protein digestion + absorption
1/9
Earn XP
Description and Tags
week 12, lesson 2
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
proteins
more complex in structure than carbs
they can be composed of 20 diff amino acids
which can be folded into high older structures
proteins also vary in how many amino acids they are composed of, and how the protein folds upon itself
the sequence of amino acids in a row is called a primary structure
proteins arent usually a linear, straight form and the primary sequence of amino acid folds upon itself
proteins can be unfolded by
low pH in the stomach, a process which is known as denaturation
it allows for the protein digesting enzymes to recognize the amino acid sequences and break them apart
more enzymes are required to achieve the job of creating individual amino acids (the preferred absorbable form of proteins)
each protease (protein digesting enzyme) has a diff sequence of amino acids in the primary protein sequence that it will recognize and break apart.
dipeptide
2 amino acids bonded together
tripeptide
3 amino acids bonded together
peptide
4-50 amino acids bonded together
polypeptide
more 51 amino acids bonded together
In all of these types of protein strands, one end of the strand.. and the other end of the strand is called the
is called the amino terminus. carboxy terminus, based on the sequence of molecules that are exposed at either end of the amino acid sequences.
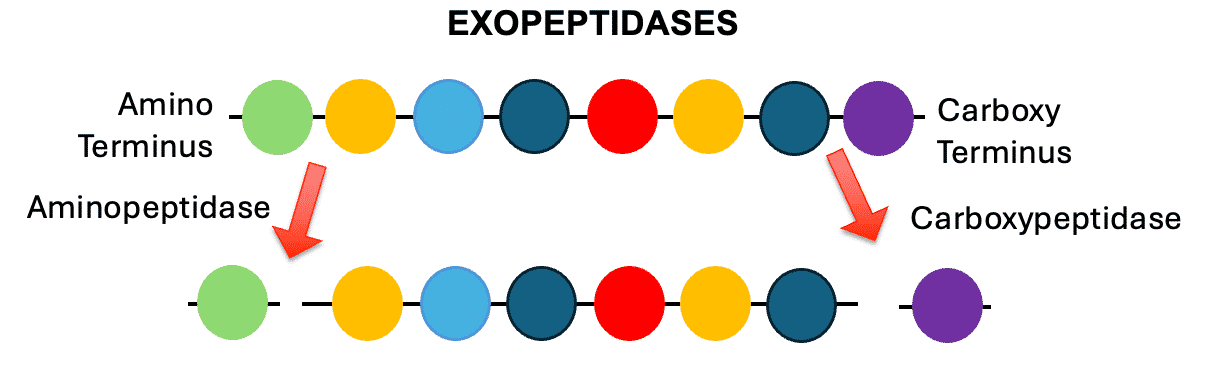
Exopeptidases
enzymes that recognize either end of a polypeptide/peptide. aminopeptidase recognizes the amino terminus of a polypeptide/peptide
it will remove whatever amino acid is in the last position of the strand
carboxypeptidase removes the last amino acid on the carboxy terminus of the polypeptide/peptide
endopeptidase
the rest of the proteases (enzyme) cut polypeptides/peptides based on amino acid sequence
these sequences usually reside somewhere in the internal portion of the polypeptide/peptide
for example, trypsin will cut the carboxy side of either the amino acid lysine or an arginine if it isnt followed by a proliine
other proteases can be less specific than this
Enterokinase is highly specific, recognizing a specific sequence that is found in trypsinogen, resulting in a smaller polypeptide, which is the active enzyme trypsin.
protein absorption
the preferred way to absorb protein is by individual amino acids
an apical Na amino acid symporter recognizes many amino acids and can move them across the apical membrane
However, amino acids come in various sizes due to their chemical composition and have different properties such that not all can be recognized by a single protein carrier.\
di- and tri- peptides can be absorbed across the apical membrane of enterocytes by an apical H+ symporter.
Once in the cell, cytosolic peptidases can digest these to single amino acids, for basolateral transport.
An amino acid uniporter on the basolateral membrane, exports the amino acids into the interstitial fluid, from where the amino acids can be absorbed and transported in the blood.