CARBONYL COMPOUNDS
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
OXIDATION of Primary & Secondary Alcohols
oxidation of primary alcohols and aldehydes
Oxidising agents: acidified K2Cr2O7 or acidified KMnO4 , both need reflux condition (heating)
Oxidising agents themselves get reduced → causing solutions to change color:
+ In K2Cr2O7, the orange dichromate ions (Cr2O72-) → green Cr3+ ions
+ In KMnO4, purple maganate ions (MnO4-) → colorless Mn2+ ions
(1) Primary alcohol [o] → (2) Aldehyde [o] → (3) Carboxylic acid
(1) Secondary alcohol [o] → (2) Ketones
[o]: acidified K2Cr2O7 or acidified KMnO4
![<ul><li><p><strong>oxidation </strong>of <strong>primary alcohols and aldehydes</strong> </p></li></ul><ul><li><p><strong>Oxidising agents: acidified K<sub>2</sub>Cr<sub>2</sub>O<sub>7 </sub>or acidified KMnO<sub>4 </sub>, both need reflux condition (heating) </strong></p></li><li><p>Oxidising agents themselves get reduced → causing solutions to change color: </p></li></ul><p>+ In K<sub>2</sub>Cr<sub>2</sub>O<sub>7, </sub>the orange dichromate ions (Cr<sub>2</sub>O<sub>7</sub><sup>2-</sup>) → green Cr<sup>3+</sup> ions </p><p>+ In KMnO<sub>4</sub>, purple maganate ions (MnO<sub>4</sub><sup>-</sup>) → colorless Mn<sup>2+ </sup>ions </p><p></p><p>(1) Primary alcohol [o] → (2) Aldehyde [o] → (3) Carboxylic acid </p><p><strong>(1) Secondary alcohol [o] → (2) Ketones </strong></p><p>[o]: <strong>acidified K<sub>2</sub>Cr<sub>2</sub>O<sub>7 </sub>or acidified KMnO<sub>4</sub></strong></p>](https://assets.knowt.com/user-attachments/9a5c5469-0219-4940-9a4a-68d7f9204359.png)
SYNTHESIS of Aldehydes
(1) Warm primary alcohol is slowly added to the oxiding agent
(2) The formed aldehyde has a lower boiling point than the alcohol → distilled off as soon as it forms
(3) Condenses into liquid and collected
→ primary alcohol + [o] (heat) → aldehydes + water
![<p>(1) Warm primary alcohol is slowly added to the oxiding agent </p><p>(2) The formed aldehyde has a lower boiling point than the alcohol → distilled off as soon as it forms </p><p>(3) Condenses into liquid and collected </p><p>→ primary alcohol + [o] (heat) → aldehydes + water </p>](https://assets.knowt.com/user-attachments/77975e91-a8cf-4448-8c99-40a685e8ab08.png)
SYNTHESIS of Ketones
(1) warm secondary alcohol is slowly added to the oxidising agent
(2) The ketone cannot be further oxidised → it does not need to be distilled off straight away after it has been formed.
→ secondary alcohol + [o] (heat)→ ketones + water
![<p>(1) warm secondary alcohol is slowly added to the oxidising agent </p><p>(2) The ketone cannot be further oxidised → it does not need to be distilled off straight away after it has been formed. </p><p>→ secondary alcohol + [o] (heat)→ ketones + water </p>](https://assets.knowt.com/user-attachments/1c6833ae-6f01-4eb1-9208-f3c55eaa781e.png)
REDUCTION of Aldehydes & Ketones
to form Primary Alcohol
Reducing Agents or [H]: NaBH4 OR LiAlH4
Aldehydes + [H] → Primary Alcohol
Ketones + [H] → Secondary Alcohol
![<ul><li><p>to form Primary Alcohol </p></li><li><p><strong>Reducing Agents or [H]: NaBH<sub>4</sub> OR LiAlH<sub>4</sub></strong></p></li><li><p>Aldehydes + [H] → Primary Alcohol </p></li></ul><ul><li><p>Ketones + [H] → Secondary Alcohol </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/d478a527-623f-4d04-9db5-04582977c259.png)
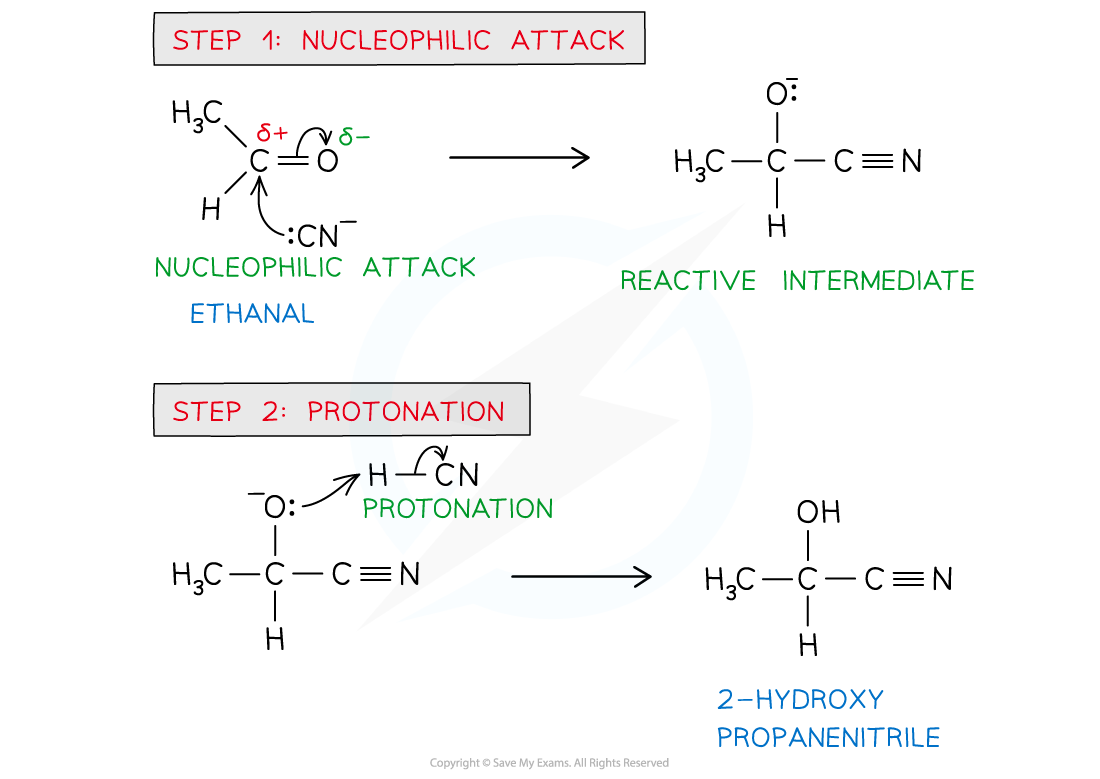
Aldehydes & Ketones with HCN (Hydrogen cyanide)
NUCLEOPHILLIC ADDITION
CN- acts as nucleophile and adds across the C=O bond
Catalyst: KCN
Condition: Heat
Product: Hydroxynitriles (nitriles containing a hydroxy, -OH, group)

Mechanism of nucleophilic addition of HCN with aldehydes and ketones
Test for Carbonyl compound
Reagent: 2,4-dinitrophenylhydrazine (2,4-DNPH)
Condensation - H2O is eliminated
Positive = deep-orange ppt, can be purified by recrystallization

Identifying Carbonyls: Fehling’s solution
Agent: Fehling’s solution
Clear blue, alkaline solution containing copper(II) ions acts as oxidising agent
Oxidisation
Warmed with aldehydes → aldehydes oxidised to carboxylic acid → Cu2+ is reduced to Cu+
Positive: turns opaque, red ppt formed
Ketones cannot be oxidised → negative result
Identifying carbonyl: Tollen’s reagent
aqueous alkaline solution of silver nitrate in excess ammonia
Oxidation
When warmed with aldehydes, aldehyde is oxides → carboxylic acids → the Ag+ are reduced to Ag atoms
Positive = silver ‘mirror’
Ketones cannot be oxidised → negative result