Acids,alkalies and titration
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
what is an acid?
A solution that reacts with alkalis, turns litmus red and has a pH less than 7. They are substances that act as a source of hydrogen ions in a solution. They are proton donors
what is an alkali?
They are soluble bases.A solution that reacts with acids, turns litmus blue and has a pH more than 7. Are a source of hydroxide (OH-) ions in solution. eg all group 1 hydroxide
what is a base?
a base is a substance that can neutralise an acid by combining with the hydrogen ions in them They are proton acceptors. They are usually metal oxides, metal hydroxides or ammonia NH3
what is neutral?
A substance that is neither acid or alkaline and has a pH of 7.
what si an indicator?
A substance that can change colour depending on the pH of a solution.
what is concentration?
The amount of solute dissolved in a certain volume of solvent.
what is the pH scale?
The pH scale ranges from 0 to about 14 and tells you how acidic or alkaline a solution is.
The lower the pH then the more acidic the solution is
The higher the pH then the more alkaline the solution is
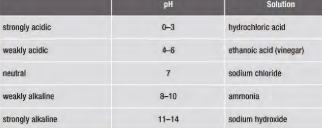
pH of acids,alkali and neutral solutions?
Acidic, alkaline and neutral substances all have a pH value:
- Acids can have a pH value between pH1-6.
- Alkaline substances can have a pH value between pH8-14.
- Neutral substances have a pH value of 7.
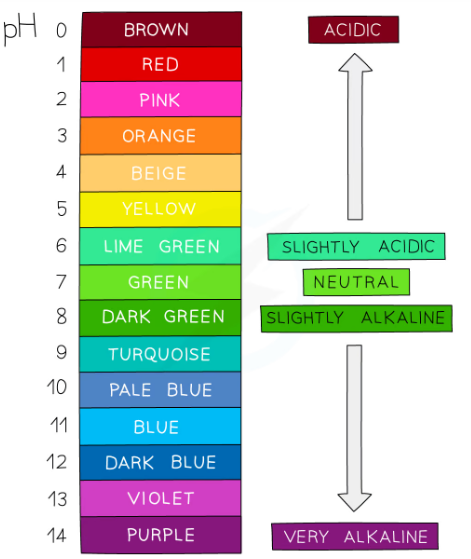
what is universal indicator?
Universal indicator is a wide range indicator and can give only an approximate value for pH
It is made of a mixture of different plant indicators which changes oclours in a gradual way over a range of pH values and is useful for estimating the pH of an unknown solution.
A few drops are added to the solution and the colour is matched with a colour chart which indicates the pH which matches with specific colours.Universal indicator colours vary slightly between manufacturer so colour charts are usually provided for a specific indicator formulation
it is red in acidic and blue in alkali
what are two colour indicators?
Two colours indicators are used to distinguish between acids and alkalis
litmus paper colour in acid and alkali
acid-red
alkli-blue
what are synthetic indicators?
Synthetic indicators are organic compounds that are sensitive to changes in acidity and appear different colours in acids and alkalis
Phenolphthalein and methyl orange are synthetic indicators frequently used in acid-alkali titrations
Synthetic indicators are used to show the endpoint in titrations as they have a very sharp change of colour when an acid has been neutralised by an alkali and vice-versa
colour of phenolphthalein in acid and alklai
Acid-colourless
alkali- pink
colour of methyl orange in acid and alkali
acid- red
alkali-yellow
why is litmus not used in titration?
Litmus is not suitable for titrations as the colour change is not sharp and it goes through a purple transition colour in neutral solutions making it difficult to determine an endpoint
Litmus is very useful as an an indicator paper and comes in red and blue versions, for dipping into solutions or testing gases
what are ions?
Ions are charged particles formed through gaining or losing electrons.
Alkalis produce excess of hydroxide ions (OH-) in water. Eg. Solid NaOH, dissolved in water, splits into Na+ and OH- ions.
what happens when acids are added to water?
when Acids are added to water they break apart to produce an excess of positively charged hydrogen ions (H+) in water. Eg. HCl gas, dissolved in water, splits into H+ and Cl- ions.
HCl + H 2O → [H 3O] + + Cl.
what happens when alkalis are added to water?
bases that dissolve in water to form solutions containing hydroxide ions are called alkalis.Alkalis produce excess of hydroxide ions (OH-) in water. Eg. Solid NaOH, dissolved in water, splits into Na+ and OH- ions.
how ions affect acidity and how alklaine a solution is?
The higher the concentration of H+ ions, the more acidic, the lower the pH.
The higher the concentration of OH- ions, the more alkaline, the higher the pH.
Neutral substances have an equal proportions or low amounts of both H+ and OH- ions. This gives them a pH of 7.
are metal carbonates alkalis?
there are some other substances such as soluble carbonate alkalis that react wiht water ot form hydroxide ions. Sodium carbonate and potassium carbonate are both alkalis with a pH greater then 7. This is due to the OH- ions in the solution. ONly some of the carboante ions react wiht water so these solutions are only weakely alklai

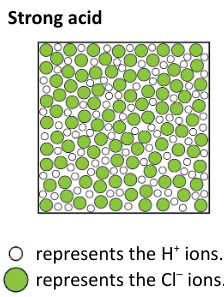
what are strong acids?
strong acid is one that completely dissociates (splits up) into ions when it is dissolved in water.
Hydrochloric acid is formed when hydrogen chloride gas dissolves in water.
HCl(g) + aq → H+(aq) + Cl–(aq)
(aq) on the left hand side of the equation represents a large amount of water.
The reaction is 100% complete, so every hydrogen chloride molecule dissociates in water to form ions.
example of a strong acid
Hydrochloric acid is a strong acid. All of the hydrogen chloride molecules split up to form ions when it dissolves in water.
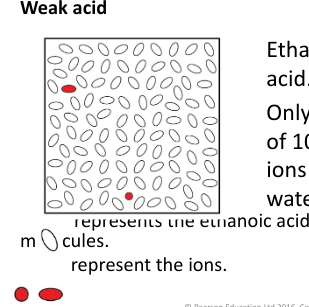
what is a weak acid
A weak acid only partially splits up into ions in dilute solution, and many unchanged molecules remain.
exampel of a weak acid reaction
Ethanoic acid is the acid found in vinegar and it is a weak acid.
CH3COOH(l) + aq ⇌ CH3COO–(aq) + H+(aq)
⇌ is a reversible arrow and means that the reaction can go forwards and backwards.
Only one molecule out of 100 splits up to form ions when dissolved in water.
what is a concentrated acid?
A concentrated acid contains a large number of particles dissolved in a given volume of solution. It is possible to have a concentrated solution of a strong acid and
a concentrated solution of a weak acid.

what is a dilute acid?
A dilute acid contains a smaller number of particles in the same volume of solution.It is possible to have a dilute solution of a strong acid and a dilute solution of a weak acid.You can dilute an acid by adding it to water.

what is a neutralisation reaction?
A neutralisation reaction is where an acid reacts with a base/alkali to produce a neutral solution of a salt and water.
neutralisaiton reaction formula
When these substances react together in a neutralisation reaction, the H+ ions react with the OH– ions to produce water. The acid donates protons which are then accepted by base
The net ionic equation of all acid-base neutralisations and is what leads to a neutral solution, since water has a pH of 7:
H+ + OH– ⟶ H2O
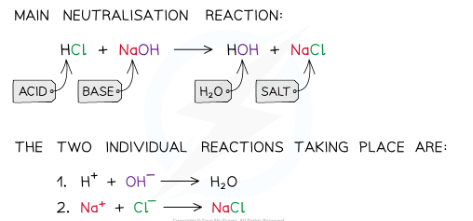
why is neutralisation important
what is titration?
Titration cna be used to find out exactly how much acid is needed to neutralsie a quanittiy of alkali (or vise versa)Titrations are a method of analysing the concentration of solution. They can also eb sued to prepare salts.
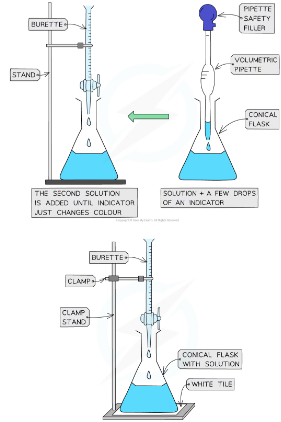
how to carry out titration?
1) Using a pipette and pipette filler, add some alkali (usually about 25cm3) to a conical flask,
2) Add two or three drops of indicator to the solution in the conical flask
3) Fill a burette with the acid. Make sure you do this below eye level-so acid doesnt spill when yorue looking up
4) Place the conical flask on a white tile so the tip of the burette is inside the flask
5) Perform a rough titration by taking the burette reading and adding in the acid to the alkali in 1 – 3 cm3 portions, while swirling the flask vigorously/regularly. Thsi gives you a rough idea of volume needed
6) The indicator changes colour when all the alkali has been neutralised eg phenolpjtahelin is pink in alklais but colourless in acids. Quickly close the tap when the colour changes
7) record the volume of acid used to neutralsie the alkali, palcing your eye level with teh meniscus.Find voluem by subtractign initial reading with final reading. Record the volume to the nearest 0.05 cm3 . Second decimal point is 0 if on line and 5 if between the lines 8) Now repeat the titration with a fresh batch of alkali
9) As the rough end-point volume is approached, add the solution from the burette one drop at a time until the indicator just changes colour
10) Repeat until you achieve two concordant results (two results that are within 0.1 cm3 of each other) to increase accuracy/reliability
difference between burette and pipette?
pipette measure only one volume of solution. Burette measure different volumes and elt youa dd the solution drop by drop
how ot find concentration of the acid in titration?
1) Work out hte number of moels of the ‘known’ susbtance you have (moles=concentration x volume)
2) Write down the equation for the reaction and work out how many moels of hte unkown stuff you ahve eg
2NaOH + H2SO4→ Na2SO4 + 2H2O
for every two moles of sodium hydroxide you have one mole of sulfuric acid
3) Work out the concentration of the unkown studd using concentration = number of moles/ volume
are metals basic a=or acidic
all metals are basic and form basic oxides and all non metals are acidic