Chemistry - Organic chemistry
1/152
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
153 Terms
What are hydrocarbons?
molecules consisting of only carbon and hydrogen
What are the trends of the hydrocarbons?
As the molecules get bigger, the:
- boiling point increases
- viscosity increases
- flammability decreases
- they burn with a smokier flame
- Volatility decreases
Why does the boiling point increase as hydrocarbons get bigger?
larger molecules have greater surface area, resulting in stronger intermolecular forces
What is viscosity?
The resistance of a liquid to flow
Why does the viscosity increase as hydrocarbons get bigger?
longer carbon chains have more surface area, resulting in stronger intermolecular that cause greater internal friction
Why does the flammability decrease as hydrocarbons get bigger?
As hydrocarbon chains get longer because stronger intermolecular forces make them less volatile
What is volatility?
how easily a liquid evaporates
Why does the volatility decrease as hydrocarbons get bigger?
the intermolecular forces between the molecules become stronger, requiring more energy to turn the liquid into a gas
What are alkanes?
saturated hydrocarbons
What is the general formula for alkanes?
CₙH₂ₙ₊₂
What are the first five alkane molecules?
- methane - CH₄
- ethane - C₂H₆
- propane - C₃H₈
- Butane - C₄H₁₀
- pentane C₅H₁₂
What does saturated mean?
- A hydrocarbon that contains the maximum number of hydrogen atoms possible, leaving no room for addition reactions, making them relatively stable
- They only have single bonds
What is a homologous series?
a family of organic compounds with the same functional group and general formula
What type of hydrocarbon is most common in crude oil?
Alkanes
What are crude oils?
a finite, non-renewable fossil fuel found in rocks, formed over millions of years from buried plankton
How are crude oils formed?
Over millions of years, heat and pressure turn the buried remains of ancient plankton into a mixture of hydrocarbons
What is fractional distillation?
A process used to separate mixtures of substances with different boiling points
How does fractional distillation work?
1. crude oil is heated to around 350°C, so all the crude oil vapourises
2. The vapourised crude oil enters the fractional column
3. The column is hotter at the bottom and cooler at the top (around 30°C)
4. Different hydrocarbons condense at different heights depending on their boiling points
- small molecules (like refinery gases and petrol) have low boiling points, so they condense at the top
- large molecules(like bitumen) have a high boiling point and condense at the bottom
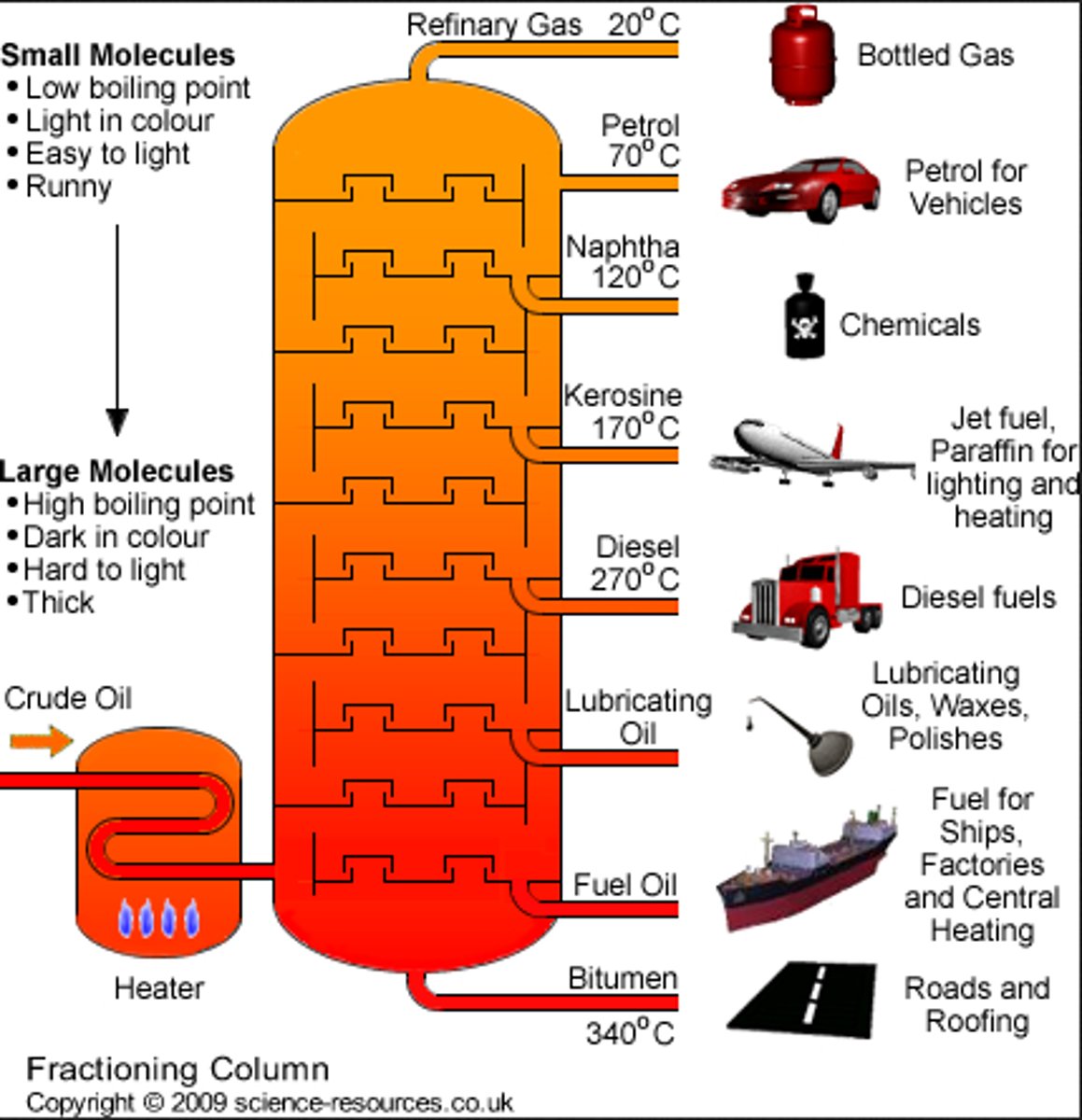
What are smaller hydrocarbons used for?
fuels such as petrol, diesel, and liquefied petroleum gas because they ignite easily and release significant energy.
What are larger hydrocarbons used for?
- they are less useful as fuels due to their high viscosity and low flammability.
- Instead, they are primarily used as feedstock to make lubricants, detergents, and polymers
What is complete combustion?
- There is enough oxygen available for all the carbon atoms to burn and form carbon dioxide
- it is safe and releases the maximum amount of energy
What is incomplete combustion?
- When there is not enough oxygen to burn so the carbon and hydrogen atoms have not been fully oxidised
- it produces less energy than complete combustion
Complete combustion of hydrocarbons
Hydrocarbon + oxygen + heat →carbon dioxide + water
Incomplete combustion of hydrocarbons with limited oxygen
Hydrocarbon + limited oxygen →carbon monoxide + water
Incomplete combustion of hydrocarbons with very limited oxygen
Hydrocarbon + very limited oxygen →carbon + water
Why is incomplete combustion dangerous?
- carbon monoxide is poisonous and replaces the oxygen the red blood cells are carrying
- soot (carbon) causes breathing problems and damages buildings
What is cracking?
a chemical process used to break down long-chain hydrocarbons into shorter, more useful ones
What does cracking make?
a shorter alkane and an alkene from a larger alkane
How does cracking work?
cracking requires
- heat
- sometimes a catalyst to speed up the reaction
Why are shorter hydrocarbons more useful?
They are more volatile, easier to ignite, and burn cleaner than longer chains
Example of a cracking reaction
C₁₀H₂₂ →C₈H₁₈ + C₂H₄
What are alkenes?
- Unsaturated hydrocarbons.
- Contain at least one carbon to carbon double bond in their structure.
What is the functional group of alkenes?
C=C
What is the functional group of a molecule?
a specific arrangement of atoms within a molecule responsible for its characteristic chemical reactions and physical properties
What is the general formula of alkenes?
CₙH₂ₙ
What is the displayed formula of ethane?
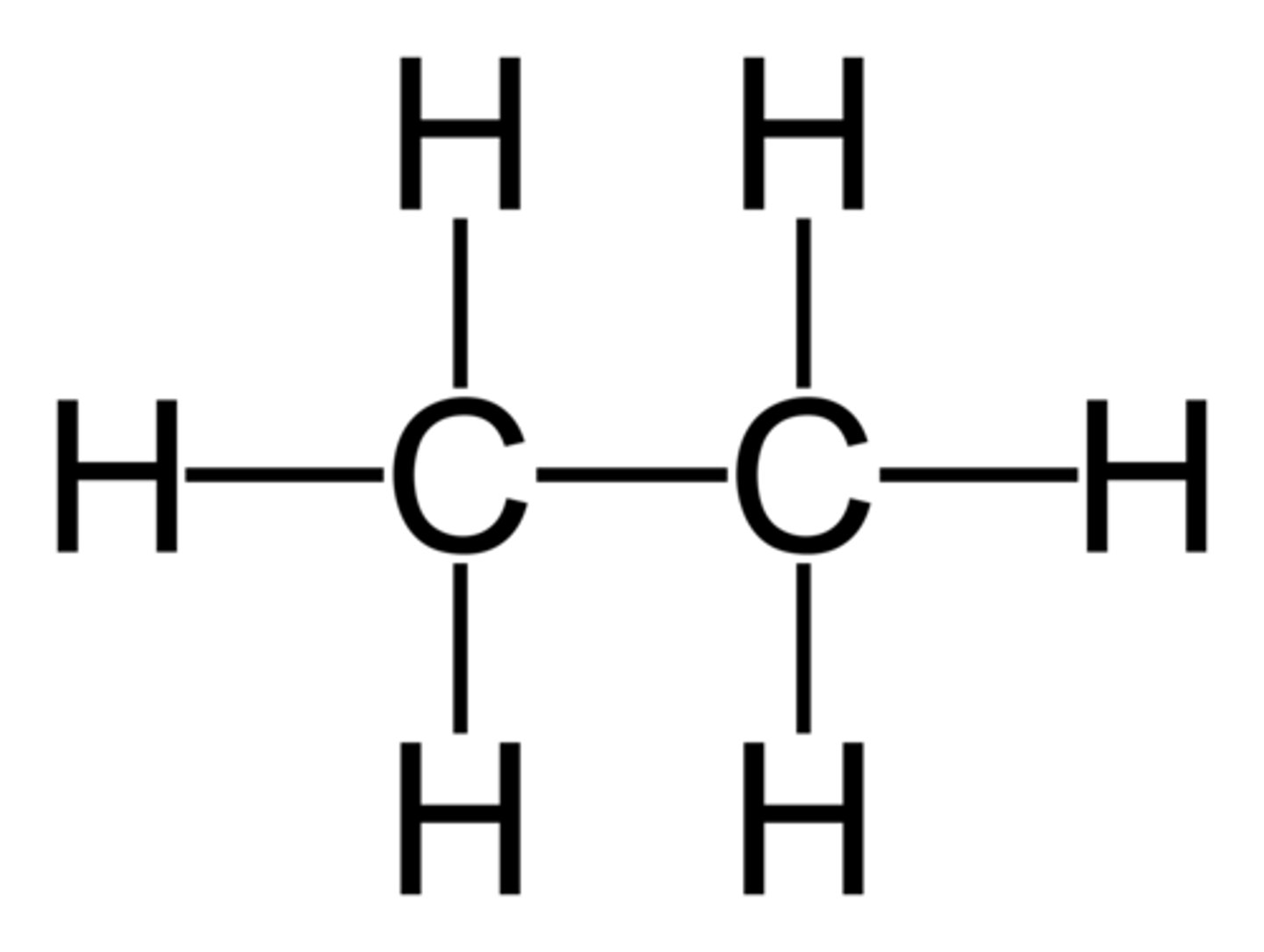
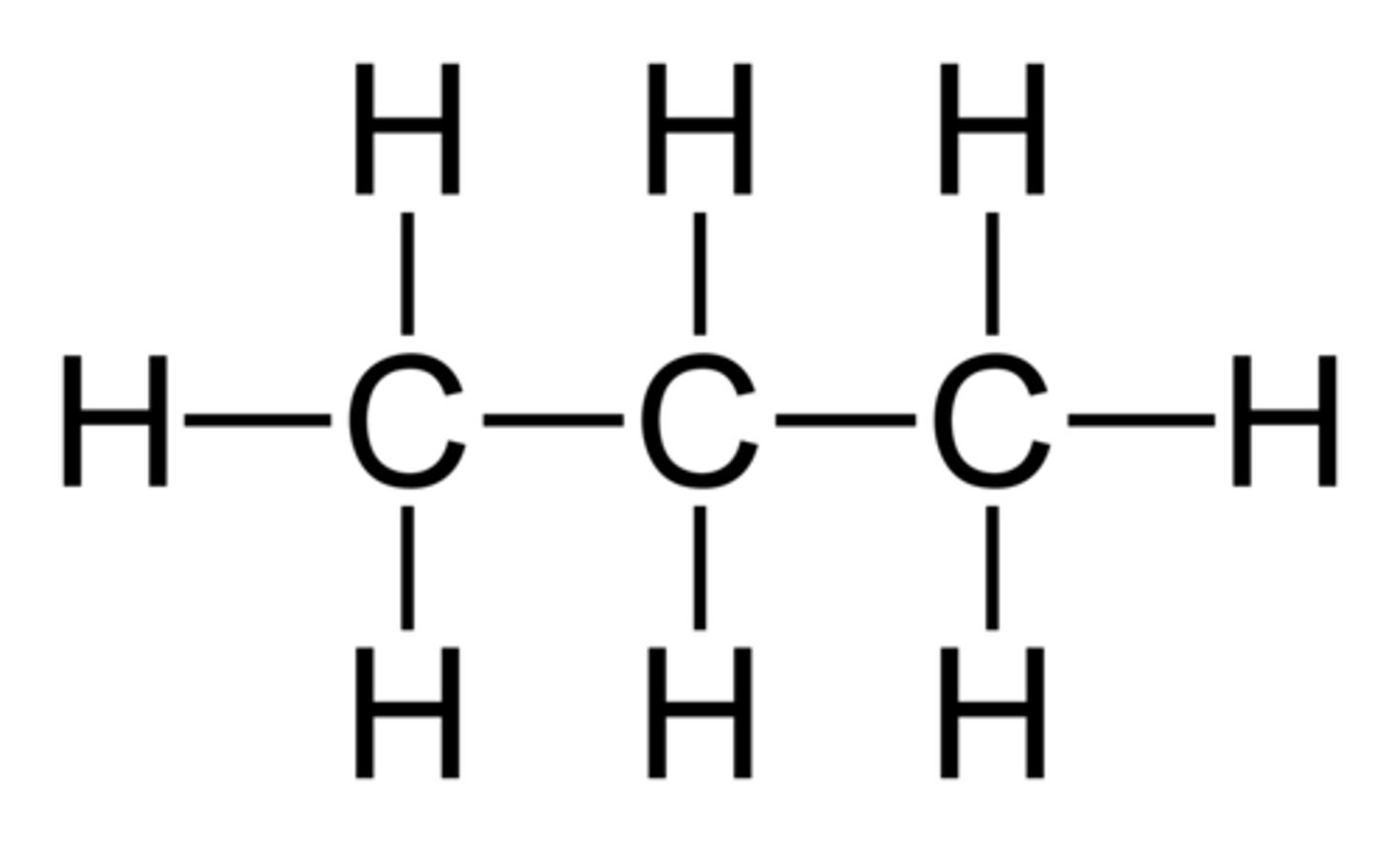
What is the displayed formula of propane?
What are the properties of alkenes?
- they are more reactive than the alkanes
- they burn with a smokier flame
- they are more likely to undergo incomplete combustion
- they can be used to make polymers
why are longer chain hydrocarbons more likely to undergo incomplete combustion
they require a higher ratio of oxygen to carbon for complete oxidation
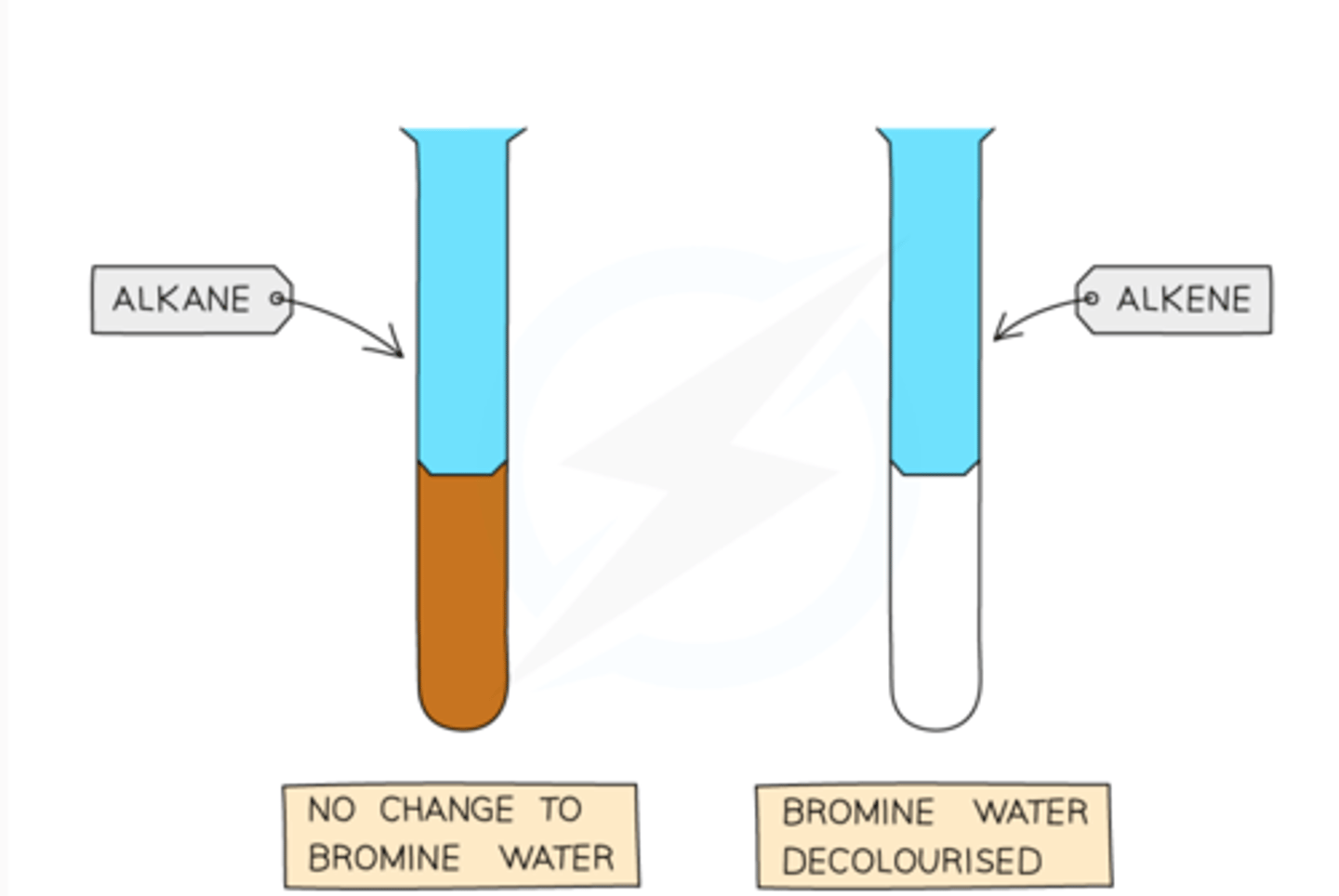
How can you test for alkenes?
- testing with bromine water
- the orange bromine will turn colourless when it reacts with an alkene
Why are alkenes more reactive than alkanes?
The carbon double bonds are more likely to break than C-H bonds and are therefore more reactive
Why are alkenes more likely to undergo incomplete combustion?
because they have a higher carbon-to-hydrogen ratio. Due to this higher proportion of carbon, they require more oxygen for complete combustion
Why do alkenes burn with a smokier flame than alkanes?
- because they have a higher ratio of carbon to hydrogen, leading to incomplete combustion.
- When burned in air, this higher carbon content causes unburnt carbon particles (soot) to form, creating a yellow, smoky flame compared to the clean blue flame of alkanes.
What are the first 5 alkenes
- ethene C₂H₄
- propene C₃H₆
- Butene C₄H₈
- Pentene C₅H₁₀
- hexene C₆H₁₂
Why does methene not exist?
because alkenes need a double bond
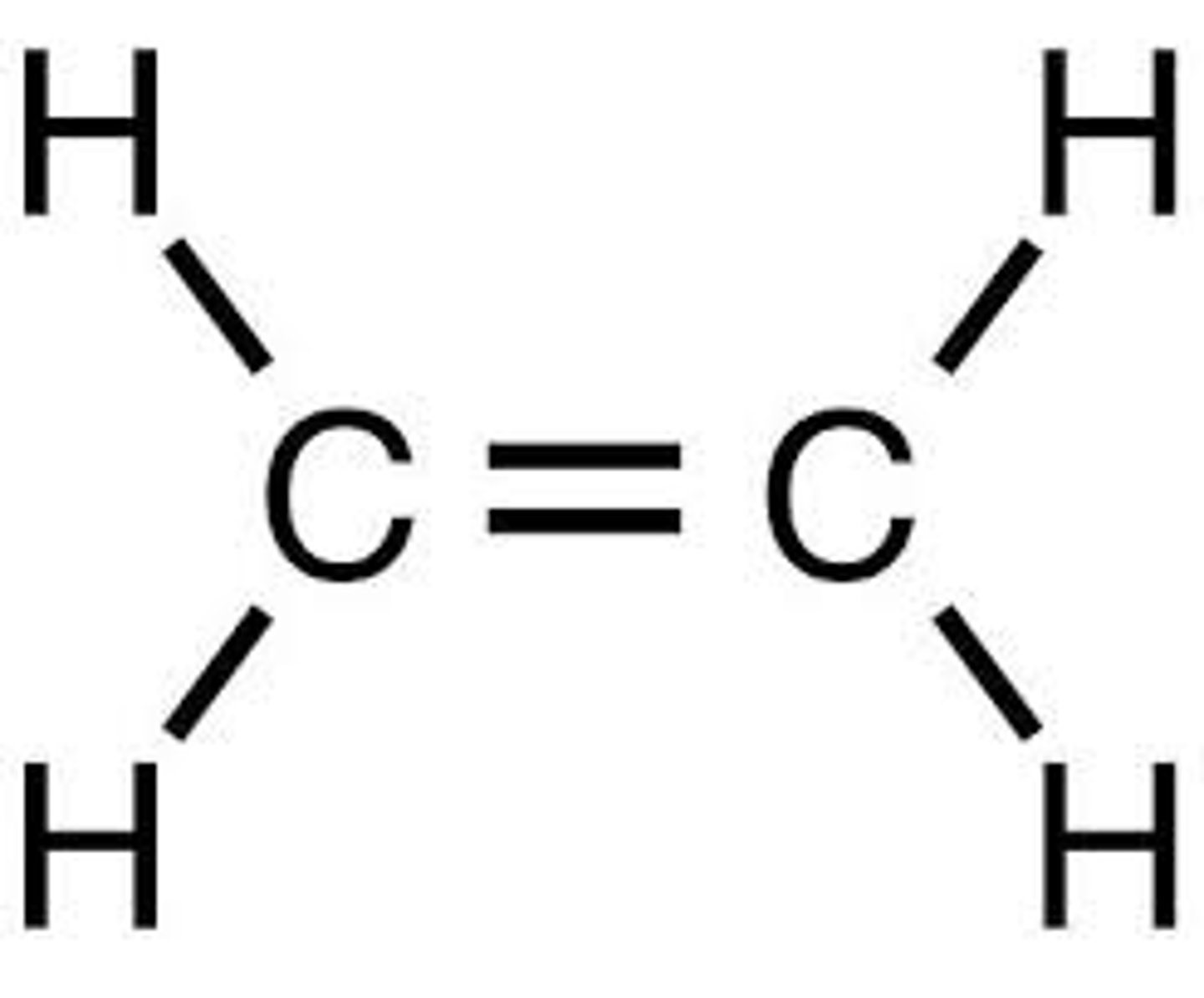
What is the displayed formula for ethene?
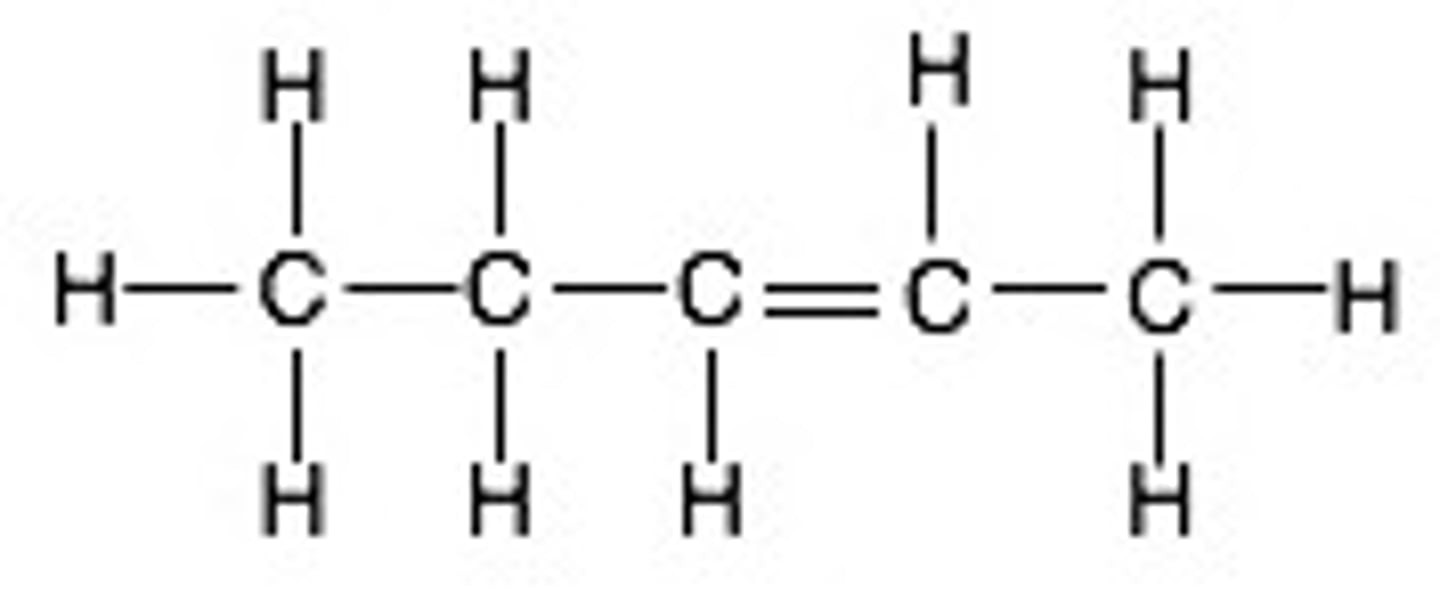
What is the displayed formula for pentene?
How many bonds does each carbon have in the alkenes?
Each carbon atom has four covalent bonds
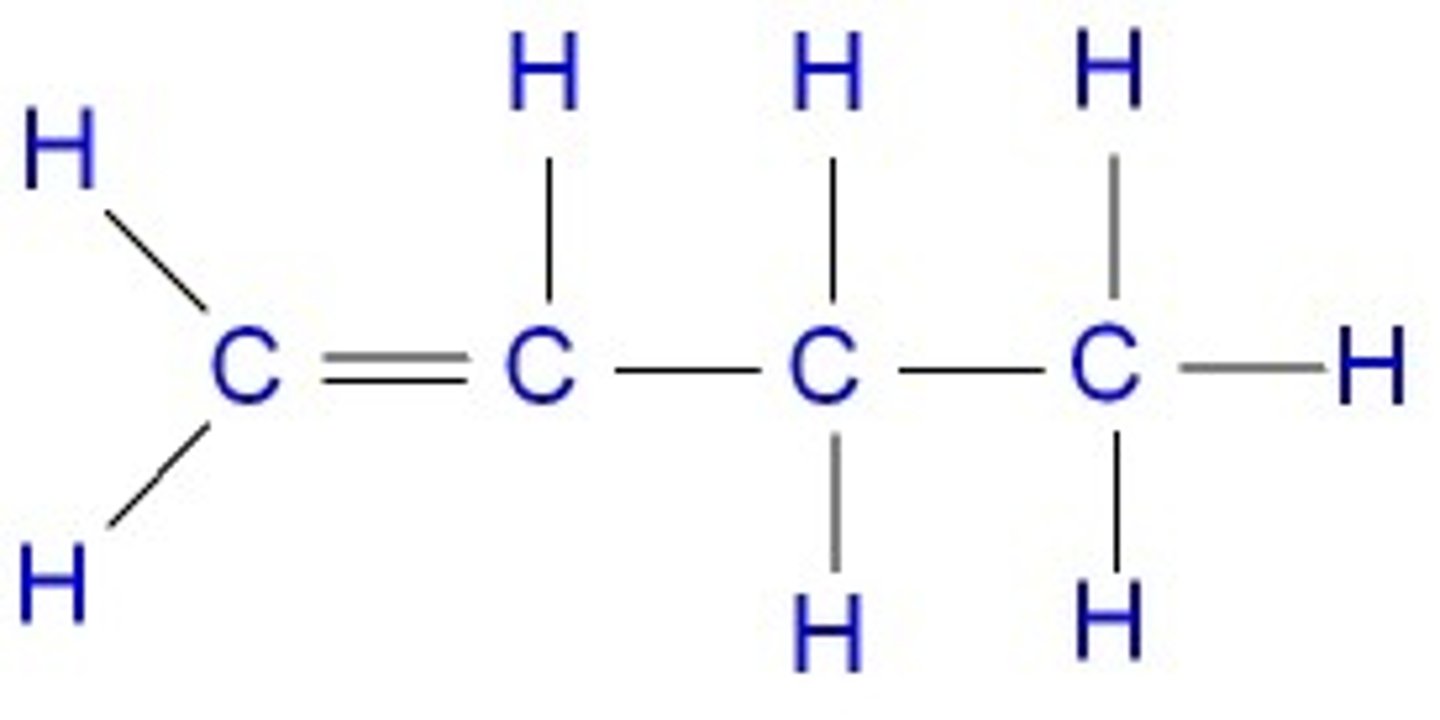
What is the displayed formula for butene?
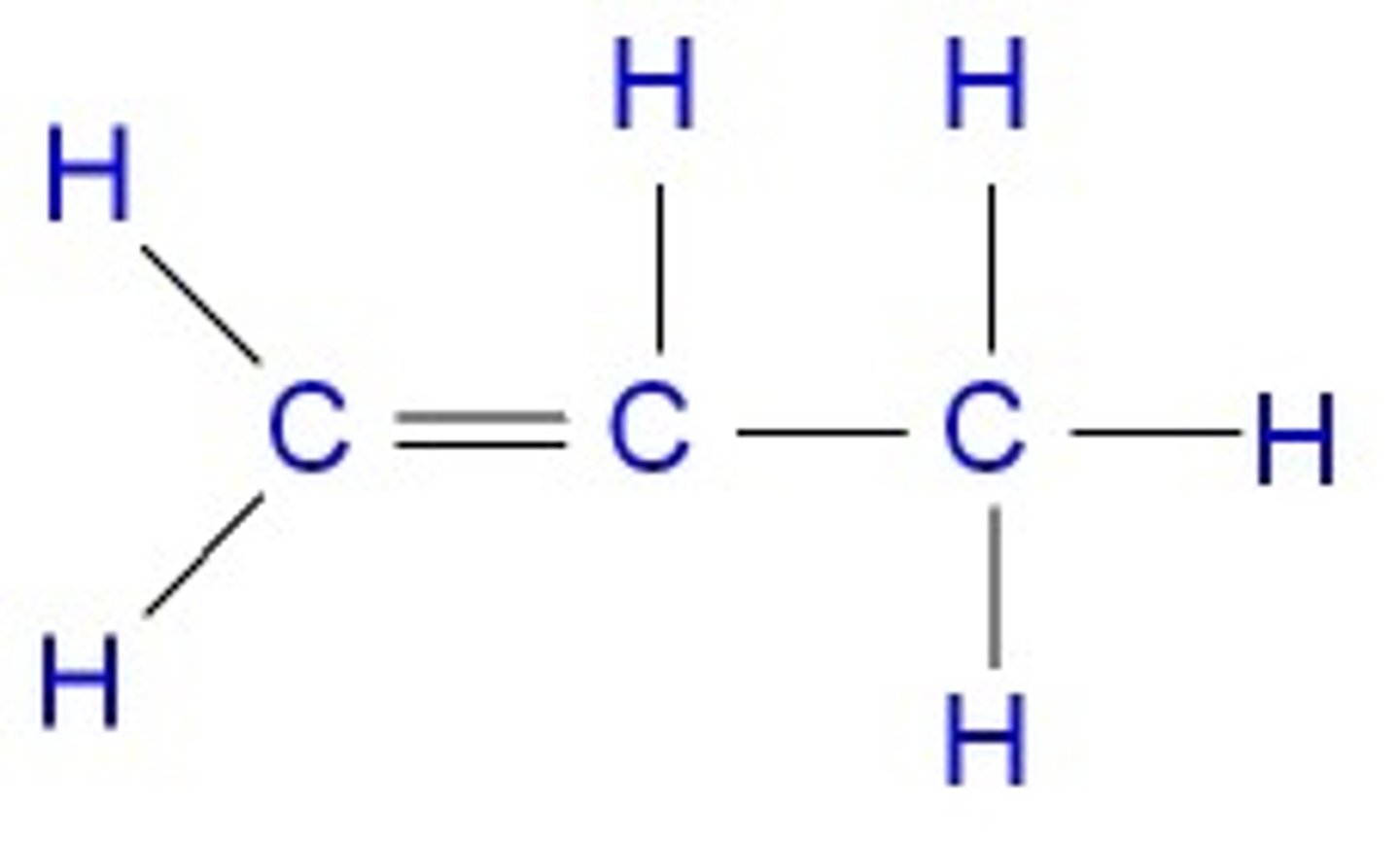
What is the displayed formula for propene?
What are addition reactions?
where two or more molecules combine to form a single, larger product
Why does bromine water turn colourless when it reacts with an alkene?
- because the orange bromine molecules react with the alkene's carbon-carbon double bond in an addition reaction.
- The double bond breaks, and the bromine atoms add across it to form a colourless dibromoalkane
How do alkenes react with hydrogen?
The carbon-carbon double bond breaks, and hydrogen atoms add to each carbon, forming and alkane
What is hydrogenation?
is a chemical reaction that adds molecular hydrogen to another compound
Formula for hydrogenation of alkenes
alkene + hydrogen→alkane
What are substitution reactions?
a chemical reaction where one atom, ion, or functional group in a molecule is replaced by another atom, ion, or group
What does the hydrogenation of alkenes require?
- heat
- a nickel catalyst
Formula for ethene + hydrogen
ethene + hydrogen→ethane
C₂H₄ + H₂ →C₂H₆
What are elimination reactions?
An organic reaction where two atoms or groups (often a hydrogen and a leaving group) are removed from adjacent carbon atoms, forming a double or triple bond
Formula for alkene + steam
alkene + steam →alcohol
What is a hydration reaction?
adding a water molecule to an unsaturated organic substrate
What is needed for the hydration of alkenes?
- a phosphoric acid catalyst
- high pressure and heat
Formula for ethene + steam
ethene + steam→ethanol
C₂H₄ + H₂O→C₂H₅OH
Formula for alkene + halogen
alkene + halogen→dihaloalkanes
Formula for ethene + bromine
ethene + bromine→dibromethane
C₂H₄ + Br₂→C₂H₄Br₂
What are alcohols?
a homologous series of organic compounds
What is the functional group of the alcohols?
-OH (hydroxyl group)
What is the general formula of the alcohols?
CnH₂n+₁OH
What are the first 4 alcohols?
- methanol - CH₃OH
- ethanol - C₂H₅OH
- propanol - C₃H₇OH
- Butanol - C₄H₉OH
What happens when alcohols are added to water?
they completely dissolve
What are the properties of alcohols?
- flammable
- neutral pH
- shorter chains dissolve in water
What does flammable mean?
a substance that can catch fire and burn easily at normal temperatures
Why are alcohols flammable?
they contain carbon and hydrogen atoms that readily react with oxygen (burn) to release large amounts of heat energy
Why do longer chains of alcohols not dissolve well in water?
because their molecular structure becomes increasingly non-polar (hydrophobic) as the hydrocarbon chain gets longer
What are alcohols used for?
- fuels
- solvents
- alcoholic drinks (ethanol)
What is fermentation?
- a reaction where glucose is broken down to form ethanol and carbon dioxide
- Fermentation is an exothermic reaction, the released energy keeps the yeast alive
Formula for fermentation
Glucose →ethanol + carbon dioxide
What are the conditions for fermentation?
- yeast
- 30°C
- anaerobic conditions
What is ethane used for?
primarily used as a feedstock
What is methane used for?
a fuel for generating electricity, heating homes, cooking food, and powering industrial processes
What is propane used for?
a fuel in domestic and industrial settings
What is butane used for?
a fuel for portable stoves, cigarette lighters, and torches
What are alkenes used for?
chemical feedstocks to manufacture plastics, alcohols, and industrial chemicals
What is feedstock?
a raw material used to provide reactants for an industrial chemical reaction, primarily derived from crude oil
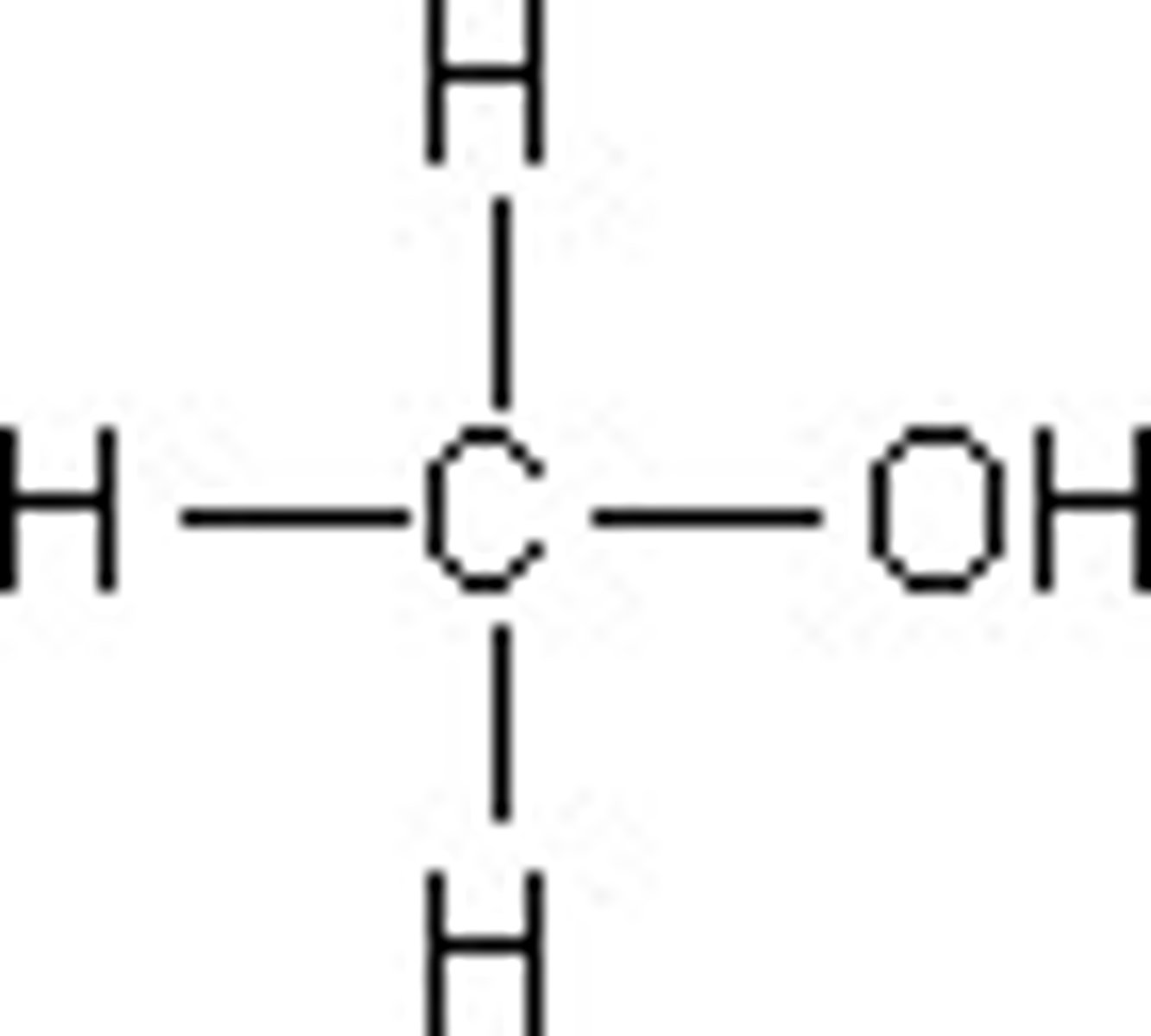
What is the displayed formula for methanol?
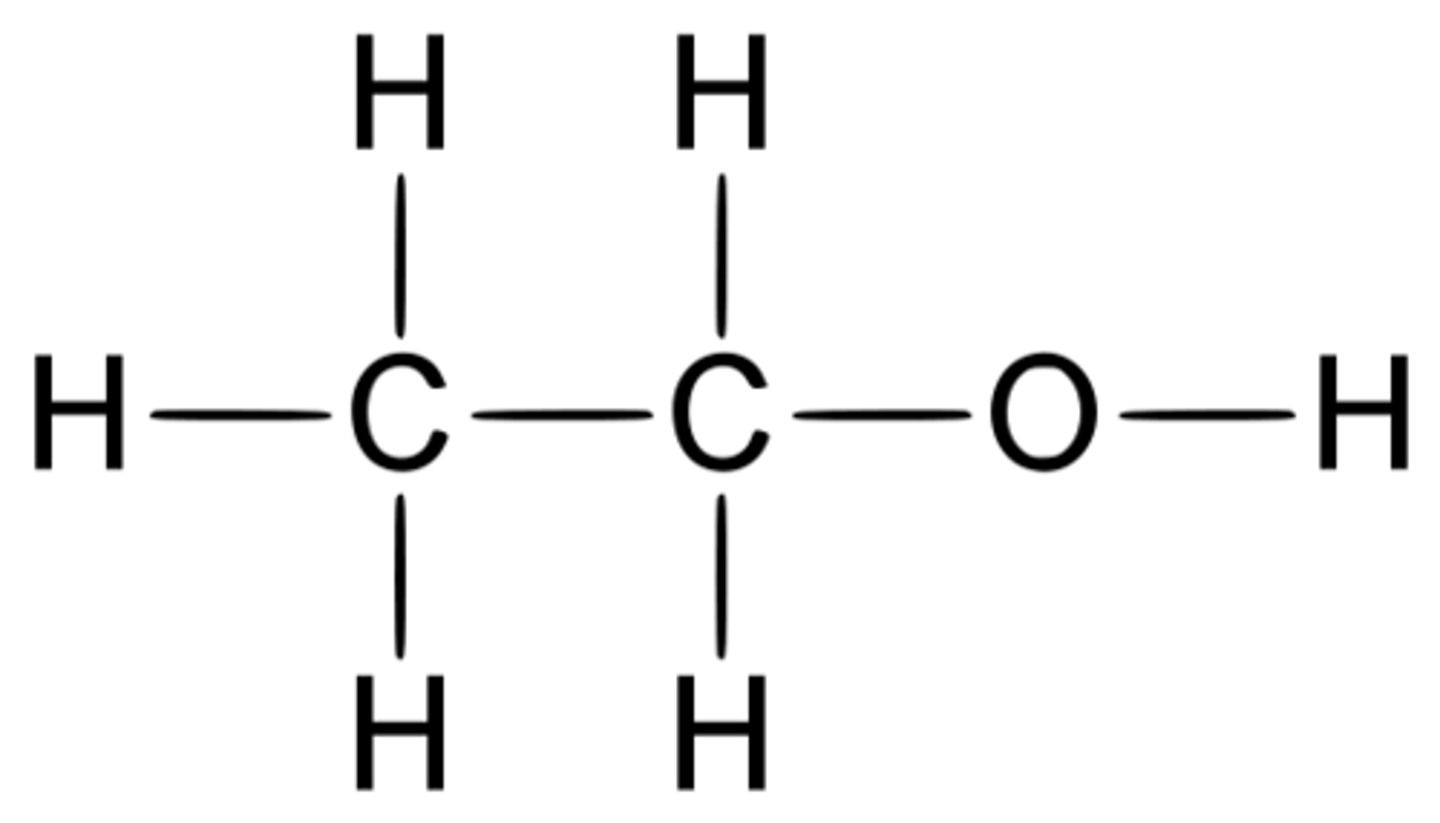
What is the displayed formula for ethanol?
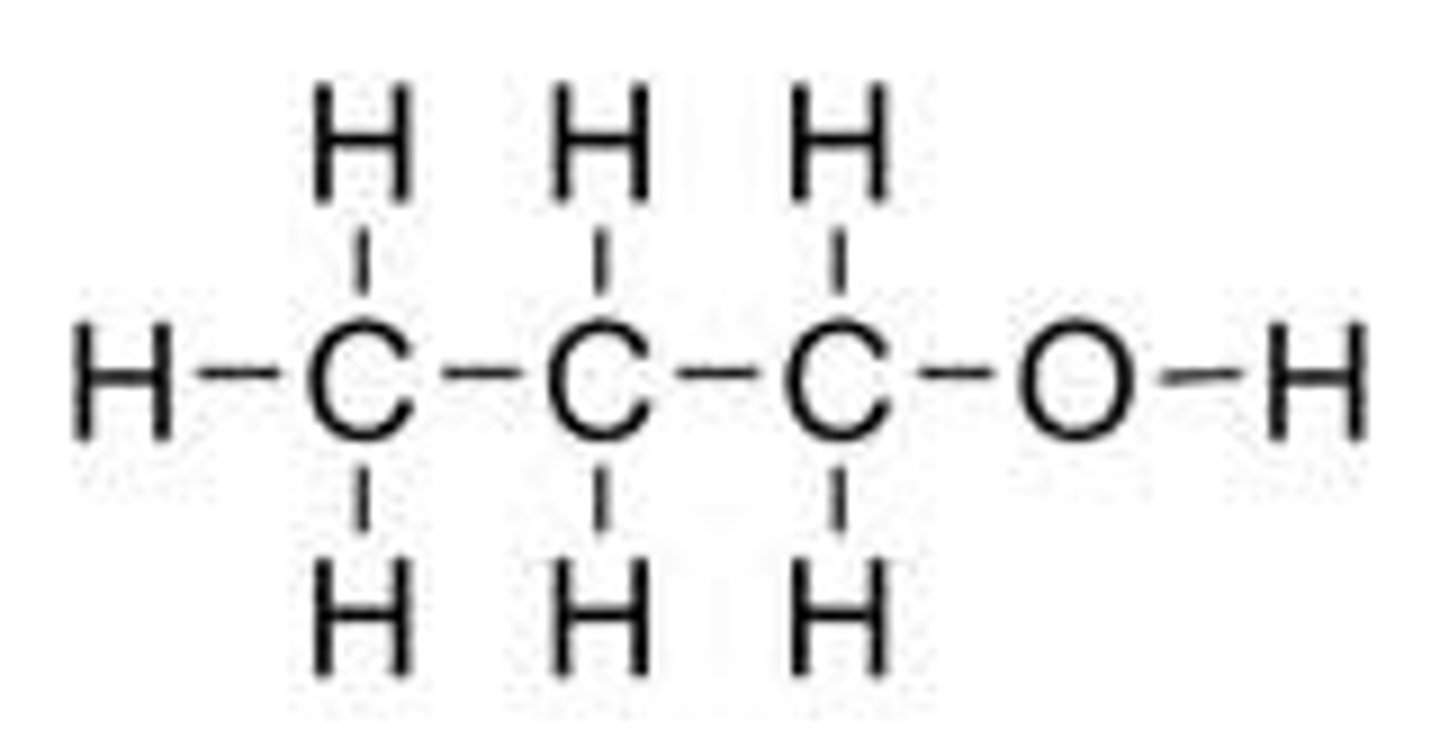
What is the displayed formula for propanol?
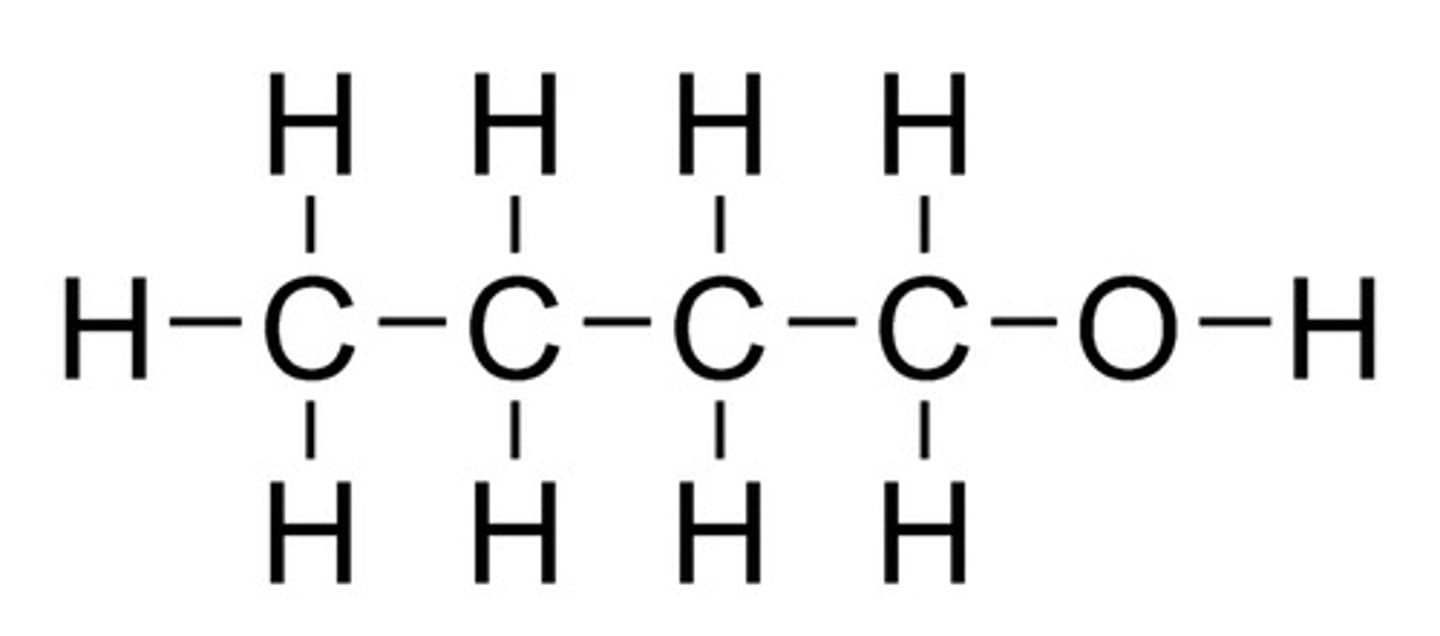
What is the displayed formula for butanol?
What is ethanol used for?
- alcoholic drinks
- solvents and fuels
Formula for the combustion of alcohols?
Alcohol + oxygen + heat →carbon dioxide + water
Formula for the combustion of ethanol
ethanol + oxygen + heat →carbon dioxide + water
C₂H₅OH + 3O₂ → 2CO₂ + 3H₂O
Formula for alcohol + sodium
alcohol + sodium→sodium alkoxide + hydrogen
What happens when sodium alkoxides dissolve in water?
They form a strongly alkaline solution
Formula for the oxidation of alcohols?
Alcohol + oxygen →carboxylic acid
Formula for the oxidation of ethanol
Ethanol + oxygen →ethanoic acid
How are carbboxylic acids made
by oxidising alcohols
What is the functional group of carboxylic acids?
-COOH
What is the general formula for carboxylic acids?
CₙH₂ₙ₊₁COOH
What are carboxylic acids used for?
to make soaps, detergents, medicinal products (like aspirin), preservatives, and vinegar (ethanoic acid)
What are carboxylic acids?
weak organic acids characterized by the presence of a carboxyl functional group (-COOH)