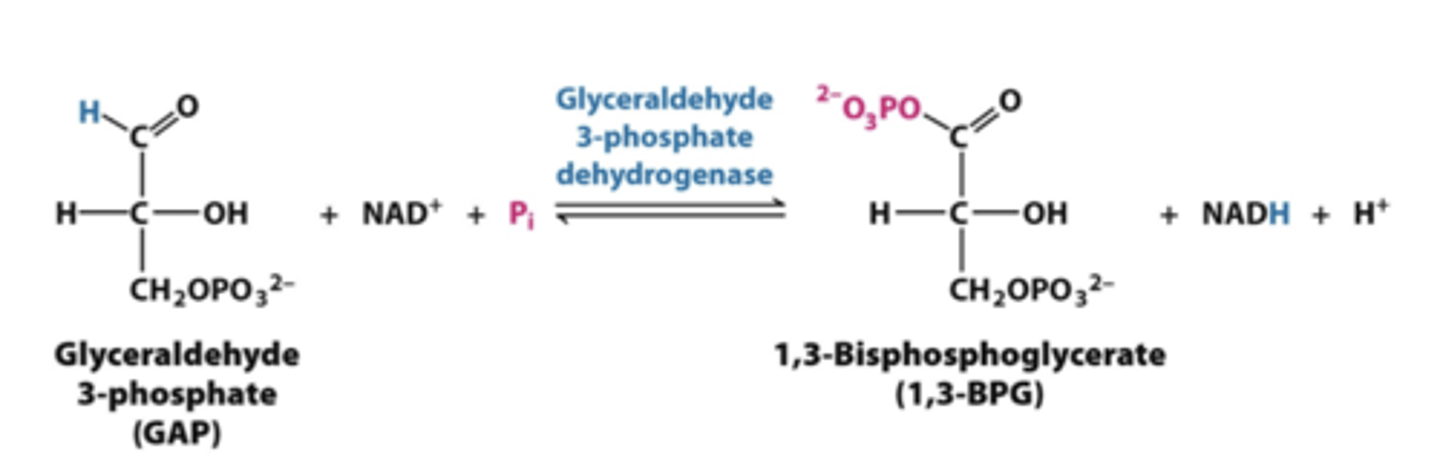
step 6 glycolysis
glyceraldehyde 3 phosphate dehydrogenase
-glyceraldehyde 3 phosphate into 1,3 bisphosphglycerate
*produces NADH
REGULATED
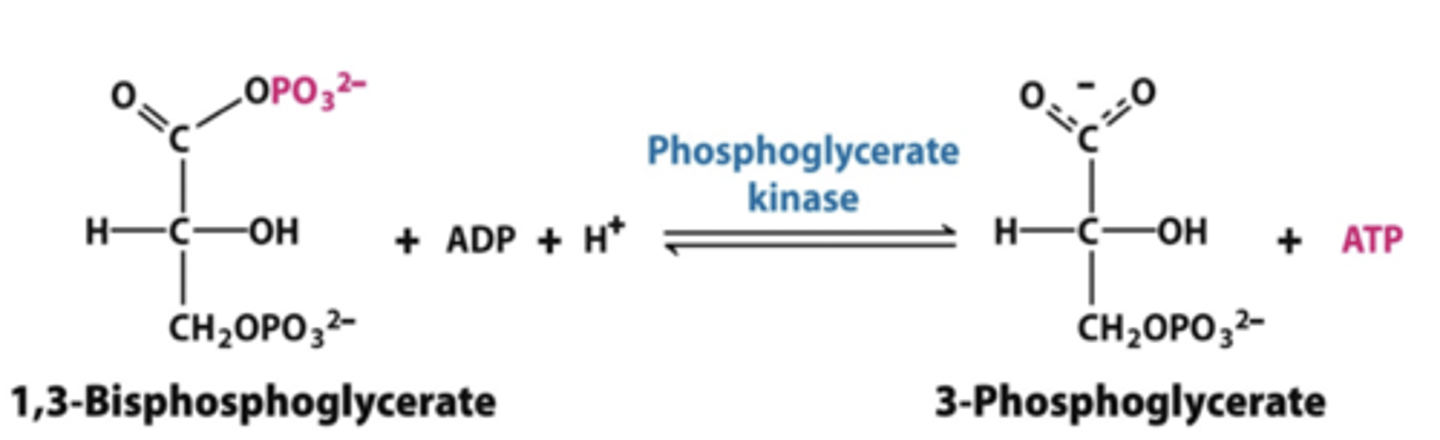
step 7 glycolysis
phosphoglycerate kinase
-1,3 bisphosphoglycerate to 3-phosphoglycerate
*produces ATP
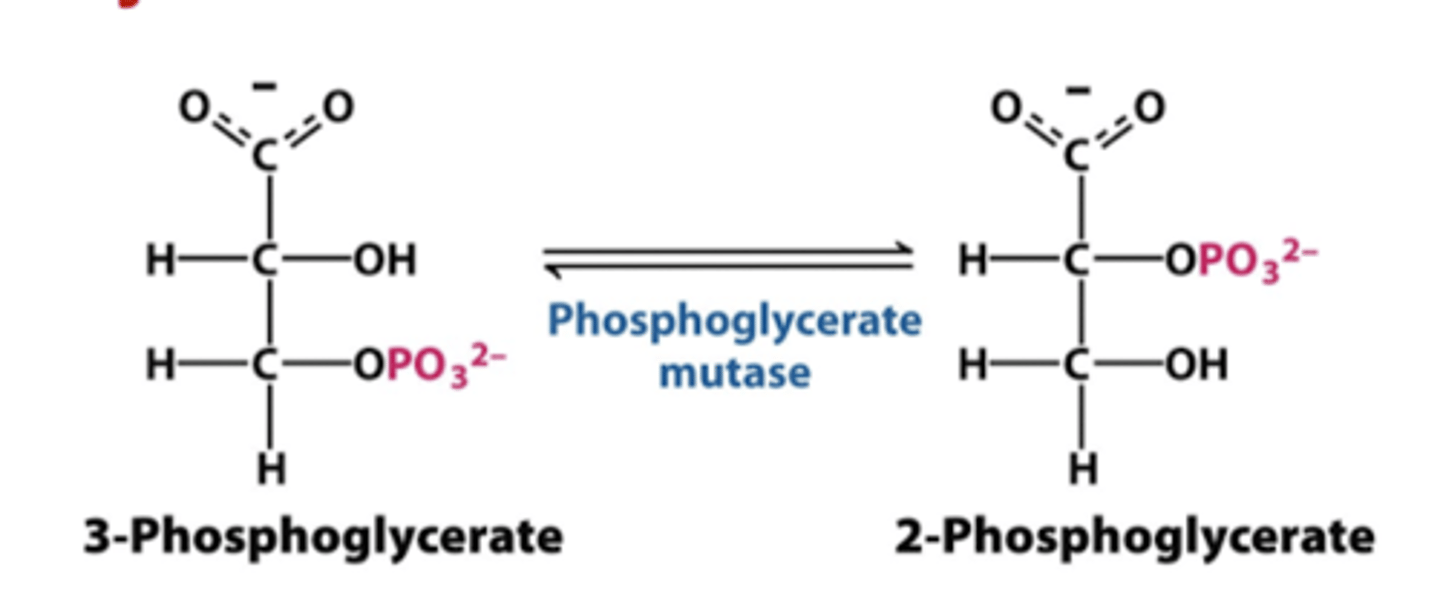
step 8 glycolysis
phosphoglycerate mutase
-3 phosphoglycerate into 2 phosphoglycerate
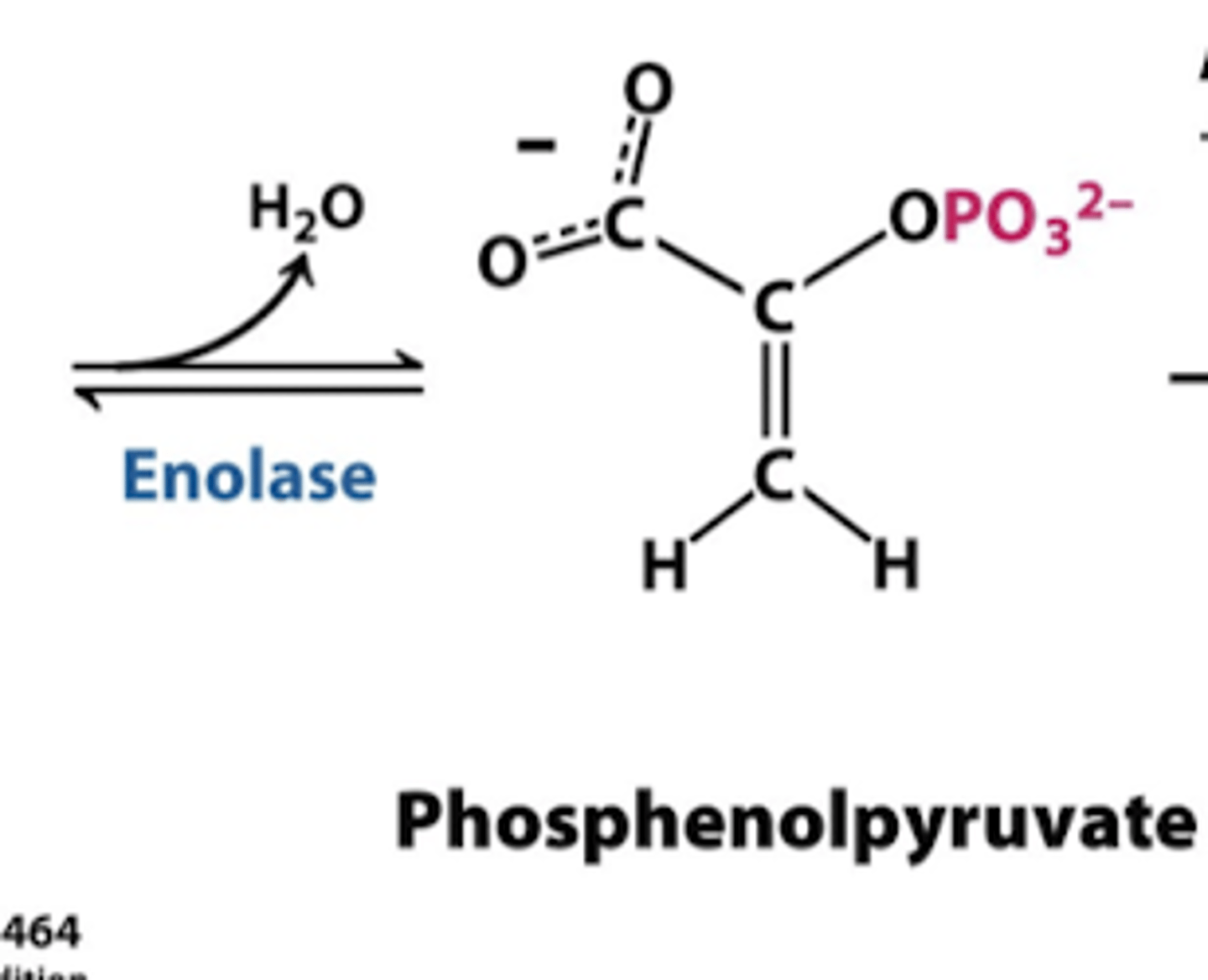
step 9 glycolysis
enolase
-takes off water
-2 phosphoglycerate into phosphenolpyurvate
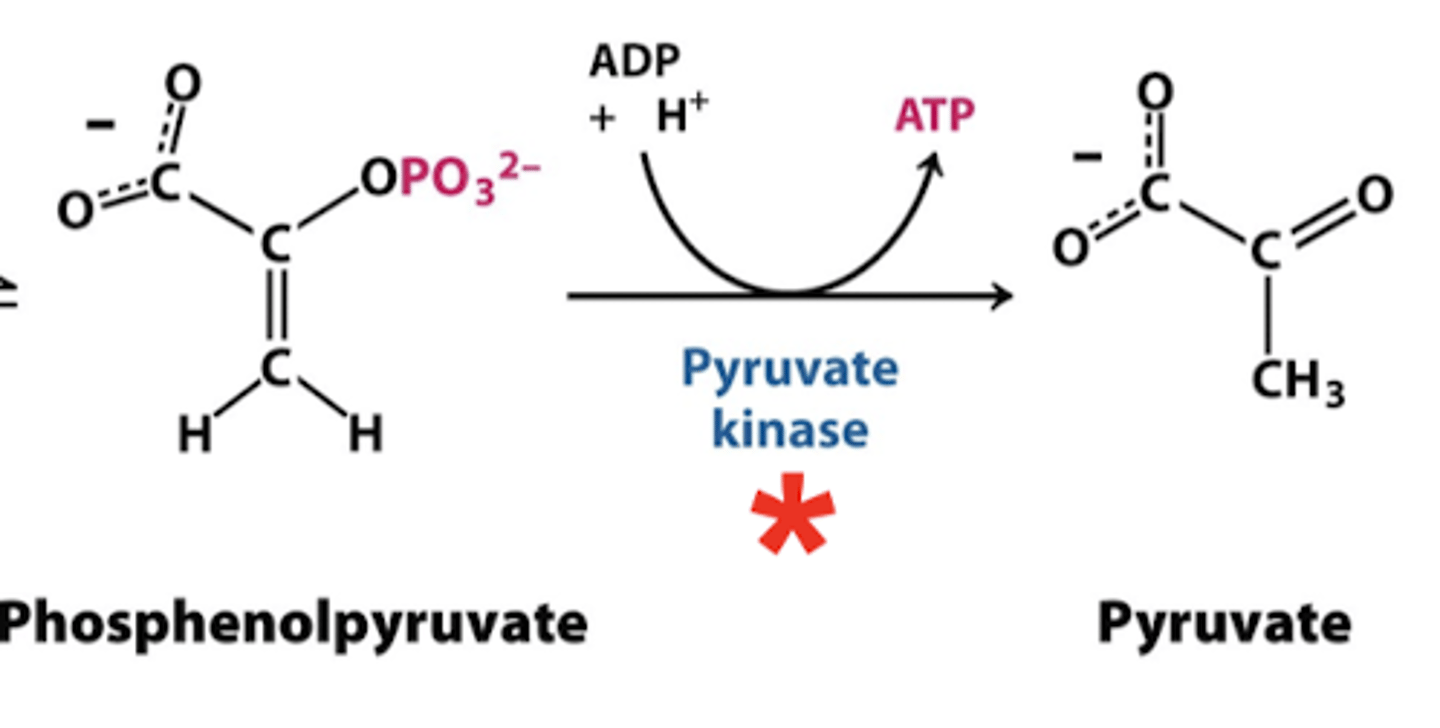
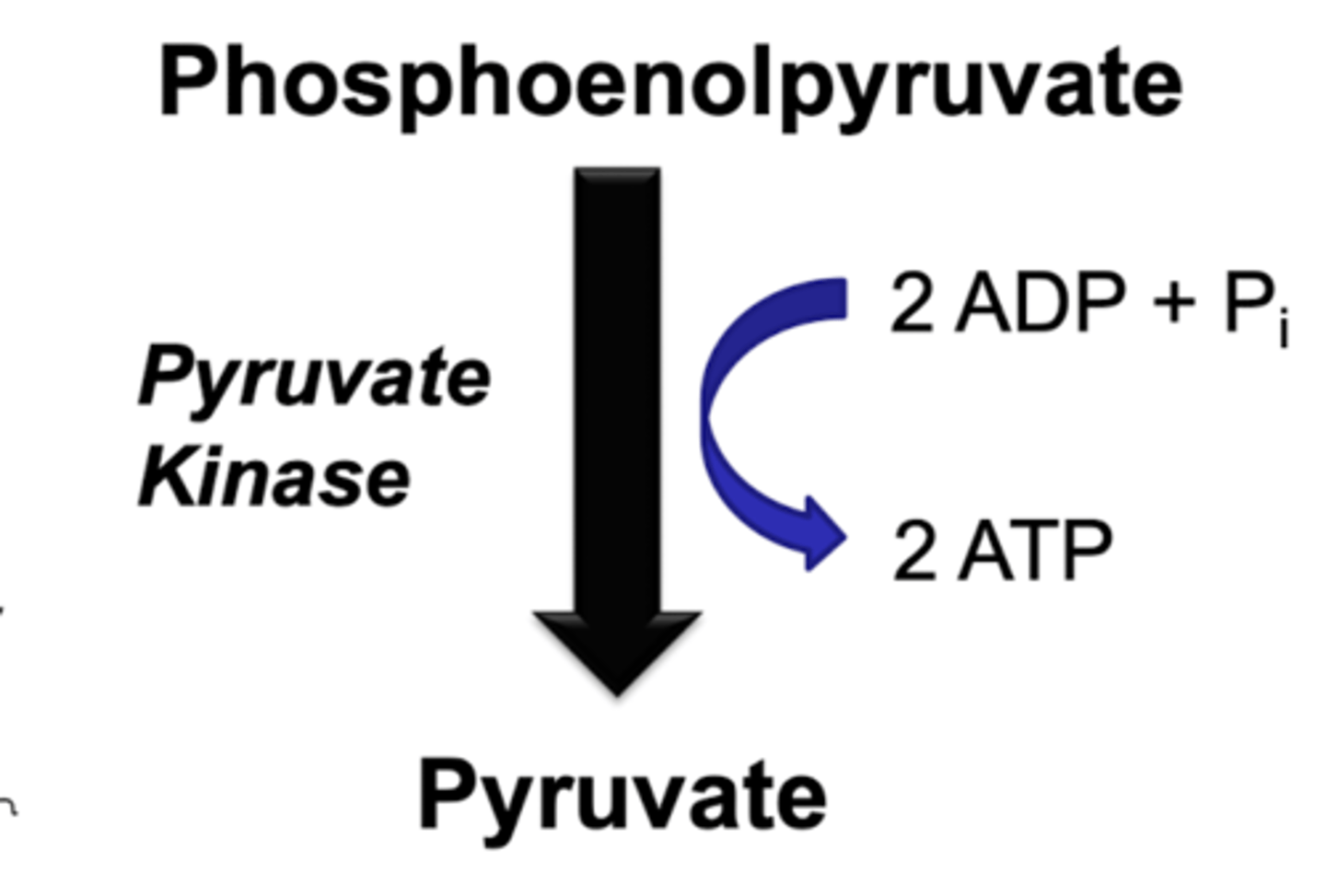
step 10 glycolysis
pyruvate kinase
-phosphenolpyruvate into pyruvate
*produces ATP
REGULATED
glycolysis net reaction
IN: glucose + 2NAD+ + 2ADP + 2Pi
OUT: 2 pyruvate + 2 NADH + 2 ATP + 2 H2O
regulation of metabolic enzymes
modulation of allosteric enzymes
-do NOT bind to active site but rather inhibit or promote binding to active site
*NADH/NAD+ ratio (high- INHIBIT)
*energy status (high ATP INHIBIT)
*other modulators
FAST ACTING
hormonal regulation
*covalent modification (phosphorylation or dephosphorylation)
*induction or genetic regulation (hormone causes gene trancription and changes in gene expression)
LONG-TERM and SLOW
directional shifts in reversible reactions
*changes in reactant or product concentrations (feedback inhibition)
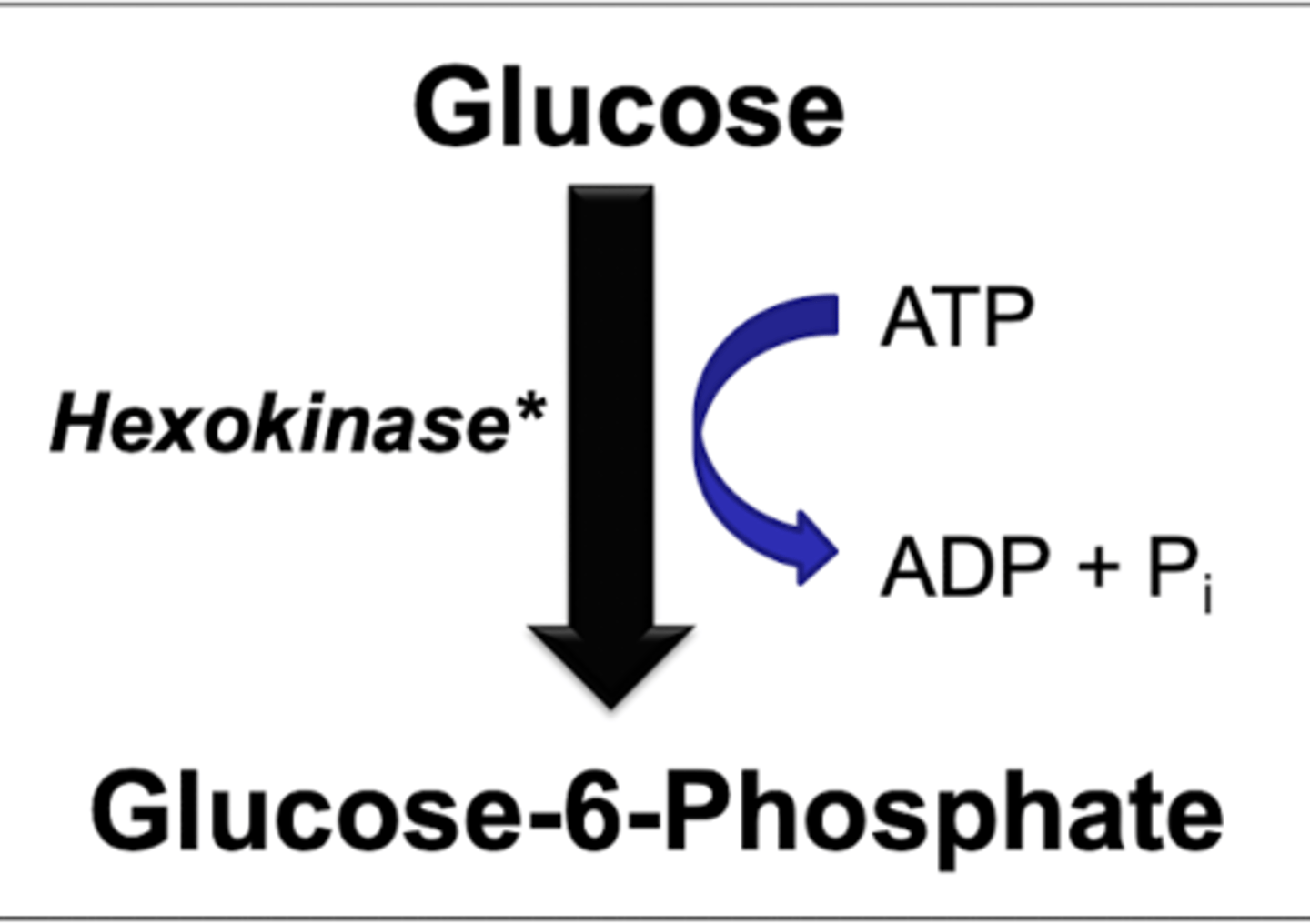
hexokinase
first step if in skeletal muscle
-phosphate traps molecule in skeletal muscle since it can not reverse reaction
allosteric regulation
-high concentrations of glucose 6 phosphate INHIBITS enzyme
-low km and low capacity- does not take much to activate
*negative feedback loop
RAPID conversion
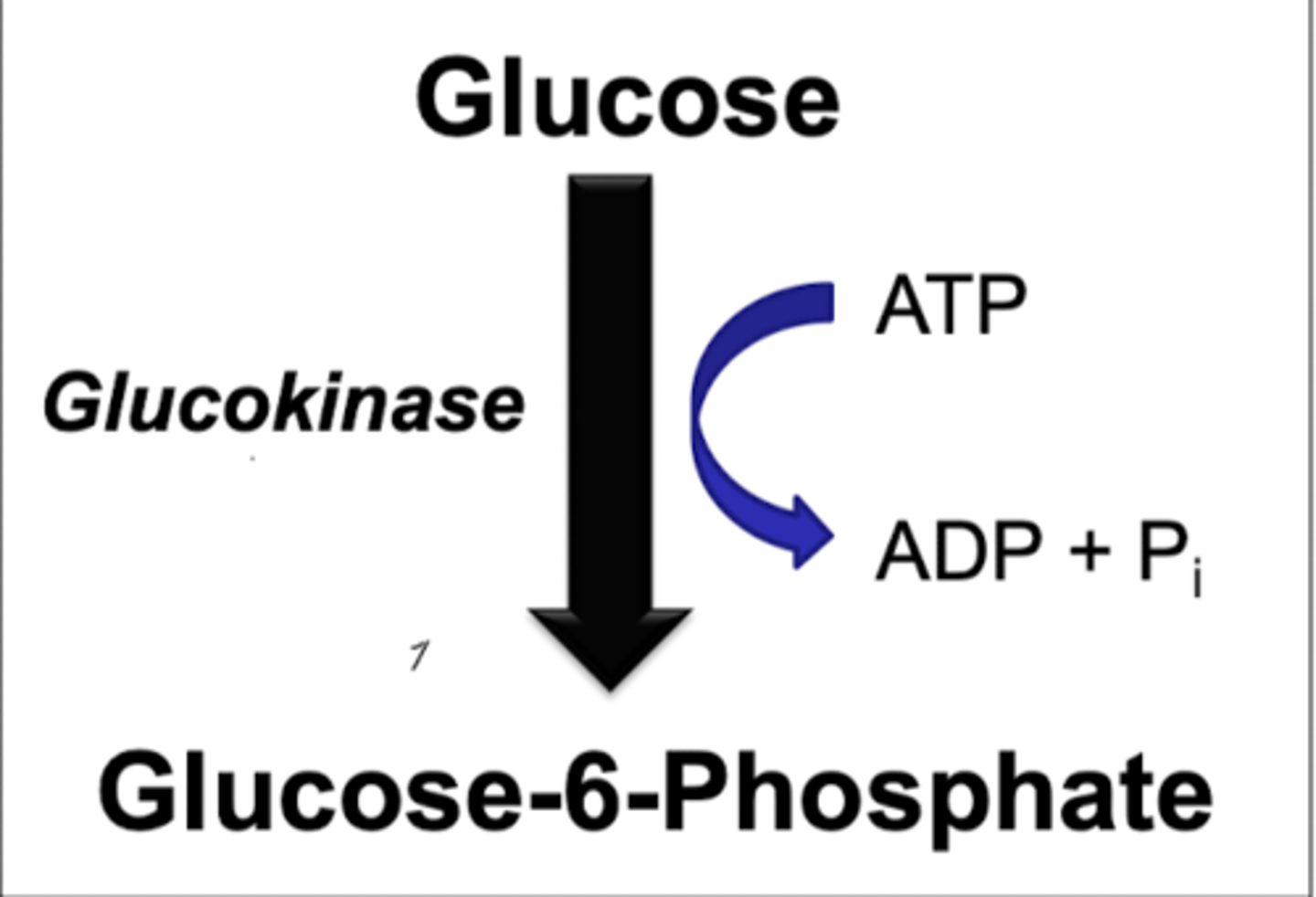
glucokinase
first step if in liver
induction
-primarily changed by the entry of glucose into the cell
-high km and high capacity- only functional when high amounts of glucose entering the liver (after a meal)
-insulin PROMOTES activity and glucagon DECREASES but not the main activity
-glucose-6-phosphate very important in liver- can be used to make glycogen stores
GLUT2 function
need conversion of glucose into glucose-6-phosphate
-need constant stream of glucose into the liver after a meal
-if glucokinase was allosteric it would stop GLUT2 from functioning and would have more sugar in blood
diabetics and glucokinase
activity of glucokinase slows down or completely stops
-signal for insulin not working- part of the way we change induction
MODY
maturity onset of diabetes in the young
-develop severe resistance to insulin
-growth issues
hexokinase summary
-located in muscle, brain, and adipose tissue
-allosterically inhibited by glucose-6-phosphate
-low km: function at maximum velocity at fasting blood glucose concentrations
-not induced by insulin in normal individuals
-not induced by insulin in insulin resistant individuals
glucokinase summary
-located in liver and pancreas
-not inhibited by glucose-6-phosphate
-high km: function at maximum velocity only when glucose levels are high
-induced by insulin in normal individuals
-not induced by insulin in insulin resistant individuals
exercise and glycogen stores
depletes glycogen stores
-helps to promote glucokinase activity
-keeps blood sugars low
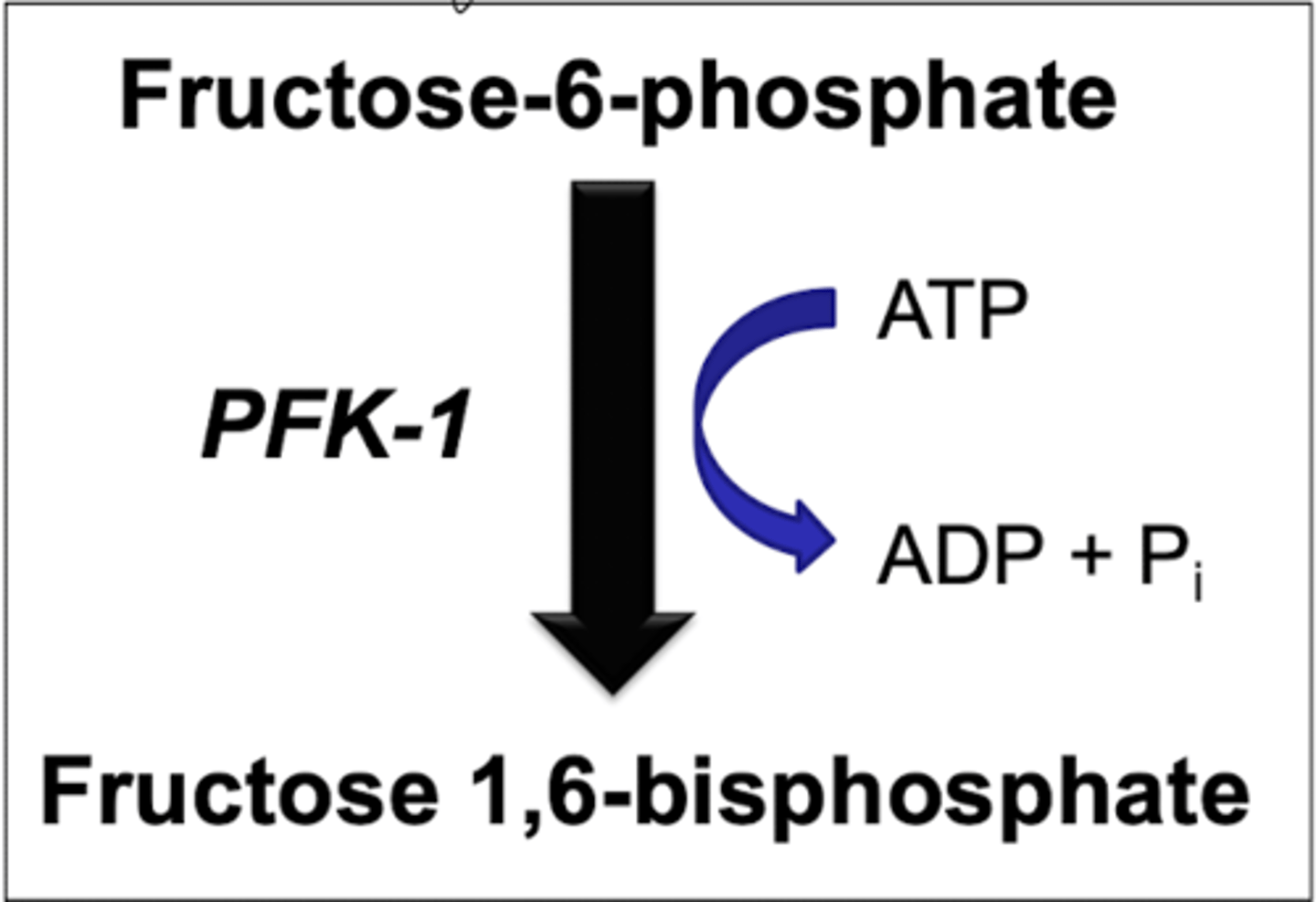
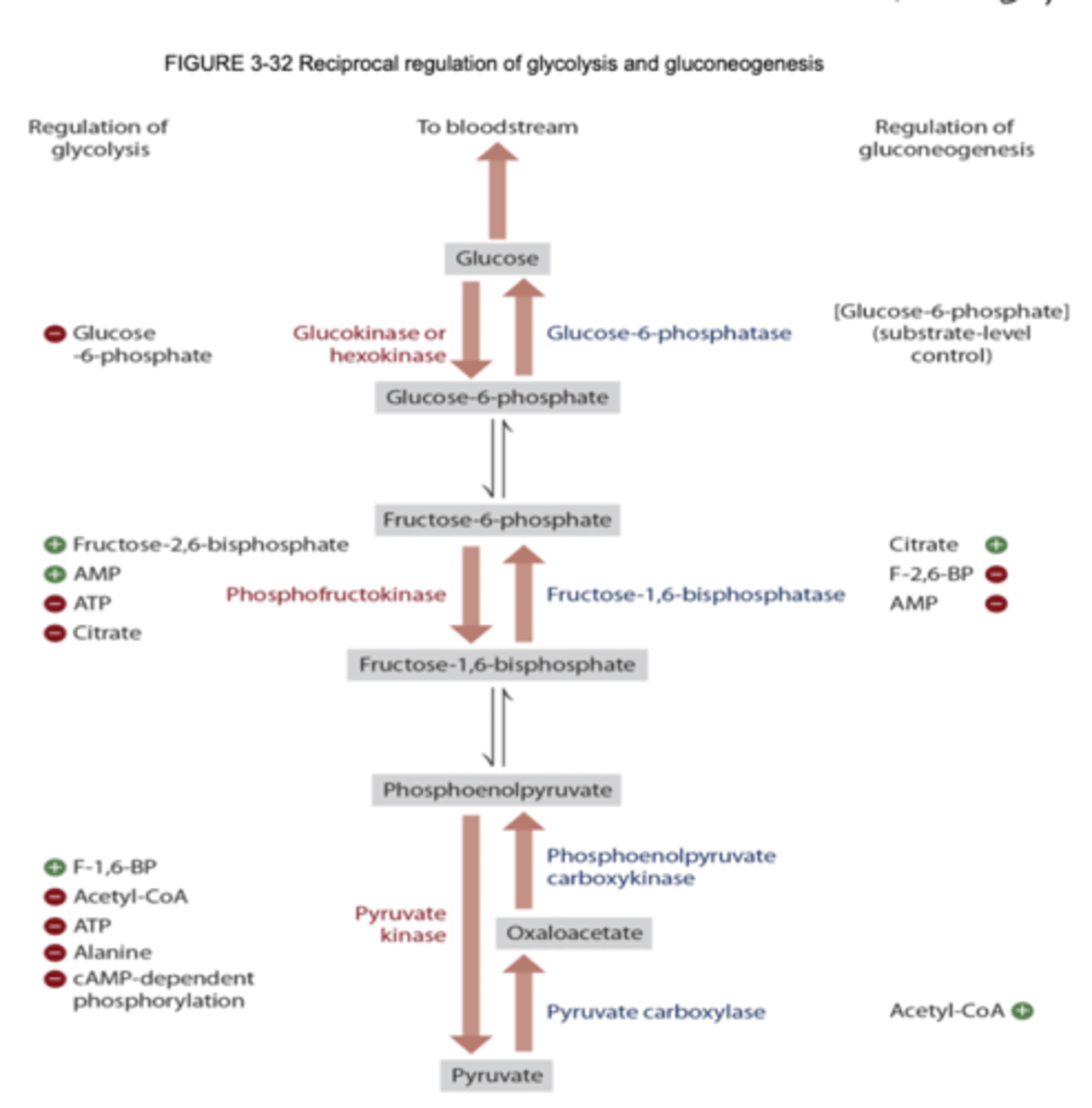
phosphofructokinase I
rate controlling enzyme of glycolysis
allosteric regulation
-energy status (ATP INHIBITS and ADP PROMOTES)
-fructose 2,6 bisphosphate (high concentrations PROMOTE enzyme)
-pH of the cell (low pH INHIBITS enzyme- lactic acid build up)
-Ca+2 concentration (influx of Ca+2 for muscle contractions PROMOTE enzyme)
hormonal status
-formation of fructose 2,6 bisphosphate
-induction (insulin will activate and glucagon will inhibit)
exercise
promotes glycolysis
ENERGETIC NEED!!!
-depletes glycogen stores in skeletal muscle
-Ca+2 influx promotes activity of PFK-1
-acts as a stress to release cortisol and glucagon (lower blood sugar and increase energy production or storage)
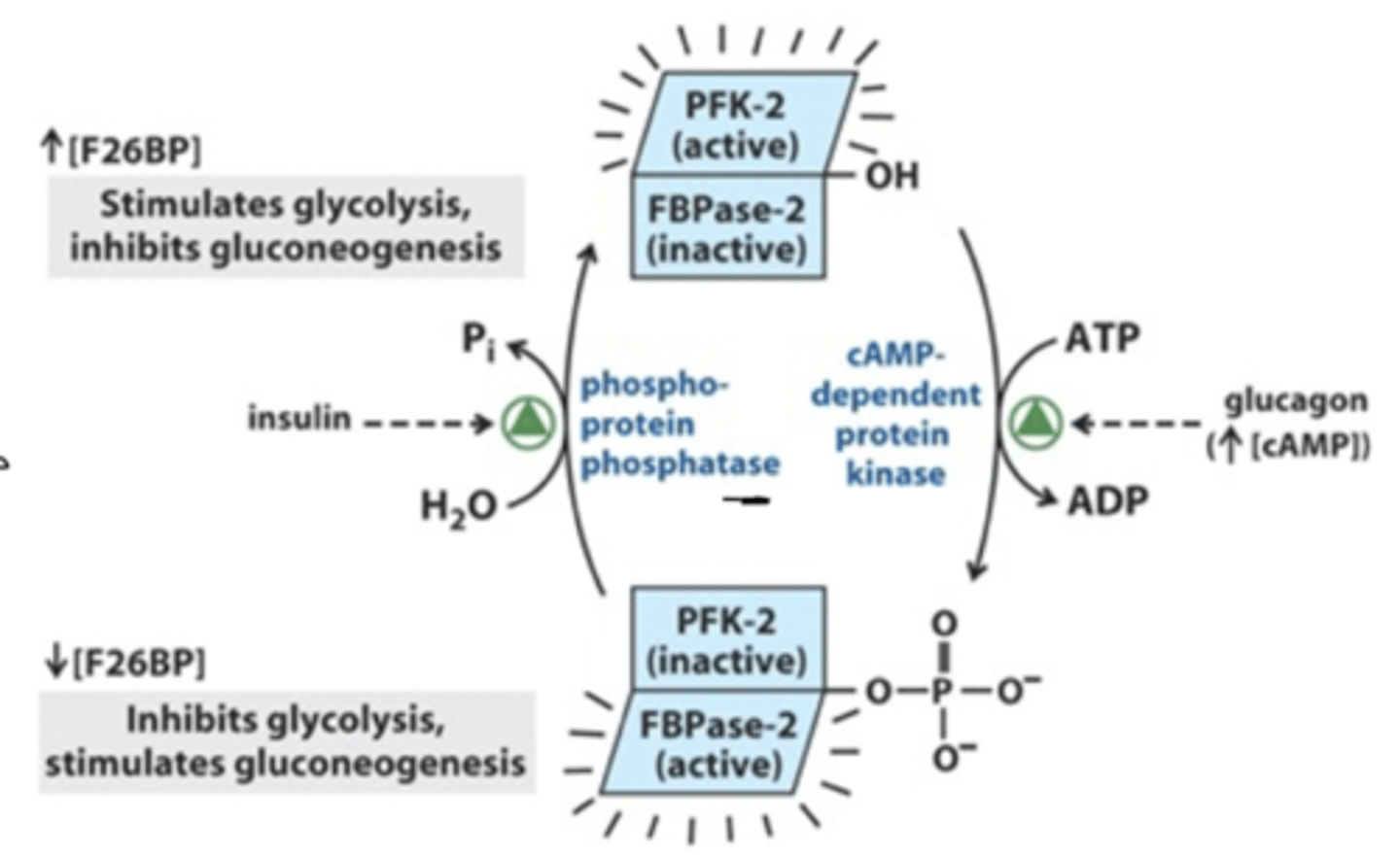
bifunctional enzyme
promotes or does not promote formation of fructose 1,6 bisphosphate
INSULIN (dephosphorylates ATP)
-activation of PFKII
-phosphate on fructose 6 phosphate to make fructose 2,6 bisphosphate
PROMOTES GLYCOLYSIS
GLUCAGON (phosphorylates ATP)
-activation of protein kinase I
-takes phosphate from fructose 2,6 bisphosphate
INHIBITS GLYCOLYSIS
*PFK-! allosterically stimulated by fructose 2,6 bisphosphate
pyruvate kinase
allosteric regulation
-energy status (ATP INHIBITS)
-product of PFK-I (fructose 1,6 bisphopshate PROMOTES)
-high levels of acetyl coA INHIBIT
-high alanine INHIBIT
hormonal status
-covalent modification (via dephosphorylation by insulin or phosphorylation by glucagon based on fasting conditions)
-induction (some gene expression)
acetyl coA and pyruvate kinase
produced by beta-oxidation which feeds into the TCA cycle
-fatty acid metabolism in fasting conditions
-need liver to be making glucose
pyruvate converted to acetyl coA (aerobic metabolism)
-TCA cycle not working- acetyl coA can build up
-used for fatty acid synthesis
HIGH ACETYL COA INDICATES FATTY ACID METABOLISM FOCUS- do not need to be breaking glucose
alanine and pyruvate kinase
high amount in liver after fasting- protein breakdown
-gluconeogenesis to make glucose
HIGH ALANINE INDICATES GLUCONEOGENESIS
- do not need to be breaking glucose
influence of NADH and NAD+ on glycolsis
*NADH is a product of glycolysis
high NADH
-pathway not needed
-inhibits glycolysis
high NAD+
-favors glucose oxidation
-now have substrate to be used for glycolysis
-promotes glycolysis
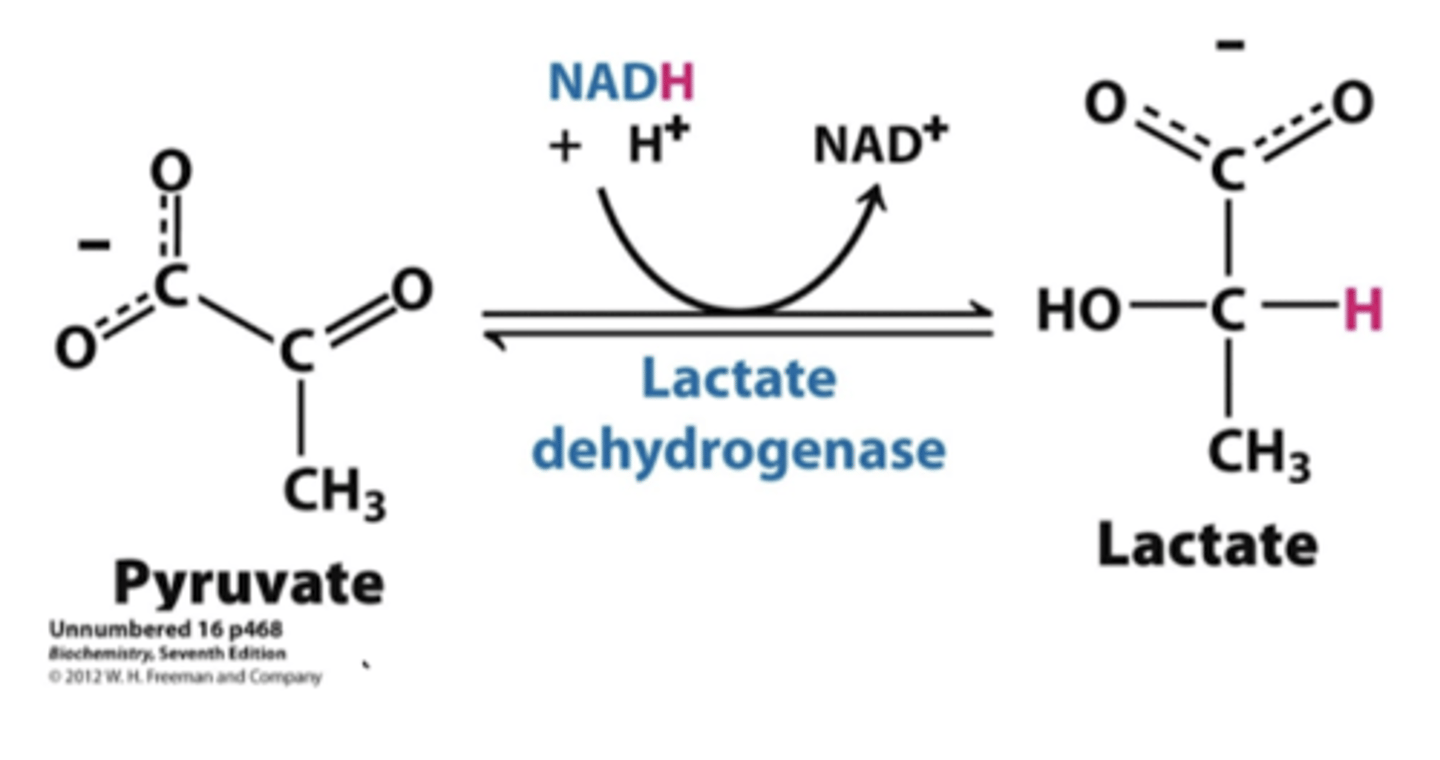
metabolism of pyruvate anaerobic
anaerobic
-generation of lactate via lactate dehydrogenase
-occurs in muscle and RBC
-generation of NAD+ without assistance
-can be recycled to liver to generate glucose- gluconeogenesis via the Cori Cycle
-build up of lactic acid will shut down the cycle
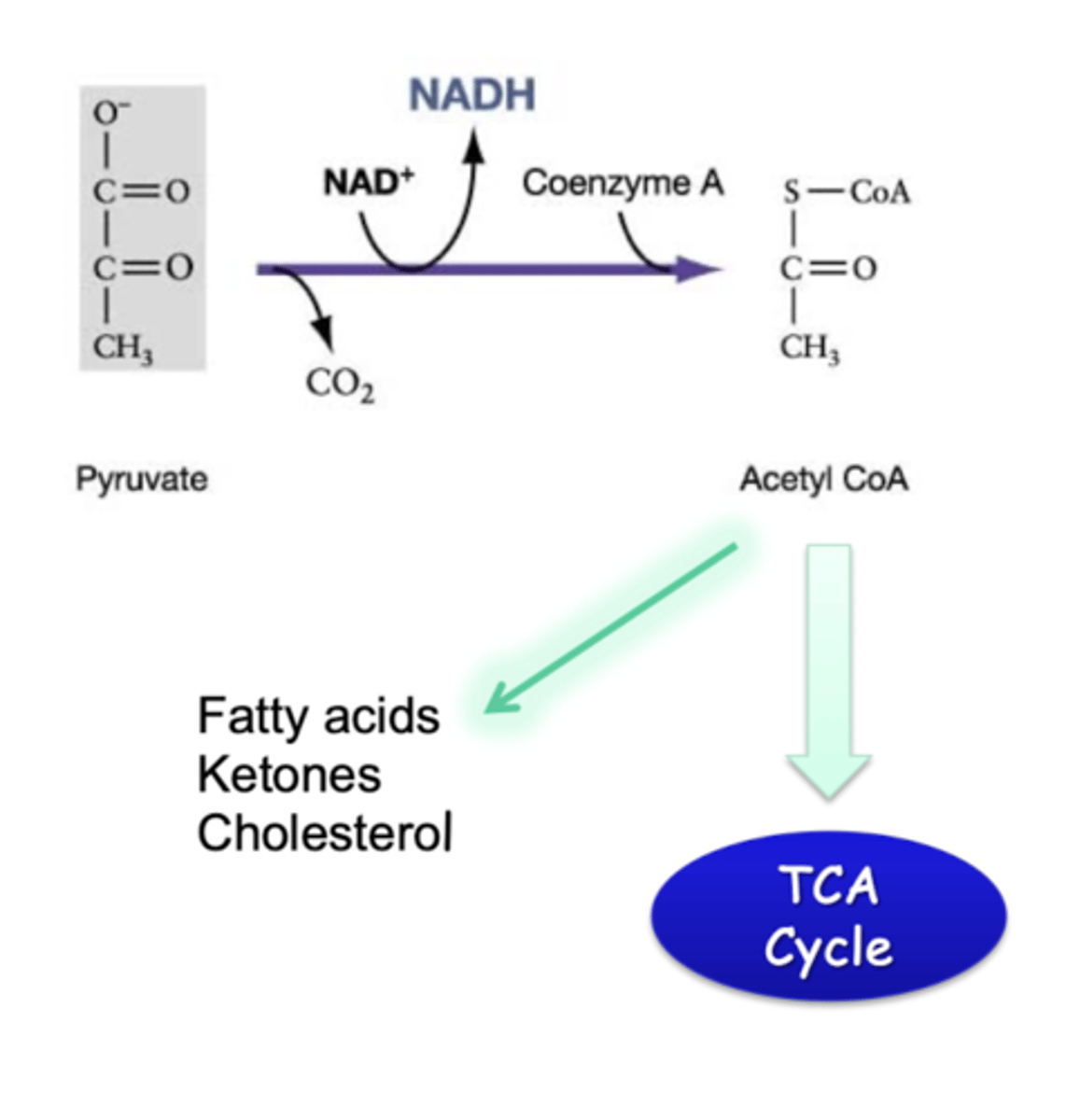
metabolism of pyruvate aerobic
aerobic
-production of acetyl coA from pyruvate
-requires mitochondria
-pyruvate dehydrogenase (another regulated complex)
-can also go to fatty acid production, ketones, or cholesterol if body does need directly need the energy
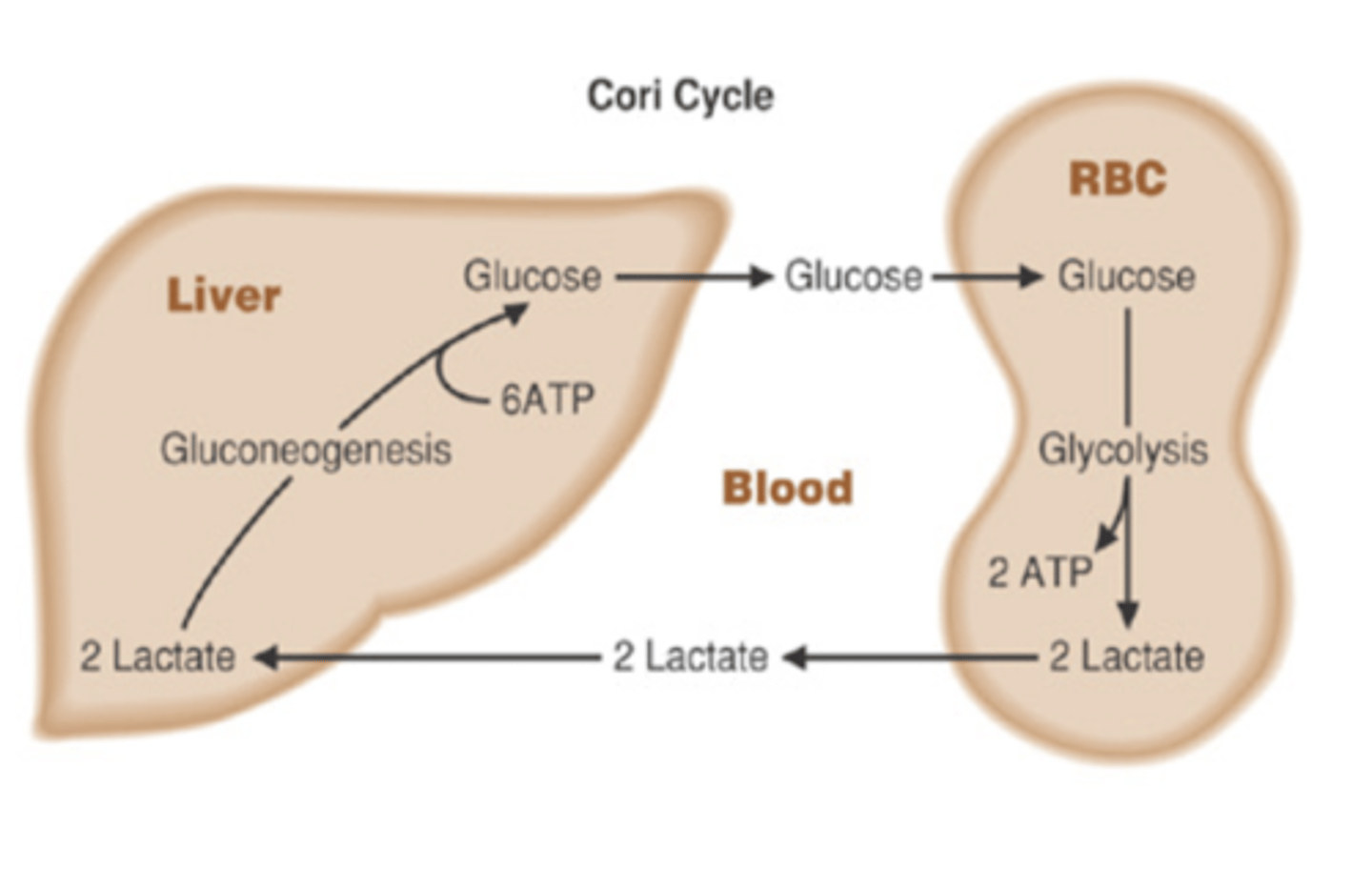
liver and RBC
Cori Cycle
-lactate from anaerobic metabolism in RBC goes to liver
-converted into glucose which can be taken up by RBC
overall regulation of glycolysis
glycogenesis summary
synthesis of glycogen (storage)
-FED state (after a meal)
-store in liver and skeletal muscle via GLUT4
-stimulated by insulin
importance:
-skeletal muscle- extra energy to break down for exercise
-liver- maintenance of blood sugar
glycogenolysis summary
breakdown of glycogen
-FASTED state
-stimulated by glucagon in liver
-stimulated by epinephrine in skeletal muscle
glycogen
-very large polymer of glucose molecules linked by a 1,4 and 1,6 bonds
-branches arise by a 1,6 bonds every 8-10th residue
-found in cytosol
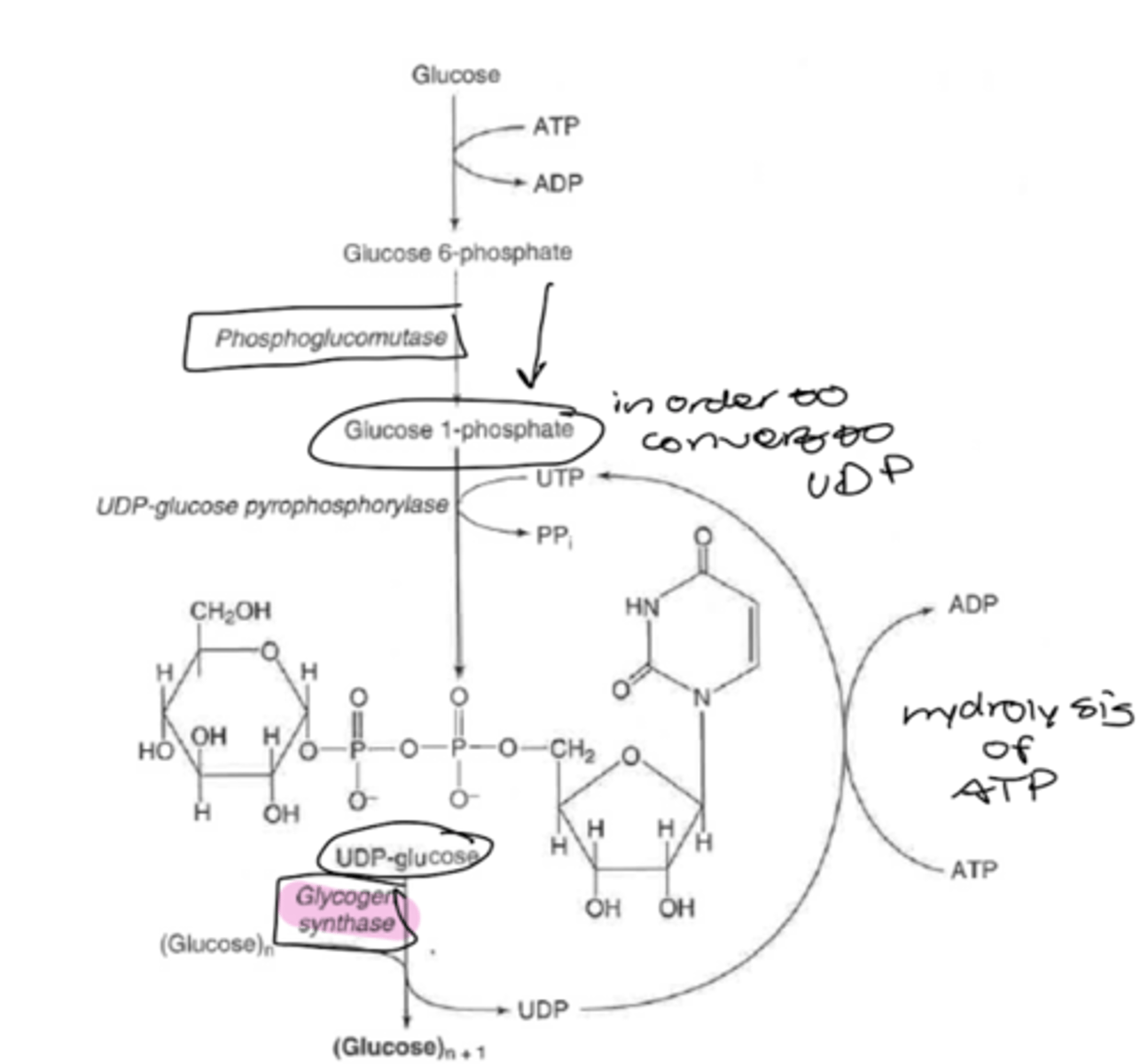
glycogenesis
-process requires energy (anabolic)
-begins with phosphorylation of glucose by hexokinase or glucokinase
*add ATP to phosphorylate glucose-6-phosphate
--> either goes into glycolysis or glycogen synthesis
two enzymes:
glycogen synthase
-creates chains of glucose molecules with a 1,4 linkages
-UDP-glucose --> glycose (n+1) +UDP
glycogenin
-primer to start glycogen chain
amylo-a (1,4--> 1,6)-glucosyl transferase (branching enzyme)
-produces a 1,6 linkages
-only regulated by how much glucose is being added
fasting impact on glycogen stores
-ability to store glycogen becomes compromised
-only "24" hours of glycogen before metabolism begins to shift
depleting skeletal muscle glycogen
resistance training
-increasing muscle mass- now have more capacity to store glycogen
anaerobic
-depletes actual stores
-prevents extra glucose from going to fat
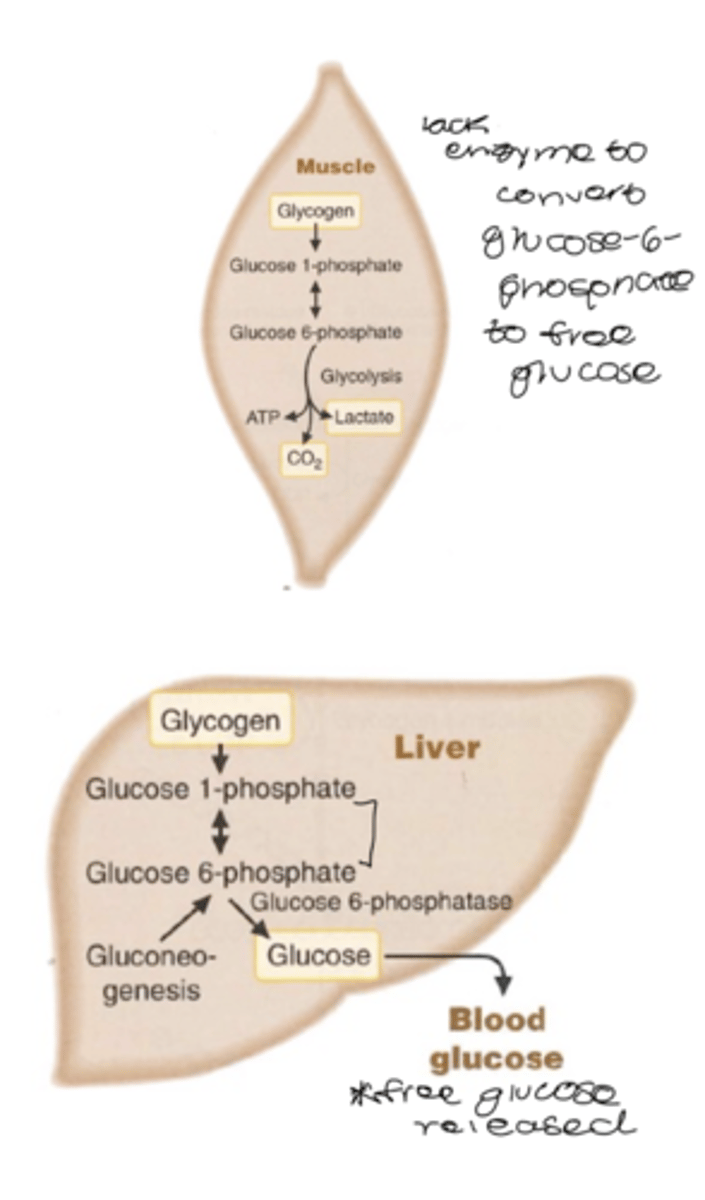
glycogenolysis
breakdown of glycogen to glucose (or glucose-6-phosphate) in respond to low blood glucose
*muscle can't convert to free glucose- goes into glycolysis
-not a reversal of synthetic reactions
-in humans, the store of liver glycogen lasts about 24 hours
glycogen phosphorylase
-cleaves a 1,4 linkages and forms glucose 1 phosphate
-G1P --> G6P
-can be converted to glucose in liver
oligo (a-1,4 --> a 1,4)-glucantransferase
-causes exposure of 1,6 branch point
amylo-a(1,6)-glucosidase (debranching enzyme)
-removal of a 1,6 branch points
-allows phosphorylase to proceed
debranching enzyme
bifunctional
-oligo-(a-1,4-->a,1,4)-glucantransferase
-amylo-a(1,6)-glucosidase
muscle glycogen synthgesis
decreased glycogen synthesis and storage in muscle in diabetics
*problem is GLUT4- can't get to membrane
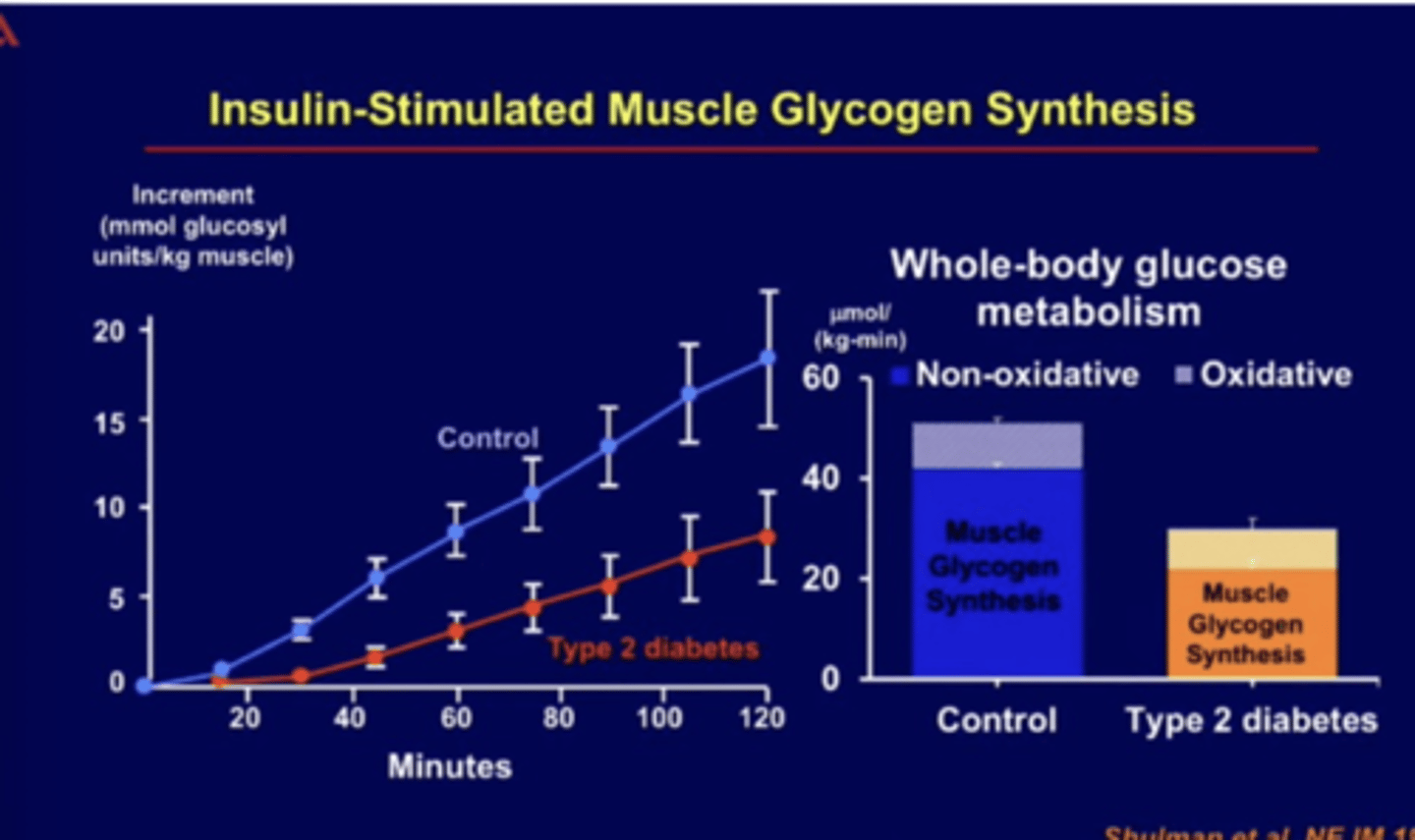
skeletal muscle and dibaetics
becomes first tissue to become insulin resistant
-glucose goes somewhere else --> fat metabolism
key enzymes in glycogen metabolism regulation
glycogen phosphorylase
-glycogen breakdown
-forms G-1-P
-liver and muscle glycogen phosphorylated activated by phosphorylation (signaling through pKA)
*glucagon in liver and epinephrine in SM
glycogen synthase
-glycogen synthesis
-addition of glucose using UDP-glucose to glycogen chain
-activity inhibited by phorphorylation
*insulin
regulation of glycogen phorphorylase
covalent modification
-phosphorylation
allosteric control
-energy charge, glucose, G-6-P
*active via phosphorylation
a-active
b-inactive
in depth regulation of glycogen phosphorylation
PROMOTES ACTIVITY OF GLYCOGEN PHOSPHORYLASE A
-epinephrine or glucagon (pka does not directly do the action)
-phosphorylase kinase PHOSPHORYLATES ATP and adds P to activate glycogen phosphorylase a
*glycogen will be broken into glucose-1-phosphate
PROMOTES ACTIVITY OF GLYCOGEN PHOSPHORYLASE B
-insulin
-protein phosphatase 1 DEphosphorylates enzyme resulting in glycogen phosphorylase b
-also signals an enzyme called phosphodiesterase that breaks down cAMP
*glycogen will remain in storage form
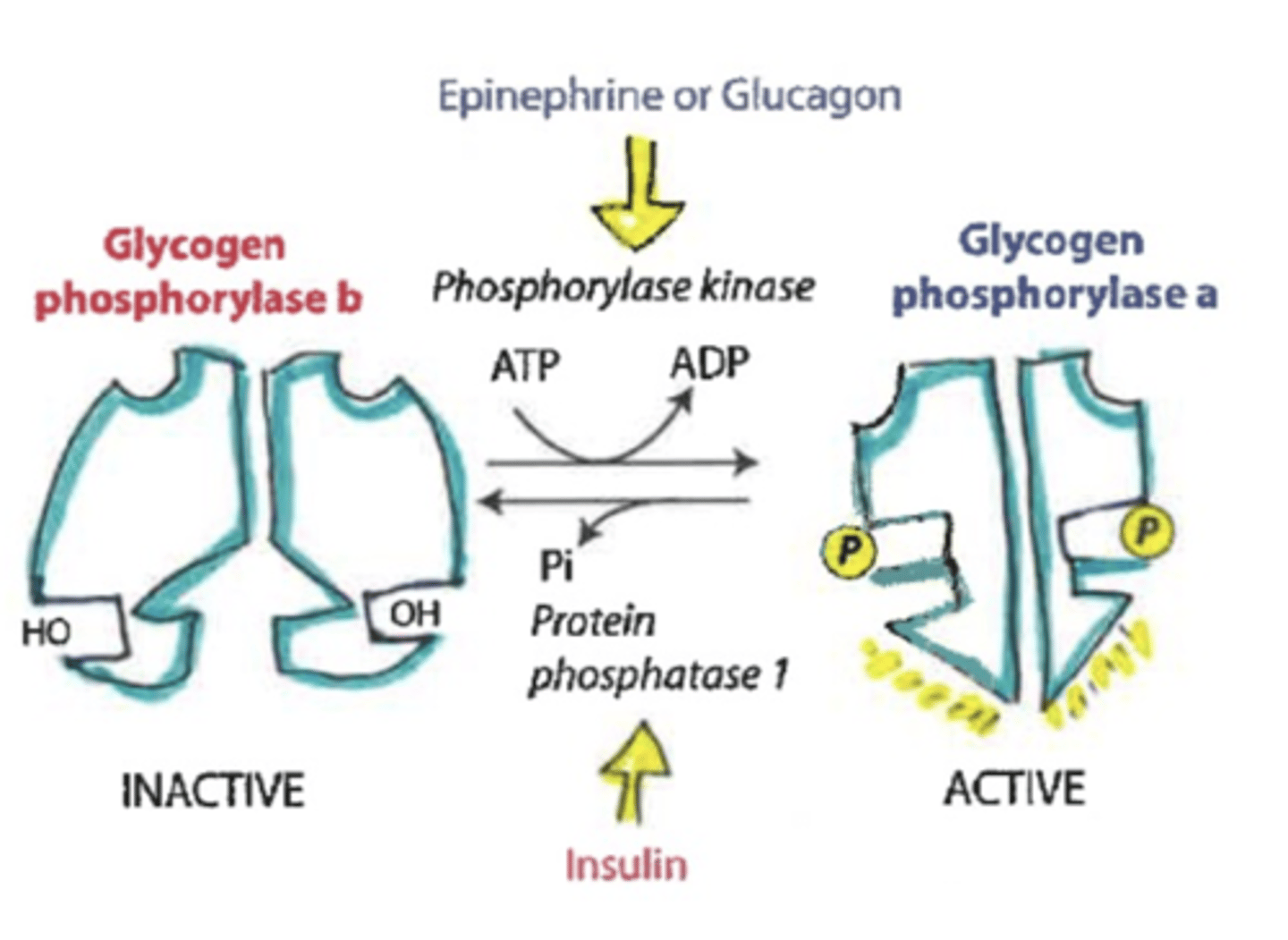
kinase
phosphorylates a molecule
-adds a phosphate from ATP to an enzyme to alter its function
muscle regulation of glycogenolysis
allosteric regulation
-ATP will inhibit
-glucose-6-phosphate will inhibit
-AMP can promote active form of glycogen phosphorylase b
*extra AMP can override normal enzymatic pathwat
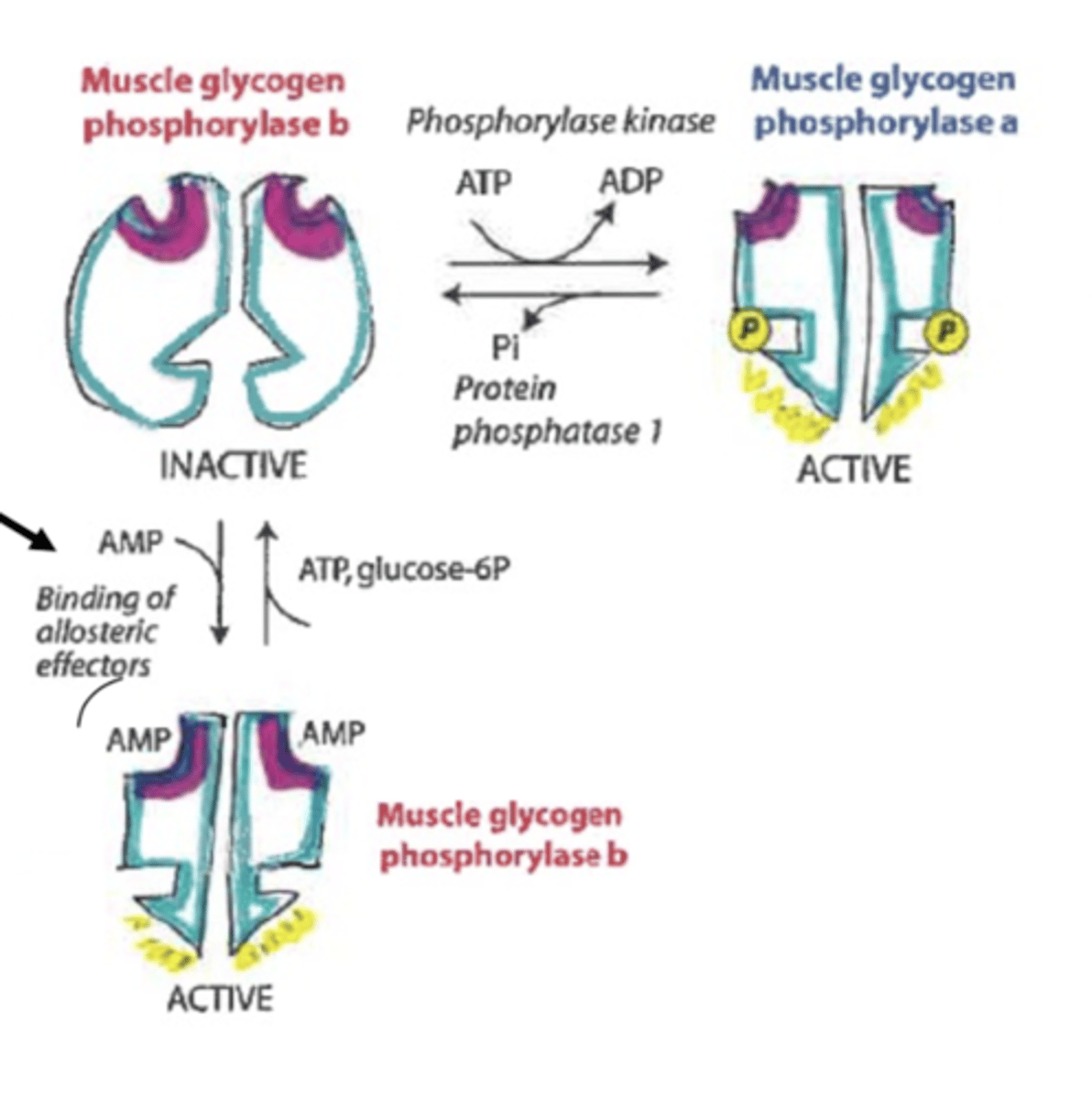
tissue regulation of glycogenolysis
allosteric regulation
-glucose will inhibit- overrides phosphorylation
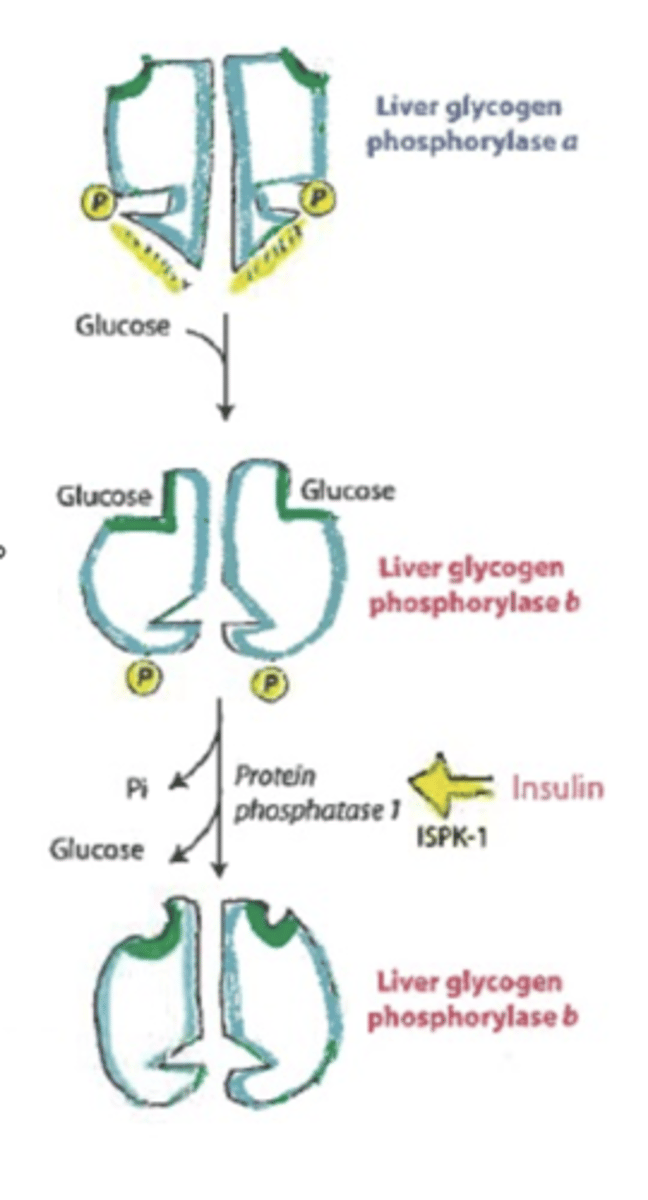
regulation of glycogen synthase
*regulated primarily by reversible phosphorylation
DEPHOSPHORYLATION activates
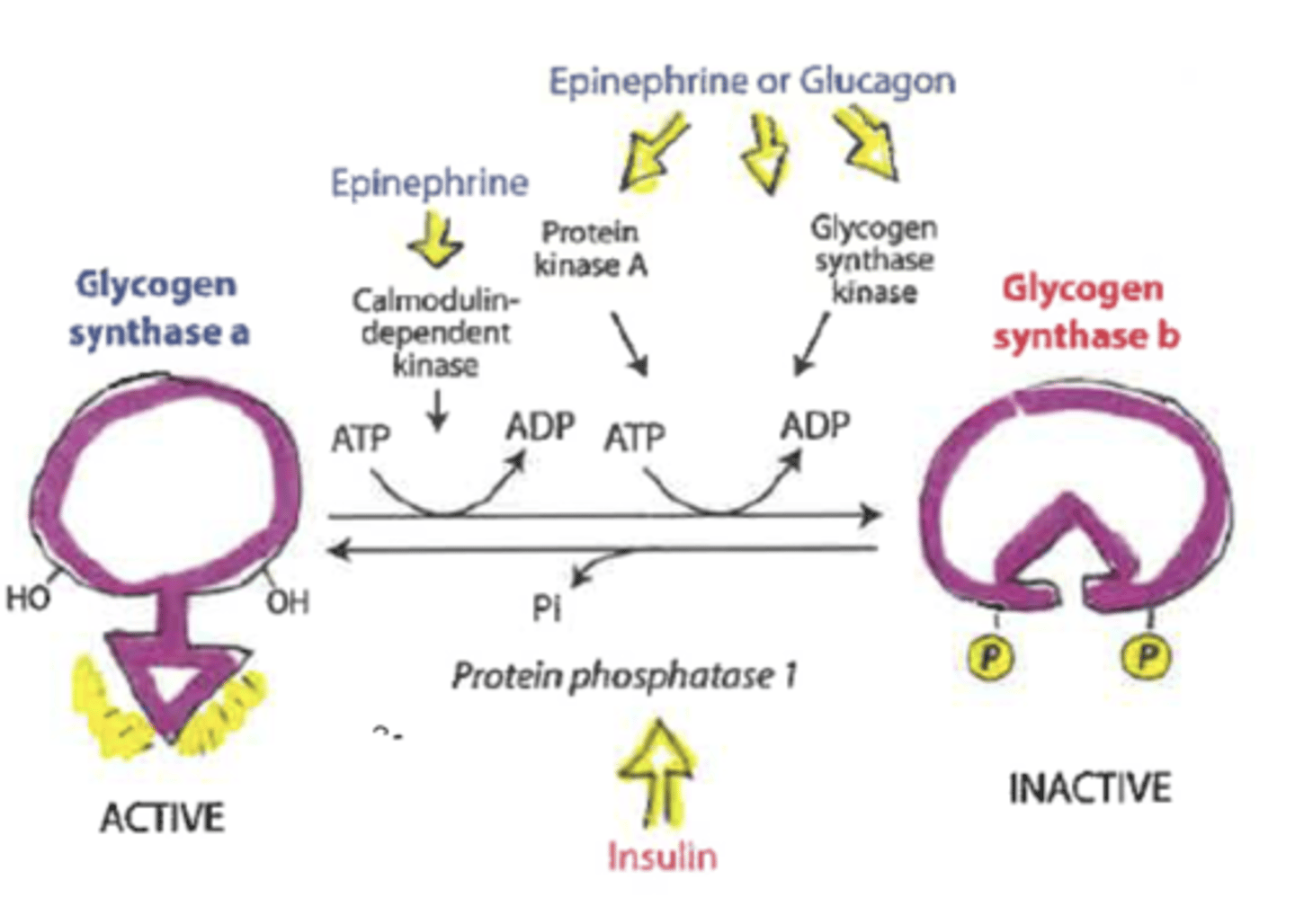
calmodulin kinase
enzyme in the skeletal muscle
-activated by Ca+2
-exercising releases
-can promote phosphorylation and activate glycogen synthase b (inactive form)
galactose summary
dietary sources:
-dairy products
-some fruits and. vegetables
intestinal absorption
-requires SGLT1 and GLUT2
liver metabolism
-galactokinase (phosphorylates)
inability to metabolize
-galactosemia- build up of byproducts which can result in organ damage
-test for day 2 after birth (can NOT consume breastmilk)
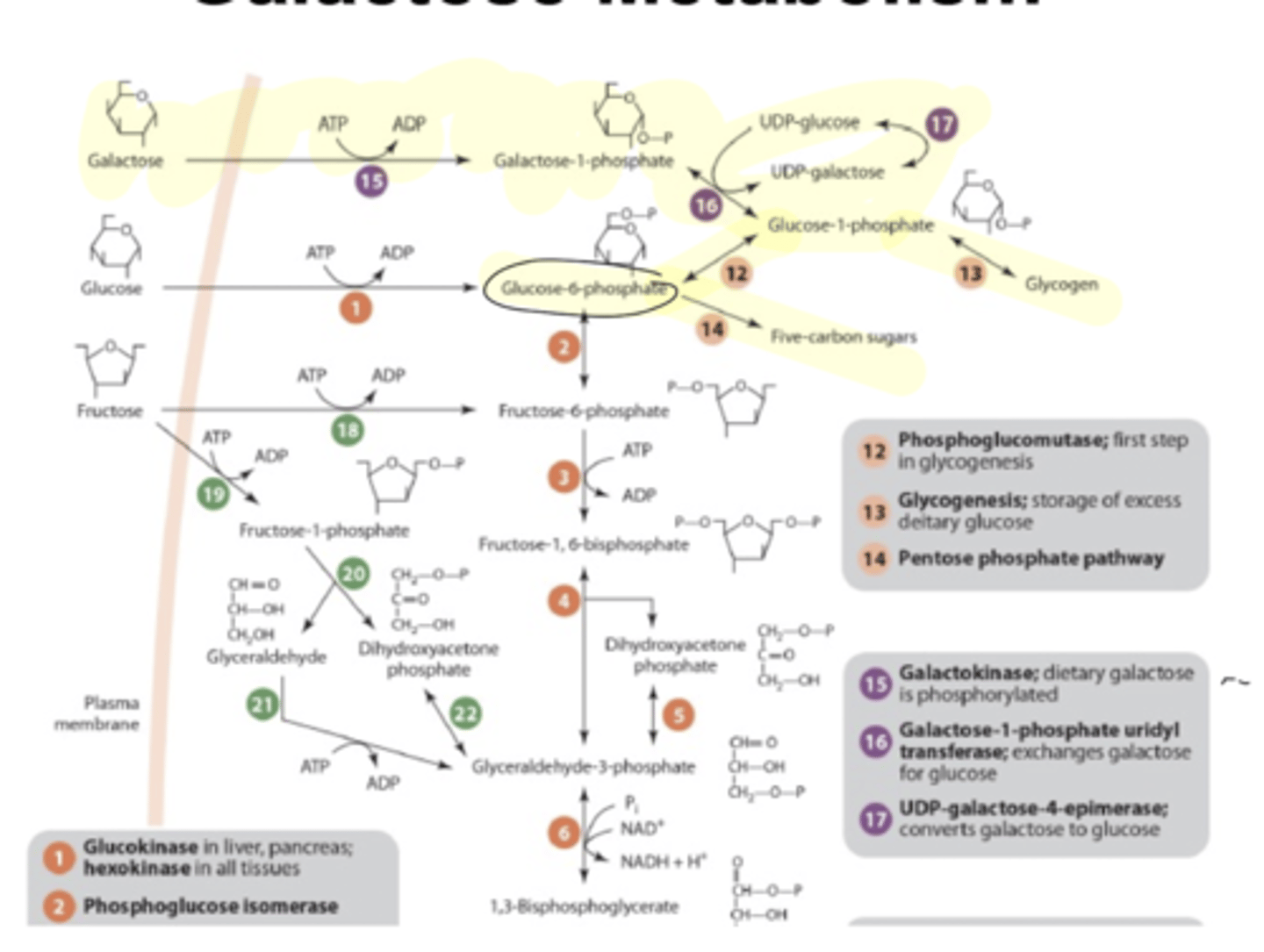
galactose metabolism
LIVER
galactose
galactose-1-P
eventually broken down into glucose-6-P
*end result is the same as glucose
can be fed into:
-glycogen production
-glycolysis
-PPP
fructose
dietary sources:
-fruits
-sweeteners (sucrose 50%, honey 40% , HFCS, added sugars)
**issue is with sweeteners- easy to overdo consumption
-fructose is 10-15% of caloric intake
-linked to some disease states due to metabolic shifts that occur
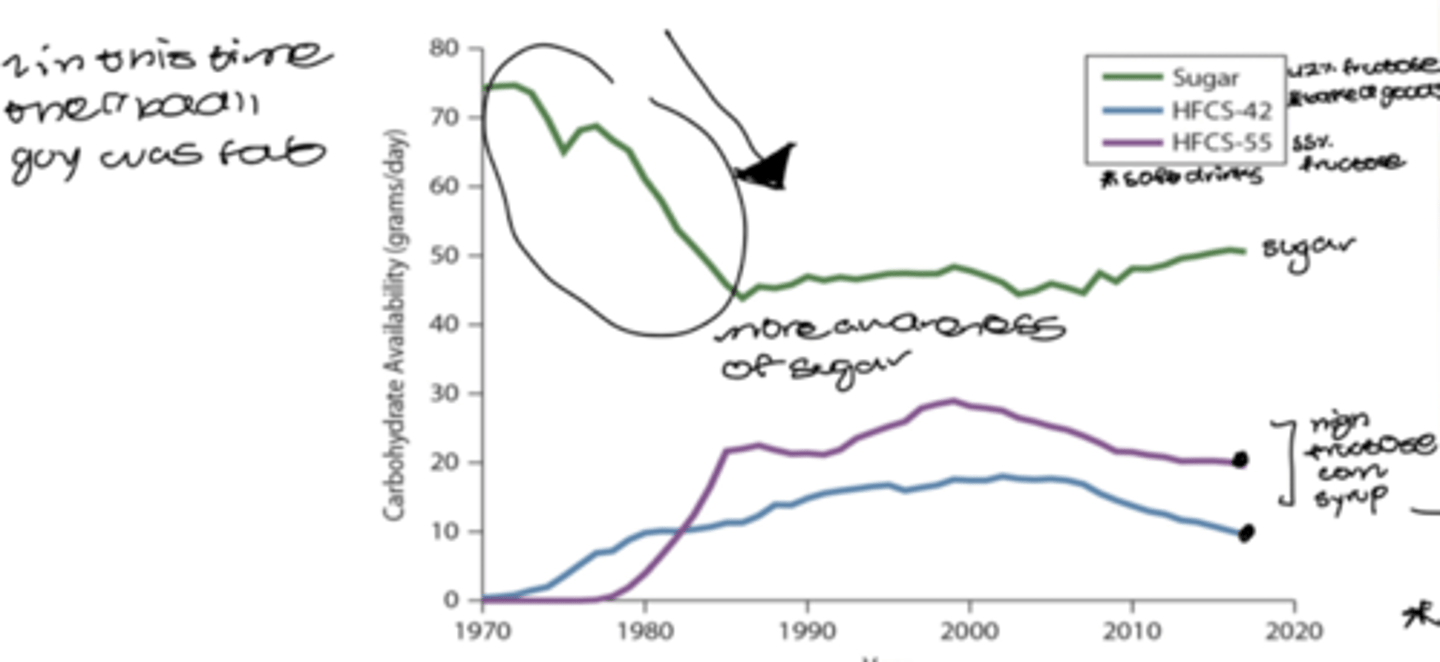
high fructose corn syrup vs table sugar
sugar
-derived from sugar cane
-fluctuates based on imports and exports
high fructose corn syrup
-source of fructose
-cheaper option
-corn is pure starch (100% glucose)
-make corn syrup and add enzymes that convert some of the glucose into fructose
*thought to be absorbed more quickly
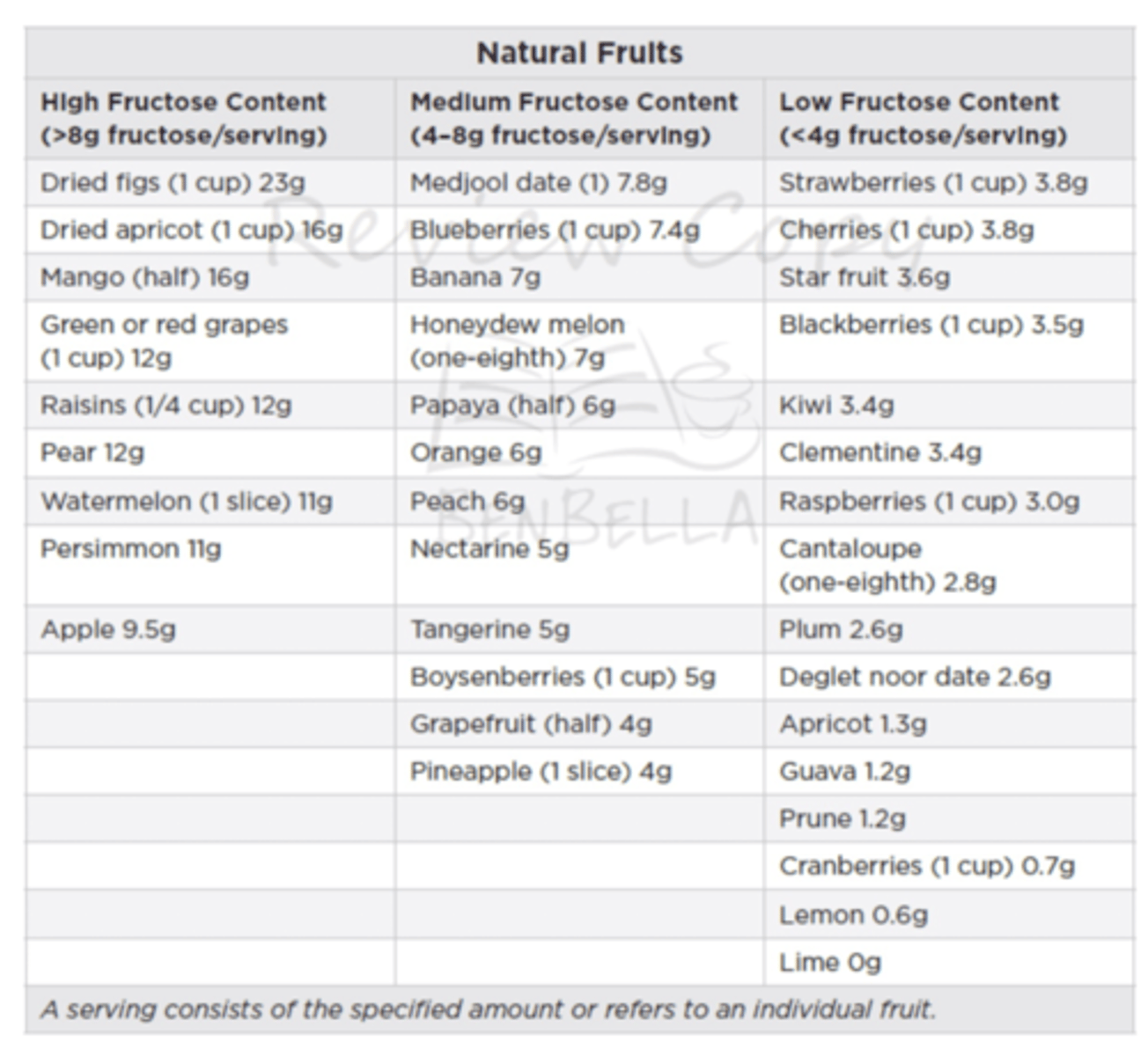
fruit and fructose
fructose content varies
-may suggest lower fructose options for certain patients
high
-dried and dehydrated fruits
-apples
-grapes
-watermelon
low
-berries
*less likely to be involved in disease as added sugars
-has beneficial constituents such as flavanols, fiber, antioxidants
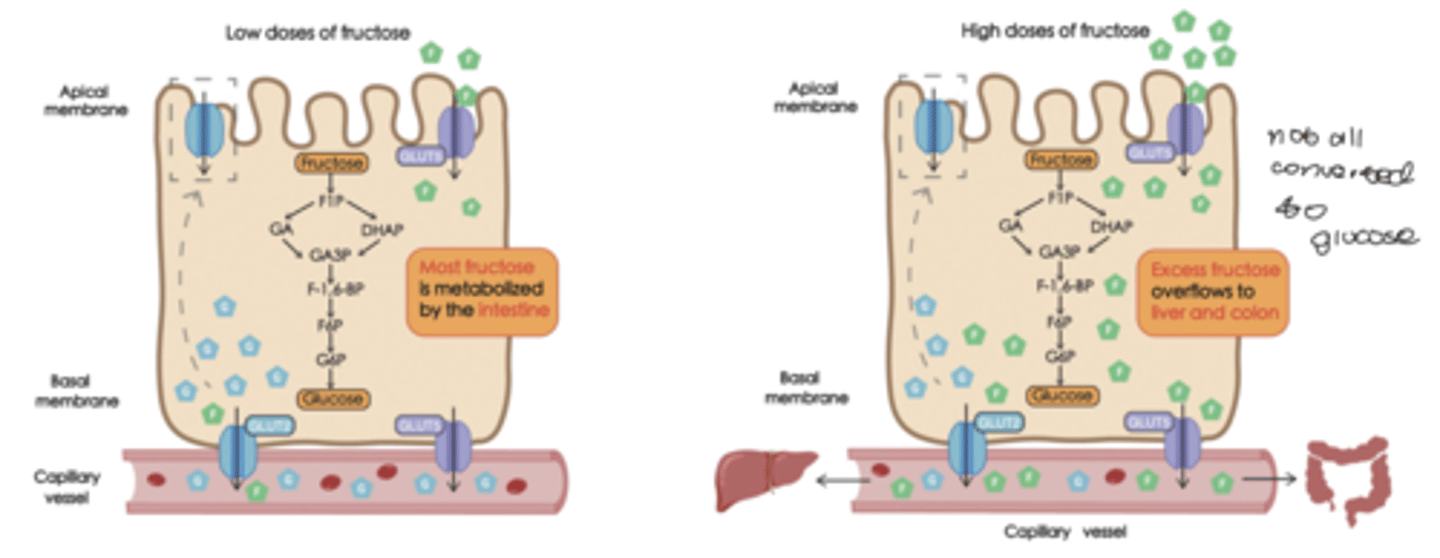
fructose metabolism
fructokinase
-adds a phosphate in the liver
*most fructose is being converted to glucose in the intestines
-hardly see any fructose coming out of liver in systemic circulation
PROBLEM IS WITH EXCESS DOSES
-the liver will not convert all of it to glucose
-more fructose
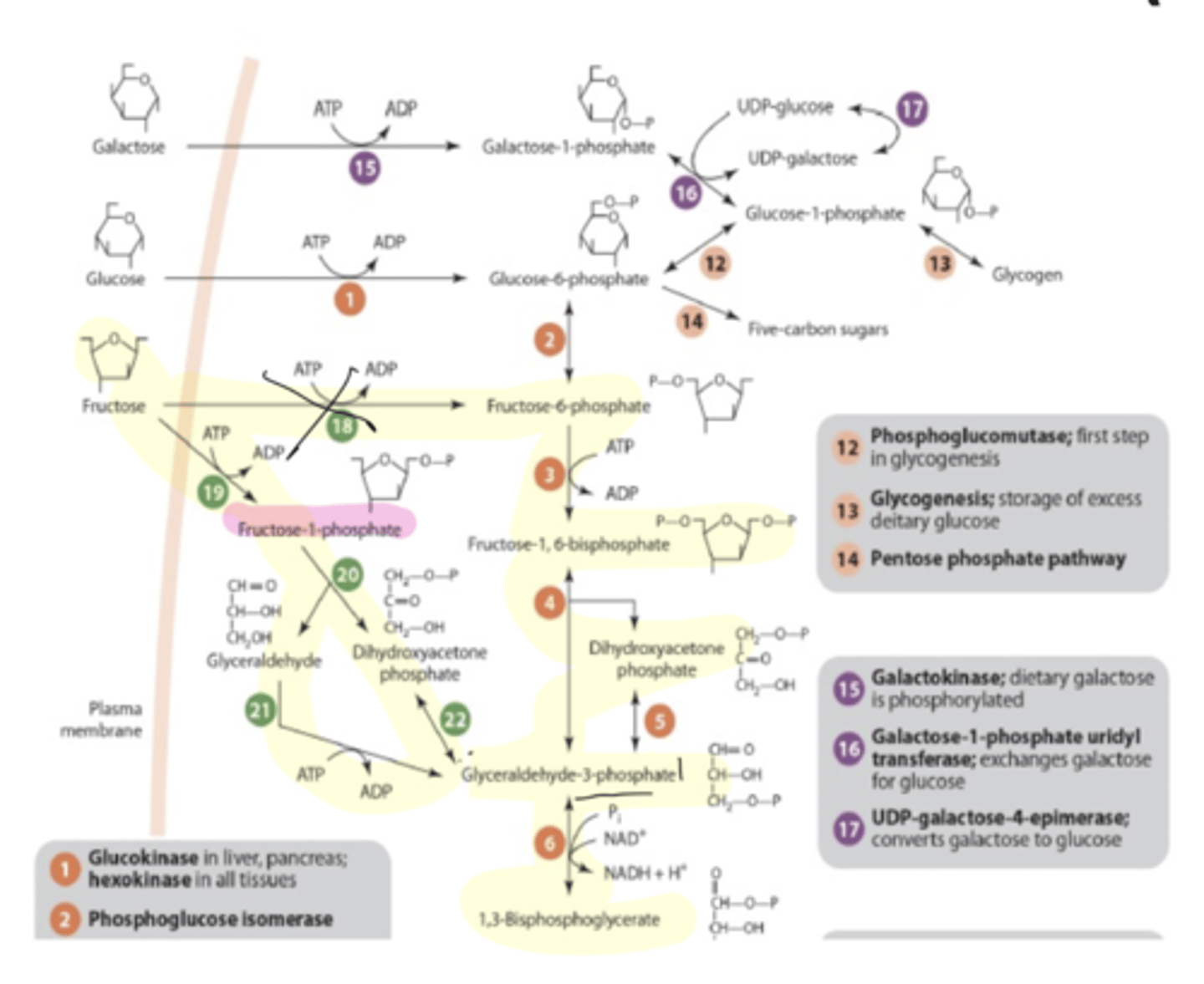
LIVER
fructose
fructose-6-P
glyceraldehyde + dihydroxyacetone phosphate
glyceraldehyde-3-phosphate
*bypasses PFK-1 but can eventually feed into the glycolysis pathwat
fructose malabsorption
highly variable in children and adults
-about 34% of adults (higher with GI disorders)
-mechanism unknown
study found that lean children has higher fructose malabsorption
fatty liver disease
greater than 5% of the liver is fat
stages:
-fatty
-steatohepatitis (fibrosis)
-cirrhosis (NOT REVERSIBLE)
-cancer
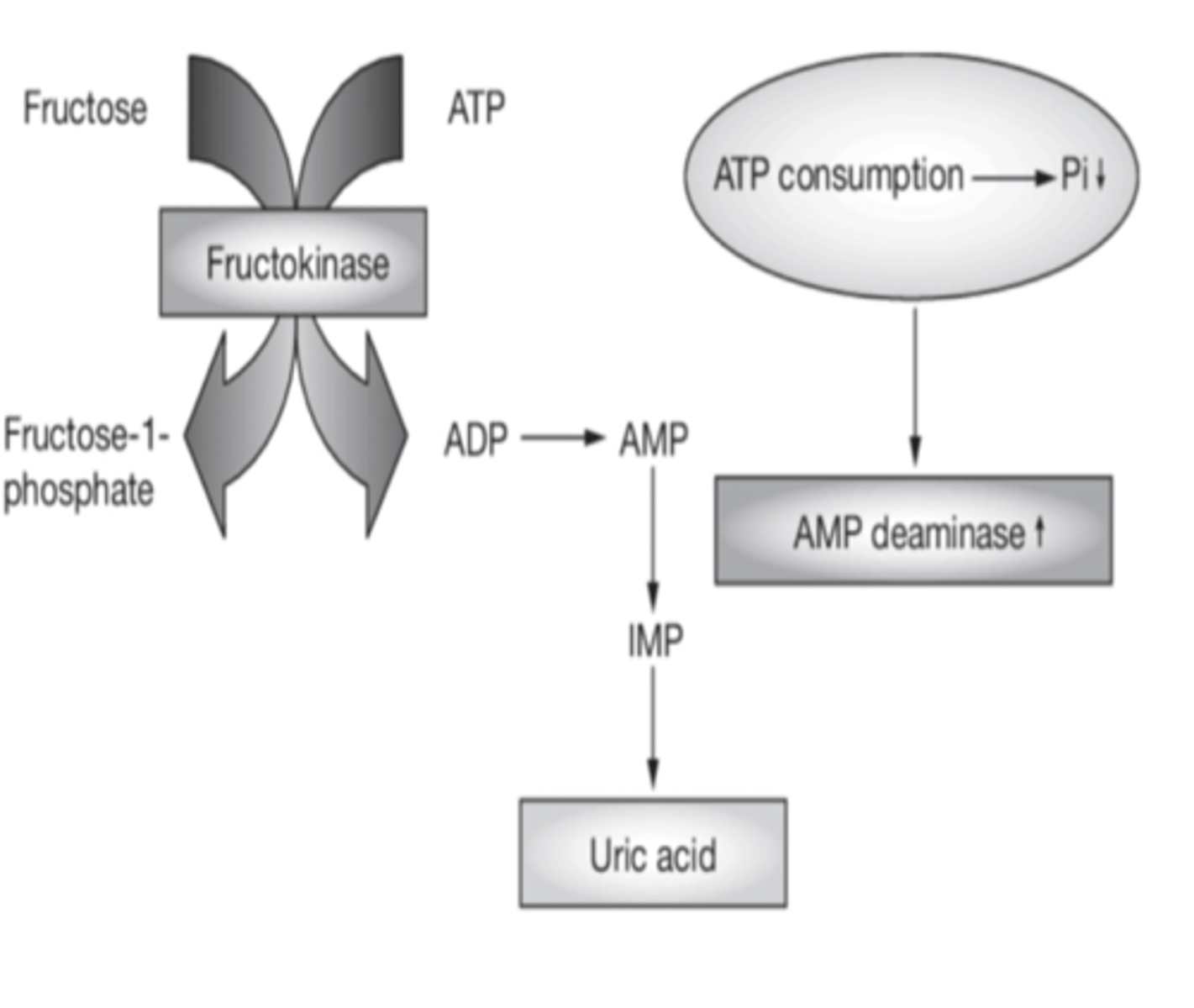
fructose metabolism and fatty liver disease
-bypasses regulation of PFK-1
-very rapid metabolism
-results in drop in ATP and P1- ATP depletion (fructokinase)
-formation of uric acid due to increased fructose coming into the liver (AMP deaminase converts AMP to uric acid)
uric acid and fatty liver
shown to
-stimulate lipogenesis
-inhibit fatty acid oxidation
-stimulate gluconeogenesis
other risk factors for fatty liver
-fructose combined with a high fat diet produces more severe fatty liver
-alcohol with fructose
-high GI diet can induce endogenous fructose production (polyol pathway makes fructose from glucose)
-high salt diet
TCA cycle
oxidation of acetyl coA to CO2 and H2O
-aerobic- requires the mitochondria
also produces:
-NADH
-FADH2
pyruvate dehydrogenase
pyruvate to acetyl coA
-produces NADH
associated with inner mitochondrial membrane
regulated:
-covalent modification (phosphorylation and dephosophorylation)
-NADH/NAD+
-ATP/ADP
-acetyl coA
activates:
-dephosphorylation (insulin)
-high NAD+
-high ADP
inhibits:
-phosphorylation
-acetyl coA
TCA functions
*produces most of CO2 made in humans
*source of reducing equivalents that drive respiratory chain to produce ATP
*converts excess energy and intermediates into fatty acid synthesis (citrate build-up)
*provides precursors used in synthesis of proteins and nucleic acids
*regulation of other metabolic pathways
TCA phases
-acetyl coA production
-acetyl coA oxidation
-electron transfer
electron transport chain
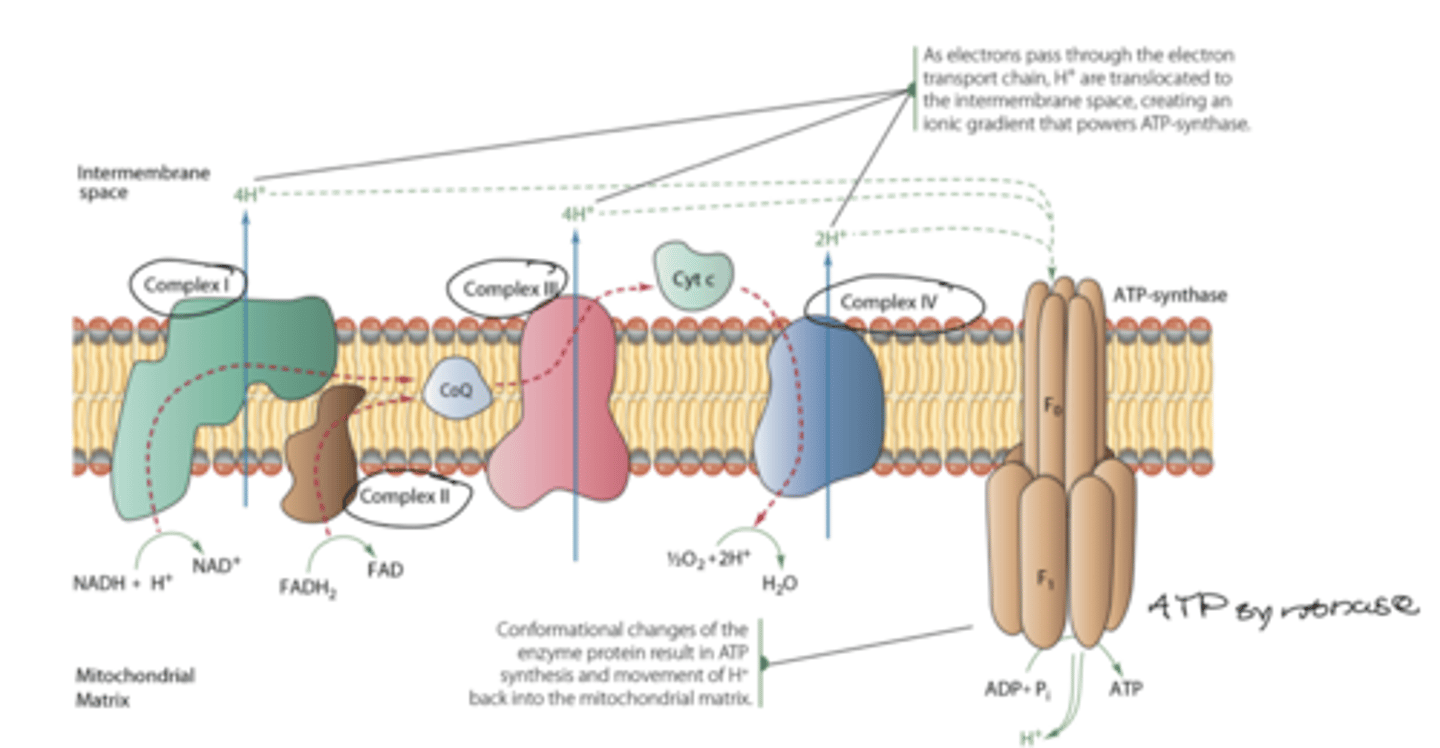
regulation of TCA cycle
flux of ETC
-state of ATP
-reduction state of NAD+
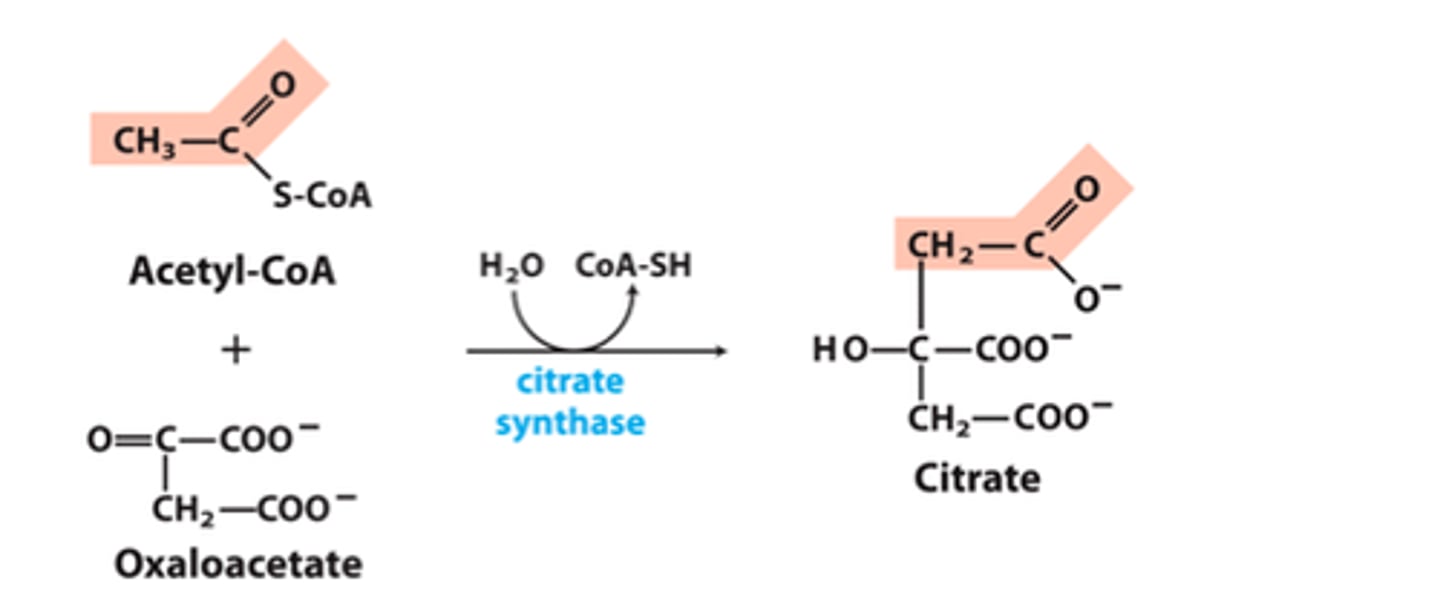
citrate synthase
oxaloacetate and acetyl coA to citrate
product and substrate concentrations
-low substrate inhibits
-a lot of citrate inhibits
allosteric
-ATP inhibits
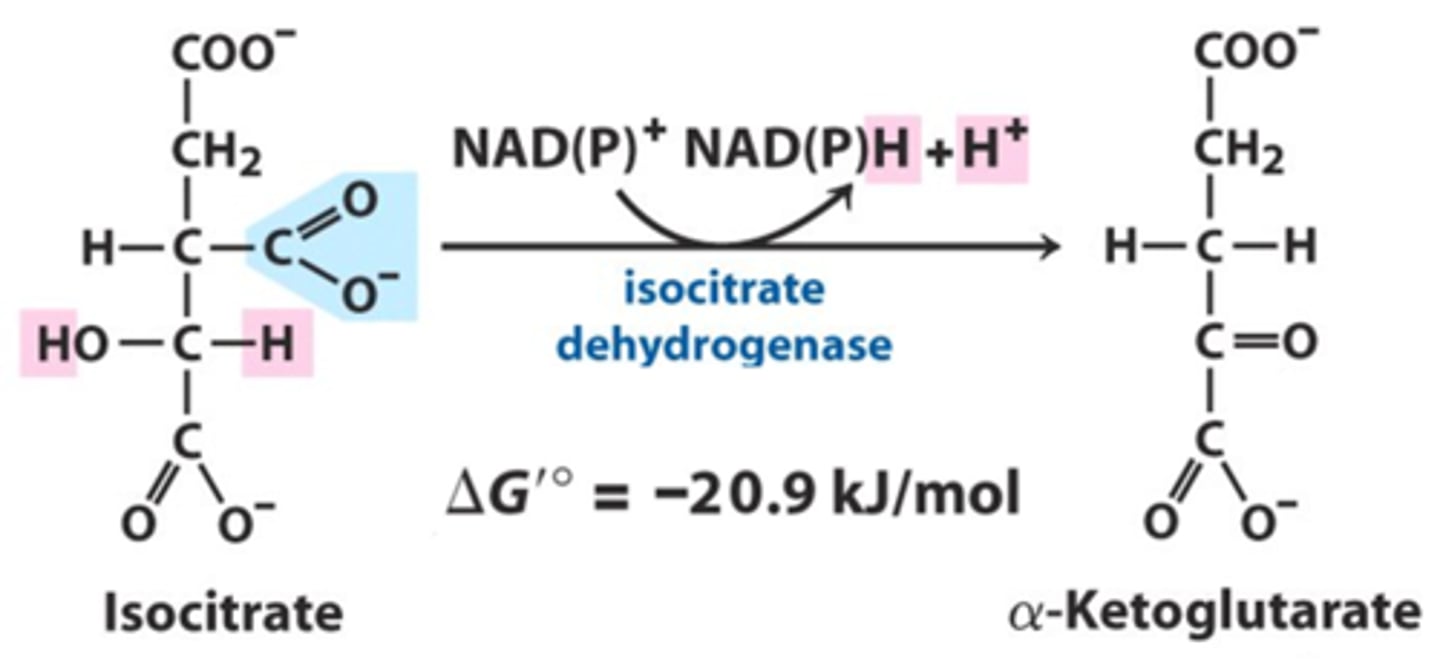
isocitrate dehydrogenase
isocitrate to a-ketoglutarate
allosteric
-NADH and ATP inhibit
-Ca+2 activates (more ATP for the muscles)
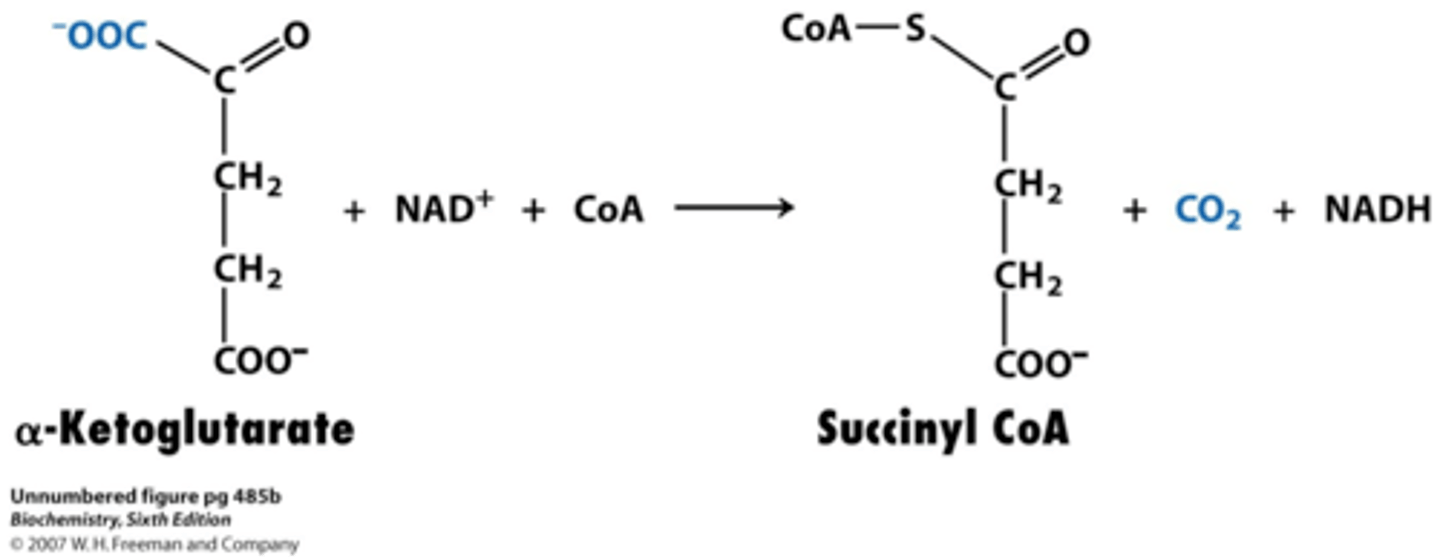
a-ketoglutarate dehydrogenase
a-ketoglutarate to succinyl coA
allosteric
-NADH inhibits
-Ca promotes in skeletal muscle
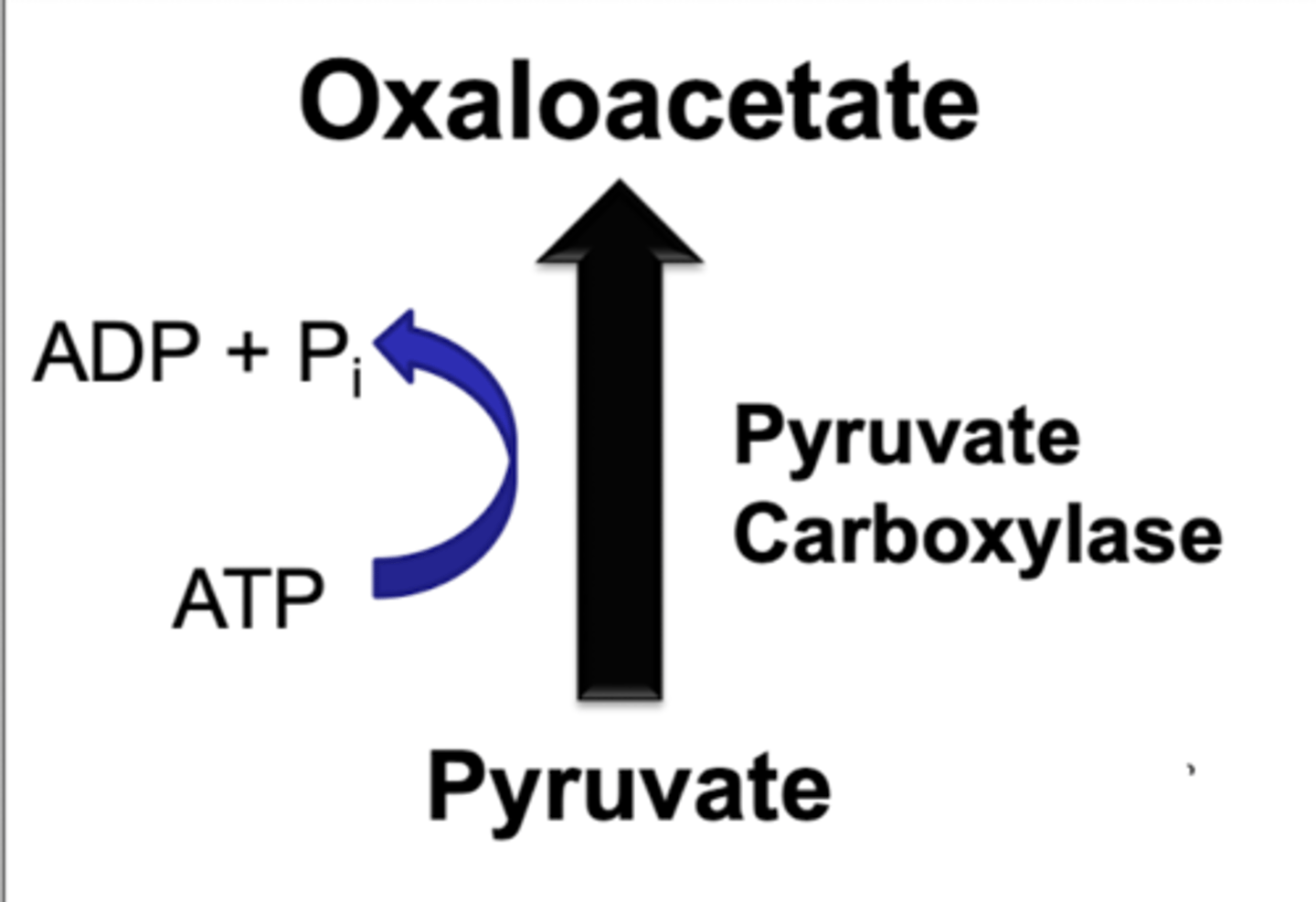
pyruvate carboxylase
pyruvate to oxaloacetate under fasting conditions
increase in acetyl coA
-regulated positively
replenishes OAA to drive TCA
*anapleurotic reaction
gluconeogenesis
production of glucose from nonhexose precursors
occurs all the time at a low level but increased under fasting conditions
*90% occurs in liver and 10% in kidney
-requires energy
-maintains blood glucose
what drives gluconeogenesis
ATP comes from the breakdown of fat
-switch from carb to lipid metabolism
ENERGY FROM FAT USED TO DRIVE GLUCONEOGENESIS
precursors for gluconeogenesis
-lactate
-glycerol
-gluconeogenic amino acids (all except lysine and leucine)
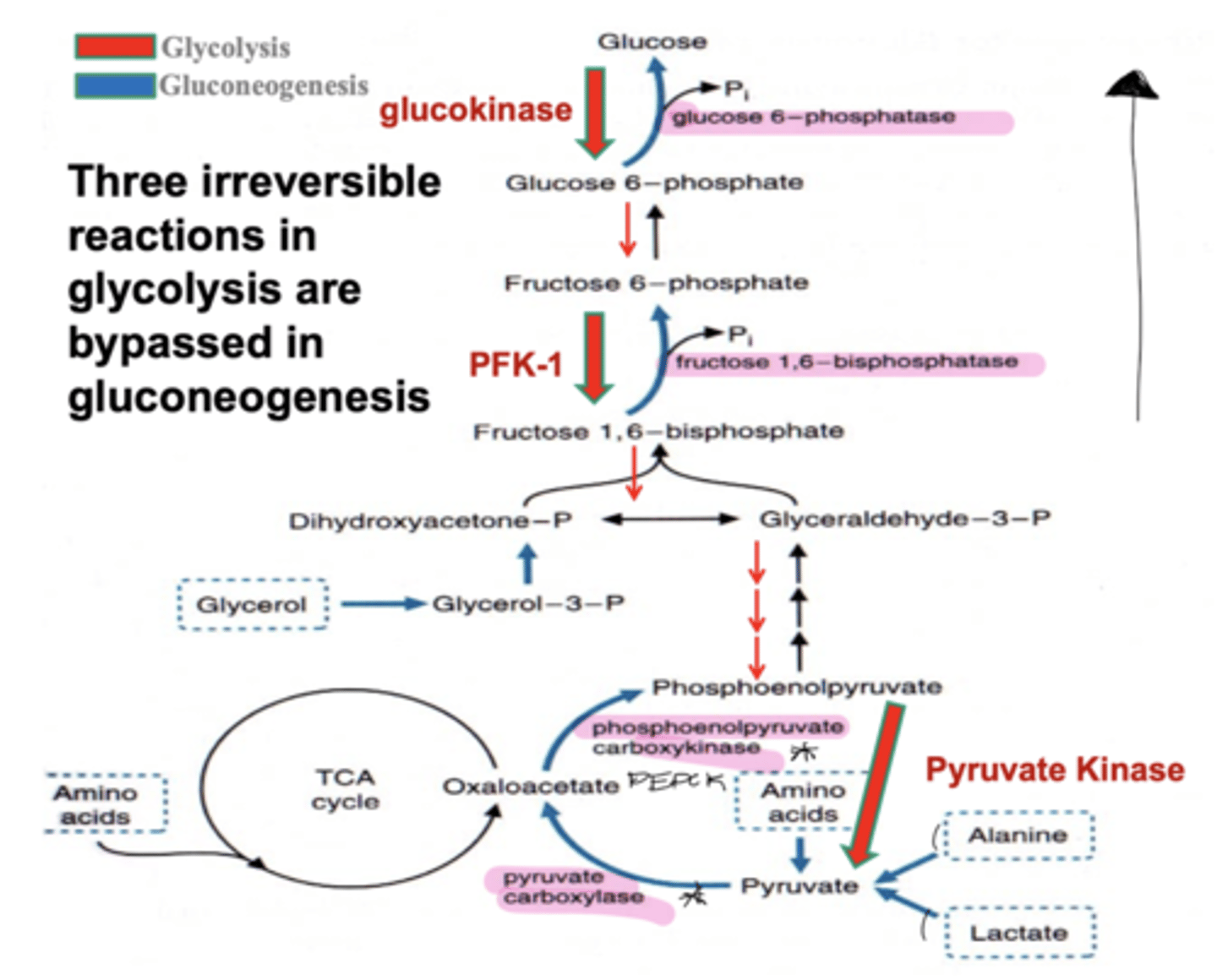
three irreversible reactions in glycolysis bypasses in gluconeogenesis
hexokinase
-glucose 6-phosphatase
PFK-1
-fructose 1,6-bisphosphatase
pyruvate kinase
-PEPCK
-pyruvate carboxylase
lactate
lactate dehydrogenase REVERSIBLE
-liver puts into pathway to make more glucose
-skeletal muscle and RBC both naturally produce a lot (produces NAD+)
*precursor for gluconeogenesis
alcohol and hypoglycemia
generates excess NADH which inhibits gluconeogenesis
amino acids
alanine aminotransferase
-alanine into pyruvate
signaled by increased protein breakdown due to fasting (takes about 24 hours to start breakdown- cortisol release)
*precursor for gluconeogenesis
glycerol
glycerol kinase
-glycerol to glycerol 3-phosphate
glycerol 3-phosphate dehydrogenase
-glycerol 3 phosphate into dihydroxyacetone phosphate
(part of gluconeogenesisi)
-glycerol is from fat breakdown
-NADH produced
*precursor for gluconeogenesis
three sites of regulation for gluconeogenesis
-glucose 6 phosphatase
-fructose 1,6 phosphatase
-pyruvate carboxylase and PEPCK
pyruvate carboxylase regulation
acetyl coA promotes
-anapleurotic
-source of OAA for TCA and carbon source for gluconeogenesis
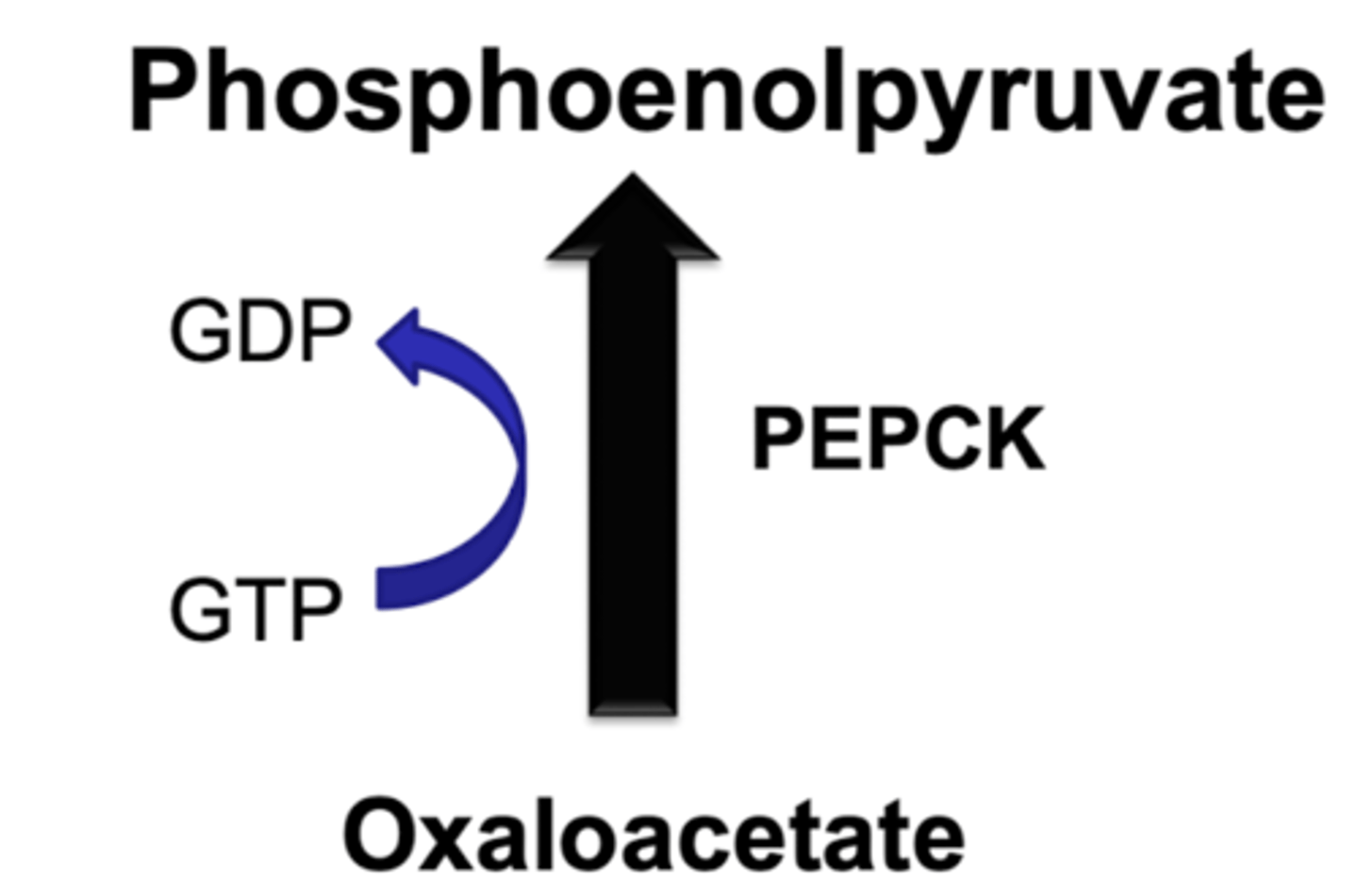
PEPCK regulation
amount of this enzyme
-liver controls expression based on fed or fasted state
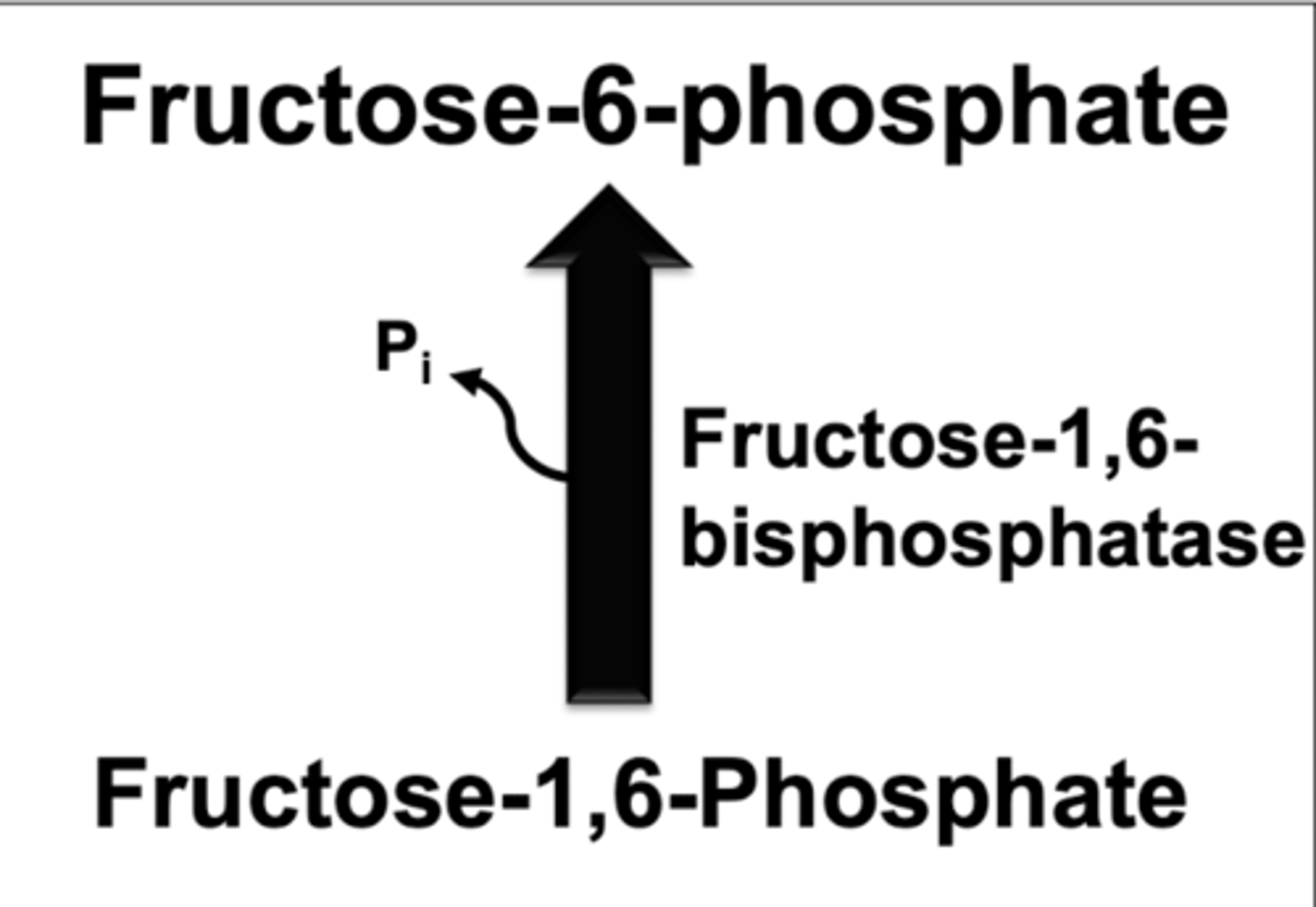
fructose 1,6 biphosphatase regulation
OPPOSITE OF PFK-1
activated by
-ATP
-citrate
inhibited by:
-fructose 2,6 bisphosphate
-AMP and ADP
glucose 6 phosphatase
*do NOT have in skeletal muscle
activated by:
-glucose-6-phosphate
-glucagon
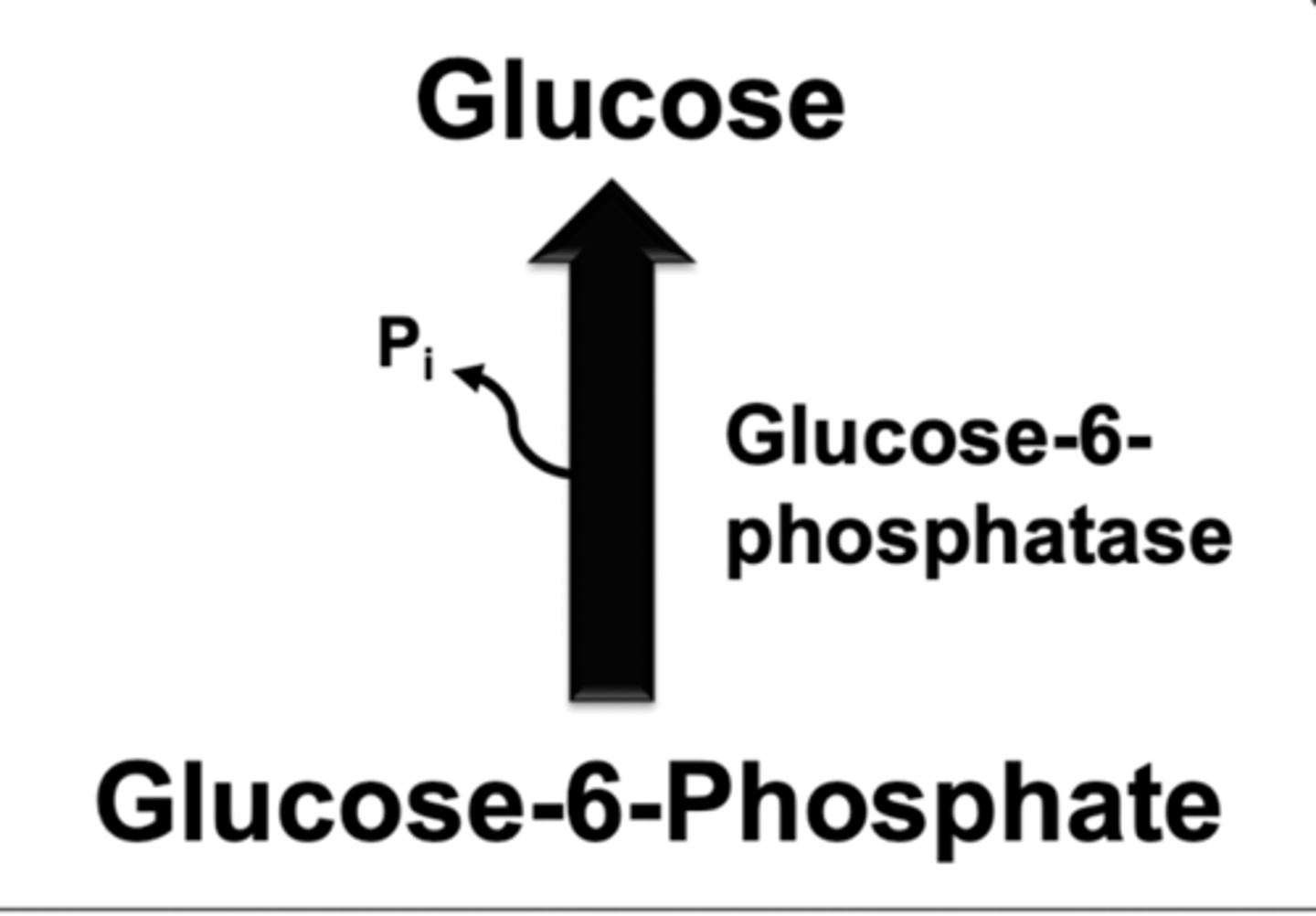
what effect does diabetes have on gluconeogenesis
insulin resistance leads to
-increased fat metabolism
-increased protein breakdown
-increased levels of glucagon
**increased gluconeogenesis- overproducing glucose
pentose phosphate pathway
production of 5 carbon monosaccharides and NADPH
-5 carbon sugars (ribose-5-phosphate) used for nucleotide and nucleic acid formation
-NADPH for biosynthetic reactions
*can take glucose-6-phosphate and shunt it into NADPH and 5C sugars
-no hormonal regulation
NADPH reactions
-fatty acid synthesis
-production of steroids
-reduction of glutathione in RBC
*used as a protective mechanism against oxidative stress- every cell needs
PPP occurs
more in the fed state
-synthesis of lipids
*utilizing extra carbons
REGULATED BY TISSUE NEED NOT HORMONES