Physiology at extremes - hyperbaric conditions eg diving
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Know the gas compositions of dry and humidified air and principles of Dalton’s law
Appreciate the relationships between partial pressure of gas in air and the gas dissolved in a solution – Henry’s law
Appreciate how gas partial pressures and volumes alter with diving
Describe the risks associated with free-diving
Describe the potential risks with descent and ascent during diving and the linked barotrauma
Appreciate the risks associated with increased partial pressures of nitrogen and oxygen when diving
Appreciate the potential problems faced when escaping from a submarine
What is standard atmospheric pressure?
760mmHg
Dalton’s Law
total pressure of a mixture of gases is the sum of their individual partial pressures
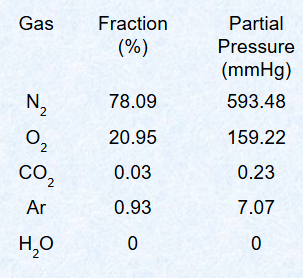
Describe gas composition of dry/atmospheric air at atm pressure
Mainly nitrogen and oxygen
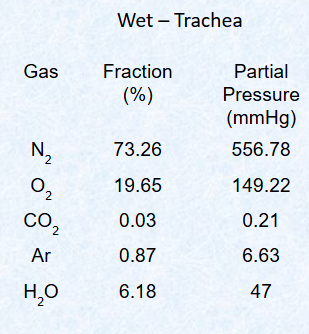
Describe gas composition of wet air at atm pressure
Using Henry’s Law, what can be determined? How?
Concentration of a gas dissolved in a solution using partial pressure information
[Gas]dis = s x Pgas (s is solubility coef and P is partial pressure of gas)
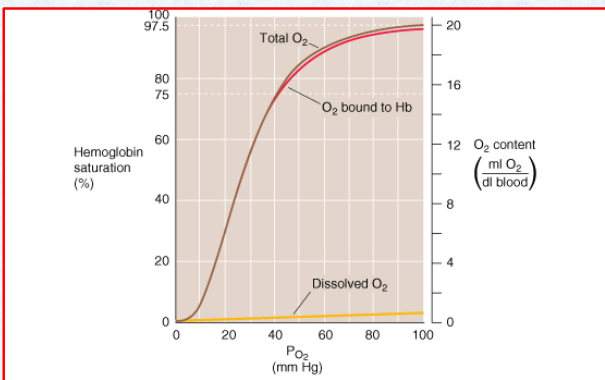
Describe the oxygen-haemoglobin dissociation curve
as pressure increases, O2 binds Hb and eventually saturates
What are problems that can arise from diving due to pressure changes? Which law is relevant in each?
increases in amount of different gases dissolved in blood as pressure increases, partial pressure of gases breathed in increase for every 10m descended by 1atm (Henry’s Law)
changes in volumes of gases as pressure changes, gas will occupy less V during descent and more during ascent (Boyle’s Law)
How much does barometric pressure increase for every 10 m descended?
1 atm
How will partial pressures of gases in air being breathed in change as pressure increases during diving? Using Henry’s Law, what can be predicted about amount of each gas dissolved in blood? What is a risk of this eg?
pressure will increase
amount of each gas dissolved in the blood will increase
nitrogen toxicity

What is the relationship between pressure and volume?
inversely proportional
During descent, how will volume occupied by a gas change?
gas will occupy less volume as pressure increases
What is the normal partial pressure of CO2 in mixed venous blood?
46mmHg
Following gas exchange, how does normal CO2 partial pressure change from 46mmHg? Imagining a 10m deep dive, how would the pressure in the gas in the lungs change? How will this affect the movement of CO2 in the body?
40mmHg
doubling of pressure in the gas in the lungs so 80mmHg
CO2 from the lungs will start to diffuse back into the body – causes increase in arterial CO2 = urge to breathe
Following gas exchange, partial pressure of O2 in alveolar air is 100mmHg, how would a 10m deep dive affect it?
doubling of pressure in the gas in lungs so partial pressure is 200mmHg
How can returning to the surface from a deep dive lead to a blackout?
going down, arterial CO2 increase, breathing urged and O2 levels not high enough for compensations to be triggered
Return to surface – pressure in lungs returns to normal = Fall in alveolar CO2 and O2 which causes a rapid fall in arterial CO2 and O2. Drop in cerebral O2 can lead to a blackout
Why is it not recommended to hyperventilate before diving?
suppresses need to breathe
Hyperventilating before diving is dangerous because it artificially lowers carbon dioxide levels without significantly increasing oxygen, removing the body’s natural urge to breathe. This hidden, rapid depletion causes a sudden, warning-free "shallow-water blackout," leading to loss of consciousness and a high risk of drowning
During scuba diving, what can lead to issues on ascent?
if breathing not controlled, careful breathing out has to happen
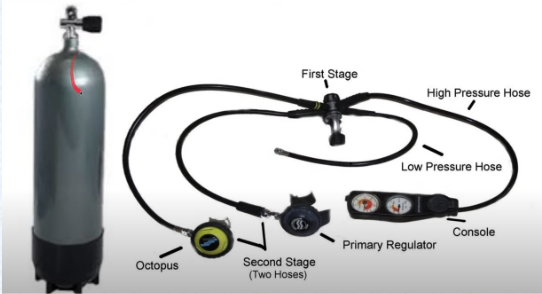
What allows diver to breathe a gas mixture equilibrated to ambient pressure?
Combination of 1st and 2nd stage regulators
tidal volume will remain the same but the pressure of the gas being breathed is higher
What is barotrauma?
injury arising from pressure changes
Considering problems can arise with air spaces, in what conditions are there no issues?
is gas pressures can equilibrate
Which areas of the body are relatively resistant to effects of high pressure?
liquid areas
Which organs are affected by pressure?
inner ear and lungs
During descent, how can ears be affected by pressure changes?
Problems arise if Eustachian tube is blocked and doesn’t allow gas movement between throat and middle ear eg during cold or infection blocking the tube - problem is the partial compensation by eardrum, blood vessels in ear might rupture or eardrum could burst if overstretched
What connects the throat to the inner ear and allows relieving pressure effects on the body?
Eustachian tube
What action allows relieving pressure change of gas in the ear?
swallowing
How can sinuses be affected by a decrease in volume of gases in them?
bleeding in sinuses to help equilibrate the pressure
What are 3 common pathologies that can arise from descent during diving due to pressure and volume changes in gases?
shallow-water blackout (10m deep), nitrogen narcosis, oxygen toxicity
At what depth could symptoms of nitrogen narcosis arise? At what stage can it be fatal?
30m
90m
What is the impact of depth increase on dissolves nitrogen concentration in the blood?
also increases
Is nitrogen more soluble in lipid or blood? How does it affect ion conductance?
in lipid
Acts like a volatile anesthetic and alters ion conductance.
What are symptoms of nitrogen narcosis?
The symptoms similar to being ‘drunk’. Sleepiness, impaired judgment, confusion, Hallucinations, severe delay in response to signals, instructions and other stimuli, tunnel vision, dizziness and often a sense of euphoria.
How can nitrogen narcosis be prevented?
limit depth and duration of dive and change in gas composition, use gas mixture replacing nitrogen with helium
What is Hb saturation like at atmospheric pressure? What would be the impact of increasing pressure?
almost fully saturated
Increasing pressure, any extra O2 is dissolved in the plasma.
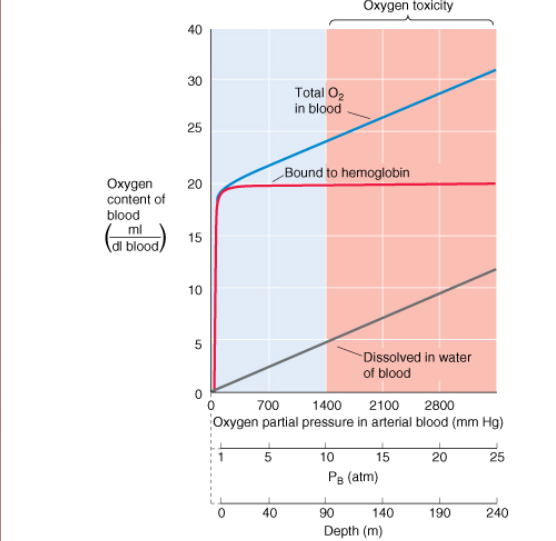
Once depth reaches 40m (5atm), how does oxygen partial pressure change?
is roughly equivalent to breathing 100% O2 at sea level
When oxygen partial pressure is roughly equivalent to breathing 100% O2 at sea level, what are the implications short vs long term?
short term ok
long term exposure leads to respiratory tract damage and possible CNS problems
Consequence of breathing air 90m (10atm). What is a solution?
oxygen toxicity causing seizures and coma due to increased levels of free radicals
reduce oxygen [] in mixture
What are problems of diving during ascent?
lung over-inflation, pneumothorax, arterial gas embolism, mediastinal and subcutaneous emphysema, decompression sickness
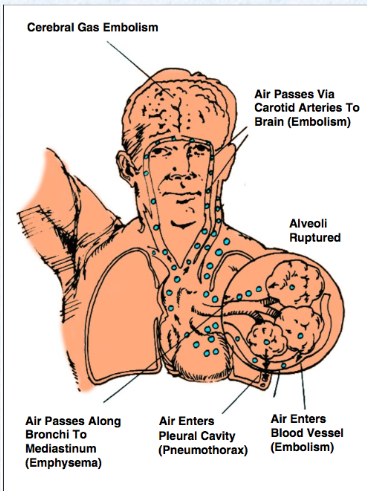
What are consequences of over-inflation of the lungs due to ascent during diving?
when breathing compressed gas at depth and failing to exhale, or when air is trapped, causing the expanding gas to rupture lung tissue = alveoli ruptured, air passes through blood vessels and pleural cavities along bronchi to mediastinum and through carotid arteries to brain = death
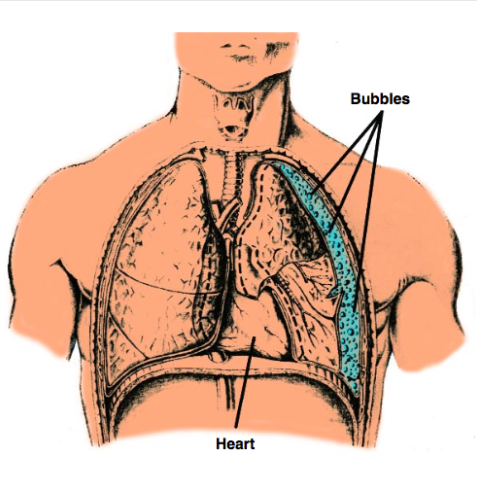
What are consequences of pneumothorax due to ascent during diving?
A pneumothorax (collapsed lung) during ascent in diving is a severe form of pulmonary barotrauma caused by the expansion of trapped air within the pleural space as ambient pressure decreases. It is a life-threatening emergency because the air bubble expands, often doubling in size when ascending from just 10 meters, which can turn a minor, asymptomatic lung tear into a fatal condition
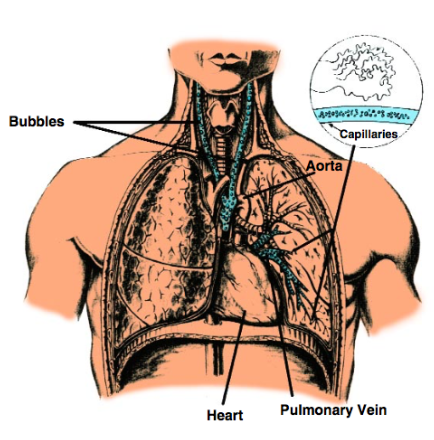
What are consequences of arterial gas embolism due to ascent during diving?
lung overexpansion, forcing air bubbles into the arterial circulation blocking flow. Symptoms usually appear within minutes of surfacing, including rapid loss of consciousness, strokes-like signs (paralysis, confusion), dizziness, seizures, or bloody froth from the mouth
What are consequences of mediastinal and subcutaneous emphysema due to ascent during diving?
lung rupture due to rapid or breath-holding ascent
Subcutaneous Emphysema: Characterized by swelling (puffiness) in the face, neck, and chest, along with a "crackling" or "crunching" sensation (crepitus) on palpation.
Mediastinal Emphysema: Produces chest pain, typically behind the breastbone, which may radiate to the neck, back, or shoulders
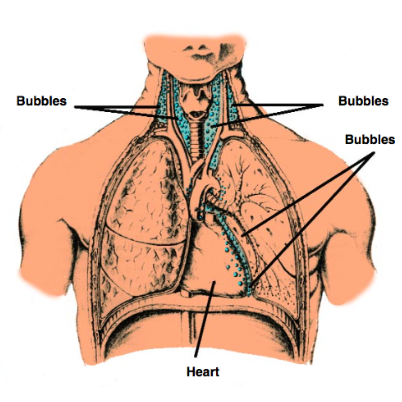
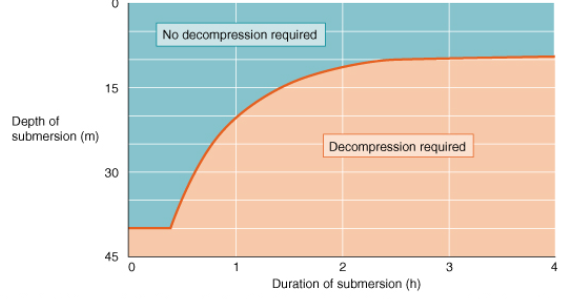
What is DCS and what are consequences of decompression sickness due to ascent during diving? How is it overcome?
Build of N2 in tissues with time at depth. If return to sea level too quickly this gas comes out of solution and forms bubbles. Overcome by slow return to normal pressures.
What are the 2 main types of decompression sickness?
type 1: linked to pains produced by bubbles forming in muscles and joints
type 2: more serious – bubbles in CNS, lungs and CVS
What is pressure like inside the submarine? What has to happen when escaping the submarine?
pressure close to atmospheric pressure at sea-level
have to enter the escape chamber partially filled with seawater at external pressure so less risk of decompression sickness at low depth eg 30m, risk increases with depth. they must exhale to avoid lung damage as they ascend - if not then have to be kicked in the stomach
What is decompression sickness in diving?
rapid ascent during diving causes dissolved nitrogen to form bubbles in tissues and blood, potentially causing joint pain, neurological issues, or rash