CHEM 1040 Forbes Exam 3 Review
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
Common-ion effect
a decrease in the solubility of an ionic compound caused by the addition of a common ion
Henderson-Hasselbalch equation
pH = pKa + log [A-]/[HA]
When [A-] = [HA], then
pH = pKa
When [A-] > [HA], then
pH > pKa
When [A-] < [HA], then
pH < pKa
What is a buffer solution?
A system that resists pH changes when small amounts of an acid or a base are added
Steps to solve buffer problems
Stoichiometry table, then either ICE tables or HH equation
Buffer capacity
the amount of acid or base that can be added without causing a large change in pH
Buffer systems are most effective when concentrations are high/low
high
A buffer becomes less effective as the difference in ___ increases
concentration
An effective buffer must have a base:acid range of
0.10-10
The more concentrated the buffer components, the more...
effective the buffer
The effective range for a buffering system is
one pH unit on either side of pKa
Buffer capacity increases/decreased with increasing/decreasing concentrations of buffer components
increases; increasing
The overall buffer capacity increases/decreases as the ratio of buffer components gets closer to ___
increases; 1
Titration
when a substance in a solution of known concentration is reacted with another substance in a solution of unknown concentration
Titration curve
a plot of the pH of a solution as a function of the volume of titrant added
Strong Acid - Strong Base Titration Curve
Equivalence point is 7
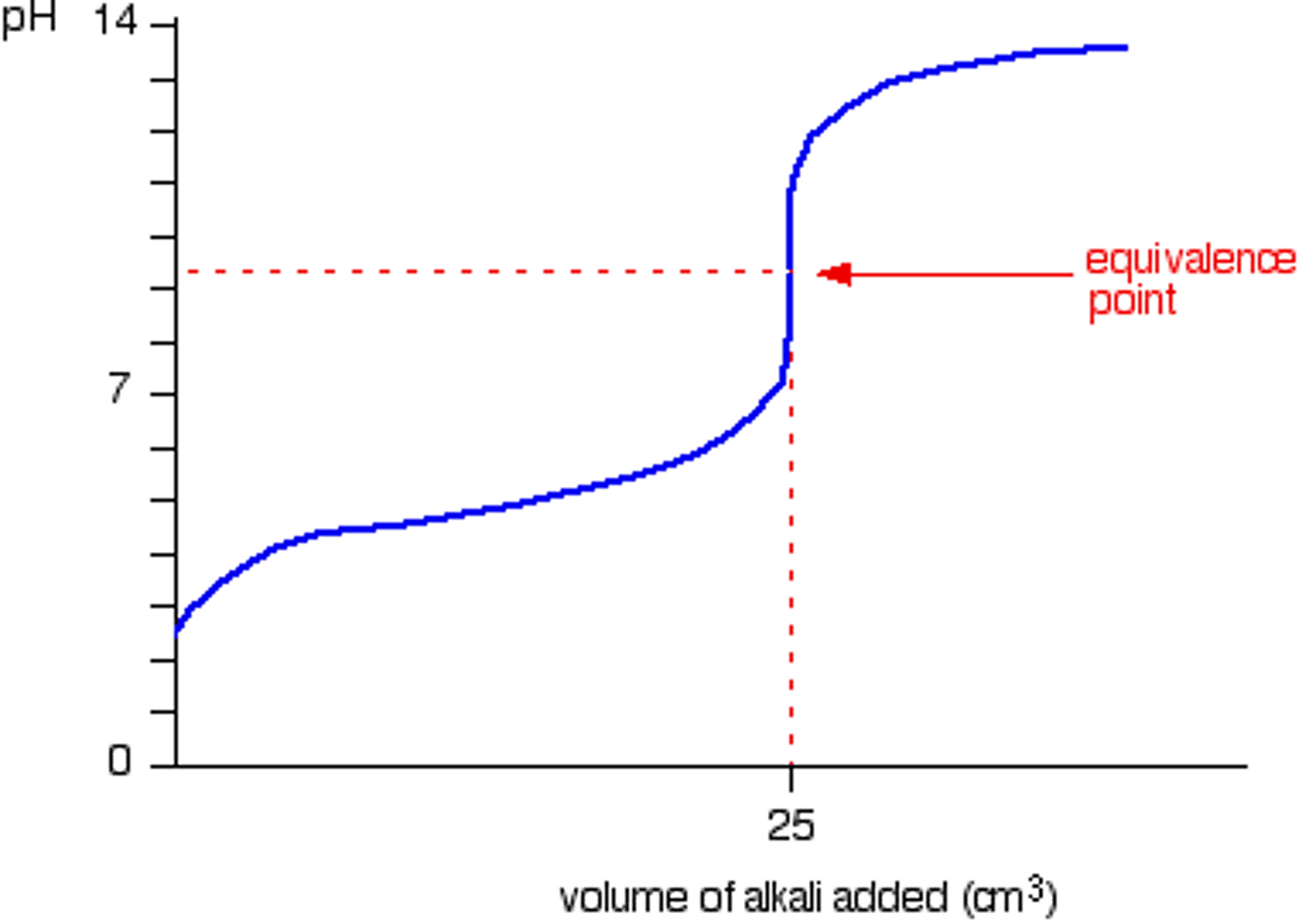
Weak Acid - Strong Base Titration Curve
Equivalence point is greater than 7, has a halfway equivalence point
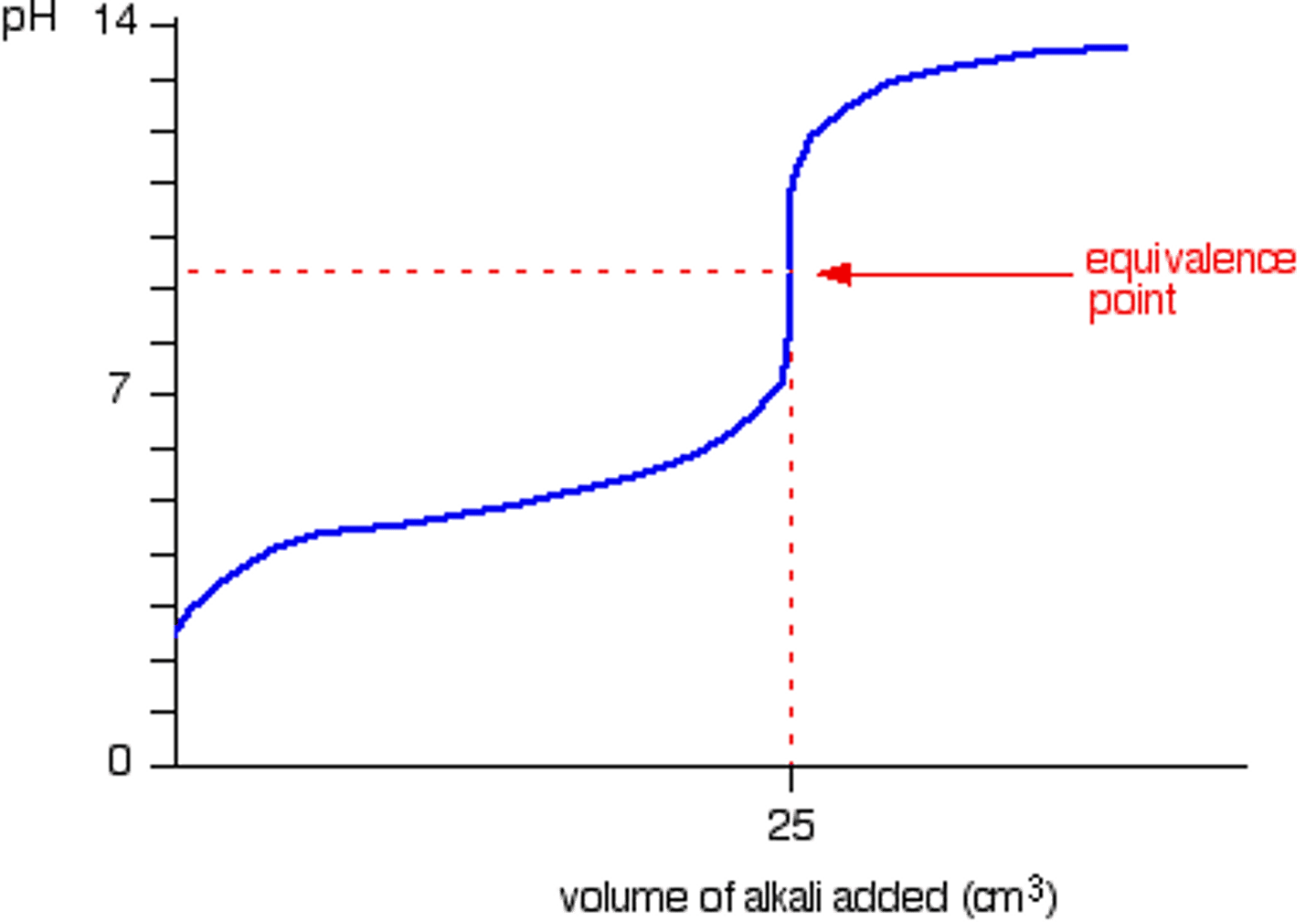
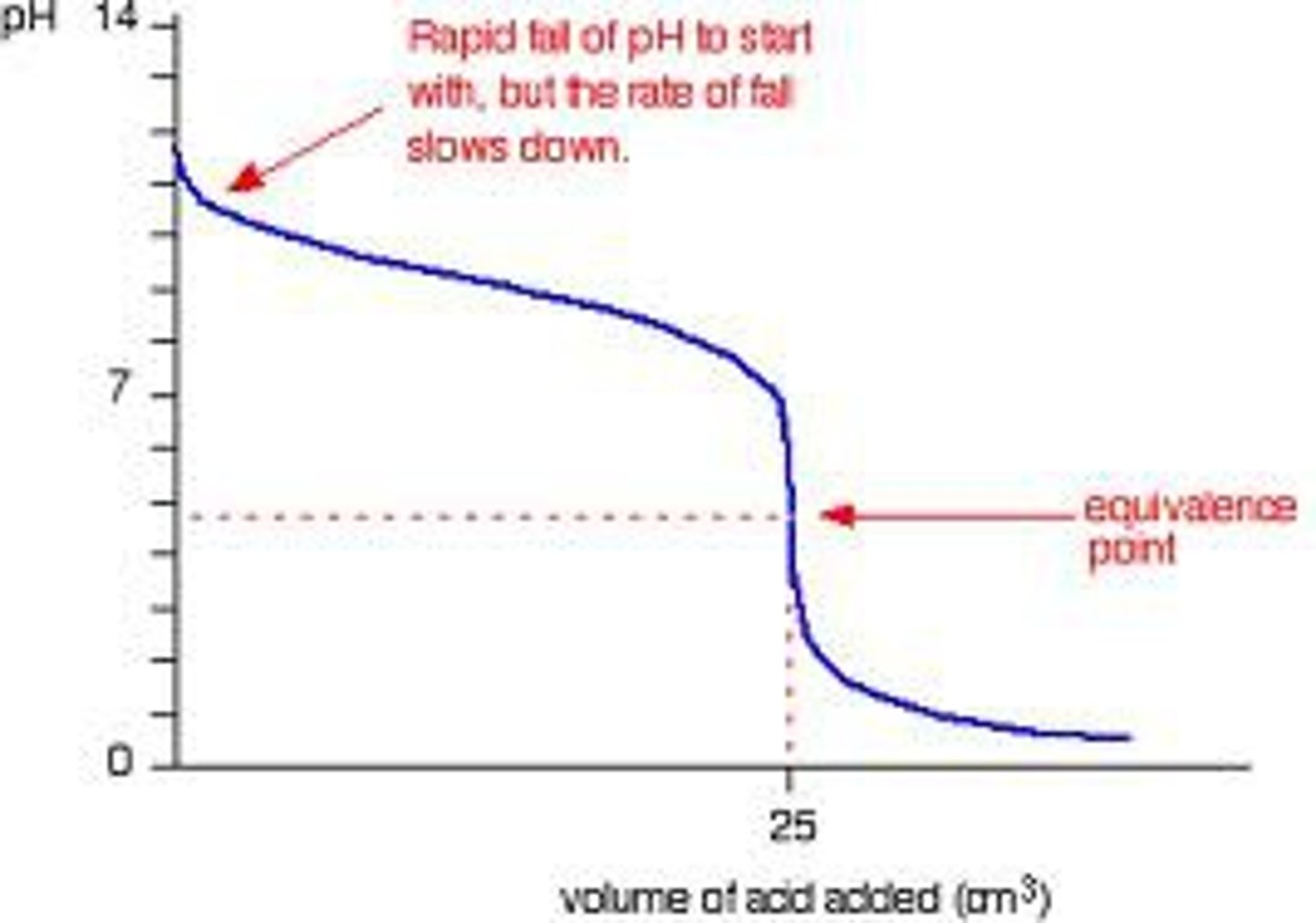
Weak Base - Strong Acid Titration Curve
Equivalence point is less than 7
Solubility product constant (Ksp) formula
Equals the product of the concentrations of the ions each raised to a power equal to the coefficient of the ion in the dissociation equation
Precipitation reaction
occurs when two aqueous solutions containing ionic compounds are mixed and one of the products is insoluble
If Q = Ksp, the solution is
saturated
If Q < Ksp, the solution is
unsaturated
If Q > Ksp, the solution is
supersaturated
_______ tend to be good electron acceptors, which makes them a good ___ acid
Transition metals; Lewis acid
Most common Lewis bases that increase the solubility of metal cations are
NH3, CN-, and OH-
6 Strong Acids
HCl, HBr, HI, HNO3, H2SO4, HClO4
6 Strong Bases
LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
Spontaneous process
a process that proceeds without outside intervention
Nonspontaneous process
a process that requires continuous outside intervention
Not all spontaneous processes are
self-starting, fast, and exothermic
The nonspontaneous formation of solid ice from liquid water is due to...
the restriction of the freedom of motion of the molecules
Decrease temperature, increase/decrease in freedom of motion
decrease; this makes the reaction spontaneous
What are the three types of freedom of motion for liquids and gases?
vibrational, rotational, and translational
Whenever an isolated thermodynamic system undergoes a change in which the particles in the system disperse into a larger volume and/or gain freedom, the process is always ____ and the system is said to have an increase in ____
spontaneous; entropy
Entropy formula (STATE function)
△Sfinal -- △Sintial
Entropy definition
measure of energy dispersal
Substances that are mixable will mix with each other spontaneously because....
there is a higher probability of a mixed distribution
The most probable mixing pattern produces...
a uniform distribution of particles throughout the volume they occupy
Increase in disorder, increase/decrease in entropy
increase
Entropy formula
S = klnW
Accessible microstates
Probable number of arrangements of particles in a system at a given temperature
Macrostate
overall state of the system defined by a set of conditions
Microstate
snapshot of a system at an instant in time
Absolute entropy
a crystalline solid in their lowest possible energy state (zero) only has one microstate
Standard molar entropy
the entropy value for a mole of a substance in its standard state
Larger mass, larger/smaller the entropy
larger
Entropy has a larger increases between ___ and ___ than solids and liquids
liquids and gases; this is due to molecular dispersion
Heating a substance at higher temperatures produce a smaller entropy increase than...
adding the same quantity of heat at a lower temperautre
Entropy increases with what three variables?
temperature, volume, and number of atoms
Entropy formula for state change at equilibrium
ΔS = qrev/T
If the ΔSuniv > 0, then a process is
spontaneous
If the ΔSuniv < 0, then a process is
nonspontaneous
ΔSsurr formula
ΔS = -ΔHsys/T
A process that emits heat into surroundings increases/decreases the entropy
increases
A process that absorbs heat from the surroundings increases/decreases the entropy
decreases
The magnitude of the change in entropy is _____ to the magnitude of ΔHsys
proportional
ΔSuniv formula
ΔSsys - ΔHsys/T
Gibbs free energy (G)
amount of energy available to do work
ΔGsys formula
-TΔSuniv
ΔG formula
ΔH - TΔS
ΔG is proportional to the positive/negative sign of ΔSuniv
negative
A decrease in Gibbs free energy corresponds to a ___ process
spontaneous
An increase in Gibbs free energy corresponds to a ___ process
nonspontaneous
ΔG formula under standard conditions
ΔHrxn - TΔSrxn
ΔG formula at thermodynamic equlibrium
ΔGstd + RTlnQ
Redox reaction
a type of chemical reaction that involves a transfer of electrons between two species
Balancing Chemical Equations in Neutral Solution Steps:
1. Separate into half reactions
2. Neutralize charges with e-
3. Scale the reactions so the number of electrons are equal
4. Add equations together and net ionic equation
Balancing Chemical Equations in Acidic Solution Steps:
1. Separate into half reactions
2. Balance elements other than O and H
3. Add water to balance O, add H+ to balance H
4. Balance charges with e-
5. Scale reactions so the number of e- are equal
6. Add equations together and get net ionic equations
Balancing Chemical Equations in Basic Solution Steps:
1. Separate into half reactions
2. Balance elements other than O and H
3. Add water to balance O, add H+ to balance H
4. Balance charges with e-
5. Scale reactions so the number of e- are equal
6. Cancel e-
7. Add OH- to balance H+
8. Combine OH and H+ to form water
9. Cancel common terms
Electrical current
flow of electric charge
Voltaic cell
an electrochemical cell that produces electrical current from a spontaneous reaction