MBIO L2 - chemical principles
1/143
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
144 Terms
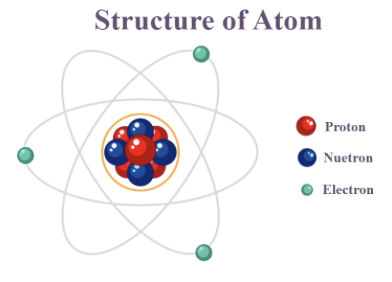
what is an atom?
the smallest unit of matter which can partake in a chemical reaction
what is an element?
give examples
a substance made up of only one type of atom that cannot be broken down into simpler substances by chemical means
ex: Carbon (C), Nitrogen (N), Oxygen (O)
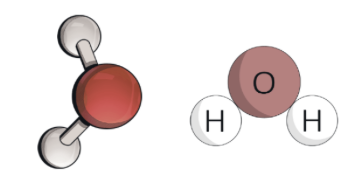
what is a molecule?
a group of two or more atoms bonded together
can the atoms in a molecule be the same?
yes, for example:
N₂ (N = nitrogen, ₂ = two nitrogen atoms)
O₂ (o = oxygen, ₂ = two oxygen atoms)
can the atoms in a molecule be different?
yes, for example, H₂O
H₂ = two hydrogen atoms
O = one oxygen atom
what is every atom composed of?
every atom is composed of a single centrally located nucleus which contains protons and neutrons and are surrounded by electrons in orbitals.
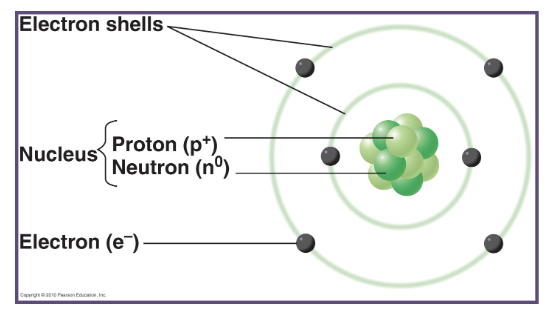
what charge do protons carry?
a positive charge
what charge do neutrons carry?
they do not carry any charge
why does the nucleus carry a net positive charge?
since neutrons do not have any charge, the nucleus will always carry a net positive charge
do nuclear contents participate in chemical reactions?
nuclear contents do not participate in chemical reactions and is thus referred to as stable
where do electrons exist in an atom?
electrons exist outside of the nucleus in specialized regions of space called orbitals
what charge do electrons carry?
a negative charge
what happens to an atom that loses an electron?
an atom that loses an electron will have a positive change
what happens when an atom gains an electron?
an atom that gains an electron will have a negative charge
when is an atom referred to as an ion?
how does this occur?
when an atom has an overall positive or an overall negative charge, it is no longer neutral and is referred to as an ion
this occurs due to the loss or gain of an electron
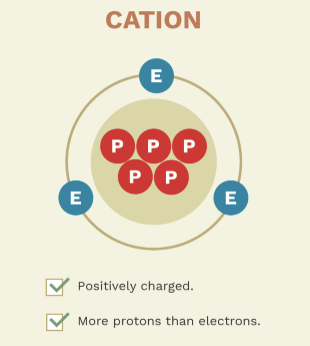
what are atoms who carry a positive charge called?
cations
give examples of cations
these — have lost —, resulting in a — charge
sodium (Na⁺)
potassium (K⁺)
calcium (Ca²⁺)
these ions have lost electrons, resulting in a positive charge.
what are atoms who carry a negative charge called?
anions
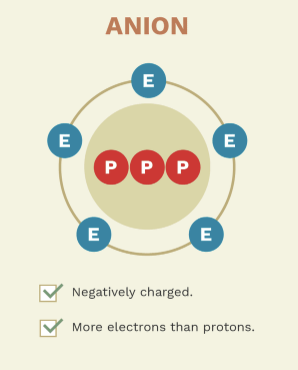
give examples of anions
chloride (Cl⁻)
sulfate (SO₄²⁻)
phosphate (PO₄³⁻)
these ions have gained electrons, resulting in a negative charge.
when do chemical bonds form between atoms?
when electrons in their outer shells interact with one another
what does every atom want to achieve?
every atom wants to achieve chemical stability by filling their outermost shell with electrons
how do atoms achieve a full compliment of electrons?
atoms achieve a full compliment of electrons (8) by combining to form molecules
how many types of chemical bonds are found in living systems?
three
what are the three types of chemical bonds found in living systems?
covalent bonds
ionic bonds
hydrogen bonds
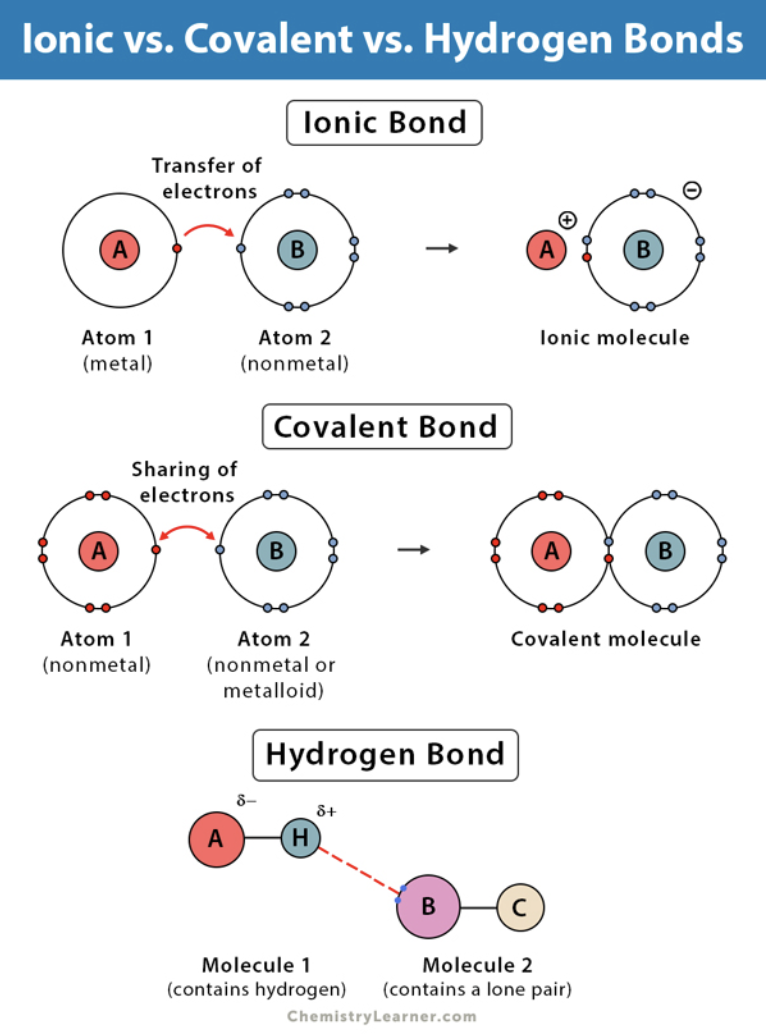
how are covalent bonds formed?
they are formed when two atoms come together and share electrons within their outer shells
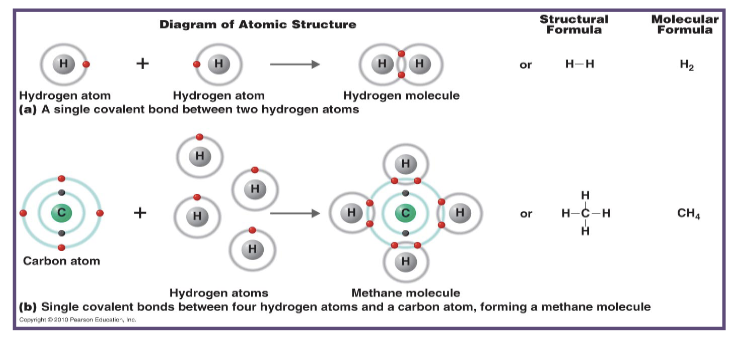
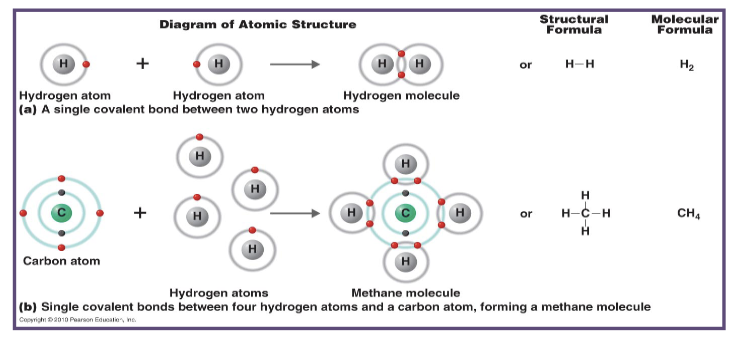
what happens to electrons in a covalent bonds?
electrons are shared within the outer shell of the atoms involved
what is formed as a result of covalent bonds between hydrogen and oxygen?
water is formed as a result of a covalent bond in between hydrogen and oxygen
how strong are covalent bonds?
very strong bonds
how are ionic bonds formed?
ionic bonds form as a result of the attractive force in between ions of opposite charge
what holds the two atoms together in an ionic bond?
the attractive force holds the two atoms together in the bond
what is an example of an ionic bond?
NaCl (sodium chloride)
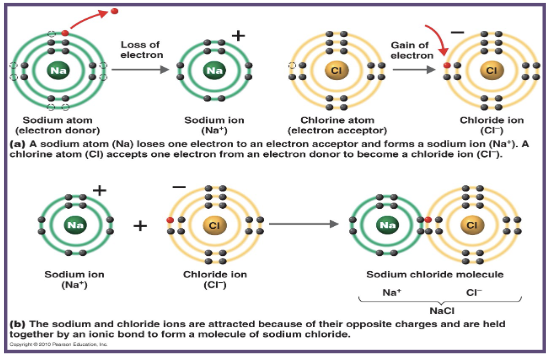
what does sodium (Na) require to achieve stability?
what does chlorine (Cl) require to achieve stability?
Na requires the loss of an electron to achieve stability
Cl requires the gain of an electron to achieve stability
how does chlorine obtain the electron in an ionic bond with sodium?
Ch (chlorine) receives the electron from Na (sodium) rather than sharing it
what ions are formed when Na and Cl form an ionic bond?
Cl becomes Cl⁻ and Na becomes Na⁺
how strong are ionic bonds compared to covalent bonds?
ionic bonds are much weaker than covalent bonds
when are hydrogen bonds form?
these bonds form when hydrogen is covalently bound to oxygen or another electron loving molecule such as nitrogen
why does the atom bound to hydrogen hold the shared electrons closer to its nucleus?
since the atom bound to hydrogen is electron loving, it holds the shared electrons closer to its nucleus than hydrogen

why are electrons not equally shared between hydrogen and oxygen?
oxygen is more electron loving than hydrogen, thus the electrons are not equally shared in between the two atoms and are held more closely to the oxygen nucleus

why are electrons equally shared between hydrogen and carbon (H : C)?
carbon is not more electron loving than hydrogen, thus the electrons are equally shared in between the two atoms
in a hydrogen bond, what charge develops on the hydrogen atom when electrons are not equally shared?
a resulting partial positive charge on the hydrogen atom (δ⁺)
in a hydrogen bond, what charge develops on the electron loving atom?
a partial negative charge (δ⁻) on the electron loving atom
what are molecules that contain unequal sharing of electrons called?
molecules that contain unequal sharing of electrons are polar molecules
what is the partial positive charge on the hydrogen atom attracted to?
the partial positive charge on the hydrogen atom is attracted to partial negative charge on atoms within other molecules
how strong are hydrogen bonds compared to covalent and ionic bonds?
these bonds are weaker than both covalent and ionic bonds
what do hydrogen bonds result in?
hydrogen bonds result in networks of molecules of different molecules
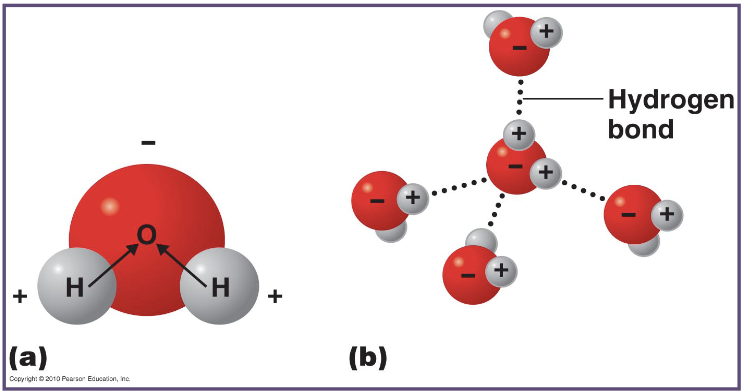
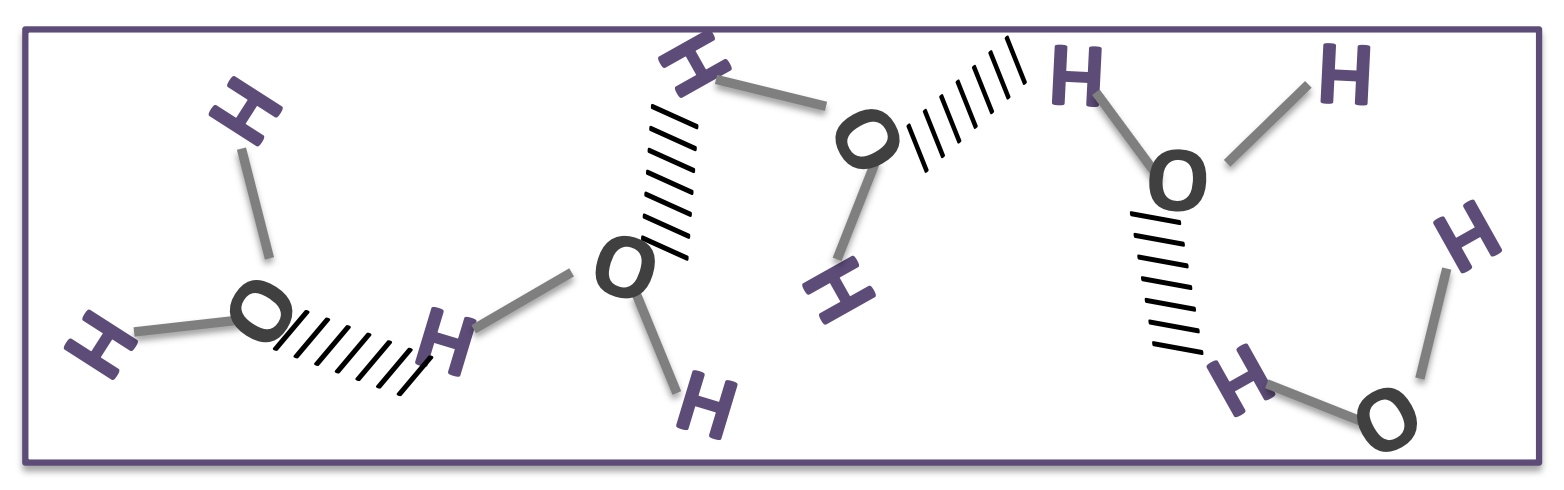
what is happening here?
what do the dotted lines represent?
it shows hydrogen bonds forming between water molecules due to attraction between partial positive hydrogen atoms and partial negative oxygen atoms in other molecules
the dotted lines represent hydrogen bonds
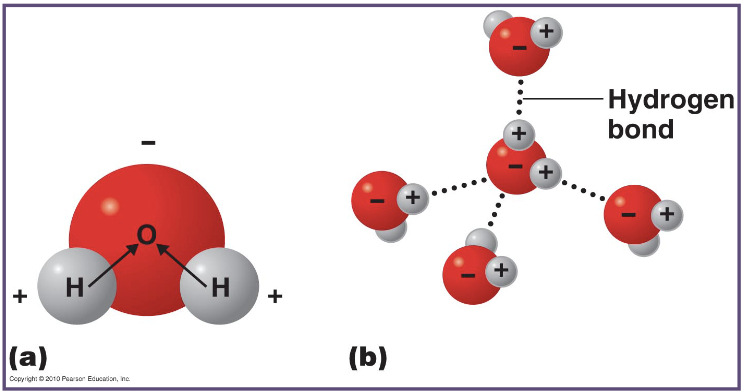
what are the main difference between covalent, ionic and hydrogen bonds?
covalent bonds shares electrons & have strong bonds
ionic bonds transfers electrons & have weaker bonds than covalent bonds
hydrogen bonds are weak attractions between a hydrogen atom and an electronegative atom & have weaker bonds compared to both covalent and ionic bonds
what are organic compounds?
organic compounds are compounds which contain carbon (C)
what are inorganic compounds?
inorganic compounds are compounds that do not contain carbon (C)
why do organisms require inorganic compounds?
organisms require inorganic compounds in order to live and reproduce
what is the most important inorganic compound in living systems?
water (H₂O)
approximately how much of all cell weight does water account for?
approximately 75% of cell weight
why is water considered a solvent?
since water is used to dissolve materials, it is a solvent
what is a polar solvent?
a solvent in which electrons are not shared equally, resulting in partial charges that allow it to dissolve materials
why is water considered as a polar solvent?
because electrons are not shared equally in between the hydrogen and the oxygen atoms, it is a polar solvent
why are living things able to make such good use of water?
because water is an excellent solvent
what does it mean that water is an excellent solvent?
it can dissolve many ionic compounds such as NaCl (sodium chloride)
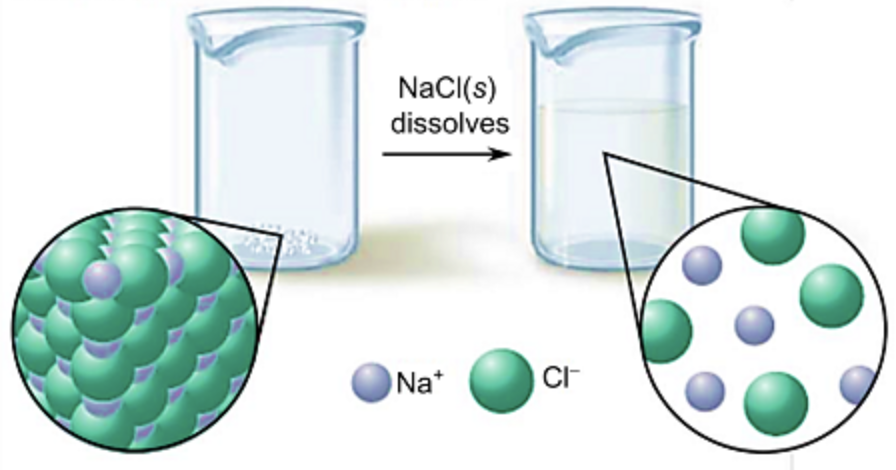
what happens to Na⁺ and Cl⁻ ions when NaCl dissolves in water?
the ions (Na⁺, Cl⁻) remain dissolved in the water and form a solution
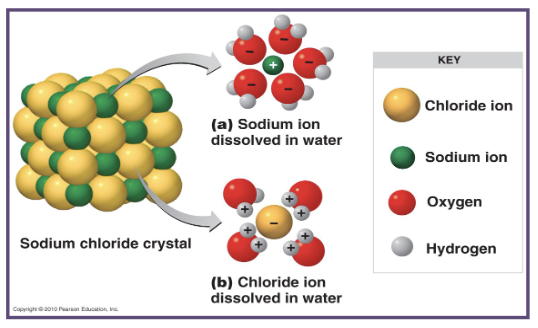
why does this solution form in water?
this solution forms because the partial positive regions within the water molecules surround the negative ions (Cl⁻) and the partial negative regions within the water molecule surround the positive ions (Na⁺), holding them in the solution
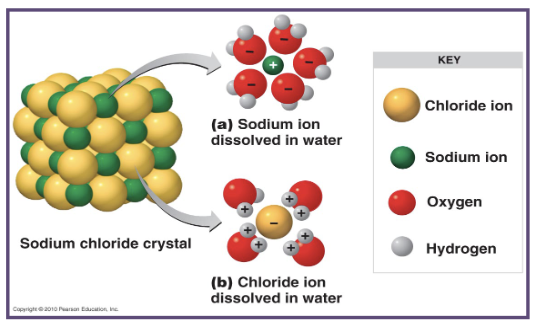
what is formed when ions remain dissolved in water?
a solution
every water molecule can form how many hydrogen bonds with other water molecules?
four hydrogen bonds with other water molecules
what does hydrogen bond allow water to form?
this excellent ability to hydrogen bond allows water to form networks of interconnected water molecules.
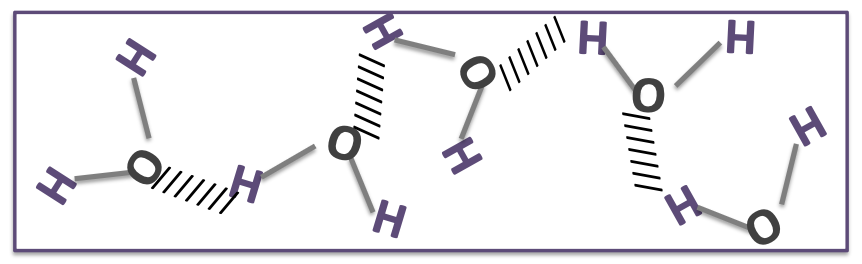
what does the formation of networks of interconnected water molecules give water?
it gives water a very high boiling point
what is water’s high boiling point?
100°C
what does water’s high boiling point (100°C) keep water in?
it keeps water in it’s liquid state
where does water remain in its liquid state because of its high boiling point?
in most of the places that it is found on earth
what are the three states of water?
solid
liquid
gas
what process occurs between solid and liquid water?
freezing & melting
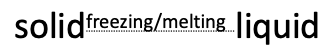
what process occurs between liquid and gas water?
boiling & evaporating

why is water a great temperature buffer?
water is a great temperature buffer because of its strong hydrogen bonds
what kind of bonds make water a great temperature buffer?
strong hydrogen bonds
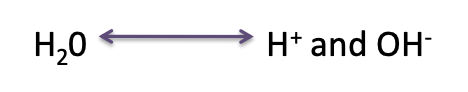
what is water easily split into?
water is easily split into its component ions H⁺ (hydrogen ions) and OH (hydroxide ions)
what are the component ions of water?
H⁺ (hydrogen ions) and OH⁻ (hydroxide ions)
what does this characteristic of splitting water into components allow it to do?
this characteristic allows water to play key roles in many chemical reactions
what equation shows water being split into its component ions?
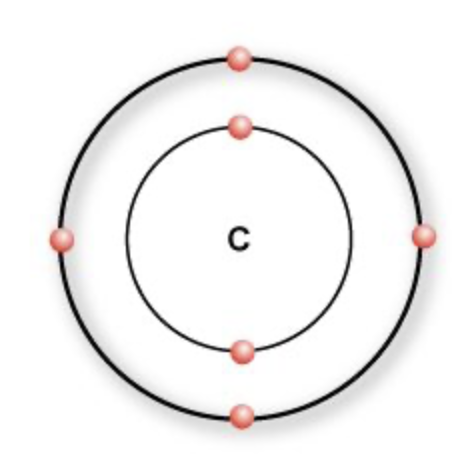
what do the four outer electrons of carbon (C) allow it to form?
four covalent bonds with a large number of different atoms
what can carbon (C) form four covalent bonds with?
with a large number of different atoms
how many major classes of organic compounds are found in living things? what are they?
four major classes:
carbohydrates
lipids
proteins
nucleic acids
what is the first major class of organic compounds found in living things?
what is the second major class of organic compounds found in living things?
what is the third major class of organic compounds found in living things?
what is the fourth major class of organic compounds found in living things?
first major class: carbohydrates
second major class: proteins
third major class: lipids
fourth major class: nucleic acids
what are carbohydrates in terms of energy?
this is an energy source that is immediately utilizable by cells
what type of compounds are carbohydrates?
polar compounds
what happens to carbohydrates because they are polar compounds?
they dissolve in water
what elements do all carbohydrates have?
what is the usual ratio of elements in carbohydrates?
C (carbon)
H (hydrogen)
O (oxygen)
usually in a 1:2:1 ratio (C₁H₂O₁)
how many major groups do carbohydrates have? what are they?
three:
monosaccharides
disaccaharides
polysaccharides
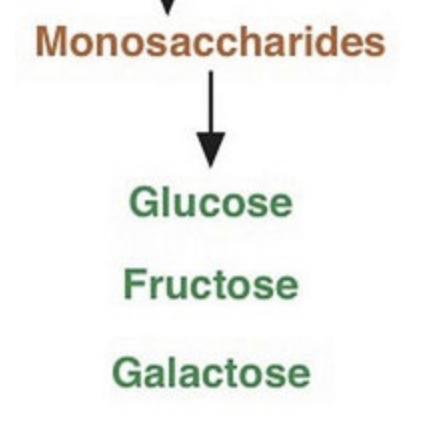
what does monosaccharides mean?
mono = one
saccharide = sugar
what are some examples of monosaccharides?
glucose, fructose and galactose
what does disaccharide mean?
di = two
saccharide = sugar
how are disaccharides formed?
when two monosaccharides come together to form a disaccharide
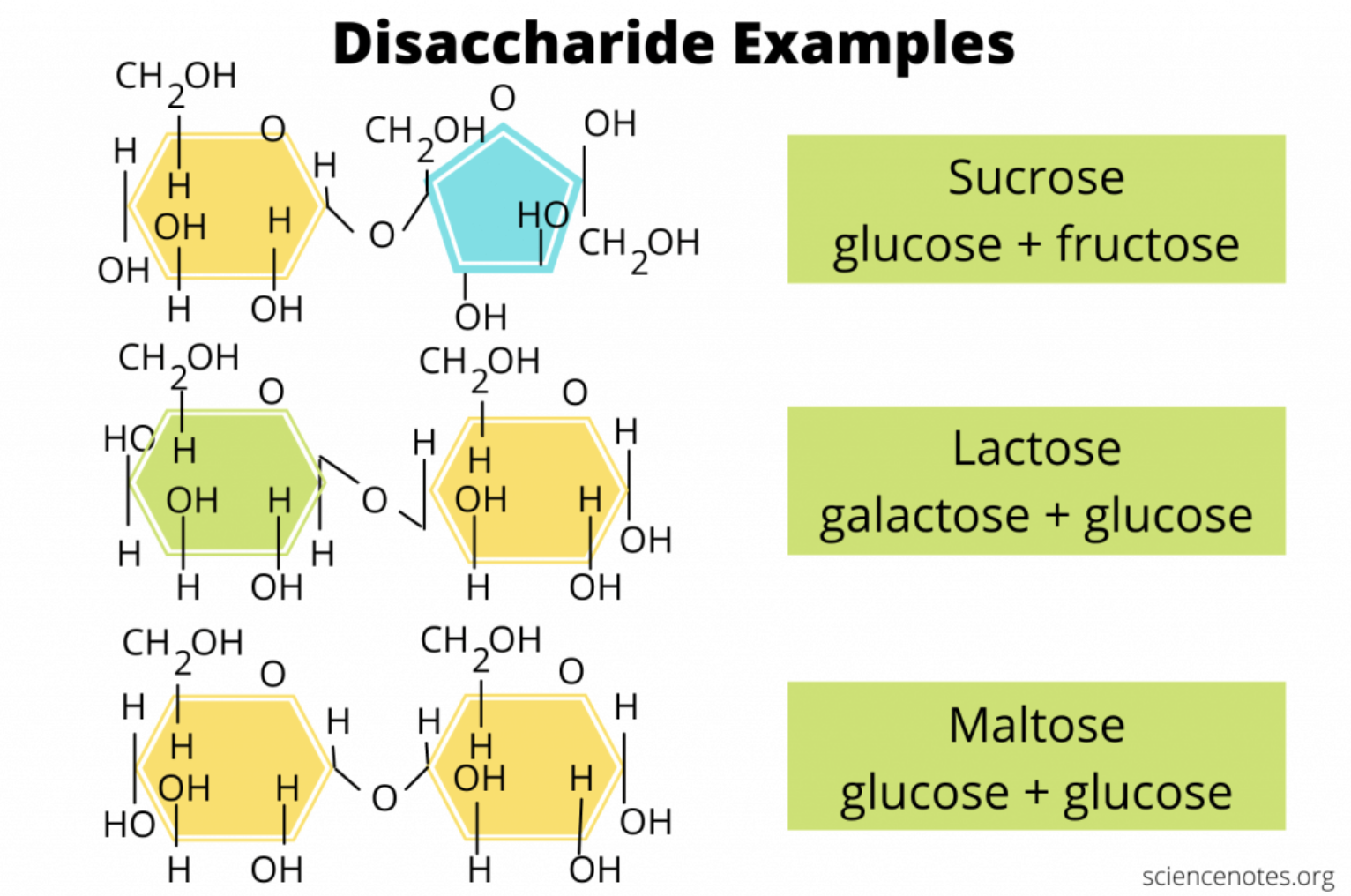
what are some examples of disaccharides?
glucose + fructose = sucrose
galactose + glucose = lactose
glucose + glucose = maltose
what does polysaccharide mean?
poly = many
saccharide = sugar
how are polysaccharides formed?
when hundreds of monosaccharides come together
are polysaccharides water soluble? explain
no, they are not water soluble because they are too large to be water soluble
what are some examples of polysaccharides?
starch, cellulose and glycogen
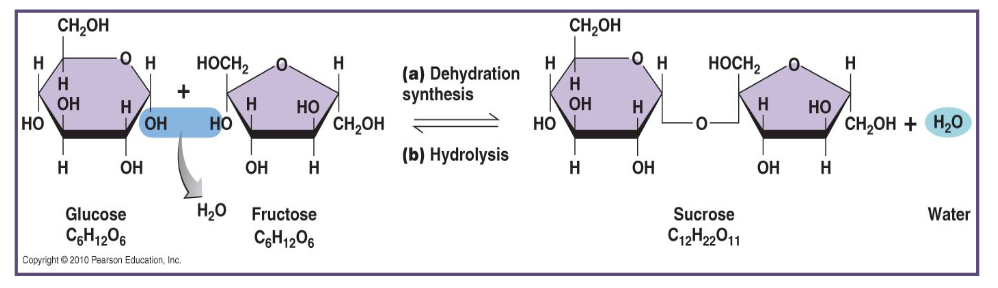
using this image, answer the following questions:
what two monosaccharides are shown coming together?
what is the chemical formula of glucose?
what is the chemical formula of fructose?
what process is shown when glucose and fructose comes together?
what molecule is released during dehydration synthesis?
what disaccharide is formed from glucose and fructose?
what is the chemical formula of sucrose?
what process breaks sucrose back into glucose and fructose?
what molecule is used during hydrolysis?
glucose and fructose
C₆H₁₂O₆
C₆H₁₂O₆
dehydration synthesis
H₂O (water)
sucrose
C₁₂H₂₂O₁₁
hydrolysis of sucrose
H₂O (water)
define:
dehydration synthesis
hydrolysis
dehydration synthesis:
a process where monosaccharides come together to form a larger molecule & releases water
hydrolysis:
a process where a larger molecule is broken apart & water is added
what does the lipid group include?
fats
phospholipids (complex lipids)
steroids
what are lipids essential to?
lipids are essential to the structure and function of membranes
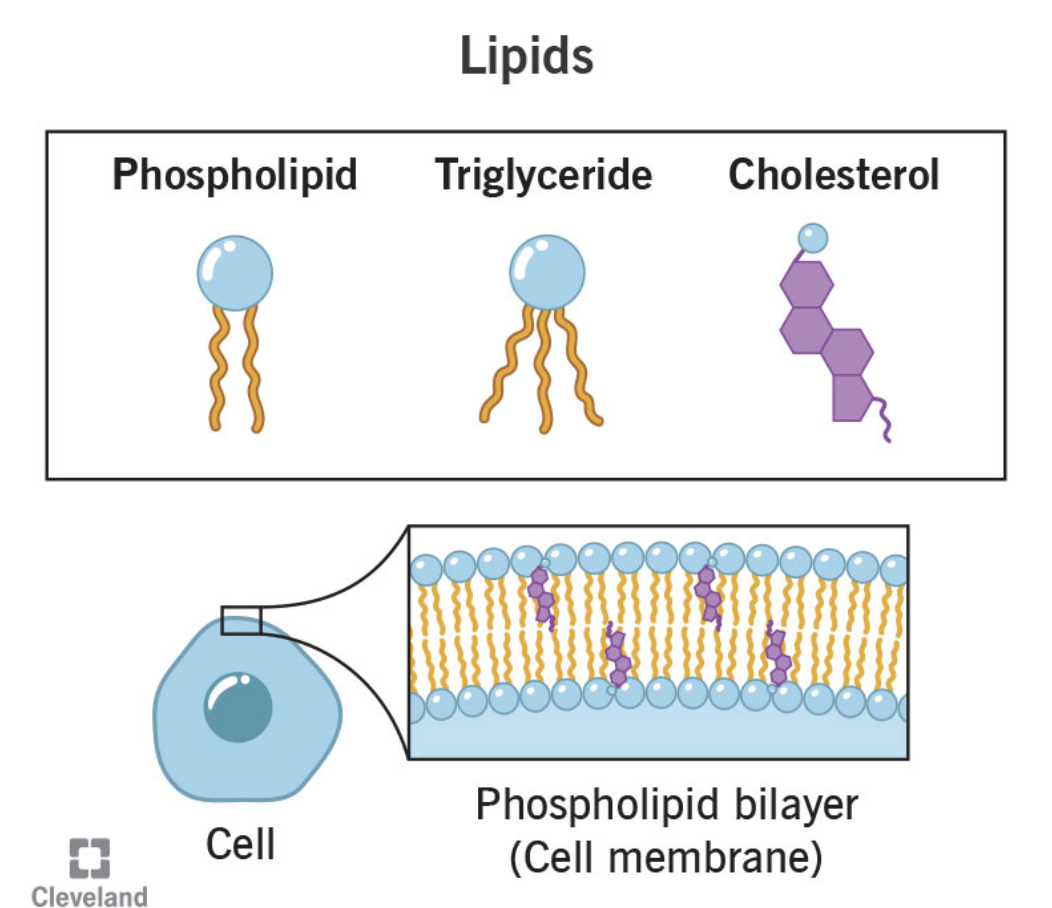
what part of the membrane do lipids comprise?
lipids comprise the phospholipid bilayer, which forms the fundamental structure of cell membranes.