Biochem ugh
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
Monosaccharides
1 sugar
disaccharides
2 sugars
oligosaccharides
few sugars
polysaccharides
Many sugars
What are carbohydrates?
A carbonyl group, and several hydroxyl groups
carbonyl group
carbon atom double bonded to an oxygen
carbohydrates are repeats of
(CH2O)n
hydroxyl group
one oxygen atom bonded to one hydrogen atom
2 types of monosaccharides
aldose, ketose
aldehyde group
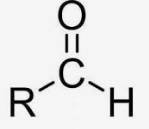
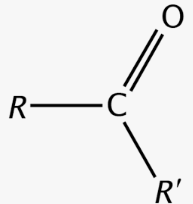
ketone group
Aldose
Sugars containing an aldehyde group (CHO)
Ketose
Sugars containing a ketone group (CO)
Where variety in monosaccharides come from
Number of carbons, chirality of each carbon atom
Isomer subtypes
COnstitutional isomers, stereoisomers
stereoisomer subtypes
Enantiomers, diastereoisomers
Diastereoisomer subtypes
Epimers, anomers
Isomers
Molecules w/ the same formula, but different structures
Constitutional isomers
differs in order of attachment of atoms
stereoisomers
atoms are connected in the same order, but differ in spatial arrangement
enantiomers
Mirror image
Diastereoisomer
uhhhhh
Epimers
Differs at one of several asymmetric carbon atoms
Anomers
Isomers that differ at a new asymmetric carbon atom formed on ring closure
Chirality
can be placed on its mirror image and not align perfectly
only chiral molecules have…
enantiomers
D or L designation
Look to the OH attached to the farthest chiral carbon from carbon 1, if it points to right, D, if left, L
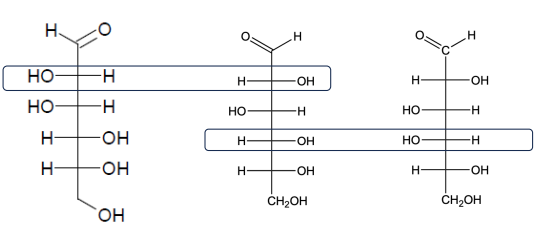
mannose is the 2-epimer of glucose, galactose is the 4-epimer of glucose
Ring of monosaccharides
when a pentose or hexose are placed in an aqueous solution, and a hydroxyl group towards the end bonds with the carbonyl carbon
pyranose
a monosaccharide with a six membered ring containing an oxygen
Anomeric center
extra chirality center in the ring form than in the open-chain form of a monosaccharide
α-anomer
a stereoisomer with the hydroxyl group below the ring
β-anomer
a stereoisomer with the hydroxyl group above the ring
glycosidic bonds
how sugars link to other sugars
WHat happens when a glycosidic bond forms
condensation reaction
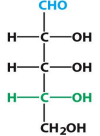
D-ribose
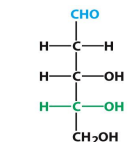
D-deoxyribose
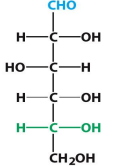
D-Glucose
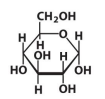
D-Glucose
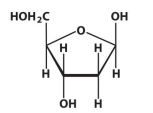
β-D-deoxyribose
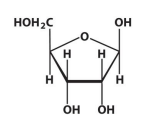
β-D-ribose
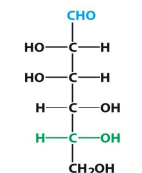
D-Mannose
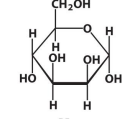
α-D-Mannose
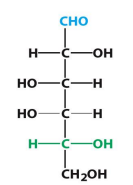
D-Galactose
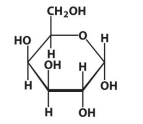
α-D-Galactose
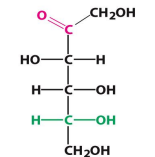
D-fructose
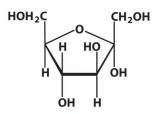
α-D-fructose
disaccharides
When 2 monosaccharides join
Sucrose is composed of
Glucose and fructose
lactose is composed of
Galactose and glucose
Maltose is composed of
2 glucose molecules
Polymers of glucose
Cellulose, starch, glycogen
Cellulose
straight chain that can be packed in tightly with other chains, dense, rigid
Starch
How glucose is stored in plants
Starch subtypes
Amylose, amylopectin
Amylose
linear polymer of glucose units, linked by α-1,4-glycosidic bonds
amylopectin
branched polymer, with an α-1,6-glycosidic bond for every 30 α-1,4-glycosidic bonds
Glycogen
How glucose is stored in animal cells—highly branched
Lipids
biological substances that do not dissolve in water but are soluble in organic solvents
Lipids have roles in…..
energy storage, structure, compartmentalization, and signaling
Fatty acids
chains of hydrogen-bearing carbon atoms that have a carboxylic acid at one end
Saturated fatty acids
Contains no carbon double bonds
Saturated fatty acids melting point
Increases as molar mass increases
Unsaturated fatty acids
Contains carbon double bonds
Unsaturated fatty acid subtypes
monounsaturated, polyunsaturated, trans-fat
Monounsaturated fatty acids
one double bond
Polyunsaturated fatty acids
2+ double bonds
Unsaturated fatty acids melting point
increases with molar mass but decreases with the degree of unsaturation.
Unsaturated fats have a lower surface area than saturated fats
true
triglycerides (TAGs) are made up of
3 fatty acids, glycerol
triglycerides (TAGs) bond
Ester bond (O-C=O)
TAGs are soluble/insoluble?
Insoluble
Cholesterol is a precurser to
bile salts, vitamin D, steroid hormones
Bile salts
Make dietary lipids more accessible for digestion
steroid hormones…
bind to specific intracellular receptor proteins that then regulate gene expression
Glucocorticoids (steroid hormones)
regulate glucose metabolism, have anti-inflammatory properties and immunosuppressant activity
mineralcorticoids (steroid hormones)
regulate electrolyte balance tissue
sex hormones (Steroid hormones)
Progestins, estrogens, androgens—puberty/development
how cholesterol and triacylglycerols are transported
lipoprotein particles
lipoprotein particles
consist of a protein part (solubilize) and various lipids (direct particles to targets)
Low-density lipoprotein (LDL)
major carrier of cholesterol in the blood.
High-density lipoprotein (HDL)
carries cholesterol released into the blood back to the liver (reverse cholesterol transport)
what the cell membrane is composed of
phospholipids, glycolipids and cholesterol