enthalpy changes
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
what is enthalpy (H)?
a measure of the heat energy in a chemical system
what does a chemical system refer to?
the atoms, molecules, or ions making up the chemicals
what is enthalpy sometimes thought of as?
the energy stored within bonds. it cant be measured, but enthalpy changes can
what are the reactants and products likely to have in a chemical reaction?
different enthalpies
what is enthalpy change?
the difference in the enthalpies of the reactants and products. it can be + or - depending on whether the products contain more or less energy than the reactants
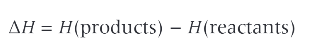
what is the conservation of energy?
states that energy cant be created or destroyed
what happens when a chemical reaction involving an enthalpy change takes place?
heat energy is transferred between the system and the surroundings
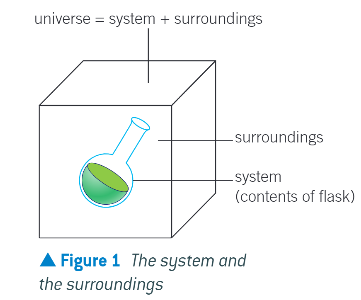
features about that statement:
the system is the chemicals (reactants and products)
the surroundings are the apparatus, the lab, and everything not in the chemical system
the universe is everything, and includes both system and surroundings
how can enthalpy change be determined?
by measuring the energy transfer between the system and surroundings
what are the 2 directions that energy transfer can be in?
exothermic change - from system to the surroundings
endothermic change - from surroundings to the system
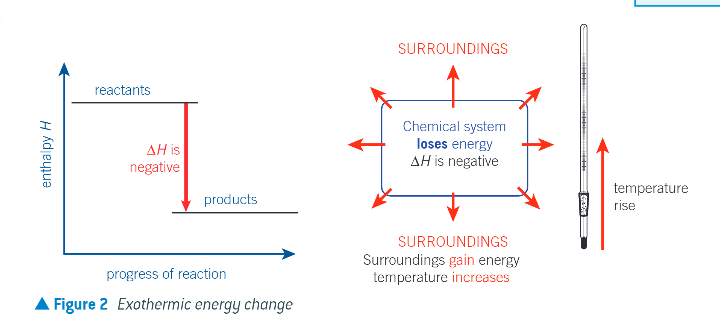
enthalpy profile diagrams with the relative enthalpies of the reactants and products and the enthalpy change: EXOTHERMIC:
the chemical system releases heat energy to the surroundings
any energy loss by the chemical system is balanced by the same energy gain by the surroundings
enthalpy change is negative
the temp of the surroundings increases as they gain energy
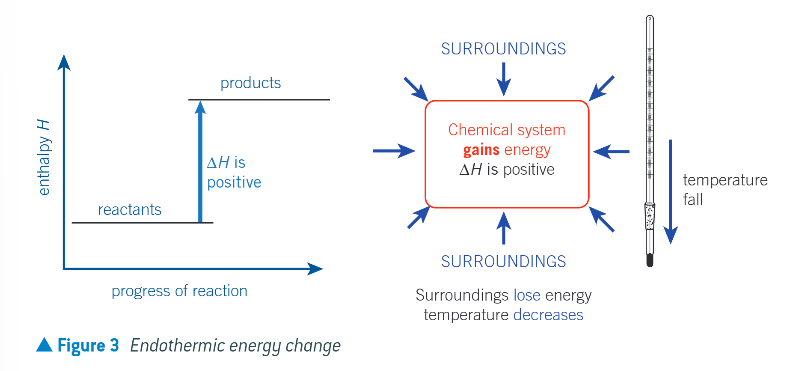
enthalpy profile diagrams with the relative enthalpies of the reactants and products and the enthalpy change: ENDOTHERMIC:
the chemical system takes in heat energy from the surroundings
any energy gain by the chemical system is balanced by the same energy loss by the surroundings
enthalpy change is positive
the temp of the surroundings decreases as they lose energy
what needs to happen during chemical reactions?
the bonds in the reactants need to be broken by an input of energy. new bonds in the products can then form to complete the reaction
what is activation energy?
the energy input required to break bonds and start the reaction
both enthalpy profile diagrams together with Ea:
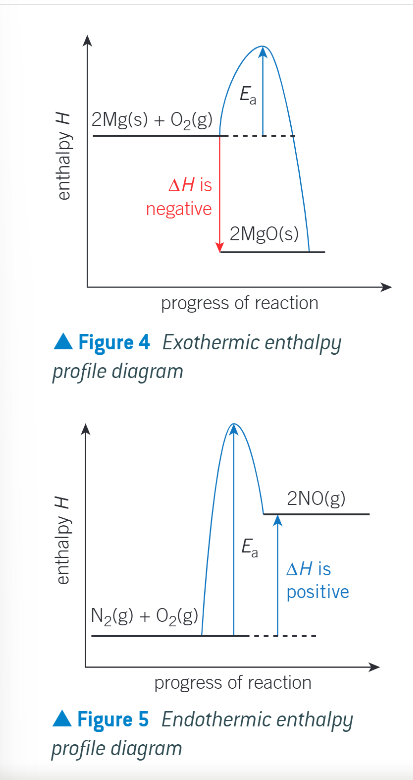
why do reactions with small activation energies take place very rapidly?
because the energy needed to break bonds is readily available from the surrounding
what may be the case with reactions with very large activation energies?
there may be such a large energy barrier that a reaction may take place very slowly or not at all
what do chemists do considering that the enthalpy change for a reaction can vary slightly dependant on the conditions used?
use standard conditions for physical measurements, close to typical conditions of temp and pressure (tables of data always include standard conditions)
how is a standard physical value shown in data tables?

what are these standard conditions?
pressure: 100kPa
temp: 25/ 298K
conc: 1moldm-3
state: physical state of a substance under standard conditions
units: kJmol-1
what is the standard enthalpy change of a reaction?
the enthalpy change that accompanies a reaction in the molar quantities shown in a chemical equation under standard conditions, with all reactants and products in their standard states
example with enthalpy change of a reaction:

what is the standard enthalpy change of formation?
the enthalpy change that takes place when 1 mole of a compound is formed from its elements under standard conditions, with all reactants and products in their standard states
compounds in enthalpy change of formation:
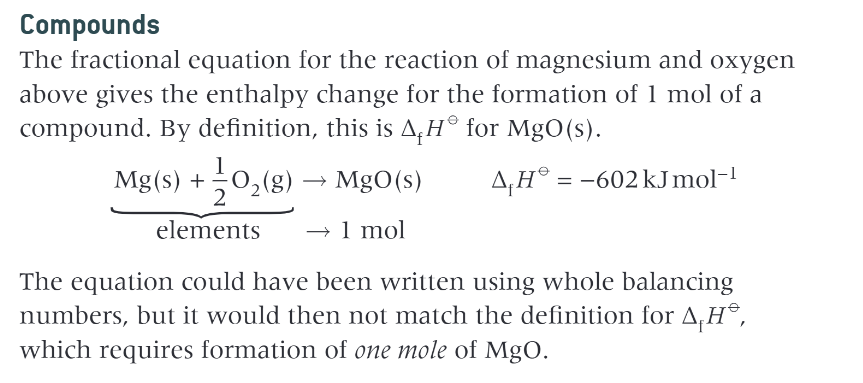
what is the enthalpy change of formation for elements?
0kJmol-1

what is the standard enthalpy change of combustion?
the enthalpy change that takes place when 1 mole of a substance reacts completely with oxygen under standard conditions, with all reactants and products in their standard states
what happens when a substance reacts completely with oxygen?
the products are oxides of the elements in the substance

enthalpy change of combustion with equation:

what is the standard enthalpy change of neutralisation?
the energy change that accompanies the reaction of an acid by a base to form 1 mole of H2) under standard conditions, with all reactants and products in their standard states
equation and example of enthalpy change of neutralisation:

extra info: what about calories:

what is a thermometer part of in a chemical reaction?
the surroundings
facts about the Kelvin scale of temperature:

how do you calculate the energy change of the surroundings?
using mass, specific heat capacity, and temp change
mass:
measured by weighing
have to identify the materials that are changing temp
mass usually measured in grams
specific heat capacity:
different materials require different quantities of energy to produce the same temp change
the energy required to raise the temp of 1g of a substance by 1K
good conductors of heat = small c
insulators of heat = large c
most experiments = 4.18g-1K-1
temp change:
determine from final temp and initial temp
how do you calculate energy change?

whats one of the easiest enthalpy changes to determine experimentally?
combustion
spirit burners:

worked example: determination of enthalpy change of combustion of methanol:

further:

why may experimental data not be the same as a data book value?
heat loss to the surroundings other than the water, including the beaker and the air surrounding the flame
incomplete combustion of methanol, with CO and C being produced instead of CO2 (would see black layer of soot on the beaker)
evapouration of methanol from the wick. the burner must be weighed as soon as possible after extinguishing the flame, otherwise some methanol may have evapourated from the wick. spirit burners usually have a cover to reduce this error
non standard conditions. the data book is a standard value, and the experimental conditions are unlikely to be identical
how can you minimise errors from heat loss and incomplete combustion?
use of draught screens and an input of oxygen gas
accuracy comparing with the previous methanol example:

how can you determine enthalpy change of reactions using 2 solutions/ solid and solution?
using a polystyrene cup, which is cheap, waterproof, lightweight, and offers some insulation against heat loss
what is the case when carrying out reactions between aqueous solutions?
the solution itself is the immediate surroundings
the chemical particles within the solution may react when they collide, and any energy transfer is between the chemical particles and water molecules in the solution
thermometer can measure temp change, allowing heat energy change to be calculated

worked example: determination of enthalpy change of a reaction for a solid and a solution:
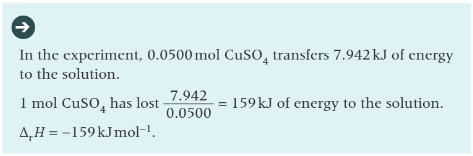
extra info: cooling curves:
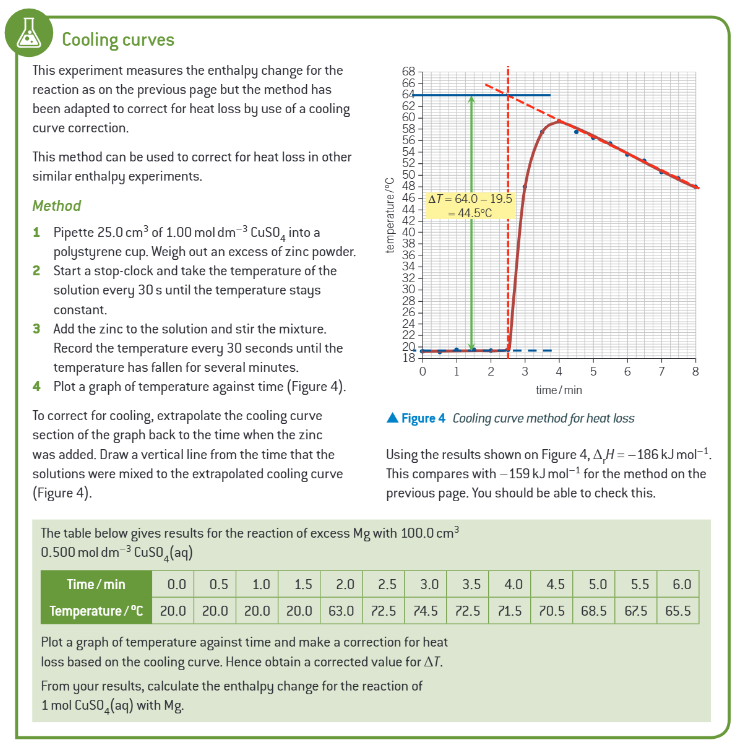
worked example: determination of enthalpy change of neutralisation:

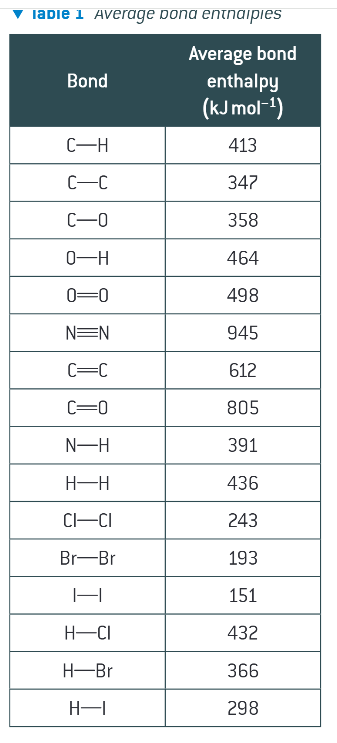
what is average bond enthalpy?
the energy required to break 1 mole of a specified type of bond in a gaseous molecule
facts about bond enthalpies:
energy always required to break bonds
bond enthalpies always endothermic
bond enthalpies always have a positive enthalpy value
average bond enthalpies for some common bonds:
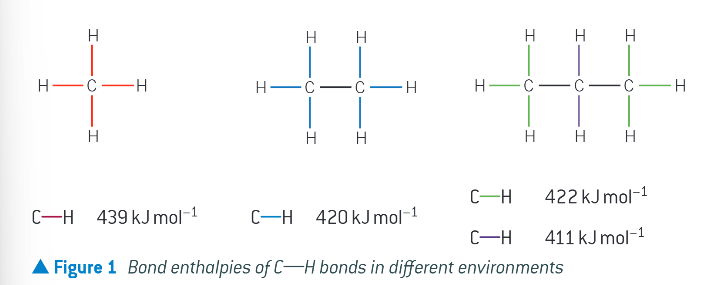
why can the actual bond enthalpy vary?
dependant on the chemical environment of the bond. figure shows example of this with the same bond in different environments
how is an average bond enthalpy calculated?
from the actual bond enthalpies in different chemical environments
energy is required to break bonds, so bond breaking is…
endothermic
energy is released when bonds form so bond making is…
exothermic
what does the difference between the energy required for bond breaking and the energy released by bond making determine?
whether an overall reaction is exothermic or endothermic
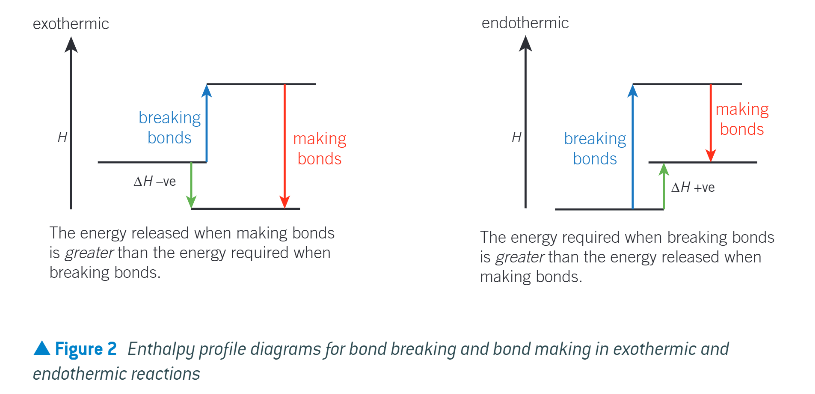
diagram to show enthalpy profile diagrams for bond breaking and bond making in exothermic and endothermic reactions:
how can you find the enthalpy change of a reaction?
calculate the bond enthalpies of the bonds in the reactants and the products
equation for enthalpy change:
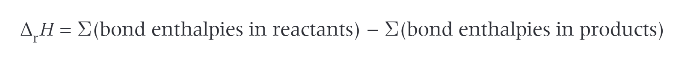
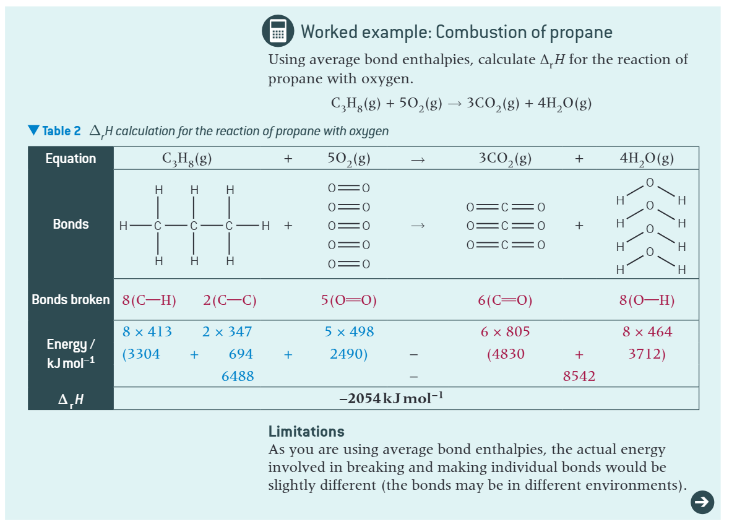
worked example: combustion of propane:

extra info: bond enthalpies and combustion:

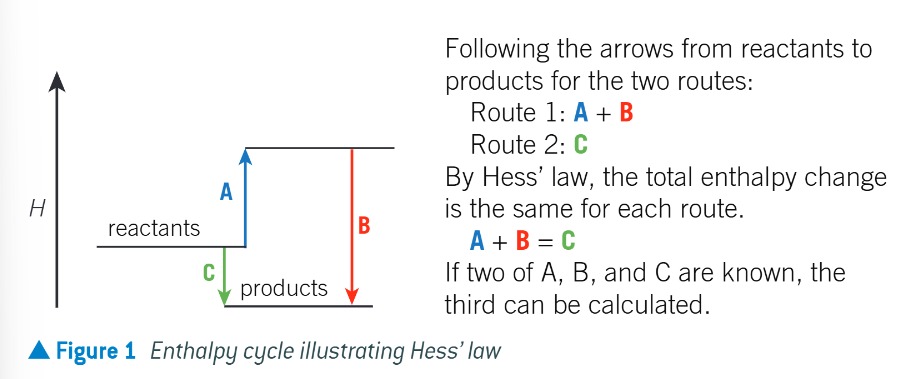
what does Hess’ law do?
allow enthalpy changes to be detemined indirectly. this is good as the enthalpy change of many reactions are very difficult to determine directly
what does hess’ law state?
if a reaction can take place by 2 routes, and the starting and finishing conditions are the same, the total enthalpy change is the same for each route
what does hess’ law come from?
conservation of energy
how can you visualise hess’ law
via an enthalpy cycle or a hess cycle
diagram to show enthalpy cycle illustrating Hess’ law:
worked example: an enthalpy cycle:
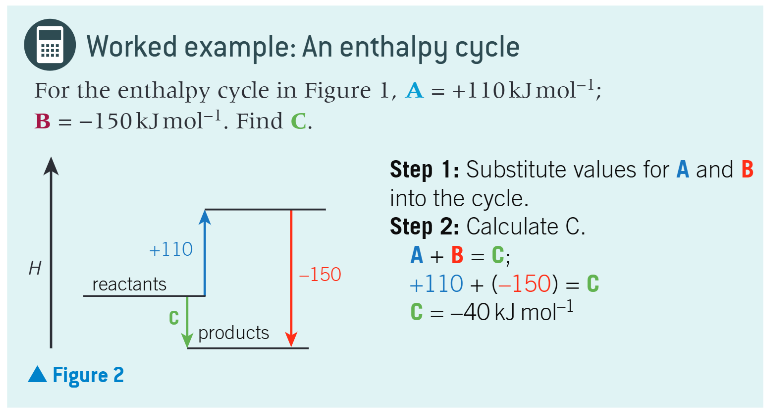
what can this principle be extended to?
any number of enthalpy changes. provided that all enthalpy changes are known except one, the unknown can always be determined
indirect determination of enthalpy changes:

worked example: enthalpy changes from enthalpy change of formation:

worked example: enthalpy changes from enthalpy change of combustion:

SUMMARY:

extra info: unfamiliar enthalpy cycle:
