C3 : Quantitative Chemistry
1/68
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
What is relative atomic mass (Ar)?
The number of neutrons and protons (mass number)
What is relative formula mass (Mr)?
The sum of all of the atomic masses of each element present in the formula
What is the Mr of MgCl2?
( Ar of Mg = 24 ) ( Ar of Cl = 35.5)
95
What is the formula for percentage mass of an element in a compound?

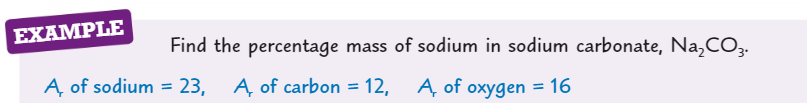



What is a mole?
An amount of a substance
What is Avogadro’s constant + what does it show?
6.02 × 1023
How many atoms there are in a molecule

3 moles of atoms

5 moles of atoms

5 moles of atoms

2 x 6.02 × 1023 = 1.024 × 1024

3 × 6.02 × 1023 = 1.806 × 1024





6.02×1023

1.806 × 1024
What is the unit for moles?
mol
What is the mass of one mole of an atom in relation to its relative formula mass (Mr) ?
Mass of one mole of an atom is equal to Mr the of that atom

What is the mass one mole of carbon?


What is the mass one mole of nitrogen?
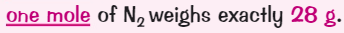

What is the mass one mole of carbon dioxide?

What is the formula for the number of moles in a given mass?
Moles = mass/Mr


( Ar of Carbon = 12) ( Ar of Oxygen = 16)
1.5mol

( Ar of Carbon = 12) ( Ar of Oxygen = 16)
48g
What is the formula for the mass of products and reactants in a chemical reaction in terms of mass?
Mass of products = Mass of reactants
Why would mass decrease in a chemical reaction?
Because one of the reactants or products is a gas




What is a limiting reactant + why is it called this?
The reactant is the reactant that is completely used up during a reaction
It limits the amount of product made
What does it mean when a reactant is in excess + why are reactants used in excess?
A reactant that has not been used up when the limiting reactant has been used up
This is to make sure the limiting reactant has been used
What is the relationship between the amount of product and the amount of limiting reactant?
The amount of product formed is directly proportional to the amount of limiting reactant

159.5g
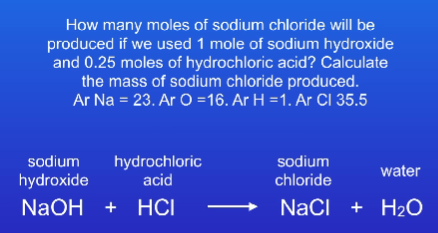
14.6(25)g

31.75g

272g

190g

426g

136g
At room temperature and pressure what does one mole of any atom occupy?
24dm3
What is the formula for the volume of a known mass of any gas at room temperature?


(Cl2)
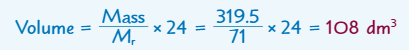

120 dm3

48 dm3


What is concentration?
The amount of substance (mass or the number of moles in a substance) in a certain volume of a solution
What is a solute + what is the relationship between the amount of solute + the concentration?
Solute → Substance that is dissolved
More solute there is in a given volume means more concentrated the solution is
What is the formula for the concentration in terms of mass of solute?
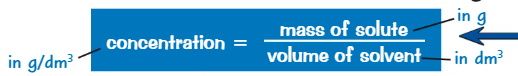

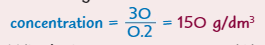
What is the formula for the concentration in terms of number of moles of solute?




0.1 moles

88.8g

34 mol/dm3

0.71 mol/dm3
What is atom economy?
The measure of the amount of starting materials that end up as useful products
What is the formula for atom economy?

Why is a higher atom economy better? (4)
Less waste
Low atom economy use resources very quickly
Materials are expensive
Waste is hard to get rid of
What do scientist try to do with unwanted side products?
They try to repurpose them in other reactions
What is the relationship between the amount of products and the atom economy?
The more the products, the lower the atom economy
What is a yield?
The amount of product made
What is the formula for percentage yield?

What does 100% yield mean?
All the products expected to be made were made
What is the benefits of a high percentage yield? (2)
Reduce waste
Reduce costs
What are 3 reasons as to why there will never be 100% yield?
Reversable reactions
Side reactions
Product lost from separation (filtration)

240g
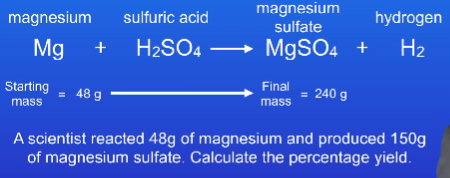
62.5%

20%

60%