water as a medium of life
1/22
Earn XP
Description and Tags
a1.1-a4.2
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
water as the medium of life
hardly ever changes chemcially, without it most chemcial reactions within organisims would not take place- remains liquid at a wide range of temperatures allowing it to keep constat and dissolve and transport substances
water is made up of
two hydrogen atoms covalently bonded to an oxygen atom (h2O)
Polarity
describes the slight difference in charge that occurs at the different poles of water molecule
oxygen(due to having higher electronegativity) attracys the aelectron more strongly, forming a slightly negative charge
the hydrogen atoms have a weaker attraction towards the electrons resulting in a slightly positive charge
polarity of water
allows it to form weak associations with other polar molecules or charged ions
slightly negative poles will attract slightly positive poles of other molecules and vice versa
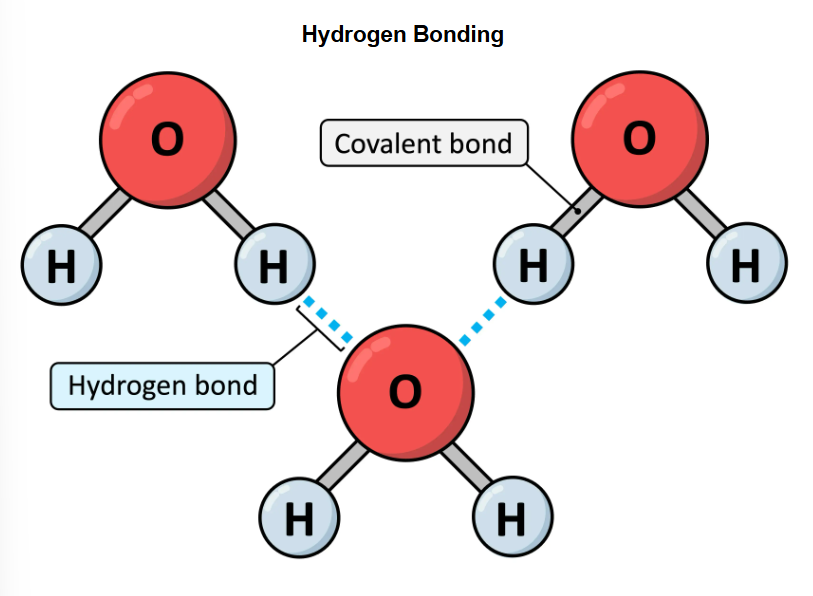
Hydrogen bonds
intermolecular associations fromed as a consequences of the polar covalent bonds within a water molecules
hydrogen bonds part two
these bonds forms when a hydrogen atom is attracted to a fluorine, oxygen or nitrogen atom of another molecule
hydrogen bonds are stronger than standard polar associations due to the particularly high electronegativity
hydrogen bonding
responsible for several unique properties of water including thermal cohesive and solvent properties
cohesion in water
water molecule is able to stick to other water molecules allows the liquid to resist low levels of external force(it creates surface tension)
Surface tension in water
high surface tension o water makes it sufficiently dense for certain smaller organisims to move along its surface
water strides
small insects that can move along the surface of water due to its high surface tension
they distribute their weight via long legs that contain thousands of microscopic hairs to trap air and increase buoyancy
high surface tension of water makes it sufficiently dense for certain smaller organisims to move along its surface
Adhesion in water
water molecule is able to stick to other polar or charged molecules via the formation of polar associations
attraction to charged polar surfaces (such as cellulose cell walls) allows water to flow in opposition to gravitational forces (capillary action)
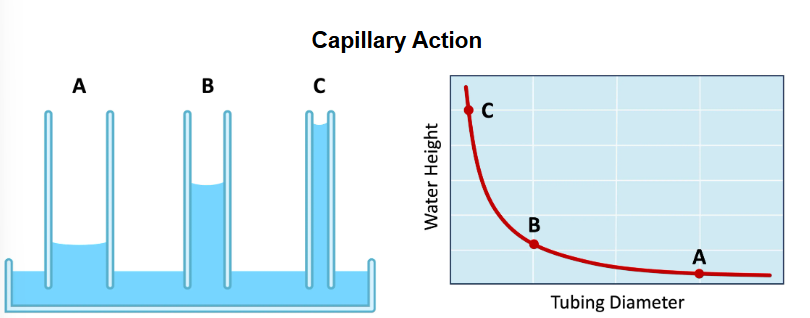
Capillary action in water
necessary to allow water to be transported up plant stems via transpiration stream
the loss of water vapour from the leaves (via ecaporation) and the absorption of water into the roots (via osmosis) creates a pressure gradient
water will move along the gradient by using capillary action and cohesion to be transported up the stem of the plant via narrow xylem vessels
Capillary action
responsible for the movement of water through the soil from the deeper water table
the type of soil will influence the strentgh of the capillary action dependant on how porous it is affection the efficact of aricultural practices
Solvation
process by which solvent molecules sorround and interact with solute molecules
a solute is any substance (usually a solid) that is dissolved by a solvent molecule to from a solution
solute that are able to be dissolved by a solvent are considered to be soluble (insoluble substances will not form a solution)
Water as a universal solvent
due to its capactiy to dissolve a large number of substances
water can dissolve any substance that contains charged particles (ions) or electronegative atoms (polarity)
The polar regions of the water molecule associate with molecular surfaces that have an opposing charge, forming dispersive hydration shells
Metabolic medium
The capacity of water to dissolve a large varity of substances makes it an important medium for metabolic reactions
solutes dissolved into aqueous solutions are more likely to colled with enzymes and undergo necessary chemical reactions
water can also promote enzyme activity b absorbing heat from exothermic reactions and maintain acid-base neutrality
Hydrophilic and hydrophobic
substances that are involved in metbolic reactions can be described as being either hydrophilic or hydrophobic
hydrophilic-substances that freely associate and readily dissolve in water and includes polar molecules and ions
hydrophobic0 do not freely associate of dissolve in water and inclues all large non polar molecules (lipis ie fats and oils)
Transport medium
the movement of water soluble substances in plants and animals involves different transport systems
in aniamls the blood plasma transports, amina acids simple sugars , urea and small amounts of gases
in vascular plants, mineral ions are transported bia xylem vessels while dissolved nutrients are transported via the phloem
non soluble substances that are transported in aqueous enviroments
animals-lipids are packages with proteins to form a water soludle lipoprotein that can be transported via the blood
the mechanisims of lipid transport is not well understood in plants but may involved conjugation of amino acids and transport via the phloem
Buoyancy
the upward force applied to an object in a medium and is determined by the density of the medium
as water is more dense than air it applied a great upward force which allows objects to float in water- the capacity of an object in water will be determined by its relative weight
viscosity
a measure of a fluids tendancy to flow (more viscous- more resistance)
water is more viscous than air as it can form hydrogen bonds which increase the friction fo flowing molecules
Thermal conductivity
a measure of a mediums ability to move heat across a temperature gradient
water absorbs and transfers heat more readily than air because water particles are packed more tightly together
specific heat capacity
the amound of energy require to raise the temperatur of 1g of substance by 1c
the high specific ehat capacity of water makes it an effective coolant in sweat (sweat absorbs a lot fo thermal energy resulting in a high latent heat of vaporisations)
remember loon and seal