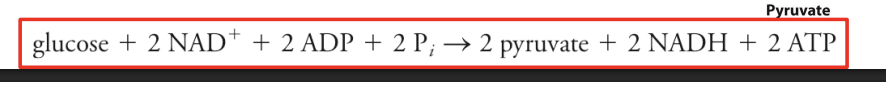Carbs You have to memorize + hints + biochem general test #2
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
D-Dihydroxyacetone (ketose or aldose) (draw the fisher projection and turn it to a hawthorn projection) - no hawthorn
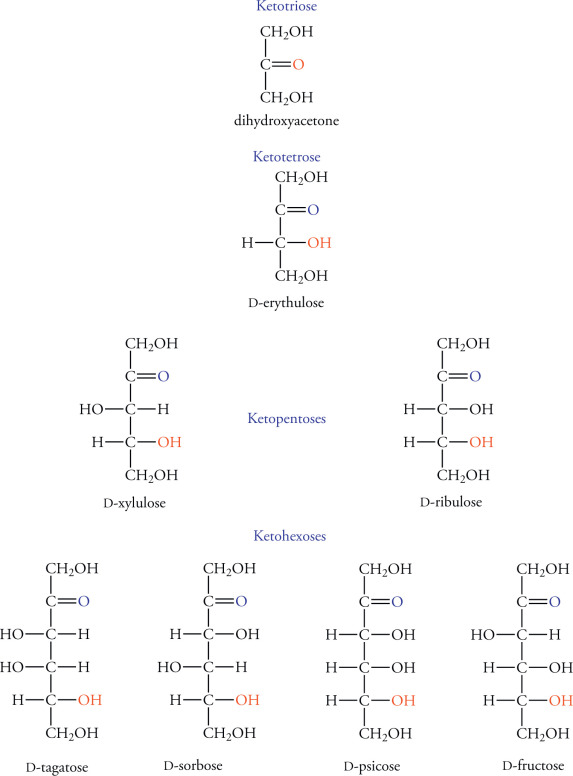
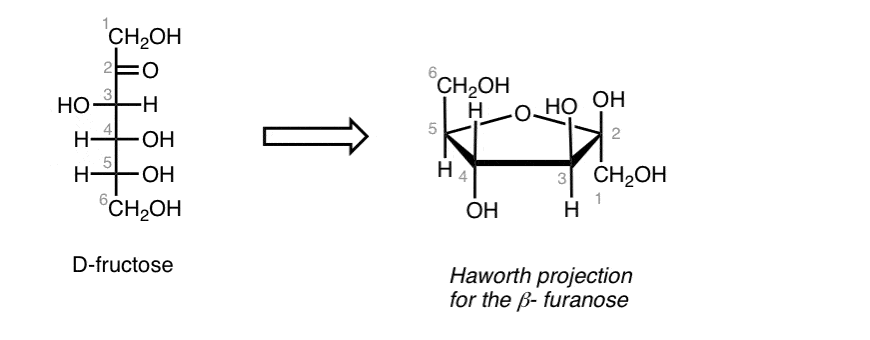
D- Fructos (ketose or aldose)(draw the fisher projection and turn it to a hawthorn projection)
didi
D- Aldoese (draw the fisher projection and turn it to a hawthorn projection)- okay what are teh differnt types of alsdos and how do you c harachterise them triose to hexose-
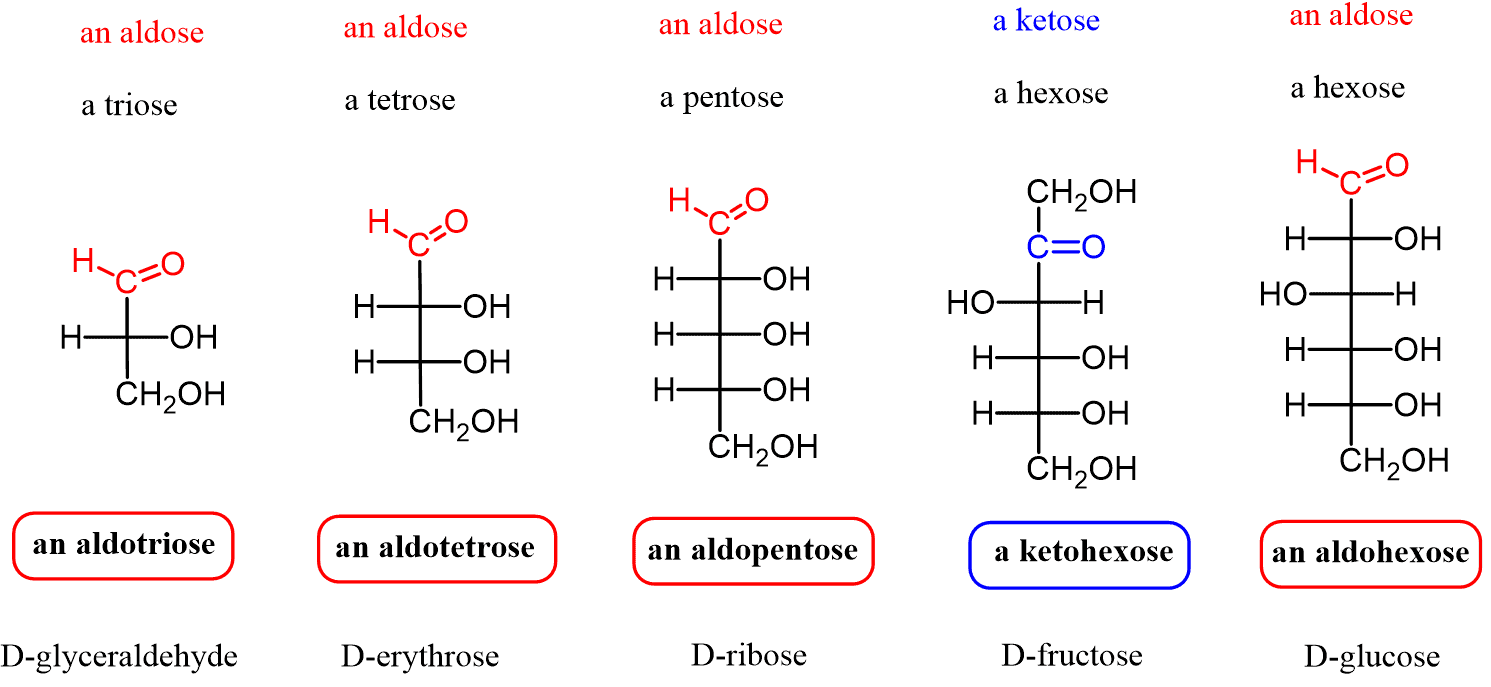
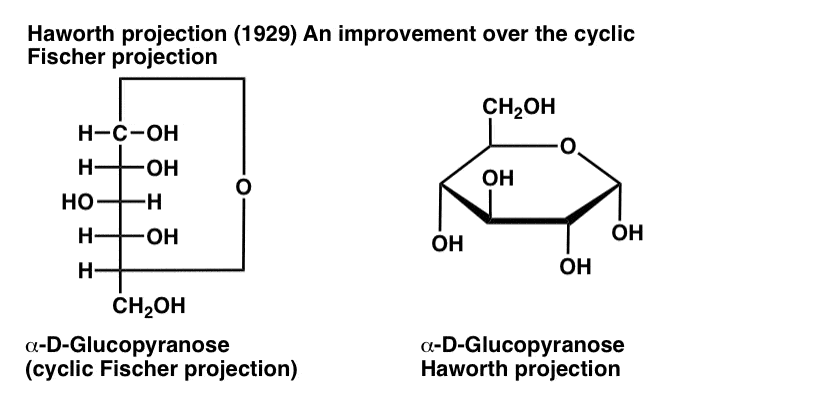
D- Glucose (draw the fisher projection and turn it to a hawthorn projection)
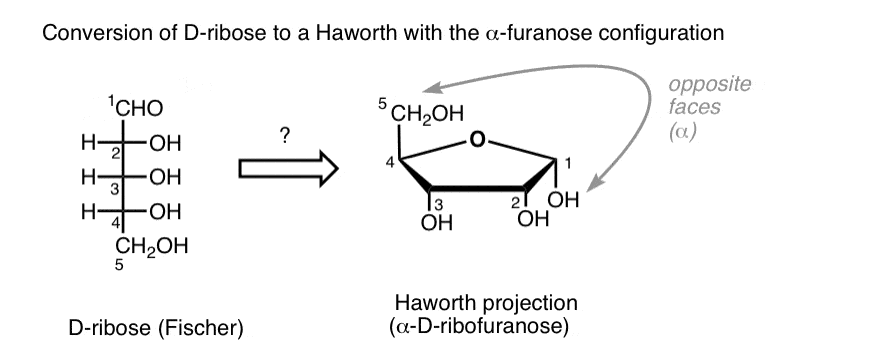
D- ribose (draw the fisher projection and turn it to a hawthorn projection)
whwhw
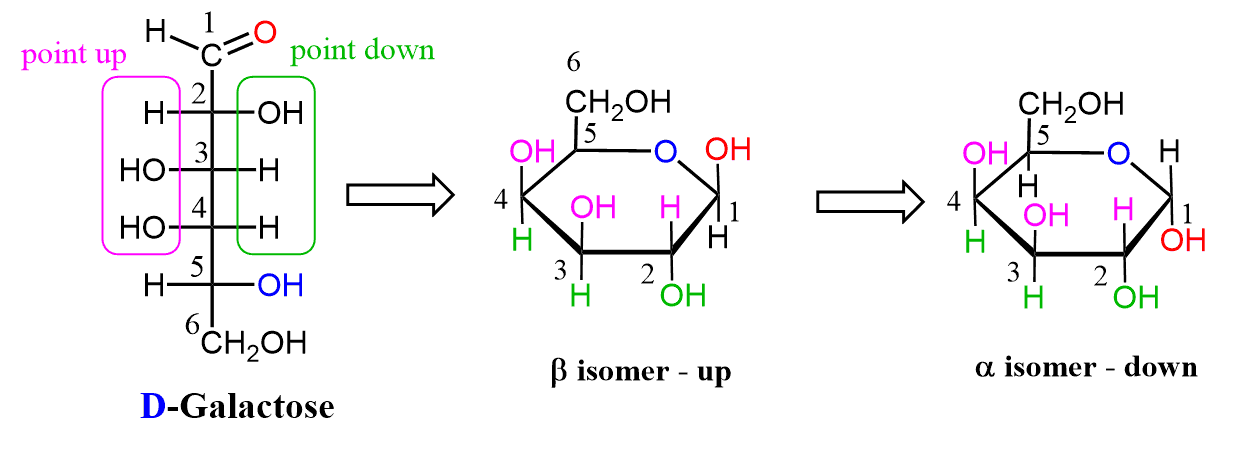
D- galactose(draw the fisher projection and turn it to a hawthorn projection)
pray for me
How to tell teh difference between a ketose and an aldose

How do you tell th diference betwen a d/l projection for fisher and hawthorn projection
fisher
D the trminal oh is to the right
L the terminal oh is to the left
Hawtorn
D- the terminal Ch2oh is facing Upwards
L- the terminal Ch2oh is facing Downwards
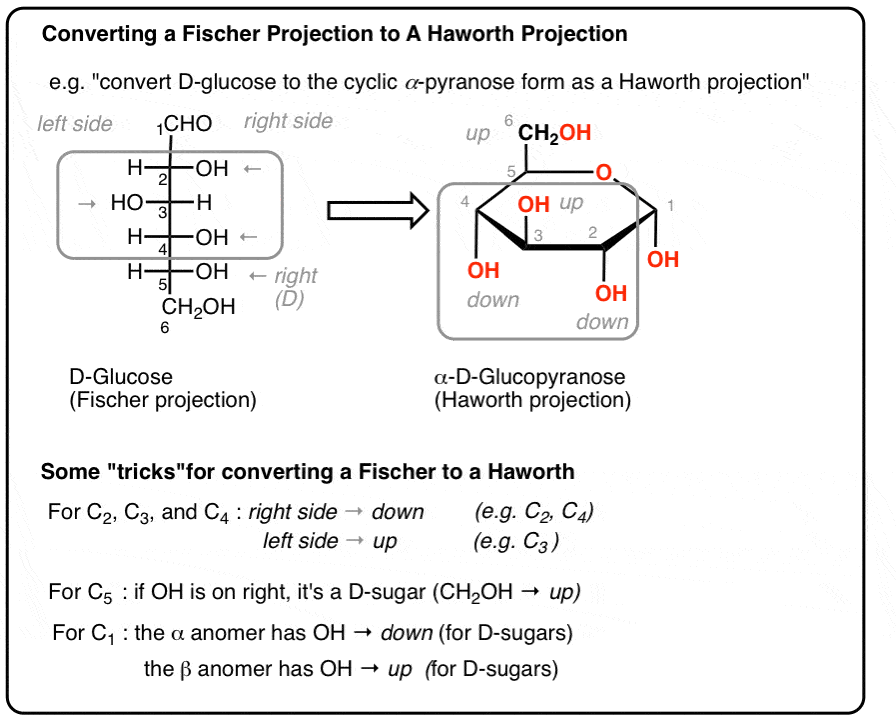
note this is the opposite for regular stuff
What is the difference between alphas and beta in the hawthron form
alphas, the oh on the anomer carbon is facing opposite direction ofr the end group
beta - anomer oh is facing same directino f the end group
What is the difference between aldoses and ketone anomeric carbons
aldoses
the anomeric carbbon s bound to two oxigens (C1)
ketoses
the anomeric carbon is bound to an oxygen and a cooh (C2)

What directino do non trminal and non anomeric carbons face (the oh grops when convirting to hawthorn frm fisher)
d = down

L = up
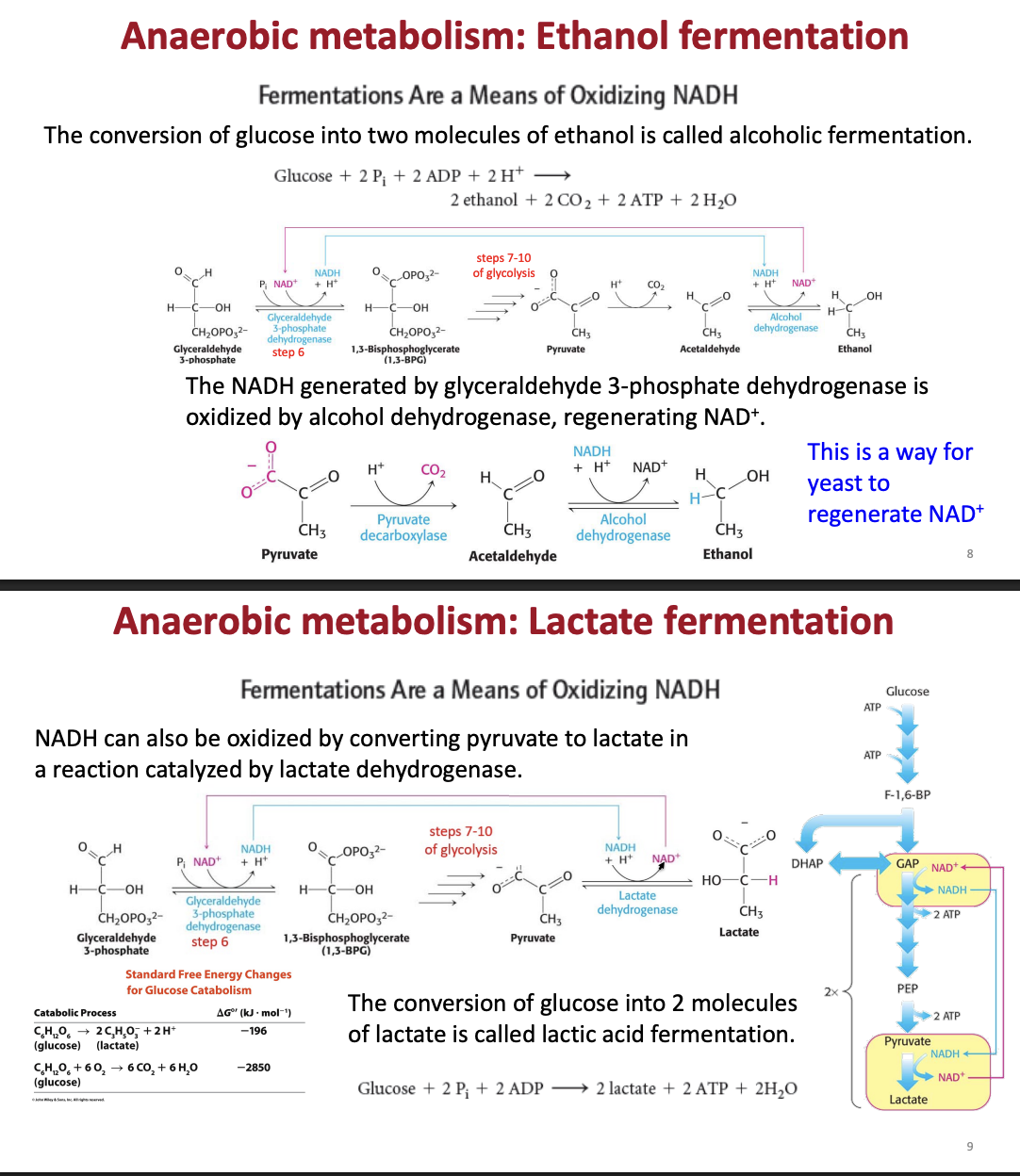
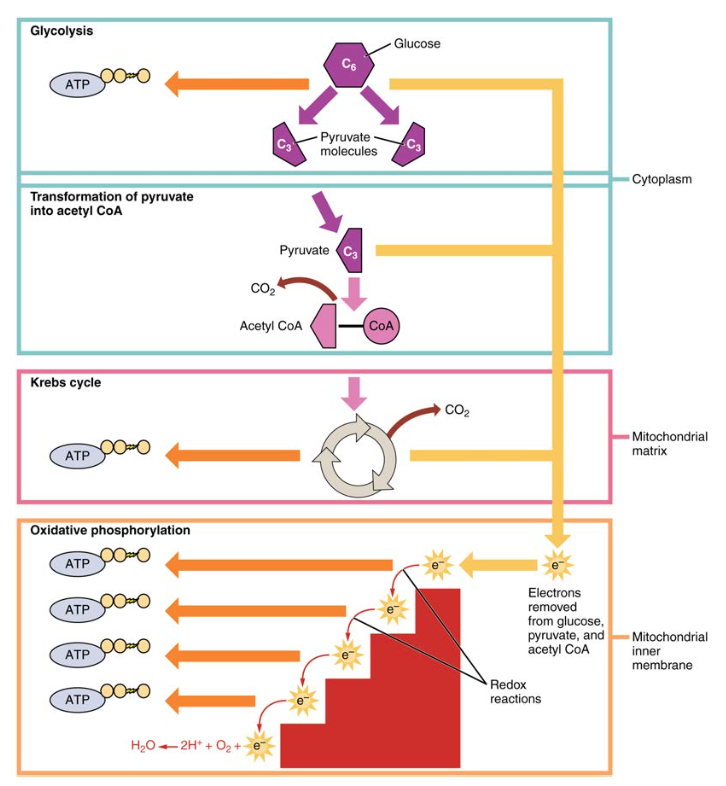
What is glucose metabolis, what is formed, what are the parts, and what conditions do they take place (where does it take place)
glucose = metabolized insied the cytoplasm
into what = 2 pyruvate later broken down inthe krebs cycle), NADH, ATP
there are arobic and anarobic parts = pyruvate becomes lactate or ethanol (
What parts of glucose metabolism are aerobiv and anarobic
anarobic glycolisis in the cytoplasm
anarobic - lactic acid fermentation
earobic = in the mitochondria for the citric acid cdyle and oxidative pohsophrilation
What are the 3 stages of glucose metabolis and how many stepas are in there
Stage oen - traps glucose in the cell ( destabalization + investment stage)
steps 1-3
stage 2 - breakdown of lguocse (into 2 components)
steps 4-5
step 3 - atp and pyruvate production (oxidation (nadh) = pay off stage
Step 6-10
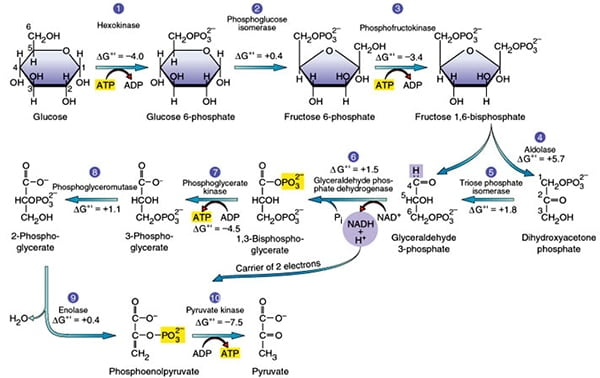
What are the steps to glycolosis by stage, steps, what is produced, what enzymes are there, what are the kinetic regulatory stages. what are the relative energies of each)
Stage 1: - trapping glucose in teh cell for (consumption)
Step 1. phosphate addition to glucose - destabilization and clevage (allosteric inhibition)
glucose + atp; —-(hexokinase/glucokinase) (in the liver)—→ Glucose 6 phosephate + atp + h+
energy = (delta)G -33.5 kg/mol - spontaneous and irreversible) -kinetic regulatory stage
Step 2- glucose to furctose (creation of symmetry)
Glucose 6-phosephate —(phosphoglucose isomoerase)—> fructose 6-phosphate
energy= (Delta) G =-2.5kj/mol
Step 3- addition of the second phosphate group (further desttabaliation)
fructose 6-phosephate + ATP —-(Phosphofructokinase)—> Fructoose 1, 6 biphosphate + adp + h+
energy = (delta)G -33.5 kg/mol - spontaneous and irreversible + committed step) -kinetic regulatory stage
Stage 2: Gap production (cleavage)
Step 4- Split of fructose into DHAP + GAP
Fructose 1-6 biphosphoros —-(aldolase)—> DHAP( not convertible) + GAP
Energy- (Delta G) = -1.3 kj/mol
Step 5 - Conversion of DHAP to GAP
DHAP —- (Triose phosphate Isomerase) —→ GAP
Energy- Delta G = +2.5 kj.mol (unfavorable to favorable)
Stage #3 - ATP production (note happens twice) (allosteric activation)
Step #6 - NADH prodution + phosphorilation of GAP
GAP + NAD+ —-(Gap Dehydrogenase)—> 1,3 BPG (has 2 phosphate groups) + NADH +h+
Energy = -1.7kj/mol)
Step #7- Phosphate group transfer to ATP (2 atp produced total)
1,3 BPG + ADP + H+ —-(Phosphoglycerate Kinase)—> 3 phosphoglycerate (3-PG)
Energy- (delta) G = +1.3kj/mol
Step #8 - intermolecular phosphaete transefer (Destabalization)
3-PG —-(phosphoglycerate mutase)—> 2 phosphate glycerate (2pg)
Energy: +0.8kj/mol
Step #9 - Enol formation
2-PG —(Enolase)—> phosphenolpyruvate (pep) (very high energy intermediate)
Energy: Delta G = -3.3 kj/mom
Step #10 - pyruvate formation + ATP formation (allosteric inhibition)
Pep + adp + H+ —-(pyruvate kinase)-→ pyruvate + ATP
energy = -61.8 kj per mol ( irrevrsible, spontaneous, kinetic regulatory step)
Net atp producced =4 atp
total energy = -96.32
What are the different types of isomers (constitutional sterioisomers, Diasterimers, Epimers, Anomers, Conformers)
constituttional isomers - same formula but different structures
sterioisomrs = different in spacial arrangemnt
Enantomers
non souperimposible mirror images (D or l form for carbs)
Diasterioisomers
Isomers that are not mirror images
epimers
they differ at one of several asymettric carbon atoms
Anomers
differe at a new asymetctruc carbon + fixed ring of carbon
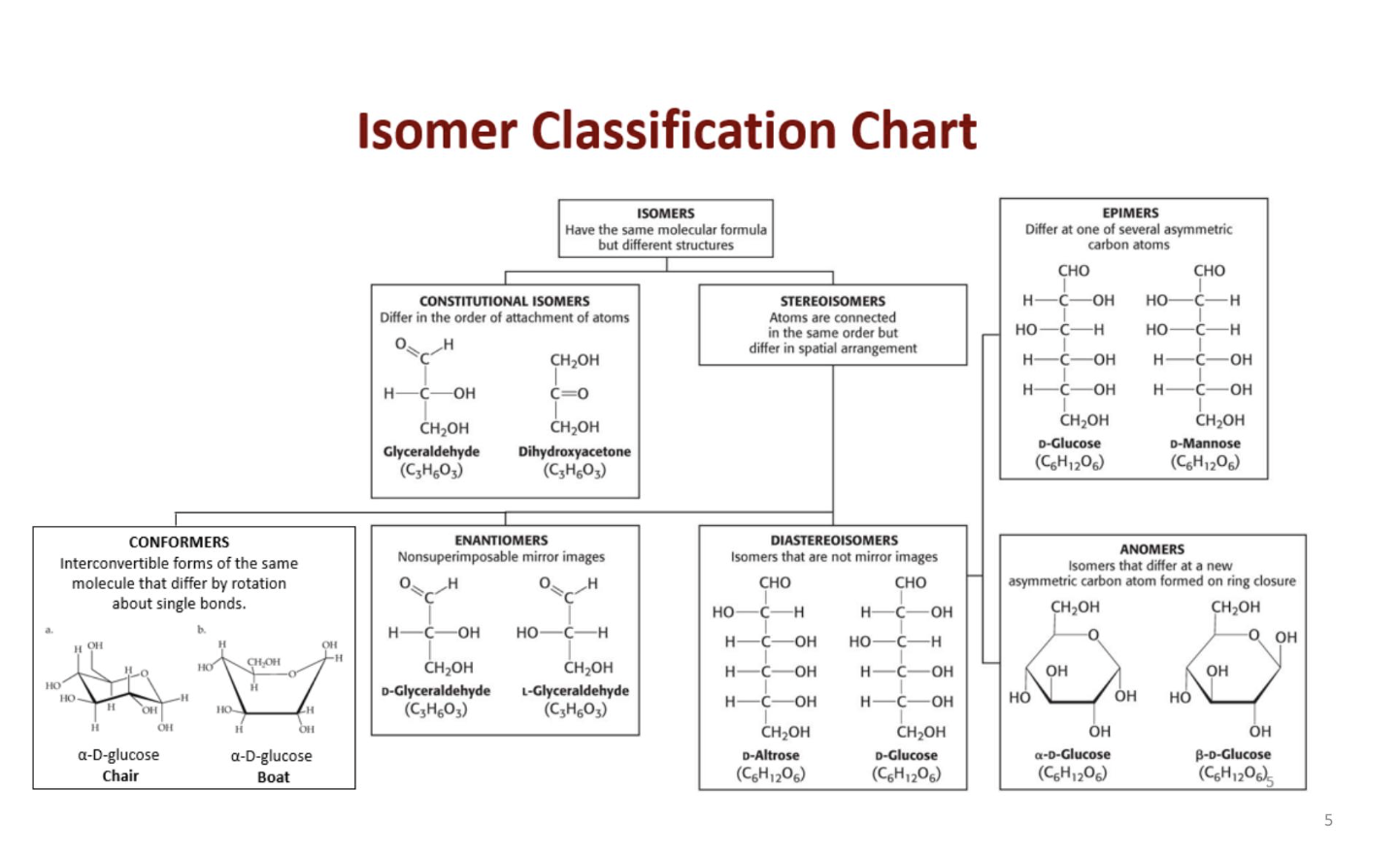
How is ring formation formed bteween carbohydrates and what is the difference in fformation for ketones and aldoses
6 membered rings = pyranose
5 membered rings - furanose
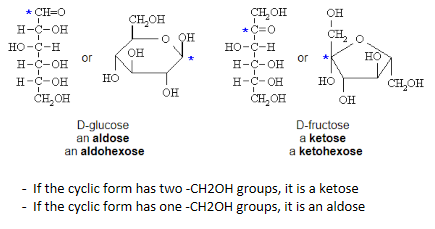
What are the different monosaccharide derrivatives and how are they formed - sugar acids
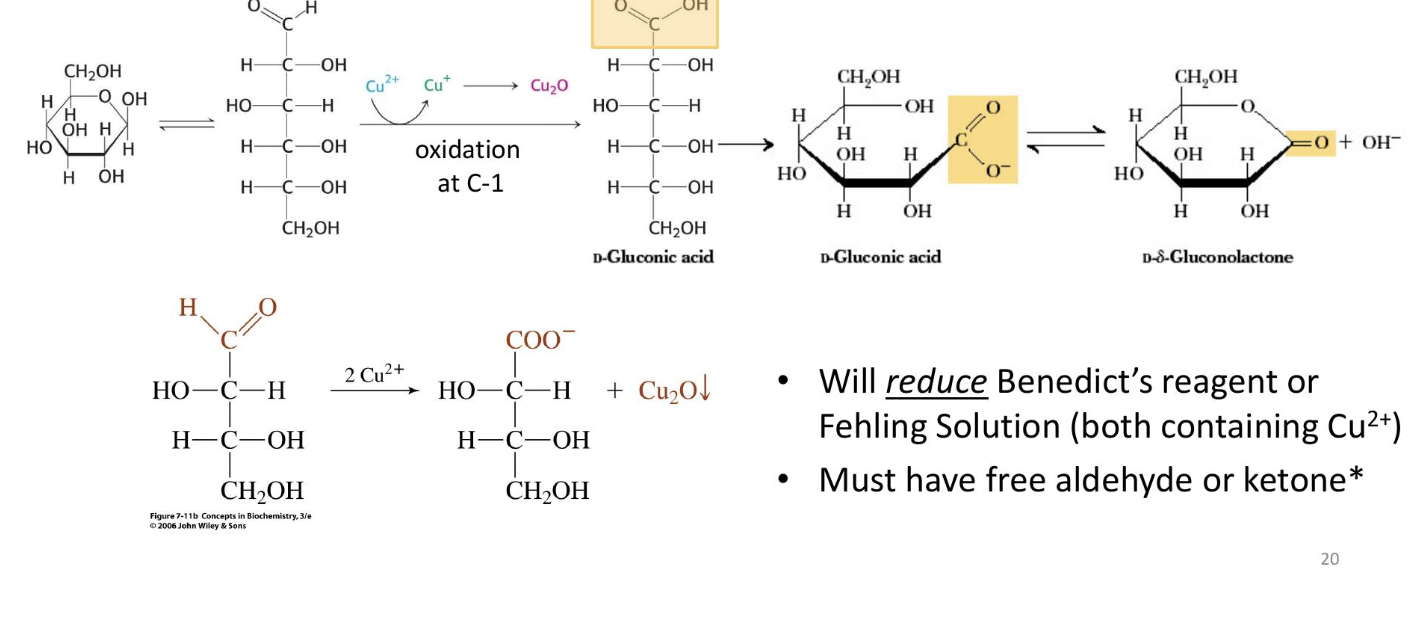
Sugar Acids: Formed from sugars with free anomeric carbons (reducing sugars) via the reduction of oxidizing agents
will reduce a benedicts reagent or fehling solution (must have a free aldehyde or kettone
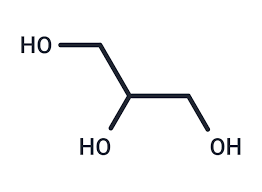
What molecule is this and what is its function
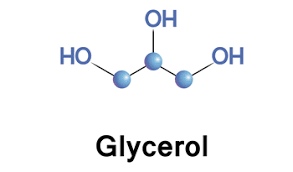
Glycerol
versitiles sugar alcohol that is used as a swetener
What molecule is this and what is its function
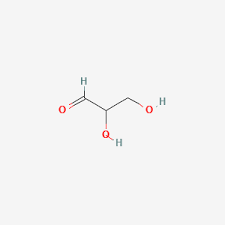
Glyceraldehyde
serving as a key intermediate in glycolysis and fructose metabolism, converting into energy-producing compounds
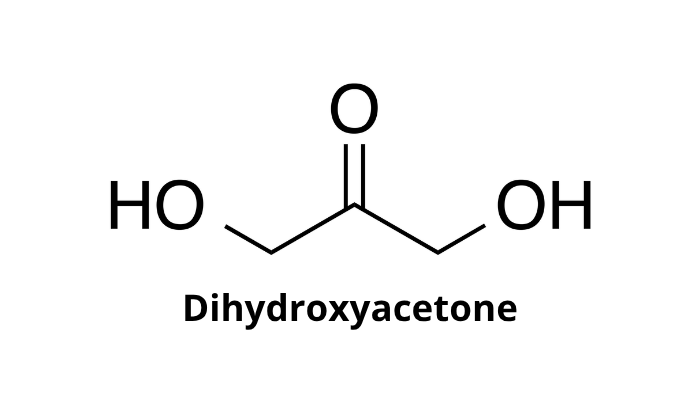
What momecule si this and what is its function
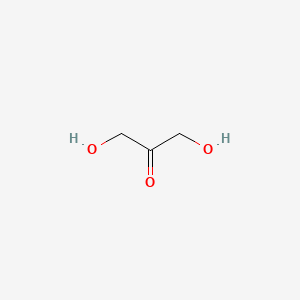
Dihydroxyacetone
acts as a carbon source for microorganisms, a metabolite in human cells, and a non-enzymatic skin-tanning agent through the Maillard reaction with amino acids
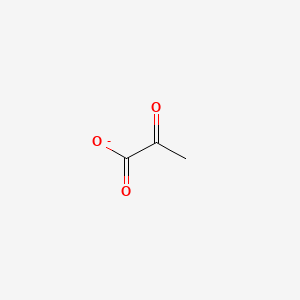
What molecule is this and what iis its function
Pyruvate
a central hub in metabolism, serving as the final product of glycolysis and a crucial branch point for energy production or biosynthesis
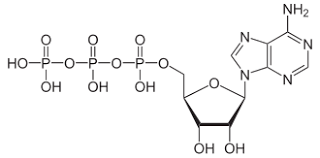
What molecule is this and what iis its function
ATP
providing readily releasable energy to drive vital biochemical reactions through the hydrolysis of its high-energy phosphate bonds into ADP and inorganic phosphate
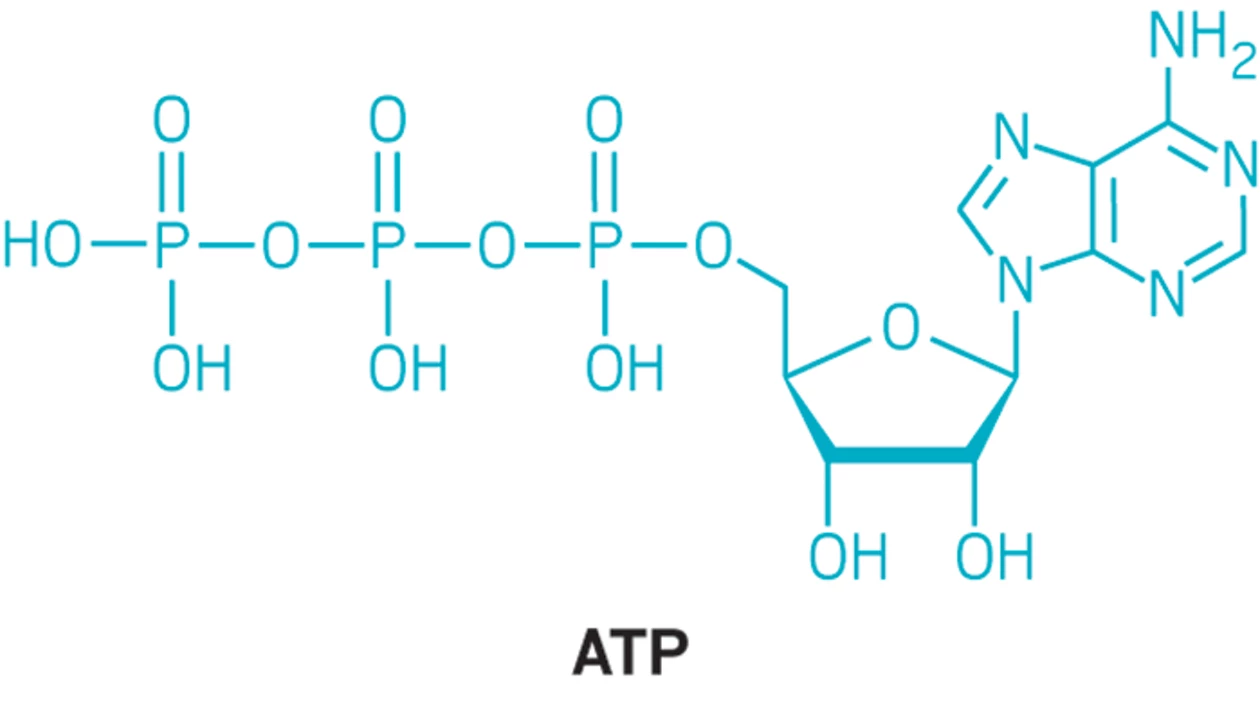
What molecule is this and what iis its function
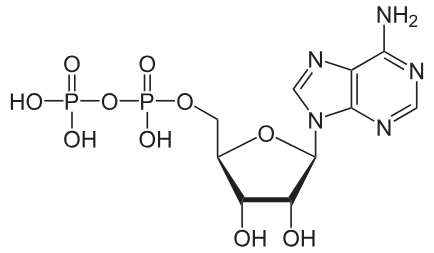
ADP
acts as the primary precursor for ATP regeneration and a product of energy-releasing reactions
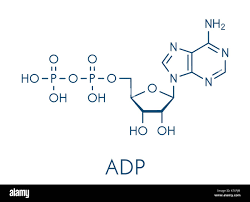
What molecule is this and what iis its function
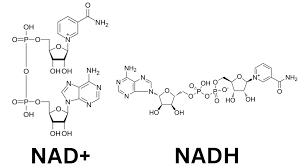
NAD+
acting as a crucial electron carrier in redox reactions to drive energy metabolism, specifically ATP production via glycolysis, the TCA cycle, and oxidative phosphorylation
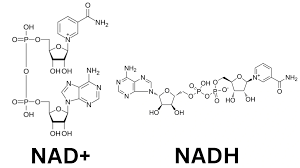
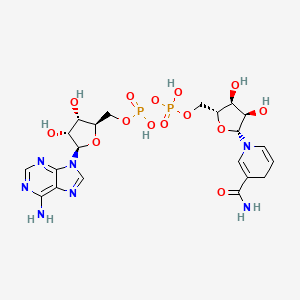
What molecule is this and what iis its function- really know the difference betwn the two pairings
NADH
a vital coenzyme acting as a primary electron carrier in cellular respiration, shuttling electrons from catabolic pathways like glycolysis and the TCA cycle to the electron transport chain (ETC
What molecule is this and what iis its function
FAD
primarily as an electron acceptor in oxidation-reduction reactions, such as the Citric Acid Cycl
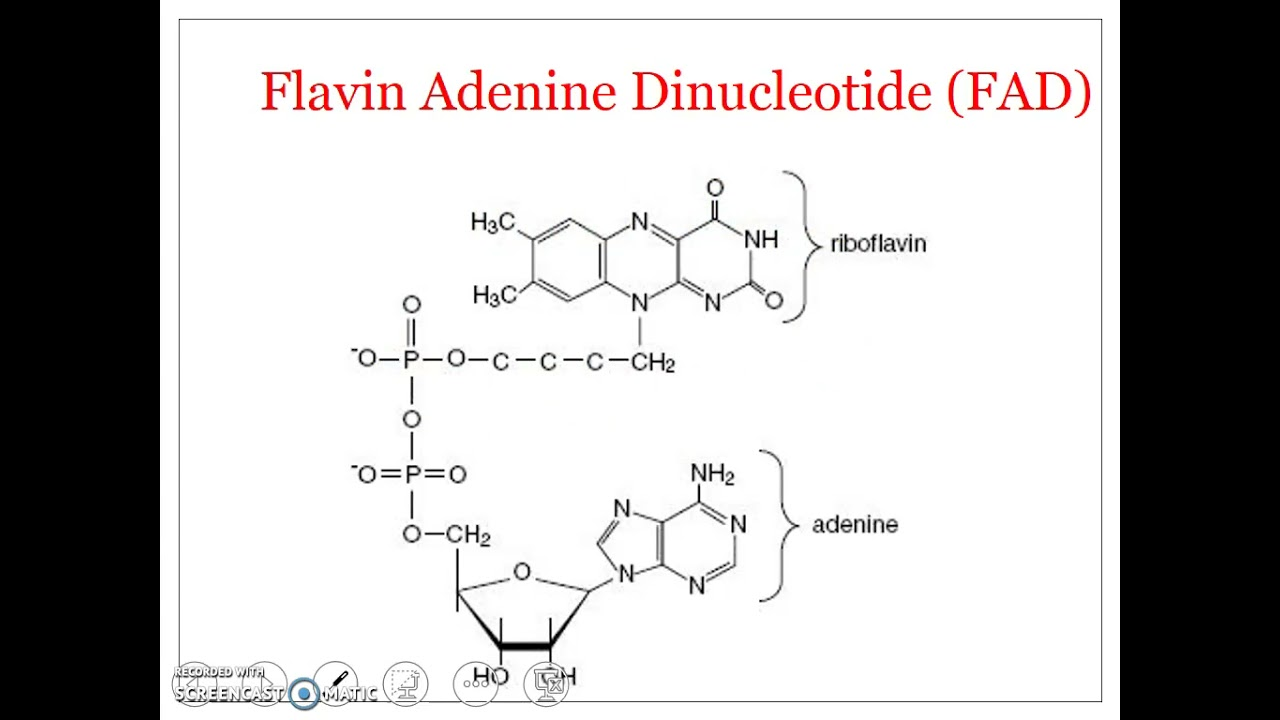
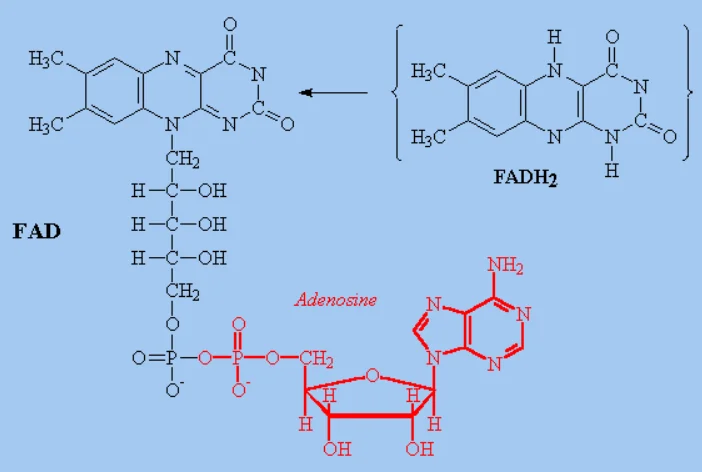
What molecule is this and what iis its function
FADH2
(flavin adenine dinucleotide, reduced form) acts as a critical electron carrier in cellular respiration and metabolism, generating approximately ATP via the electron transport chain (ETC)
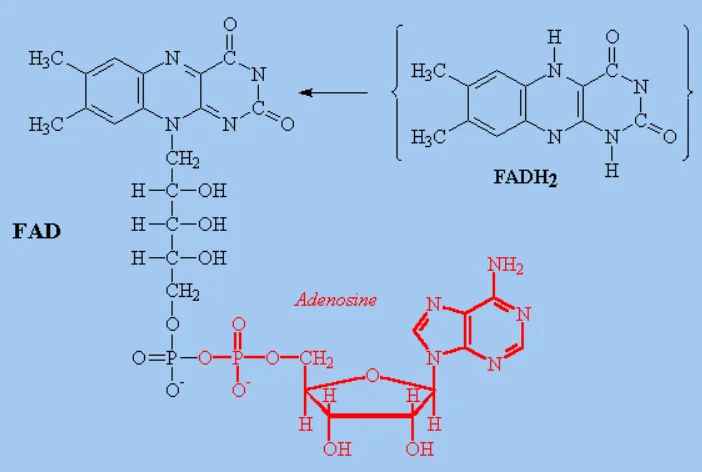
The reactants and products, the number and kind of cofactors, and the cellular locations for Glycolysis
Location
cytosol of the cell
Reactants
1 mole glucose, 2nad+, 2adp, 2pi
Products
2 pyruvate
2nadh
2atp net
2 h2o
2H+
ATP accounting
Investment phase = -2 atp
energy payoff phase +4atp
net atp 2atp
Cofactors Used
NAD⁺ → reduced to NADH
Mg²⁺ (required for ATP binding in several enzymes)
Overall reaction
Glucose + 2 NAD⁺ + 2 ADP + 2 Pi
→ 2 Pyruvate + 2 NADH + 2 ATP + 2 H₂O
What are the 3 irreversible steps of glycolisis and how do they work - what is the reaction, waht is the type of reaction, what is the purpose, and vaguely how is it regulated
Step 1 (hexokinase -muscle, Glucokinase - liver
Reaction
Glucose + ATP → Glucose-6-phosphate + ADP
Type of reaction
PhosphorylationPurpose
Traps glucose in the cell
Commits glucose to metabolism
Regulation
Inhibited by Glucose-6-phosphate
In liver: glucokinase regulated by insulin
STep 3 - phosphofructokinase-1 PFK-1
Reaction
Fructose-6-phosphate + ATP → Fructose-1,6-bisphosphate + ADP
Type of reaction
Phosphorylation
it is a rate limiting step of glycolisis
REgulation
activators
AMP, ADP, Fructose-2.6 bisphospate
inhibitors
ATP, citrate, lowph
concept
high atp levels = cell has eneryg = inhibition of glycolisis
Step 10- pyruvate kinas
reaction
Phosphoenolpyruvate (PEP) + ADP → Pyruvate + ATP
Type of reaction
substrate level phosphorilation
activated by
fructose 1,6 biphosephate (feed forward activation)
inhibitied by
atp alanine (3 memberd ring carbon), acytel co a
What are the different types of reactions inmetabolic pathwasy: what type is used for hexokinase, wwhat happens, what is the enzyme class invovled
reaction type
phosphorilation
addition of phosphate groups
enzyme class (transferases - kinases)
What are the different types of reactions in metabolic pathwasy: what type is used for glucose-6 phosphate to frucotes 6 phosephate change, what is the enzyme class invovled, what happens,
reaction type
isomerization
rearrangement of atoms
enzyme class - isomerases
Different types of reactions in metabolic pathways: reaction type, enzyme class, effect: aldolase (enzyme)
reaction type
nonhydrolitic cleavagge
splittin gof a molecule by non h ydroxyl process
enzyme type
lysases
Different types of reactions in metabolic pathways: reaction type, enzyme class, effect: G3p dehydrogenase
reaction type
oxidation reduction
electron transfer
enzyme type
oxidoreductases (dehydrogenases)
Different types of reactions in metabolic pathways: reaction type, enzyme class, effect: pyruvase kinase
reaection type
substrate level phosphorilation
atp produced directly
Different types of reactions in metabolic pathways: reaction type, enzyme class, effect: Enolase
reaction type
dehydration reactio (hydrolotic cleavage/hydrolisi_
clevage of bonds by water to remove fuctional groups to water
enzyme type
hydrolases
Different types of reactions in metabolic pathways: reaction type, enzyme class, effect: Ligases/ synthases
reaction type
bond formation using. energy
formtation of carbon-carbon and other bond s with energy from atp
What are the different. types of catalyitic stratagies used for glycolisis and how does it do it: acid baes, covalent, metal ion, what amino acids are in voled
acid base
enzyme donates or accepts protions (h+)
often involves histinde, aspartate, glutamate
covalent catalissts
temporary covalent bodn between enzyme and substrate
invovles
serein, cystine, lysine
metal ion
metal ions stablize chargs
mg2+ stabilszes many glyclisizs enzymes
What are the different monosaccharide derrivatives and how are they formed - sugar alcohols (altidolss)
formation
formed form mild reduction (gaining of electrion) of carbyonyl groups of aldoses
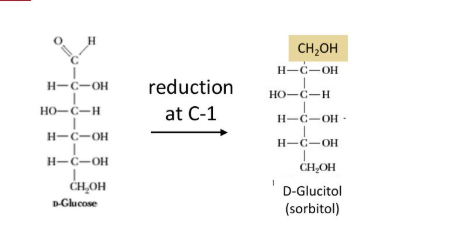
What are the different monosaccharide derrivatives and how are they formed - Sugar esther
formation
phospate esters of onosaccharides are importatt metabolic intermediates
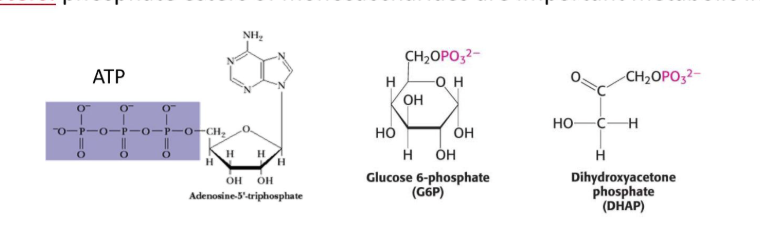
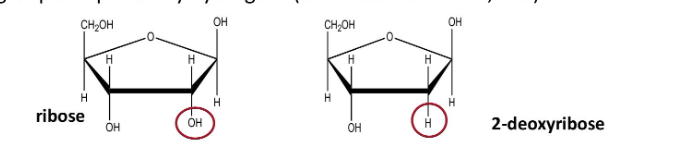
What are the different monosaccharide derrivatives and how are they formed- deoxy sugars
formation
monosaccharides with one or more hyudroxyl groups replased by hydrogens
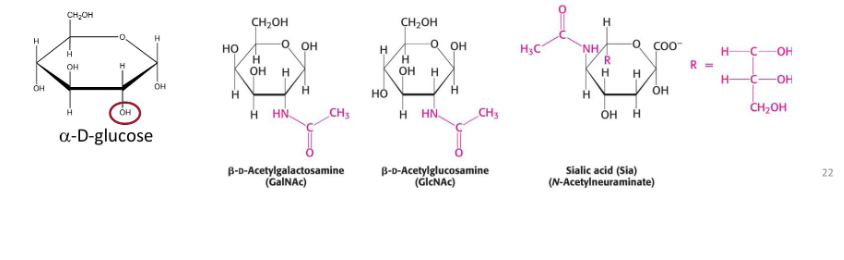
What are the different monosaccharide derrivatives and how are they formed- amino sugars
formation
contain an amio group in place of a hydroxyl grop at the c-2 position
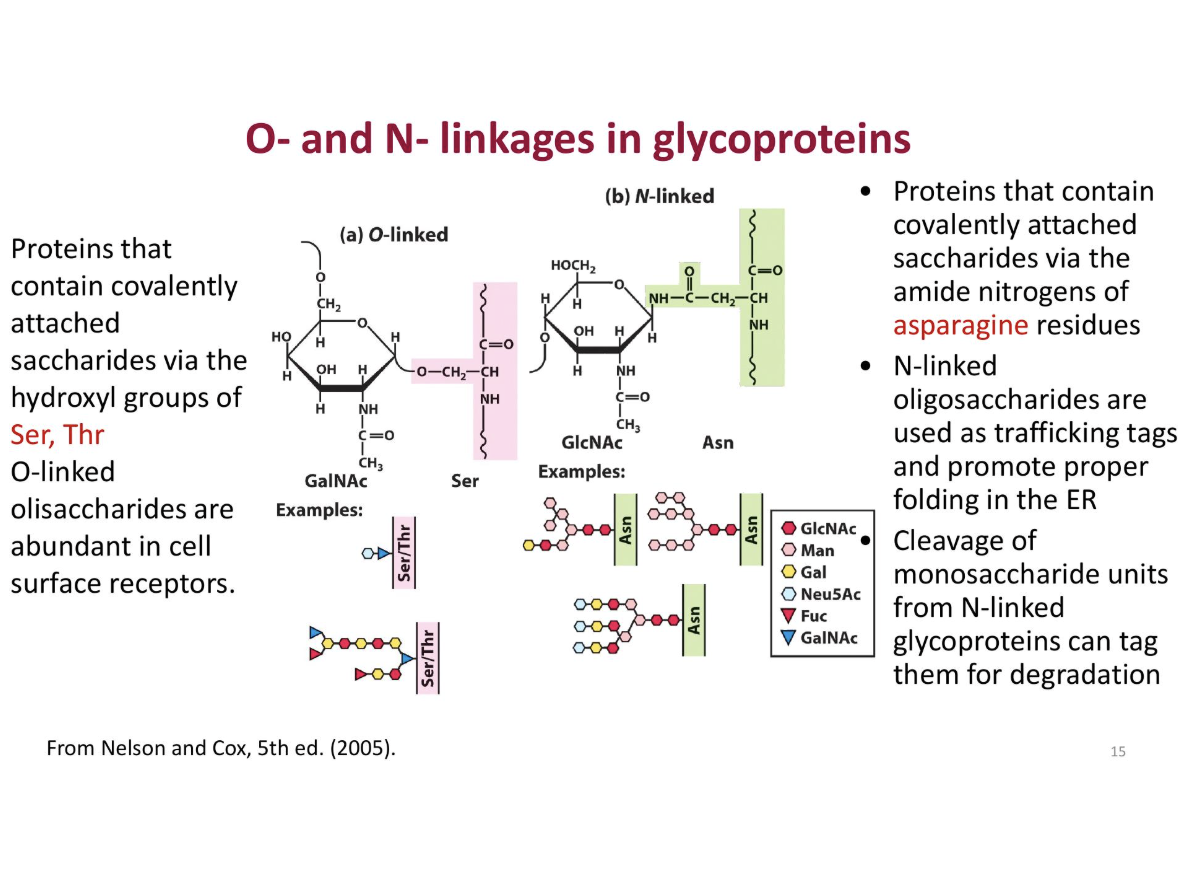
How are glycosidic bonds formed- what is the difference between an n and an o glucosidic vond, what catalises the reaction. what is the distinctive difference
o-glycosidic vond (glycosides)
formed between the anomeric carbon atom and hydroxyl group of another molecule
n - glycosidic vond
formed between teh anomeric carbon and an amine
catalized by - glycotransferase
Diffferences
o linked
proteins contain convalently attached saccharides via the hydroxyl (OH) groups of ser, thr
used for cell surface
n l inked
proteins contain covalently attached saccharides via the amide nitoriges of asparigine residues
usd o be folded er
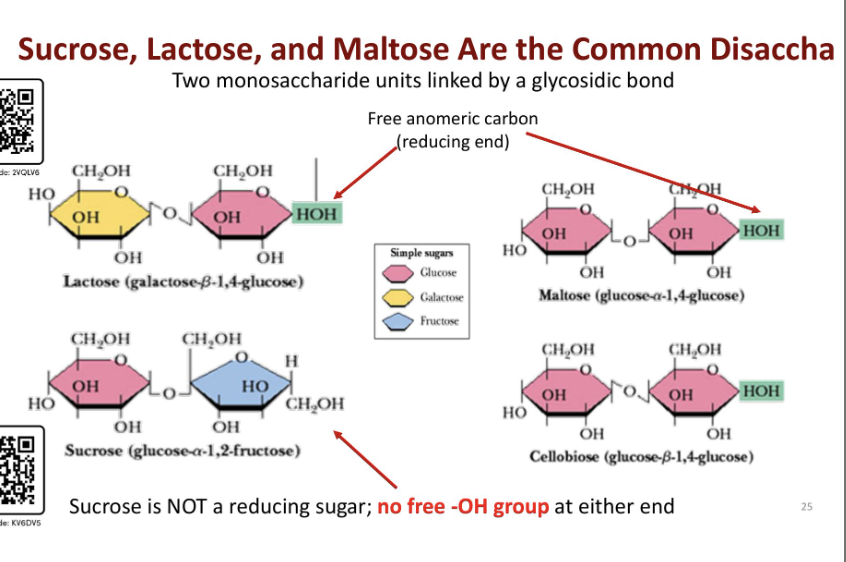
Describe the different types of glycosidic bonds for the following disaccharides: sucrose, lactose, and moltose
Lactose
bond between beta galactose (same side) and glucose
zigzag pattern (galactose beta 1,4 glucose)
Maltose
bond between alpha glucose and regular glucose (u shaped pattern)
glucose alpah 1,4 gluocose)
Sucrose
bond between glucos alpha (opposite sides) and fructose (beta 5 memberd ringO
forms u shaped structure
nnon reducing because t here is no free oh group at either end
Starch: what are teh bonding patterns for amulose an d amylopectin, and cellulose
amulose
alpha 1,.4 linkages of gluocse with one reducing end
forms helical structure
amylopectin
alpha 1.6 structures with glucose = branches in every 12-30 resuidues. more linear structure
note glycogen is similar to amylopectin iwth alpha 1,6 bonding and branches every 8 -12 residues
cellulose
lienar homopolymer of D-gluocse with beta 1-4 glycosidic bonds (fors zig zag pattern
straight chain
note its a hmopolymer of glucose
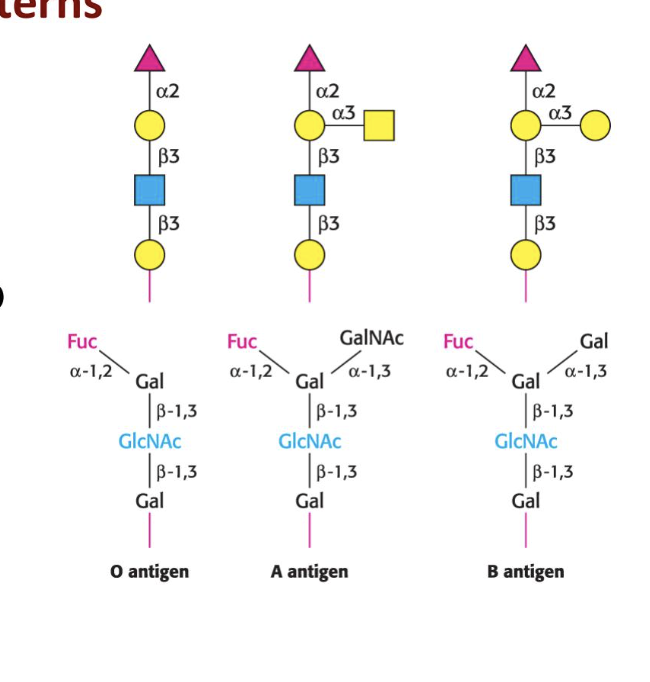
Blood types and glycosylation patternsh. what are the bonding patterns for each blood type, a, b and o
A
n-acetylgalatosamine is added to the O by a spcific glycotransferase
B
galactose is added by another transferase
O
produces no active glycotransferase
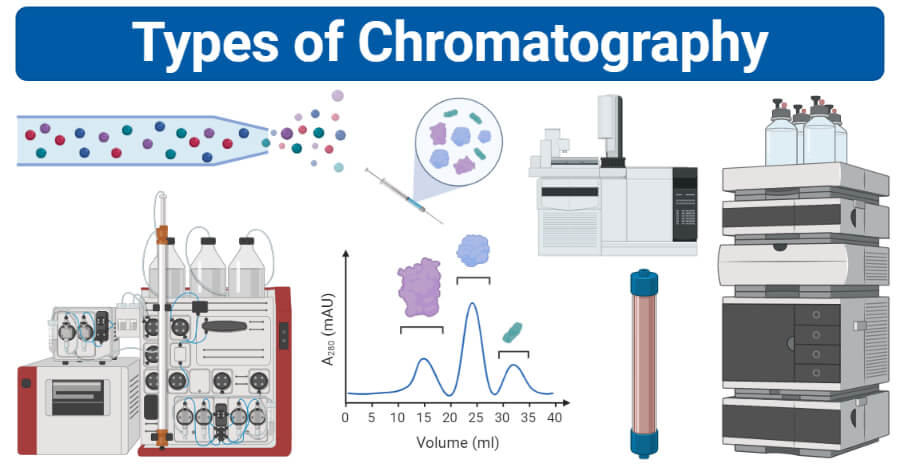
What are the different typs of cromatography and how do they work? what are the ways that you can purify proteins ( Separation by solubiliy, size, net charge, specific binding affinnity, higher performance liquid chromotryphy
Methods
solubility size charge, and specific binding affinity
Method 1. sparation by solubility
happens at high salt concentrations = proteins precipitate out of a solution
how does it happen
charges from the proteins - prefferibly negative come to react with water. Filtration methods are used to sparate the small molecules
large stuff is trapped inside while smaller moleules diffus outide
Separation by size
Gell flltration chromotrophy = molecular exculsion chromotrophy
what happens
small molecules can enter the beads but large molecules cannot = filration
larger molecuels leave the filtraiton column first followed by the bound small molecules
Separation by net charge
ion exchange chromotrhohy
neaural charged protens bind with negativley charged carboxylae groups
negatviely charged particals are unable to bind/have a difficult time binding
positively charged = couple with the positlvey charged groups on the binding plate
can work both ways
cation exchagne = pisitive proein bind to negative beads
anion exchange = negatively charged bead bind to pisitive
leftover proteins pass to the bottom and out of the solution to be filterd
Separation by specific binding affinity
affinity chromotrophy = motst powerful mehtod of protein purification
uses glucose binding
glucose binds to proteins that are able to favoribley bind to glucose
nonbinding proteins are reoved
glucose is whashed out and the required proteins are left behind
Higher performance liquid chromotrophy
its like a more enhanced version of cholumn techinechs
beads are finley dividd = more interactions = more pressure =. more rapid separation
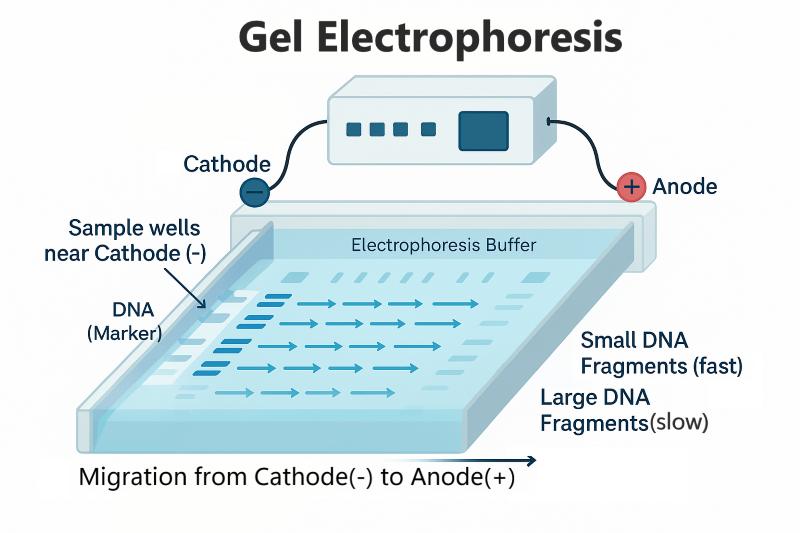
Whta is gel electrophorisis and how does it relates to chromotrophy. How does SDS used
gel electrophorisis = used to tell if purification methods are effective
what steps does it use
molecules with electric charges move towerd the feild (negatice cathode to psitive anode)
smaller moleucles move faster and furhte rwhile larger moleucels are pracitcally immoble in teh fluid
SDS is needed to separate by mase
method
the negatively charged SDS = denatures proteins = binds at a 1:2 ration sds to aino acid protein
Betamericapoethanol is added to reduce the disfulfide bond sof the amino acids = li nearization of proteins
allows for smaller molecules to move it down
Salting out: A separation technique that takes advantage of the fact that the solubility of proteins varies with the salt concentration. As the salt concentration is increased, different proteins will precipitate at different salt concentrations, a process called salting out.
Polyacrylamide Gel Electrophoresis (PAGE): allows the separation of proteins on the basis of their mass to charge ratios (m/e). Remember gel electrophoresis for DNA? This method sorts proteins using the same properties: charge (which is more variable in proteins than DNA) and mass. SDS-PAGE can be used to separate proteins based on ONLY their masses, because it disrupts proteins charges.
Size exclusion chromatography (SEC) (Gel filtration chromatography): allows the separation of proteins on the basis of size– which is only slightly different from mass. In this case, there is a porous material that slows down smaller proteins. This might be counterintuitive: larger proteins are separated first, because smaller proteins move more slowly
Ion exchange chromatography (IEC): allows separation of proteins on the basis of charge. This may seem redundant with PAGE, but it uses an entirely different technique of a column that acts as a sort of filter. There are two types, named after which type of protein will stick to the stationary phase.
Anion exchange chromatography: the resin is positively charged, so anions will stick.
Cation exchange chromatography: resin is negatively charged, so cations will stick.
Affinity chromatography (AC): takes advantage of the fact that some proteins have a high affinity for specific chemicals or chemical groups. Perhaps it is easiest to understand with an example. Maybe you want to find a protein that binds to sugar. So, you coat beads with that sugar, and filter a bunch of proteins through. The proteins that come out of the column do not bind to the sugar. So separate the bound proteins, you do a “wash”
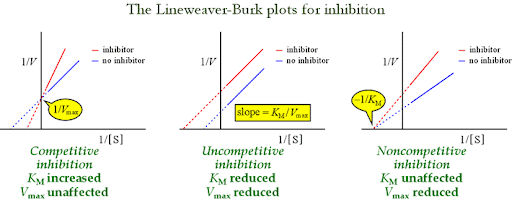
look at your notes about allosteric enzymes, differences in charting. What is the differnece between a muchalis meten graph and an alllosteric enzyme. know how to draw the charts - know abou the te 3 horsemen of enzyme inhiibtion = competative uncompetative and noncompetative inhibiton
how does the state of an allosteric enzyme play into its form
look at notes bae
allosteric enzymes have multiple activaiton sytes
the enzyme can exist in 2 states = t state and r state
concentrated modle show s that all subunits or activesites must be in teh same states (T state or R state)
note the R state is more favorable to binding while the t state is more stable - you can inhibit enzymes by forcing binding into the t state to make it more stable

What are the 4 differnet ypses of catalists used by enzymes
covalent catalists
active site contaiing a reacive groups that reacts whith incoming groups
General Acid base catalists
any thing other than water that acts like a protein donor or accptor
metal ion catalists
Serves as a neutrophile catalist = stabalizes the negative charge on the (reaction intermediate)
can also bind to the substrate by increses the interactsion with the enzyme
Catalists by approxiation
reactions with 2 distinct substrates
reaction rats must be consiterabley close by eachter to allow for conformational change
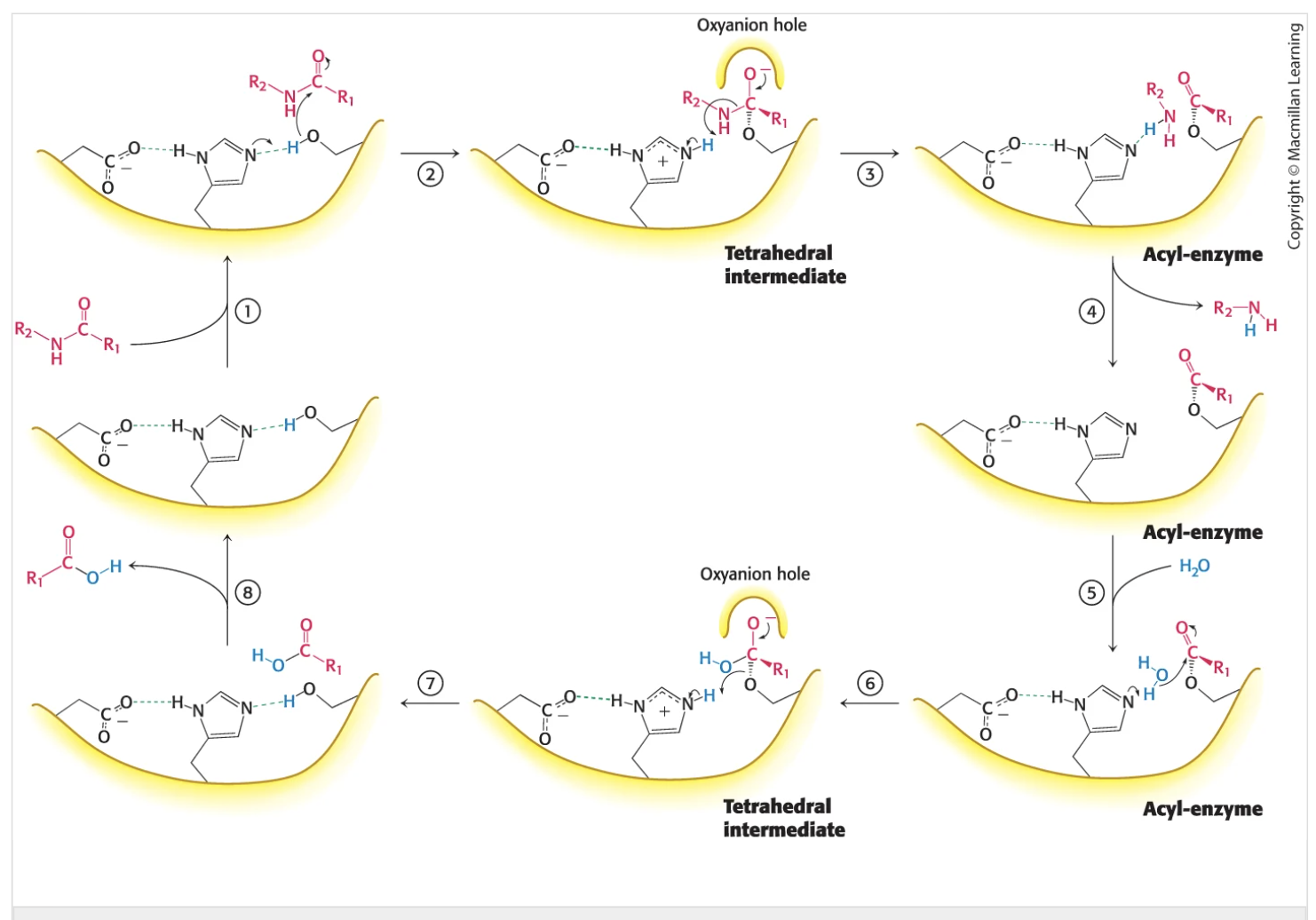
What are teh steps to chromotripsion function as a n enzyme. How does it work, how does it utilize reaction intermediates. Know the genral steps and what interactions take place. Waht is the resulting binding energies of each
Histidine attacks the serine and removes the hydrogen to. make it a good nucleophyle
substrate binds to the enzyme
the substrate binds to the sibtrate via noncolvaent forces- attaches to the serine (nuclophile)
neturophile attack (tetrahedral intermediate formation
oxygen hole binds the new found negative moleucle on the substrate as it attaches to the histidine (via its lone hydrogen)
the carbonyl part attaches to teh oxygen hole
collapse of the tetrahedral intrmeidate
the oxygen hole is released and teh amine and carbonlyl secont are cleved leaving 2 new residues
release of the amine component
amine component lost - acyl-enzyme is used to release this
water binds to open site on carbonyl
using the acyl enzyme h2o comes in and binds tothe carbonyl group
nuclophile attack of watr to the acytl enzyme (tetrahedral intermediate formation)
new oxynation hole comes to stabilize the carbonyl group = hdyrgen bond with the nigrogen on histidine
collapse of the tetrahedral intermedate
stabalization of histidine
release of the new substrate

note chromotripsin cleves petide bons selelcively on the carbonyl terminal syde of large hydrophobic amino acids
tryptophan, tyrosine, phenyl, methonine, isoleucine
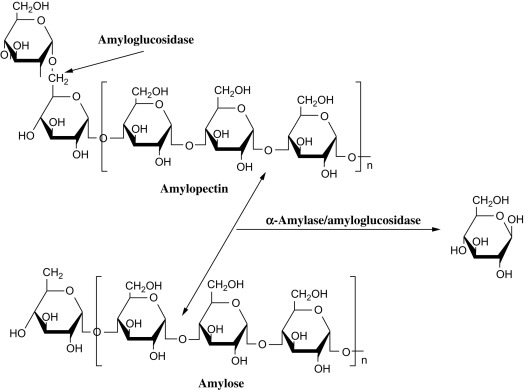
What is the function of alpha-amulase and where does it cleve and not gleve
alpha amilas e= pancreatic enzyme usd for glucose digesiton
celves at the 1,4 linkages not 1,6 linkages
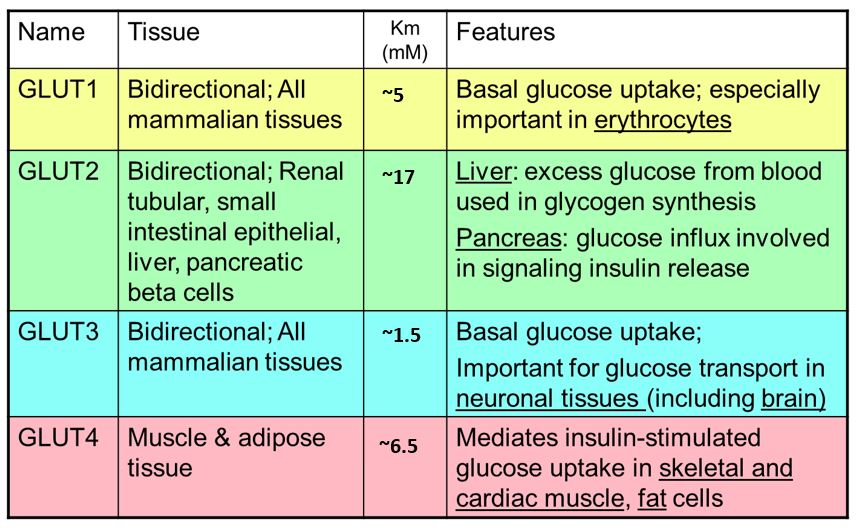
What are the differnt ypes of glucose transportors uses
Sgluts = sodiu, glucose linked transport
gluoces + galactose uptake into intestanal cells
fructose is small enough to diffuse across the cell membrane using GLUt 5
Glut 2 releases monosachharides into the blood streme
Explain what is meant by metabolism in terms of both catabolic and anabolic processes.
Catabolism = breakdown of coplex molecuels
allows for enrgy release
building block for biosyntehis ( NADH and FADH2)
Anabolyzm
= upatake of eneryg to make prdocut
usesATP + NADPH to form
Reaction coupling can help make an unfoavorable reaction fravorable
Identify factors that make ATP and other phosphoesters useful molecules for capturing and transforming chemical energy. (4 methods)
electrostatic repulsiotn
4 negative chargers present when atp is present = higer energy due to strong erpulsive forces
resonance stablaization
After ATP hydrolysis:
ATP → ADP + Pi
The inorganic phosphate (Pi) has many resonance forms, making the products more stable.
Greater stability → energy release.
increase in entropy’
more disorder = higher energy for relase
stabalization due to water
water helps stabilizes
The products of hydrolysis (ADP + Pi) interact with water more favorably than ATP.
This stabilization also contributes to a negative ΔG.
Explain how ATP can power reactions that would otherwise not occur.
the high energy output from when phospahte gorps are removed from atp = makes unfavoral reactions more favorable = reaction coupling
Describe the relationship between the oxidation state of a carbon molecule and its usefulness as a fuel.
Reduced molecules
Contain many:
C–H bonds
Few oxygen atoms
These molecules store large amounts of chemical energy.
Examples:
Fatty acids
Hydrocarbons
They release energy when oxidized to CO₂.
Oxidized molecules
Contain many:
C–O bonds
Examples:
CO₂
Carboxylic acids
These molecules already lost most of their energy and therefore cannot serve as effective fuels.
Summarize the recurring motifs and regulation principles in metabolic pathways.
Metabolic pathways are organized into stepwise enzyme-catalyzed reactions with a committed (rate-limiting) step that is often irreversible. They are regulated by allosteric control (activators/inhibitors like ATP), feedback inhibition (end product inhibits an early step), and covalent modification (e.g., phosphorylation). Pathways are also compartmentalized and use common energy carriers (ATP, NADH, NADPH) to coordinate energy flow.
Describe the following shoot: NAD+/NADH, FAD/FADH2, Coenzyme A
Differentiate among the structures and functions of glycoproteins and lectins.: What are the general function of glycoproteins (3 major classes): Glycoprotins, proteoglycnans, mucans
what are their general strcuture
Back (Answer):
General Function:
Many are membrane proteins (largest component by weight)
Often carbohydrate-rich
Roles include:
Structural support
Cell recognition (receptors)
Lubrication
Glycoproteins:
Proteins with covalently attached carbohydrates
Found especially in cell surface receptors
Types:
O-linked glycoproteins
Sugar attached to –OH group of serine (Ser) or threonine (Thr)
N-linked glycoproteins
Sugar attached to amide nitrogen of asparagine (Asn)
Formed in the endoplasmic reticulum (ER)
Proteoglycans:
Proteins attached to glycosaminoglycans (GAGs)
GAGs = long chains of repeating disaccharide units:
One amino sugar derivative
One negatively charged sugar
Function:
Provide structural support
Act as hydrated gels/lubricants
Mucins:
Heavily glycosylated proteins (mostly carbohydrate)
Carbohydrates attached via N-acetylgalactosamine (O-linked)
Function:
Form mucus
Provide lubrication and protection (e.g., in respiratory & digestive tracts)
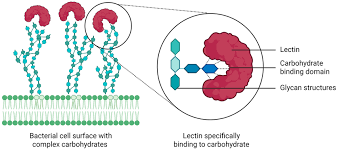
What are lectins, their structure, funciton and whatnot
Structure:
Proteins that bind carbohydrates (NOT covalently attached)
Have specific carbohydrate-binding sites
Function:
Recognize and bind specific sugars on glycoproteins/glycolipids
Mediate:
Cell-cell interactions
Immune responses
Cell targeting
Lectins — What You Should Definitely Know
Act like “carbohydrate recognition molecules”
Highly specific (can distinguish small sugar differences)
Important in:
Immune system (pathogen recognition)
Cell communication
Can cause cell agglutination (clumping) by cross-linking glycoproteins
Used in labs to:
Identify cell types
Study glycosylation patterns
Easy Way to Remember
Glycoprotein = HAS sugar
Lectin = GRABS sugar
Describe the irreversible and commited steps of glycolisis. what makes them what them what they are
use chat to fill this out- look at your notes.
Describe how and why the aldolase mechanysm works generally
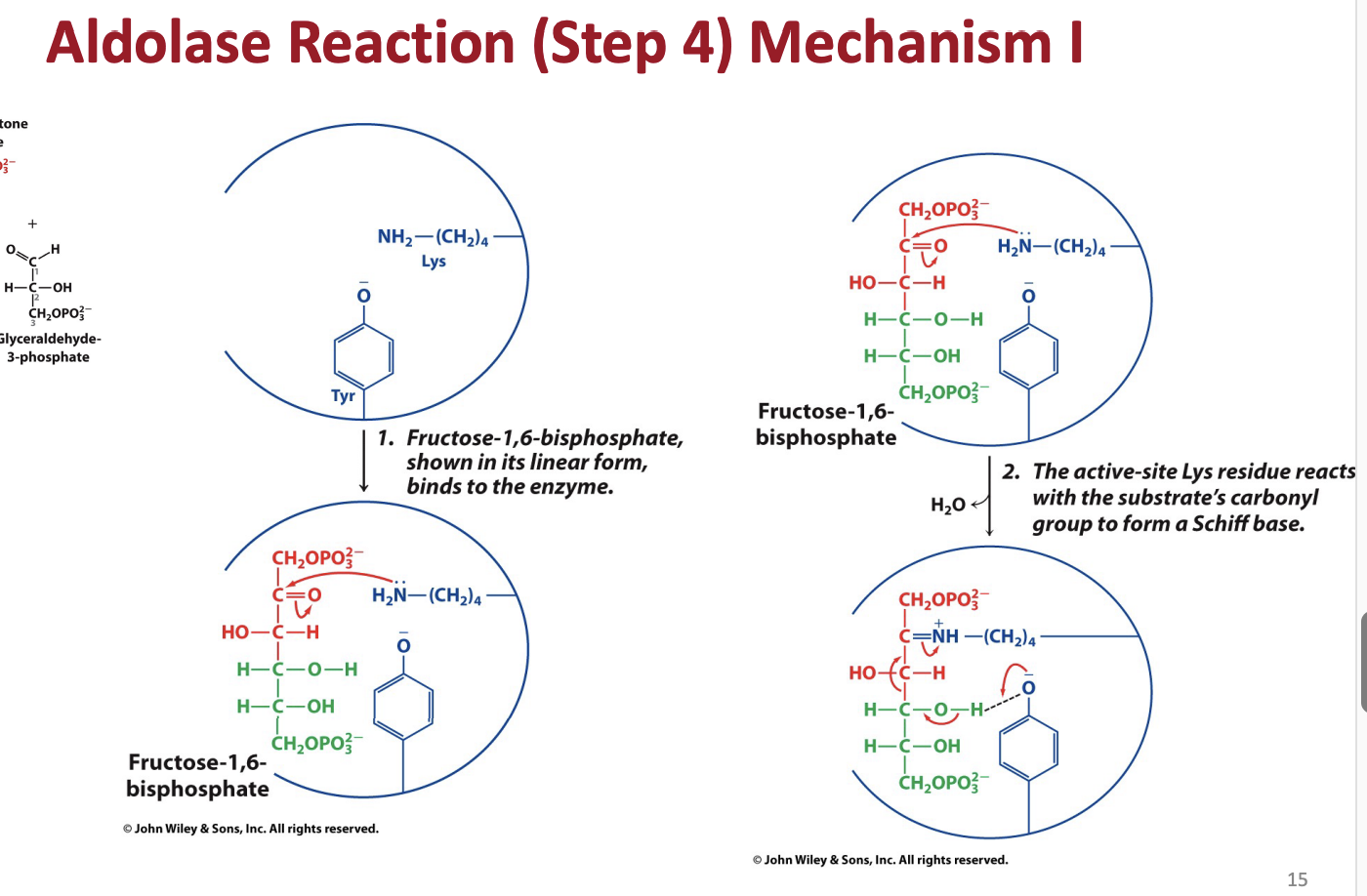
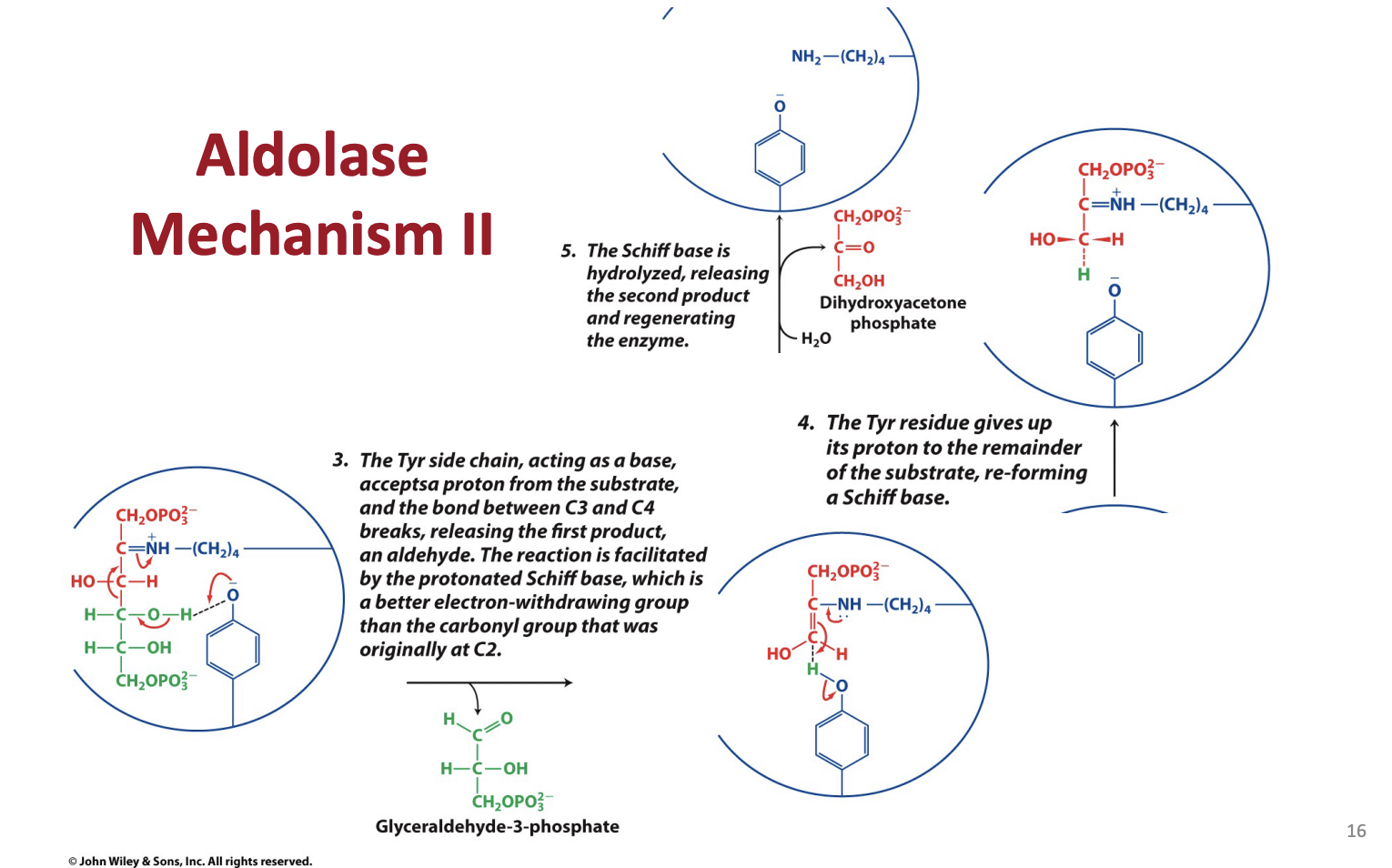
What are schiff bases and what is their role in aldolase
Describe the role of the thioesterintermediate in G3p dehydrogenates
What are the equations for glycolisis and lactate/ ethanol fermentation glycolisis