L23 - Reproductive Agein
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
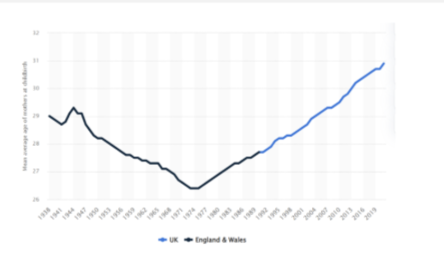
How has age at childbirth changed over time and why does it matter?
Age at childbirth has steadily increased since the 1970s
Average age ↑ from ~26 → >31 years
Driven by societal factors (education, careers, choice)
Your notes:
Increase largely due to choice
Important because fertility declines with age
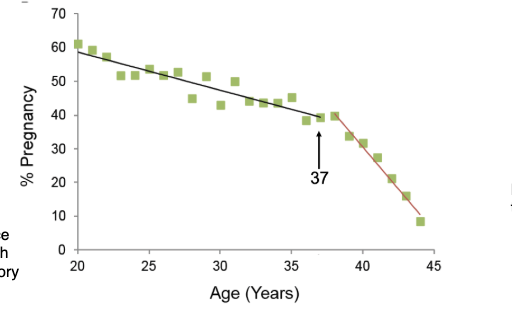
How does female fertility change with age?
Fertility gradually declines with age
Relatively stable until ~37 years
Then sharp decline
Based on natural fertility populations (no contraception)
Gradual decline until 37
Real-life fertility harder to measure (people don’t try continuously)
Data from communities with early/continuous childbearing
How does miscarriage risk change with female age?
Risk of miscarriage increases with age
Relatively low in 20s–early 30s
Sharp increase from ~37 years
Very high risk in 40s+
Your note:
At ~37 → sharp jump in pregnancies ending in miscarriage
How is nuclear DNA transmitted during fertilisation?
Egg and sperm each contribute one haploid set of chromosomes
Fertilised egg → diploid genome (one copy from each parent)
What is aneuploidy and how does it relate to female ageing?
Aneuploidy = incorrect number of chromosomes
~95% originates from the maternal side
Caused by errors during oocyte division
Increases with maternal age
Sharp rise from ~age 37
Often due to errors in female genome transmission
Dramatic increase with age
What are the reproductive consequences of aneuploidy?
Embryos may fail before implantation → infertility
Embryos may fail after implantation → miscarriage
Some survive → congenital abnormalities
Can occur at multiple stages of development

Why can aneuploidy often go unnoticed?
Early embryo loss may occur before pregnancy is recognised
Failure can happen at any stage (pre- or post-implantation)
Your notes:
Problems can occur at any step
May not even be recognised
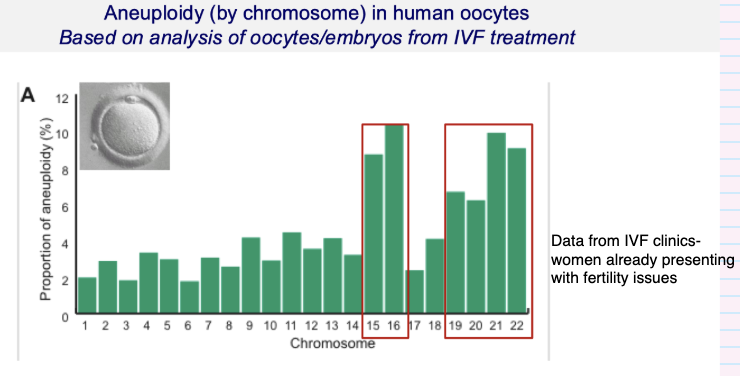
Are some chromosomes more prone to aneuploidy than others?
Yes — error rates vary by chromosome
Some chromosomes are more prone to errors (e.g. highlighted ones)
No chromosome is error-free
Based on IVF embryo/oocyte data
Identified from embryos that failed to progress
Shows which chromosomes most likely to go wrong
Data from IVF populations (fertility issues)
How does trisomy risk change with maternal age?
Trisomy = extra copy of a chromosome
Risk increases with maternal age
Sharp rise from ~37 years
Incidence increases dramatically at 37
What are the most common autosomal trisomies and their outcomes?
Trisomy 16: most common cause of miscarriage
Trisomy 18 (Edwards): often reaches term but early death
Trisomy 21 (Down’s): compatible with life
How has the prevalence of Down’s syndrome pregnancy changed over time?
71% increase in Down’s syndrome pregnancies (1989–2008)
Likely driven by increasing maternal age
Your note:
Mostly due to women having children later
What typically happens to Down’s syndrome pregnancies?
Majority end in:
Miscarriage
Pregnancy termination
Why are clinically observed trisomies considered the “tip of the iceberg”?
Most aneuploid embryos fail before implantation
Therefore never clinically recognised
Only a small fraction progress to later stages
Very few reach a stage resembling foetal life
What is the lifecycle of the female germline?
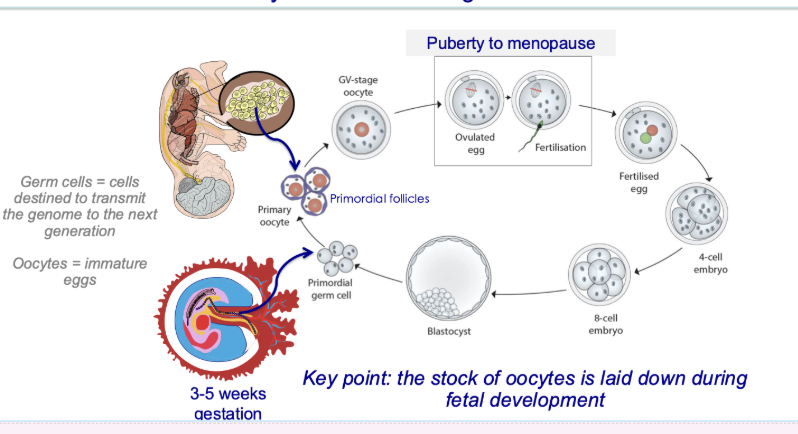
When and how are female germ cells formed?
Primordial germ cells form early (3–5 weeks gestation)
Oocyte pool established before birth (~7 million mid-gestation)
Finite supply — no new oocytes after birth
Your notes:
First cells laid down = primordial germ cells
~7 million halfway through gestation
If fetus is female → germ cells begin forming at 3–5 weeks
What happens to oocytes from fetal life to ovulation?
Oocytes remain arrested in ovary (germinal vesicle stage)
Stay there until stimulated for ovulation
Resume meiosis before ovulation:
Complete meiosis I
Enter meiosis II
Your notes:
Sit in ovary until ovulation
Sit in germinal vesicle stage until stimulated
What happens to oocytes during and after fertilisation?
Meiosis II only completed if fertilisation occurs
Fertilised egg → embryo → implantation
Your notes:
Go most of the way through meiosis II but only finish if fertilised
Healthy embryo implants in uterus
What is meiosis?
Specialised cell division producing haploid gametes from diploid cells
Involves:
1 round of DNA replication
2 rounds of chromosome segregation
Reduces chromosome number by half (“lessening”)
Compared to mitosis:
Mitosis = alternating rounds of DNA replication and segregation
What happens during mitotic cell division?
Replicated chromosomes → 2 sister chromatids
Chromatids align on spindle
Sister chromatids separate into daughter cells
Outcome:
Cells retain 2 copies of each chromosome
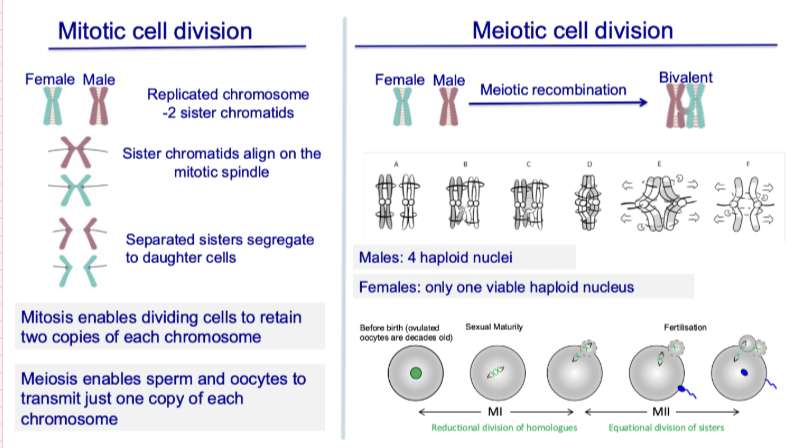
What happens during meiotic cell division?
Homologous chromosomes pair (bivalents)
Meiotic recombination occurs
Two divisions:
Meiosis I → separates homologues
Meiosis II → separates sister chromatids
Outcome:
Haploid cells (1 copy of each chromosome)
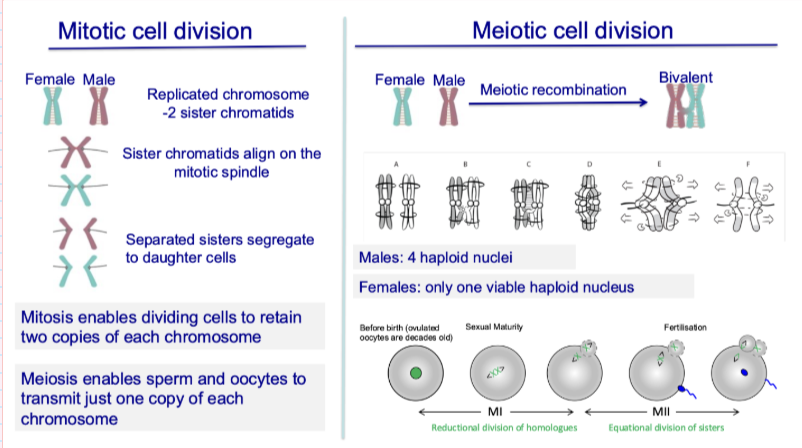
How does meiosis differ between males and females?
Males: 4 haploid sperm produced
Females: only 1 viable oocyte (others discarded as polar bodies)
Your notes:
Oocyte discards chromosomes to maintain large size
Early embryo has no blood supply → relies on stored resources
Unequal division occurs again in second meiotic division
How does the oocyte pool change across the lifespan?
Oocyte stock established before birth
Declines continuously with age (ovarian ageing)
Vast majority of oocytes undergo atresia (die)
Numbers:
Birth: ~1 million
Menopause (~45–55 yrs): <1,000
Primordial follicles are recruited throughout life
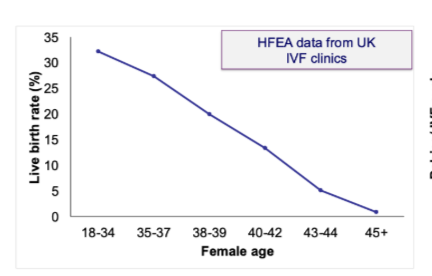
Why does fertility decline with age despite IVF?
Age-related fertility decline not rescued by IVF using own eggs
Suggests problem lies with the oocyte (egg), not uterus
Your notes:
Issue is the oocyte itself
Evidence from use of donor eggs
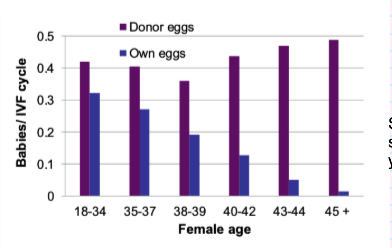
What does donor egg IVF tell us about female fertility decline?
Using donor eggs from younger women:
IVF success rates remain high across ages
Shows fertility decline is due to egg quality, not maternal environment
Your notes:
Success rate stays high with donor eggs
Confirms age-related infertility = egg problem
How are ovarian ageing and aneuploidy related?
Ovarian ageing = decline in number of oocytes
Occurs alongside ↑ aneuploidy in oocytes
Cause:
Chromosome segregation errors during meiosis
Key idea:
Fewer eggs + lower quality with age
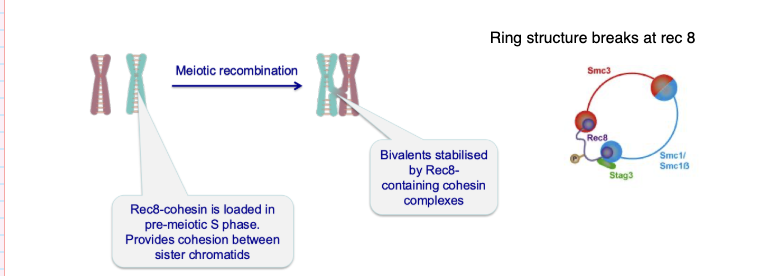
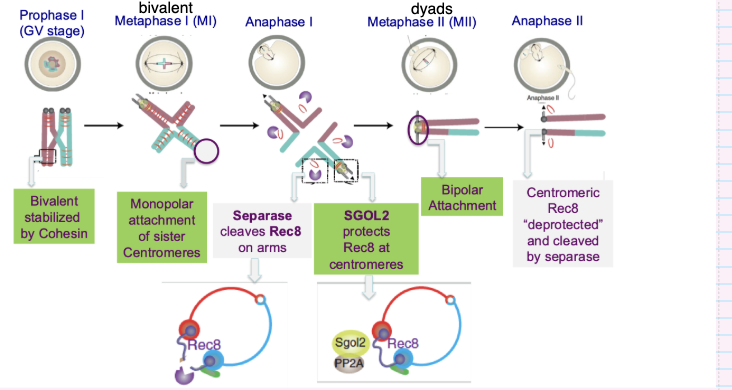
How are bivalent chromosomes held together during meiosis?
Homologous chromosomes pair via meiotic recombination
Form bivalents
Stabilised by cohesin (Rec8-containing complexes)
Cohesin holds sister chromatids together
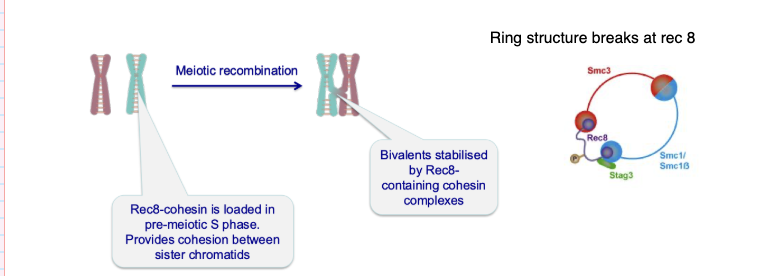
Why does cohesion failure lead to aneuploidy with age?
Cohesin is not replenished over time
Weakens during long arrest in oocytes
Leads to chromosome segregation errors
Sister chromatids must stay attached until meiosis II
Must be released at the correct stage
Errors in timing → aneuploidy
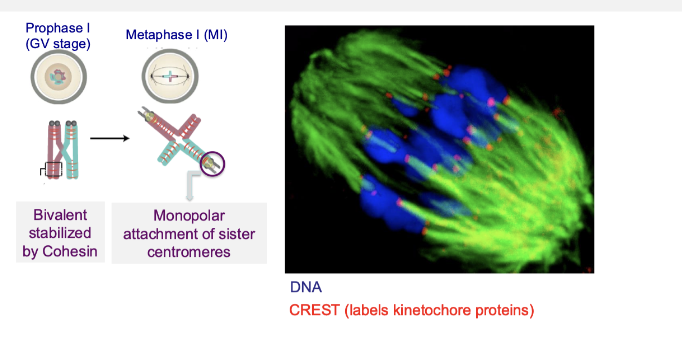
How do chromosomes attach to the spindle during meiosis I?
Sister centromeres attach to microtubules from the same spindle pole (monopolar attachment)
Bivalents stabilised by cohesin
Ensures homologous chromosomes (not sisters) separate in meiosis I
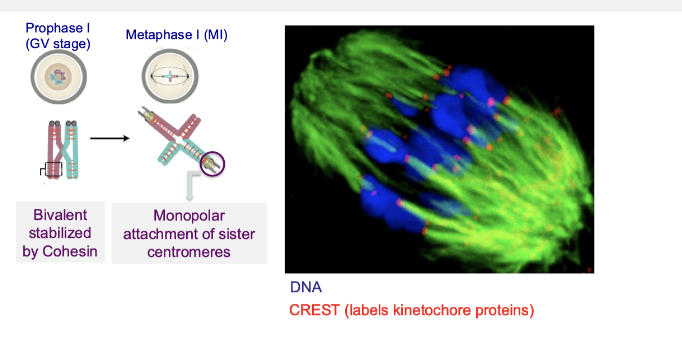
What changes in chromosome attachment and cohesion during meiotic division?
Cohesion must be removed stepwise during meiosis
At division:
Chromosomes reorient → pulled to opposite poles
Cohesion broken between homologues
Outcome:
One set lost in polar body, one retained in oocyte
Cleavage must occur stepwise
Cohesion breaking is tightly controlled
Ensures correct chromosome segregation
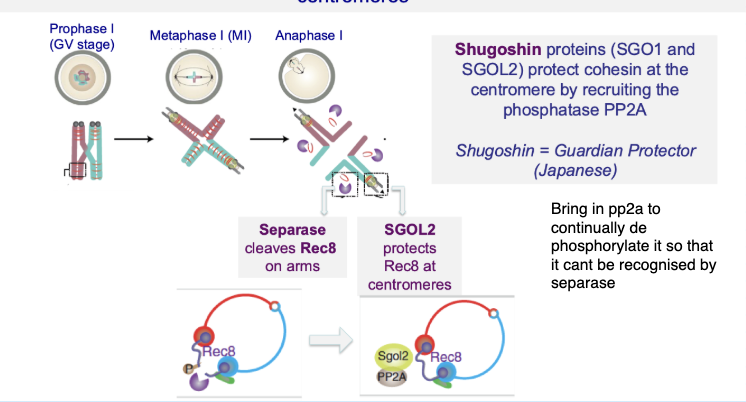
What happens to cohesin during the first meiotic division (meiosis I)?
Cohesin is removed from chromosome arms
Allows homologous chromosomes to separate
Cohesin at centromeres is preserved
Your notes:
Removed from arms but kept at centromeres
Cohesion removal is highly specific
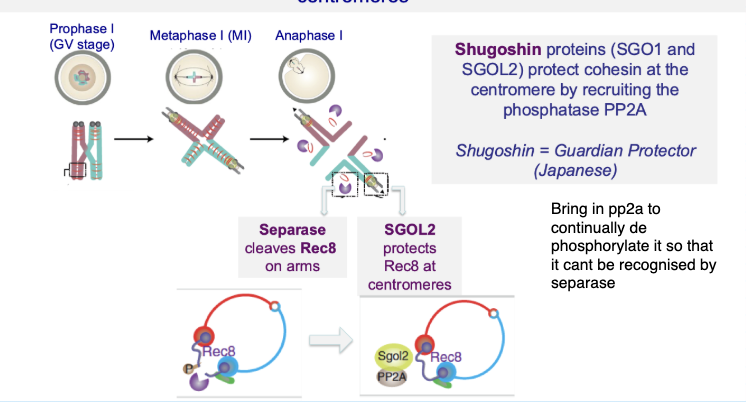
How is centromeric cohesin protected during meiosis I?
Shugoshin (SGO1/SGO2) protects centromeric cohesin
Recruits PP2A phosphatase
Prevents cohesin (Rec8) from being cleaved by separase
Your notes:
Protection depends on phosphorylation state
Centromeric cohesin cannot be recognised by separase
Ensures sister chromatids stay together until meiosis II
What is the role of centromeric cohesin in meiosis II?
Enables bipolar attachment of sister centromeres
Allows sister chromatids to attach to opposite spindle poles
Ensures accurate segregation in meiosis II
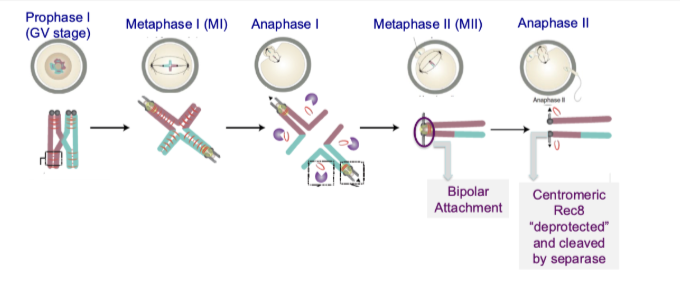
What happens to centromeric cohesin before and during meiosis II?
Shugoshin is removed/degraded between meiosis I and II
Centromeric cohesin becomes “deprotected”
Separase cleaves cohesin (Rec8)
Sister chromatids finally separate
Shugoshin removal is key step between M1 and M2
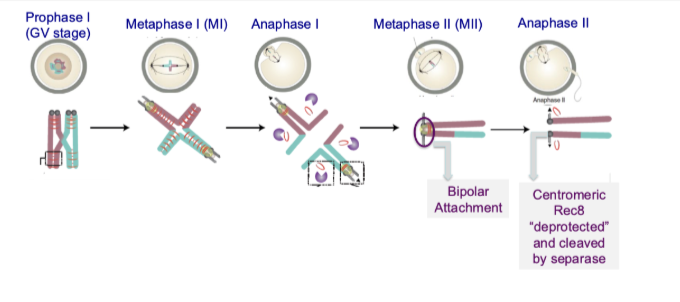
What are the key steps controlling chromosome segregation in meiosis I?
Bivalents stabilised by cohesin
Monopolar attachment of sister centromeres
Separase cleaves cohesin on chromosome arms
Homologous chromosomes separate
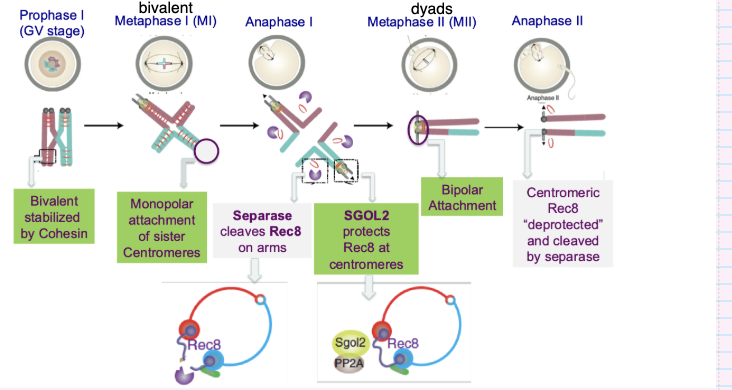
What ensures correct chromosome segregation in meiosis II?
SGO2 protects centromeric cohesin during meiosis I
In meiosis II:
Bipolar attachment of sister chromatids
Shugoshin removed → cohesin deprotected
Separase cleaves centromeric cohesin
Sister chromatids separate → haploid genomes formed
What meiotic errors occur in aged oocytes due to loss of centromeric cohesion?
What meiotic errors occur in aged oocytes due to loss of centromeric cohesion?
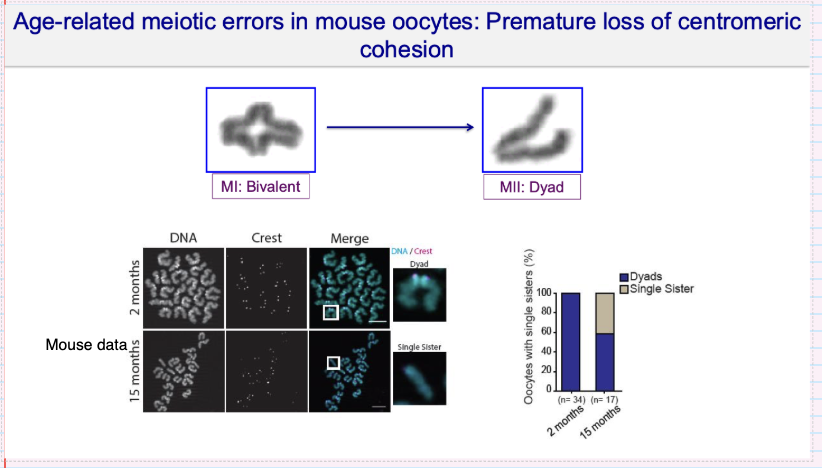
What molecular changes underlie age-related cohesion loss in oocytes?
↓ Cohesin (Rec8) levels
↓ Shugoshin (SGOL2) protection at centromeres
Reduced recruitment of SGOL2
Result:
Centromeric cohesion not maintained
↑ risk of premature chromatid separation
Your notes:
Less Rec8 and Shugoshin with age
Weakens protection of centromeric cohesion
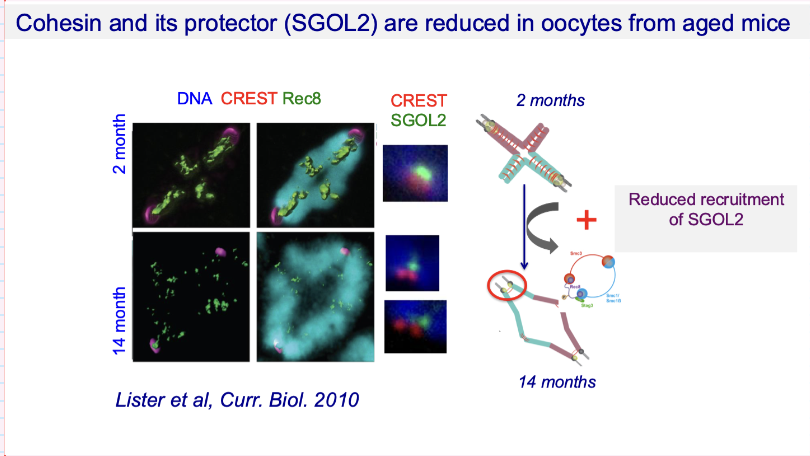
How does centromeric cohesion loss relate to female age in human oocytes?
Premature loss of centromeric cohesion ↑ with age
In women >40:
50–100% of oocytes show prematurely separated sister chromatids
Consequence:
Major cause of aneuploidy
Your note:
These prematurely separated chromatids are the ones that lead to errors
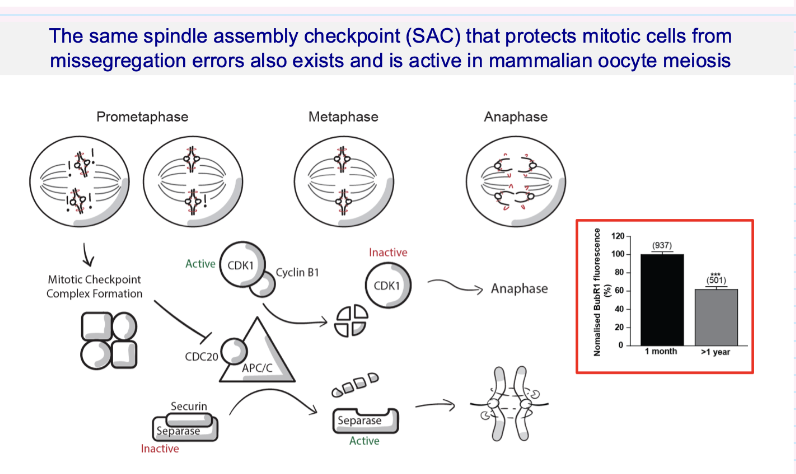
What is the role of the spindle assembly checkpoint (SAC) in oocyte meiosis?
SAC ensures chromosomes are correctly attached to the spindle before separation
Prevents chromosome missegregation
Works via:
Inhibiting APC/C → separase inactive until ready
Once satisfied → separase activated → chromosomes separate
Key point:
SAC is present and active in oocyte meiosis (like mitosis)
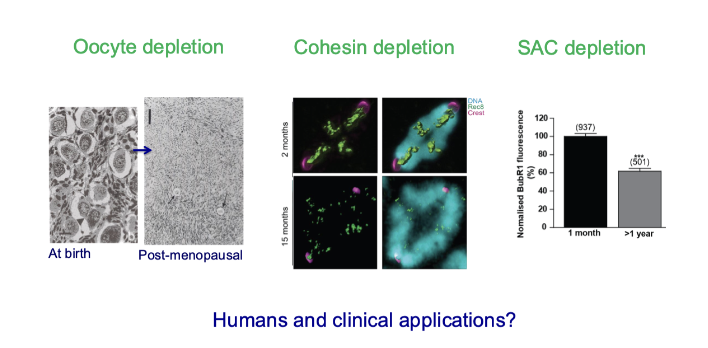
What are the “ticking clocks” of oocyte ageing?
Three parallel but independent ageing processes:
Oocyte depletion (loss of egg number)
Cohesin depletion (chromosome instability)
SAC decline (weaker error-checking)
Ovarian ageing, Chromosomal ageing, Cell cycle ageing
Occur in parallel but are mechanistically distinct