Chemistry Reaction Maps
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What are the conditions needed to convert Acyl Chloride into a Carboxylic Acid? — what does the chemical reagent act as and what inorganic byproducts does it produce?
SOCl2 — it act as a source of chloride and makes SO2 and HCl as a byproduct.
What are the conditions needed to convert Acyl Chloride into an Amide? — what is more than 1 molecule of the condition needed?
Conditions: it reacts with 2 of the same amines to form an amide. 2 Amine molecules are needed as one of the amine molecules will react with the HCl byproduct that forms and lose its nucleophilic site,
What are the conditions needed to convert Acyl Chloride into an Ester? — what is the process called?
Fischer Esterification.
Conditions: conc H2SO4 (cat) and any carboxylic acid.
What are the conditions needed to turn Acyl Chloride into Carboxylic Acid?
Water
What are the conditions needed to turn Carboxylic Acid into Acyl Chloride?
Conditions: SOCl2
What are the conditions needed to turn Carboxylic Acid into an Ester
Conditions: conc. H2SO4 and an alcohol
What are the conditions needed to turn Carboxylic Acids into Alcohols (2-step & 2 ways)
Conditions:
Convert Carboxylic Acid into an Ester using H2SO4 and an alcohol
Then, using harsh oxidizing agent LiAlCl4 and then adding an acid (H3O+) → split ester bond in half to get two different alcohols.
2nd Way Conditions:
Add LiAlCl4 then add and acid to directly change a carboxylic acid into an alcohol.
What are the conditions needed to turn a Carboxylic Acid into an Amide? Then, how do we convert it into an amine?
Conditions:
Turn the carboxylic acid back into an Acyl Chloride (add SOCl2)
Then add an amine in order to produce the Amide.
Using a harsh chemical, LiALCl4 to provide harsh conditions, then adding an acid/water → an amine will be formed and double bond to oxygen will be broken.
How many different pi-nucleophiles are there and what are they called?
Alkene, Alkyne and Benzene — they are pi-nucleophiles due to the presence of pi-bonds, making the area electron-rich.
When do major and minor products come about?
When there is an alkyne, alkene or benzene, and there can be more than one possible product that can be formed (major/minor)
What do you add when you want to add halogens to a alkene?
Add either HBr / HCl or Br2/Cl2
Why can be Br2 and Cl2 be used without a catalyst?
The pi-nucleophilic region in the alkene/alkyne/benzene is a strong enough nucleophile in order to repel and break the sigma bond between the bromine/chlorine atoms.
What are the few possible ways to break the double bond?
Hydrogenation
Using water and H2SO4 as a catalyst
Using Br2/Cl2
Using HBr/HCl
Why can alkynes react with reagents like Br2 more than once?
They have 2 pi-bond and one sigma bond instead of just one pi-bond, hence they can react with more than one bromine/chlorine/fluorine atom to break both bonds.
How many different ways are there to break 2 pi-bonds in alkynes?
2 ways. Through Hydrogenation or the addition of halogens.
Why does benzene not undergo addition reaction?
Benzene is a overall very stable molecule with alternating patterns of single and double carbon bonds, where delocalized electrons move around freely. By going through an addition reaction, it will disrupt the delocalization of electrons, making the reaction undesirable/unfavourable.
What are the reagents and conditions needed for nitration of benzene?
Conditions: HNO3 and H2SO4 as a catalyst.
What is the reagents and conditions needed for sulphanation of benzene?
H2SO4
What is the requirements needed for bromination?
Br2 and FeBr3 catalyst
What is needed for Friedel-Crafts Alkylation?
RCl /RBr and AlCl3/AlBr3
What is needed for Friedel-Crafts Acylation?
ROCl and AlCl3 catalyst
How do you turn an alkylbenzene into benzoic acid?
By adding a strong oxidizing agent , KmNO4
How do you turn a benzene-Grignard’s reagent into benzoic acid?
By adding carbon dioxide and water
How do you make a phenol from a benzene ring with sulphate attached?
By adding NaOH @350 C with Acid/H3O
How do you make Diazonium Chloride or Food Coloring from a Benzene ring that went through Nitration?
Add NaNo3 with HCL @ 0C → This forms Diazonium Chloride
This reaction can be progressed further by adding Phenol to make bright-colored dyes for food coloring.
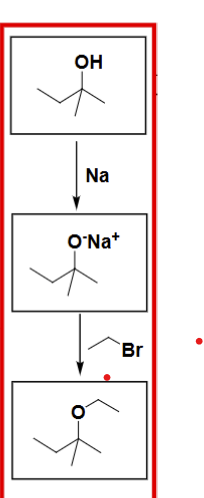
How do you carry out a Williamson Ether Synthesis reaction?
Alcohol (3rd Deg) + Na → get a salt with O- Na+
Then add your other products like e.g. CH3Br to add on CH3 to form an ether.
What do you add in order to turn this into a chloride salt/bromine alkyl halide? How do you turn it into a Grignard’s Reagent?
SOCl2 or PBr3
How to turn an existing alcohol into a alkyl halide?
Add HBr/HCl to the alcohol, and it can be converted back using NaOH to add back the hydroxide group.