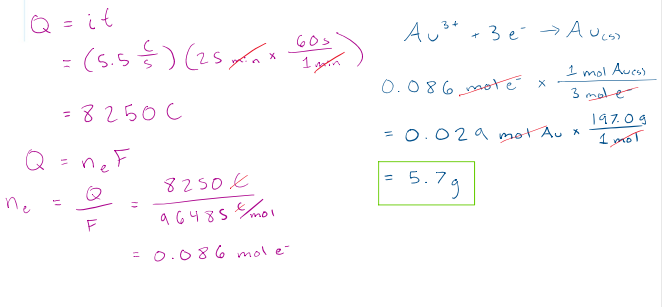Chapter 4.6 and 18: Redox and Electro chem
1/94
Earn XP
Description and Tags
Up to p.36 rn
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
Electrochemistry
studies the transformations between chemical and electrical energy.
In electrochemistry, electrical energy is generated by….
the movement of electrons from one chemical species to another in reactions
Redox reactions consist of…
oxidation and reduction half reactions
the movement of electrons from one chemical species to another in reactions known as….
oxidation-reduction (redox) reaction
Redox reactions consist of oxidation and reduction half reactions which can be identified by…
the change in oxidation state for the species involved
Oxidation and reduction must occur…
occur together.
Oxidation and reduction must occur together. If one substances loses…
electrons, another must gain the electrons.
Zn (s) + Cu 2+ → Zn 2+ (aq) + Cu (half reactions, which is reduced which is oxidized)
ox : (e lost)
Zn → Zn 2+ +2e
red: e gained
Cu2+ + 2e → Cu (s)
Each redox reaction contains an….
oxidizing agent and a reducing agent
𝑍𝑛 𝑠 + 𝐶𝑢2+(𝑎𝑞) → 𝑍𝑛2+(𝑎𝑞) + 𝐶𝑢 (𝑠) ( oxidizing agent and a reducing agent)
Zn is reducing agent, Cu 2+ oxidizing agent
In an oxidation-reduction reaction, the species reduced _____
B. Is the oxidizing agent
We can identify redox reactions through…
observation of a change in the oxidation states of the chemical species.
5 rules identify redox reactions (in order of priority:)
1) The oxidation state of a pure element is always zero.
2) The oxidation state of a monoatomic ion is equal to the charge
3) In a neutral species, the total of all charges must equal zero
4) In a complex ion, the total charges must equal the charge on the ion
5) When assigning oxidation states to the elements in a compound, follow the rules below in the sequence provided:
5) When assigning oxidation states to the elements in a compound, follow the rules below in the sequence provided:
a) Fluorine is always -1
b) Group 1 metals (Na, K, etc.) are always +1
c) Group 2 metals (Ca, Mg, etc.) are always +2
d) Hydrogen is +1 (except when bonded to a metal, then it is -1 … ex. LiH)
e) Other halogens (group 17) are usually -1
f) Oxygen (and other group 16 elements) are usually -2
g) Group 15 elements are usually -3
The oxidation state of a monoatomic ion is equal to the charge example
Na +, F-, Ca 2+, S 2-
The oxidation state of a pure element is always zero. examples
Zn, O2, Cu, Br2
In a neutral species, the total of all charges must equal zero example
HBr (1+ + 1-), H2O (2 x 1+ + -2)
In a complex ion, the total charges must equal the charge on the ion examples
NH4 - ((-3) + 4(+1)) = +1 , NO3 - (+5+3(-2))= -1
Assign oxidation states to each element in HSO4 -
+1 +6 + 4(-2) = -1
1 + X - 8 = -1
Assign oxidation states CrO4 2- .
+6
-8
= -2
-8 + X = -2
What is the oxidation state of the carbon atom in CO3 2- ?
+4
Examine the following reaction, is it a redox reaction? Which species are oxidized and reduced? Identify the oxidizing and reducing agents.
2𝑀𝑔 𝑠 + 𝑂2 𝑔 → 2𝑀𝑔𝑂(𝑠)
yes

Redox reactions must be balanced in both
mass and charge.
Generally, redox reactions are balanced by..
individual half-reactions and then recombined.
When balancing aqueous reactions, we must consider whether…
they are occurring in an acidic or alkaline environment
Balance the following redox reaction in acidic solution:
𝑀𝑛𝑂4 − + 𝐶2𝑂4 2− → 𝑀𝑛2+ + 𝐶𝑂2 1.
1. Write the two half reactions
2. Balance the coefficients for all atoms except hydrogen and oxygen
3. To balance the oxygen, add H2O to the side deficient in oxygen 4. To balance the hydrogen, add H+ to the side deficient in hydrogen
4. To balance the hydrogen, add H+ to the side deficient in hydrogen
5. Balance the charges by adding electrons to the side deficient in negative charge. The electrons should be on opposite sides of the half-reactions! 6. Multiply the equations to ensure equal numbers of electrons are transferred.
6. Multiply the equations to ensure equal numbers of electrons are transferred.
7. Add the two balanced half-reactions and cancel species present on both sides. Reduce the coefficients if a common denominator exists.
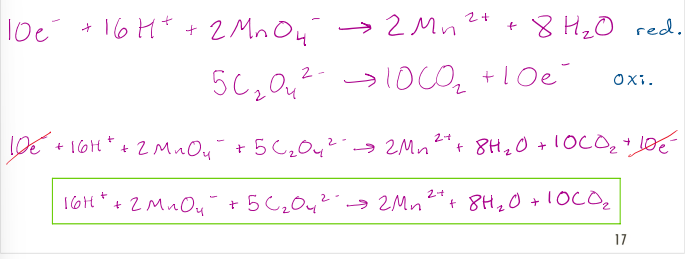
Balance the following redox reaction in basic solution: 𝑀𝑛𝑂4 − + 𝐶𝑙− → 𝑀𝑛2+ + 𝐻𝐶𝑙O
1. Write the two half reactions
2. Balance the coefficients for all atoms except hydrogen and oxygen
3. To balance the oxygen, add H2O to the side deficient in oxygen
4. To balance the hydrogen, add H+ to the side deficient in hydrogen
5. The solution is BASIC! For every H+ added, add the same number of OHto both sides!
6. Combine H+ and OHspecies to form H2O where applicable
7. Balance the charges by adding electrons to the side deficient in negative charge. The electrons should be on opposite sides of the half-reactions!
8. Multiply the equations to ensure equal numbers of electrons are transferred.
9. Add the two balanced half-reactions and cancel species present on both sides. Reduce the coefficients if a common denominator exists
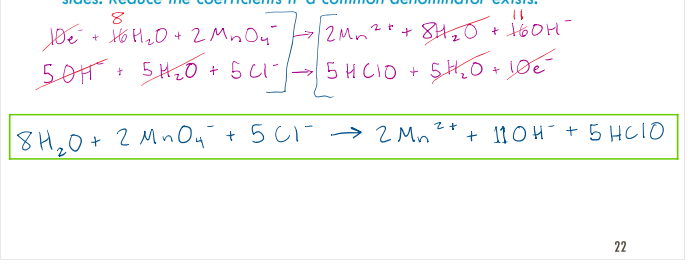
The following reaction is a disproportionation reaction:
𝐶𝑙2 → 𝐶𝑙− + 𝐶𝑙𝑂3 −
The chlorine is both oxidized and reduced. Balance this reaction in basic solution.
correct
Electrical current is the
flow of electric charge – such as the movement of electrons during a redox reaction
If you immerse a piece of Zn metal in a CuSO4 solution containing free Cu2+ ions, the Zn will…
dissolve while the Cu2+ is reduced and deposited on the surface of the Zn metal.
The electrons produced from Zn in the oxidation half-reaction are directly and immediately transferred to the…
Cu2+ ions in the reduction half-reaction.
Electrochemical cells are used to…
harness the electrical energy of the electron transfer.
Each half-reaction is carried out within a…
half-cell.
Forcing the electrons to move through an external wire creates a
useable electrical current.
Two general types of electrochemical cell exist.
Voltaic Cell, Electrolytic Cells
Voltaic Cells
A spontaneous reaction produces a flow of electrons (electrical current).
Electrolytic Cells
An electric current is used to cause an otherwise nonspontaneous reaction to occur.
Electrode:
The conductive surface through which electrons can enter and leave a half-cell.
Anode (-):
The electrode at which the oxidation occurs.
Cathode (+):
The electrode at which the reduction occurs.
Salt Bridge:
U-shaped tube containing a strong electrolyte and connecting the two half-cells. Ion flow through the salt bridge neutralizes the charge build-up of the half-reactions in each half-cell.
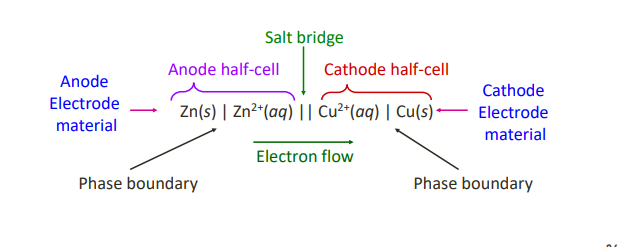
A cell diagram or line notation can be used as…
a shorthand notation for representing an electrochemical cell.
The following conventions are used when writing a cell diagram: (6)
1) The anode is written first on the left, followed by the other reactants in the order in which they occur in the cell from the anode to the cathode.
2) A phase boundary is represented by a single vertical bar.
3) The salt bridge is indicated by a double vertical bar.
4) If two or more reactants are in the same phase, separate them by a comma. 5) If inert electrodes such as graphite or platinum are involved, they are placed on the anode or cathode end of the diagram as applicable, and separated by a single vertical bar.
6) Stoichiometric coefficients and species that are not directly involved in the reaction are not shown.
Zn (s) + Cu 2+ → Zn 2+ (aq) + Cu (Draw the cell diagram)
correct
Consider the following redox reaction in which Fe(s) is oxidized and MnO4 - (aq) is reduced:
5𝐹𝑒 𝑠 + 2𝑀𝑛𝑂4 − 𝑎𝑞 + 16𝐻 + 𝑎𝑞 → 5𝐹𝑒2+ 𝑎𝑞 + 2𝑀𝑛2+ 𝑎𝑞 + 8𝐻2𝑂(𝑙)
Write the cell diagram for this reaction.
correct
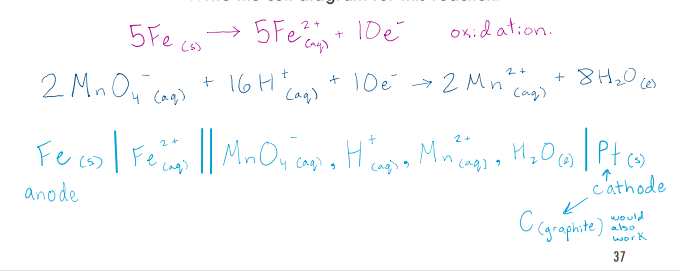
Consider the following electrochemical cell: 𝑍𝑛 𝑠 |𝑍𝑛2+ 𝑎𝑞 || 𝐶𝑢2+ 𝑎𝑞 |𝐶𝑢(𝑠) In the above cell notation, which species is the cathode?
Cu
electrons flow from states of….
high electrical potential energy to states of lower electrical potential energy
Electrical current is driven by…
the potential difference between the two electrodes of the redox process.
A large potential difference corresponds to a….
large difference in charge between the electrodes and a strong tendency for electron flow.
cell potential (Ecell)
The potential difference between the electrodes in an electrochemical cell
The cell potential is a measure of…
the overall tendency of a redox reaction to occur spontaneously.
Ecell depends on…
concentrations, temperature, and the relative tendencies for the reactants to undergo oxidation or reduction.
Combining a substance with a strong tendency to undergo oxidation with a substance with a strong tendency to undergo reduction produces….
a large difference in charge between the two electrodes.
Under standard conditions, the cell potential is known as the…
standard cell potential (E° cell).
The value of E° cell represents the…
potential energy difference between the electrodes.
The value of E° cell is determined by the…
difference in the standard electrode potentials (E°) for the two electrodes (half reactions).
When half-cells are connected, electrons flow from the….
electrode with the higher potential for oxidation to the electrode with a higher potential for reduction.
What is standard conditions:
1m, 1 BAR, 25celsius
By convention, standard electrode potentials for half-reactions are written as…
reduction potentials
The half-cell standard electrode potentials are determined against a…
defined electrode potential of zero.
The zero point is defined by the..
standard hydrogen electrode (SHE)
E° cell =
E° red + E° ox
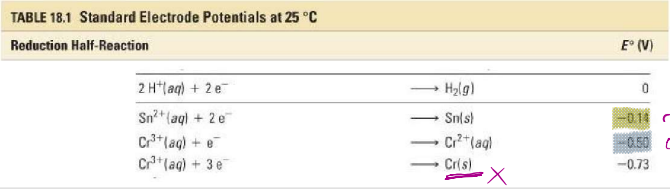
Use tabulated standard electrode potentials to calculate the standard cell potential for the following reaction:
𝑆𝑛2+ 𝑎𝑞 + 2𝐶𝑟2+ 𝑎𝑞 → 𝑆𝑛 𝑠 + 2𝐶𝑟3+(𝑎𝑞)
correct


Will the following redox reaction occur spontaneously under standard conditions? 3𝑍𝑛 𝑠 + 2𝐴𝑙3+ 𝑎𝑞 → 3𝑍𝑛2+ 𝑎𝑞 + 2𝐴𝑙(𝑠)
A spontaneous redox reaction requires a POSITIVE standard cell potential!

Which of the following metals will dissolve in HNO3 but not HCl?
Fe Au Ag
correct

In general, a redox reaction will be spontaneous as written when:
• 𝐸𝑐𝑒𝑙𝑙 𝑜 is positive (>0)
• ∆𝐺𝑟𝑥𝑛 𝑜 is negative (<0)
• K > 1
SPONTANEITY IN REDOX REACTIONS pyramid
correct
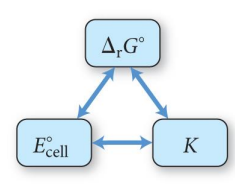
RELATING ∆𝐺𝑟𝑥𝑛 𝑜 AND 𝐸𝑐𝑒𝑙l standard
∆𝐺𝑟𝑥𝑛 𝑜 = -nFEcell 𝑜 (F is Faraday’s constant 96.485 c/mol)

Using tabulated reduction potentials, determine ΔG° rxn for the following reaction. Is the reaction spontaneous?
𝐼2 𝑠 + 2𝐵𝑟− 𝑎𝑞 → 2𝐼 − 𝑎𝑞 + 𝐵𝑟2(𝑙)
correct

The following reaction was non-spontaneous 𝐼2 𝑠 + 2𝐵𝑟− 𝑎𝑞 → 2𝐼 − 𝑎𝑞 + 𝐵𝑟2(𝑙) Which best explains this result?
A. Br has a larger electron affinity than I; therefore, we do not expect Brto give an electron to I2 .
RELATING 𝐸𝑐𝑒𝑙𝑙 𝑜 AND K R= 8.314 j/mol.k
𝐸𝑐𝑒𝑙𝑙 𝑜 = ((RT)/(nF)) * lnK

Using tabulated reduction potentials, calculate the value of K for the oxidation of iron by H+ at 25°C.
2𝐹𝑒 𝑠 + 6𝐻 + 𝑎𝑞 → 2𝐹𝑒3+ 𝑎𝑞 + 3𝐻2(𝑔)
correct

E CELL AND NONSTANDARD CONCENTRATIONS: Q < 1
correct

E CELL AND NONSTANDARD CONCENTRATIONS: Q = 1 (standard conditions)
correct

E CELL AND NONSTANDARD CONCENTRATIONS: Q > 1
correct

E CELL AND NONSTANDARD CONCENTRATIONS: Q = K (equilibrium)
correct

In an electrochemical cell, Q = 0.0010 and K = 0.10. Which statement is true?
A. Ecell is positive and E° cell is negative
A pH meter has 1.2 atm of H2 in contact with a Pt electrode. The other electrode is an Al rod in a 0.20 M Al3+ solution. If the pH of a solution is 4.00, what is the potential of the cell? The cell reaction is:
2𝐴𝑙 𝑠 + 6𝐻 + 𝑎𝑞 → 2𝐴𝑙3+ 𝑎𝑞 + 3𝐻2(𝑔)
correct

For the same pH meter, what is the pH if the Ecell is 1.17 V?
2𝐴𝑙 𝑠 + 6𝐻 + 𝑎𝑞 + 6𝑒 − → 2𝐴𝑙3+ 𝑎𝑞 + 3𝐻2(𝑔) + 6𝑒 −
correct

In an electrolytic cell, electrical current from an external source is used to…
drive a nonspontaneous redox reaction through electrolysis.
Voltaic and electrolytic cells work in…..
reverse directions.
ELECTROLYSIS 2 cell options
Discharge (voltaic cell) (+) and Recharge (electrolytic cell): (-)
In the voltaic cell, the anode (-)…… while the cathode (+)…..
is the source of electrons, draws the electrons.
In the electrolytic cell, the source of electrons is….
the power source
In the electrolytic cell, the source of electrons is the power source – which draws the electrons from….
the anode (+ terminal) driving them to the cathode (- terminal).
ELECTROLYSIS For the reaction to occur, we must supply the reaction with….
more voltage than the standard cell potential.
For the reaction to occur, we must supply the reaction with more voltage than the standard cell potential. This will….
reverse the direction of the electron flow, forcing the non-spontaneous reaction to occur.
Which statement is true for an electrolytic cell at standard state conditions?
D. The cell reaction has a positive ΔG°
If an electrolytic cell contains both molten NaBr and KCl, what would be the products of the redox reaction? Cation species:
correct

If an electrolytic cell contains both molten NaBr and KCl, what would be the products of the redox reaction? Anion species:
correct

In aqueous electrolysis, the oxidation or reduction of water at either electrode is possible. Water reduction:
correct
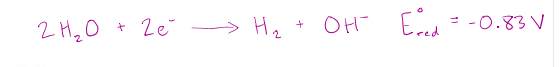
In aqueous electrolysis, the oxidation or reduction of water at either electrode is possible. Water oxidation:
correct
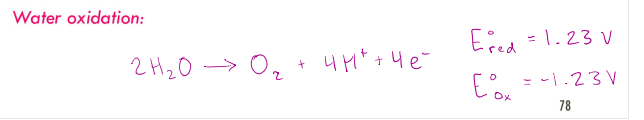
What products form from the electrolysis of an aqueous NaBr solution?
correct
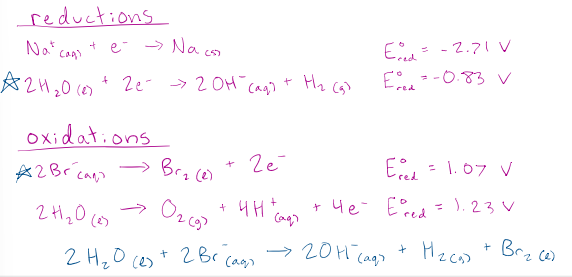
electroplating
preferential reduction., Metal ions can be plated onto other metals or substrates through a process of
Gold can be plated out of a solution containing Au3+. What mass of gold, in grams, is plated in 25 minutes at a current of 5.5 A?
correct