Transamination and Deamination
1/111
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
112 Terms
True or False: Like carbohydrate and lipid metabolism, protein degredation is primarily an energy synthesis process
False- it is not primarily an energy degredation process
What is protein breakdown a large consequence of
cellular maintenance and nitrogen balance
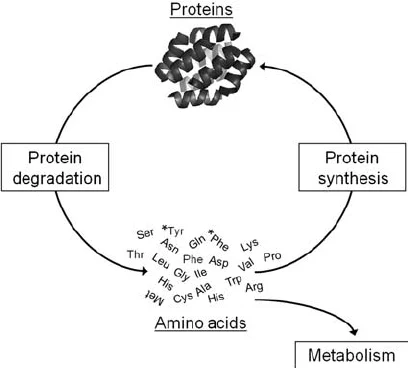
In the body, proteins are continuously synthesized and degraded in a dynamic equilibrium known as:
protein turnover
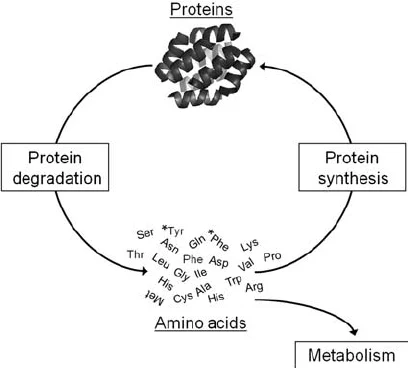
what does protein turnover ensure
that damaged, misfolded, or unecessary proteins are removed and replaced
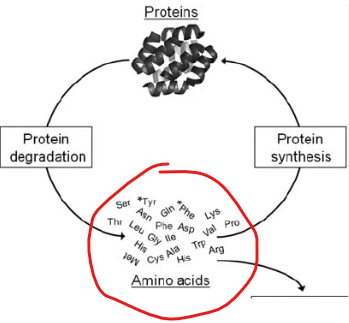
What does protein turnover inherently geenrate regardless of whether the body is in a fed or fasting state
free amino acids
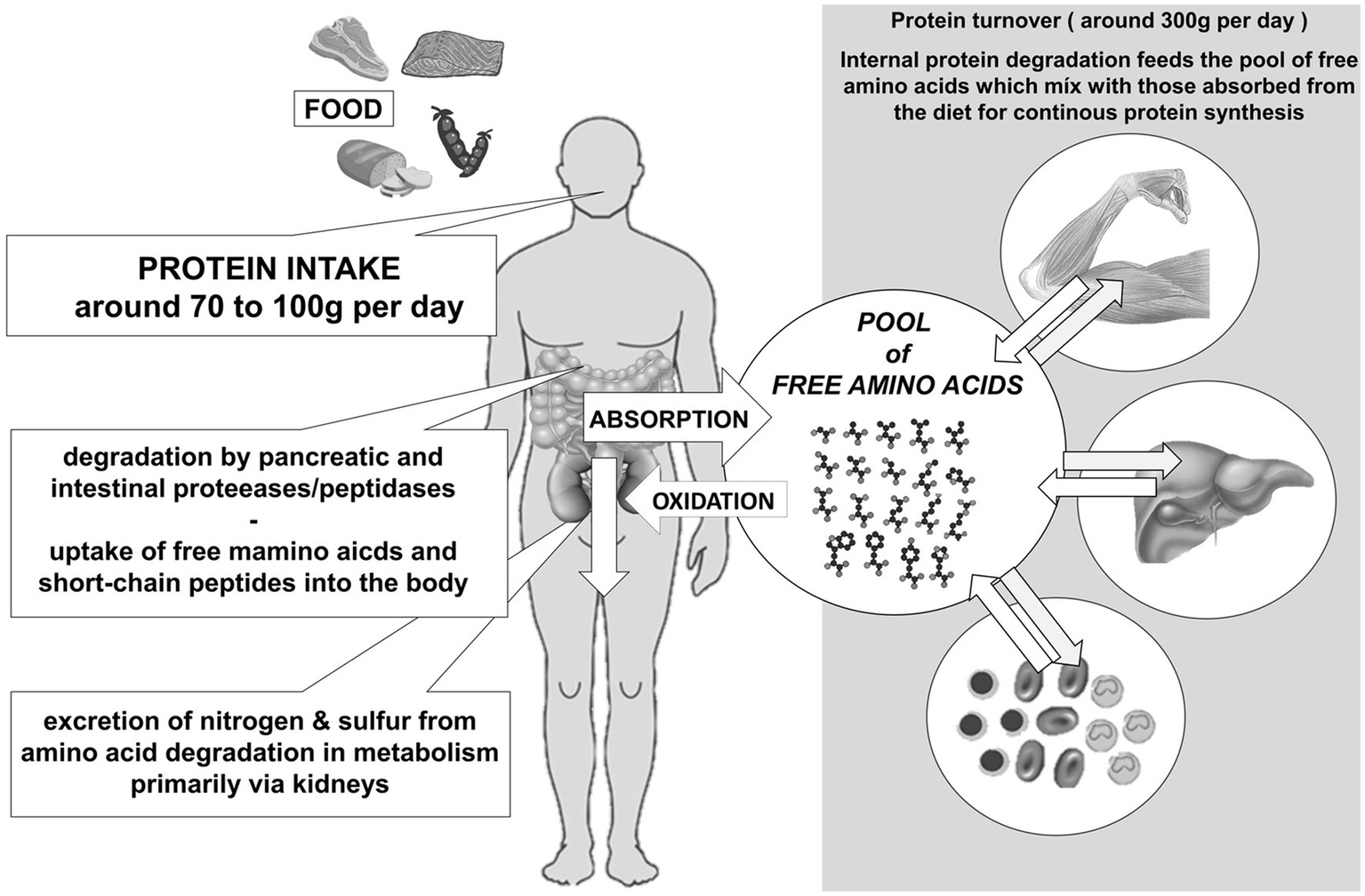
which type of proteins contribute significantly to the amino acid pool following digestion and absorption
dietary protein
why must amino acids that are not immediately required for protein synthesis must be further processed and ultimately degraded
because the human body does not maintain a storage form of amino acids
True or False: Amino acid catabolism is obligatory
true
Amino acid catabolism is not optional, but obligatory driven by what need
nitrogen homeostasis
Because the human body does not maintain a storage form of amino acids analogous to glycogen or triglycerides, any amino acids that are not immediately required for protein synthesis must be further processed and ultimately degraded. This creates a situation in which amino acid catabolism is not optional but obligatory, driven by the need to maintain nitrogen homeostasis.
Therefore,
even in energy-rich conditions, amino acid degradation occurs, not for ATP production, but to prevent accumulation of excess nitrogen, which would otherwise disrupt metabolic balance.
what are amino acids generated from
protein degredation
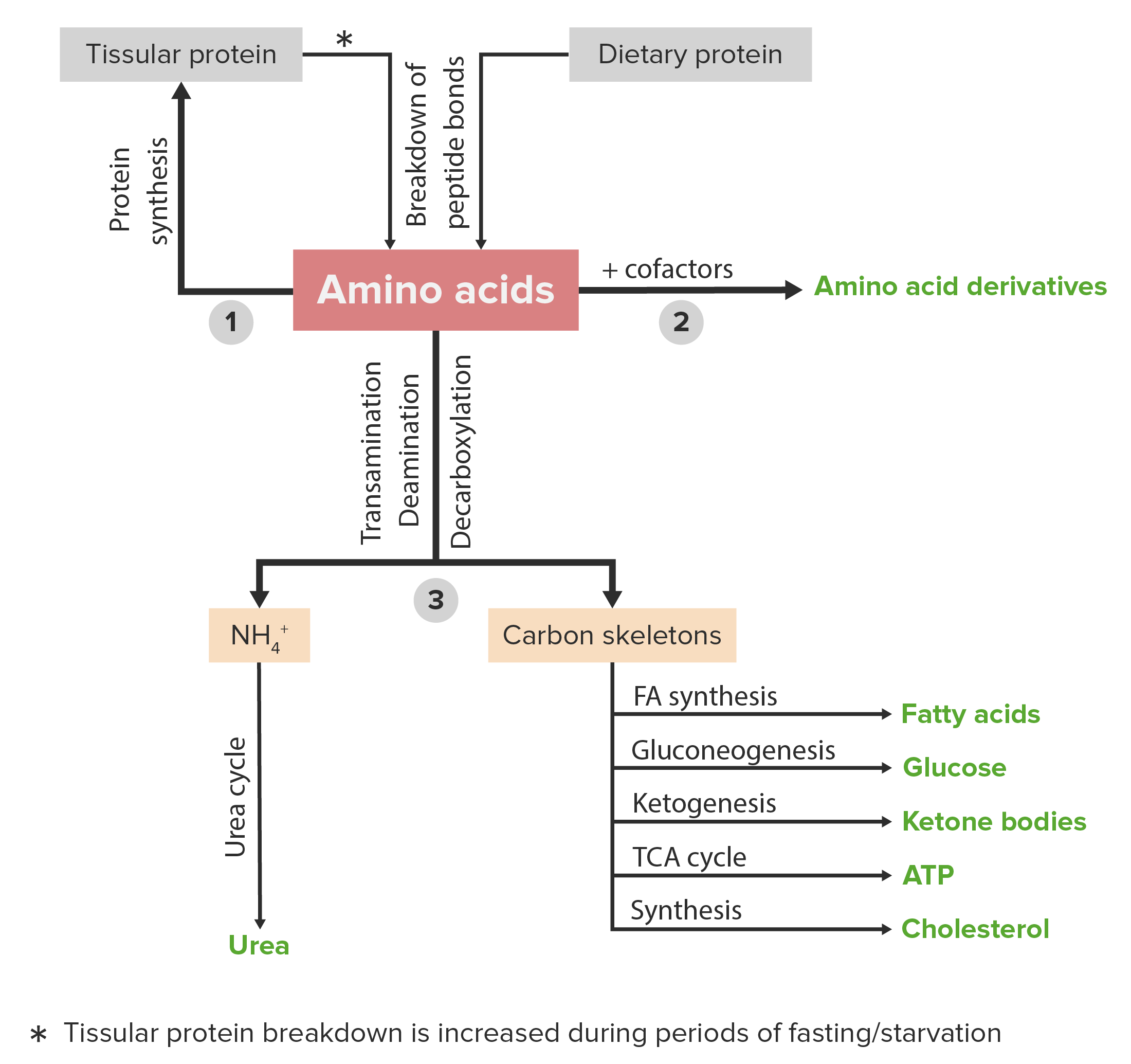
Once amino acids are generated from protein degradation, they are conceptually divided into two distinct components, what are they?
carbon skeleton
amino group
The division of an amino acid is central to understanding nitrogen metabolism because each component follows a completely different metabolic fate.
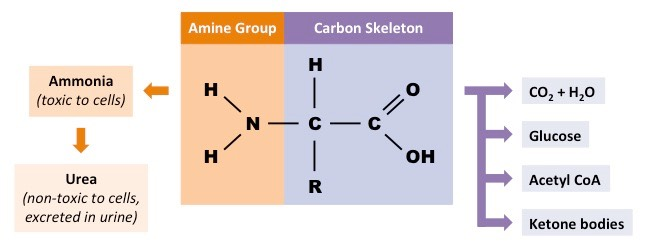
What is the carbon skeleton
essentially an alpha-keto acid after removal of the amino group, can enter central metabolic pathways such as the citric acid cycle, gluconeogenesis, or ketogenesis, depending on its structure
The division of an amino acid is central to understanding nitrogen metabolism because each component follows a completely different metabolic fate.
What is the amino group
represents a nitrogen-containing moiety that cannot be freely released due to its potential to form ammonia, a highly toxic compound
The division of an amino acid is central to understanding nitrogen metabolism because each component follows a completely different metabolic fate.
What must the body do as a result
the body must employ tightly regulated biochemical mechanisms to safely transfer, collect, and ultimately dispose of nitrogen.
What challenge does the entire framework of nitrogen metabolism revolve around
how to remove nitrogen efficiently without ever allowing toxic intermediates to accumulate.
What are the central nitrogen- carrying amino acids
alanine
glutamine
glutamate
aspartate
What makes
alanine
glutamate
glutamine
aspartate
have a disproportionately important role in nitrogen metabolism
they have an ability to both carry nitrogen and interface directly with central metabolic pathways
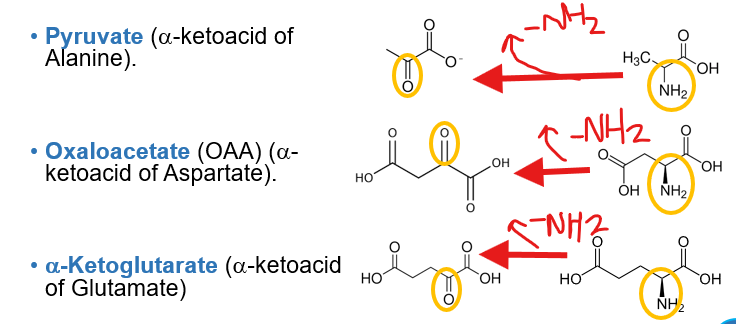
What makes the 4 central nitrogen carrying amino acids uniquely suited for this role
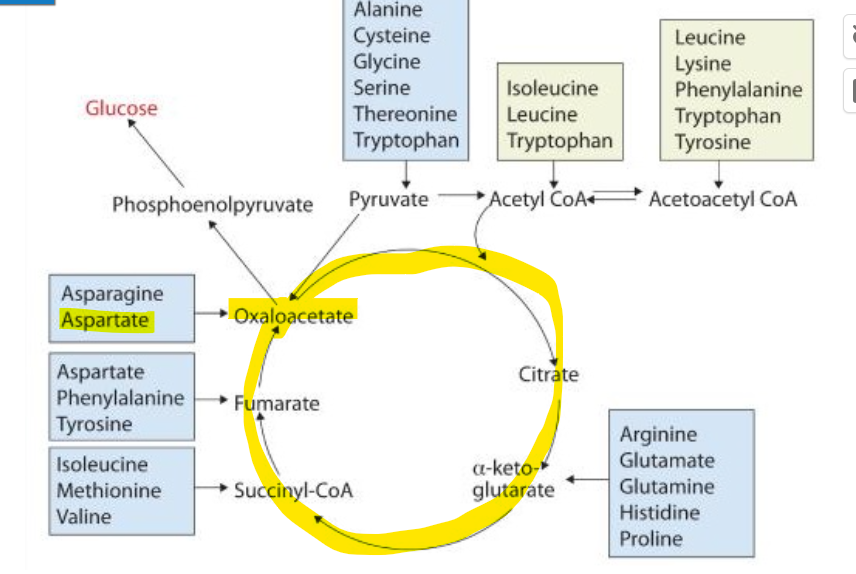
because their corresponding carbon skeletons are key intermediates in the citric acid cycle
What makes the 4 central nitrogen carrying amino acids uniquely suuited for this role is becasue their corresponding carbon skeletons are key intermediates intermediates in the citric acid cycle.
What are the key intermediates?
pyruvate
alpha-ketoglutarate
oxaloacetate
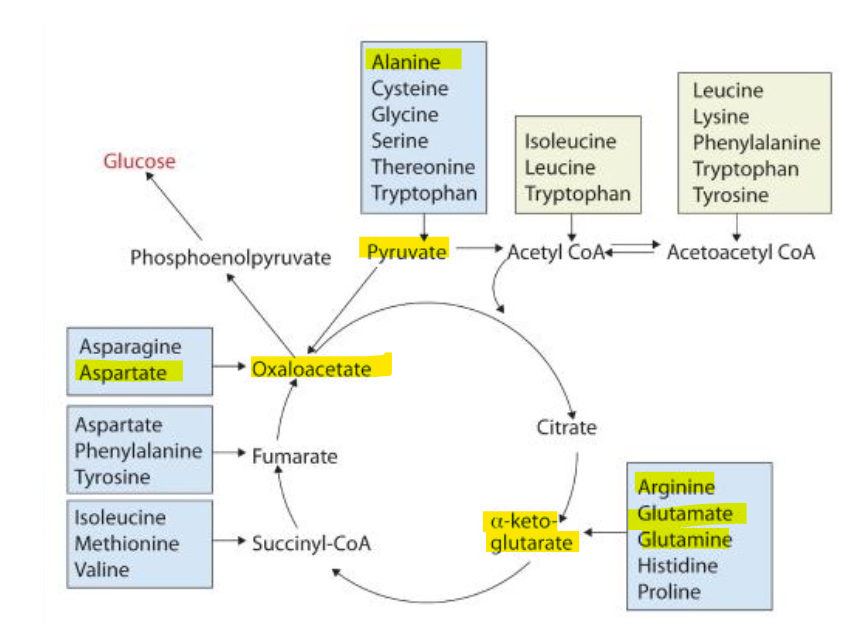
What makes the 4 central nitrogen carrying amino acids uniquely suuited for this role is becasue their corresponding carbon skeletons are key intermediates intermediates in the citric acid cycle:
pyruvate
alpha-ketoglutarate
oxaloacetate
What does this structural relationship allow for metabolism
it allows these skeletons of these amino acids to to link nitrogen metabolism with energy metabolism
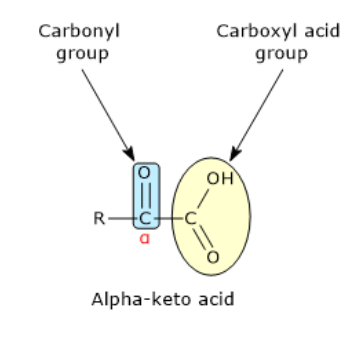
Whatare known as the metabolic counterparts of amino acids
alpha- keto acids
What is a critical structural concept underpinning nitrogen metabolism
the relationship between amino acids and their corresponding alpha keto acids
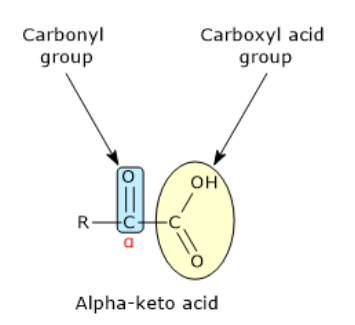
what is an alpha ketoacid
the presence of a carbonyl group at the alpha position adjacent to a carboxyl group
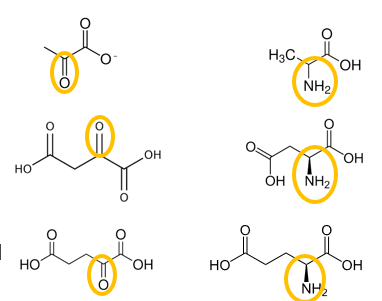
how does an alpha ketoacid differe from its corressponding amino acid
only by the substitution of an amino group with a keto group
What does the structural difference beween an alpha keto acid and the amino acid counterpart
it has profound metabolic implications, as it allows for the reversible interconversion between amino acids and TCA cycle intermediates.
The structural difference between an alpha keto acid and the amino acid counterpart allows it to have profound metabolic implications, as it allows for the reversible interconversion between amino acids and TCA cycle intermediates.
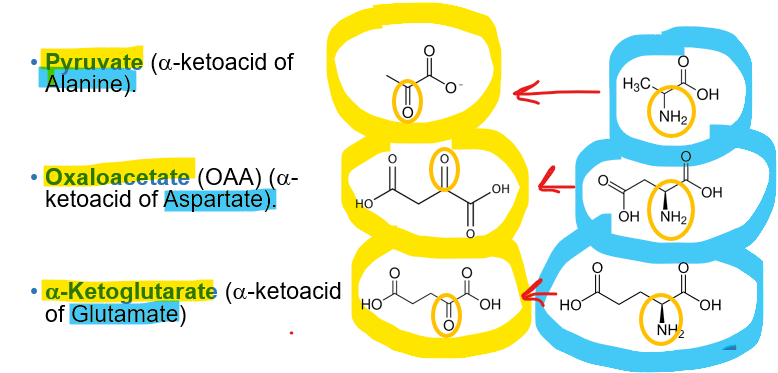
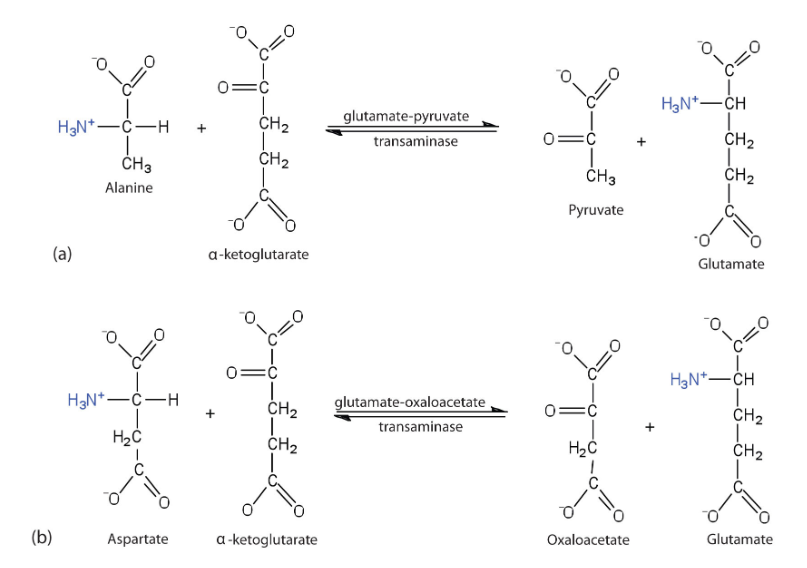
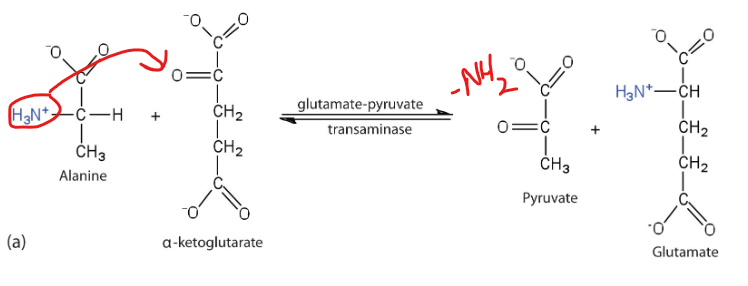
What is an example of this using alanine
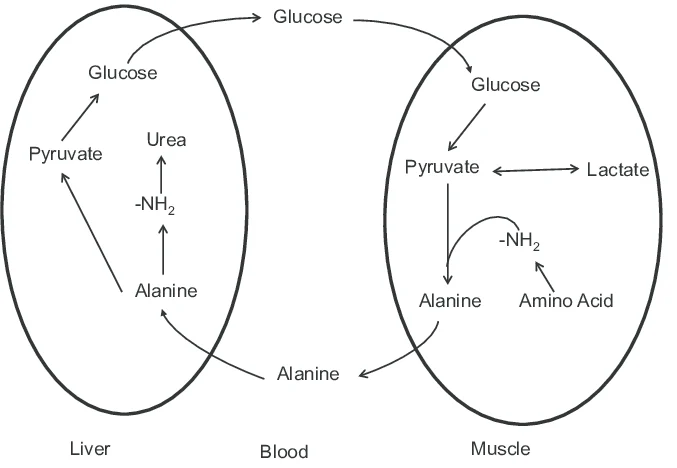
Alanine can be converted back to pyruvate through transamination, and therefore participate in the glucose- alanine cycle- transporting the nitrogen from muscle to liver
The structural difference between an alpha keto acid and the amino acid counterpart allows it to have profound metabolic implications, as it allows for the reversible interconversion between amino acids and TCA cycle intermediates.
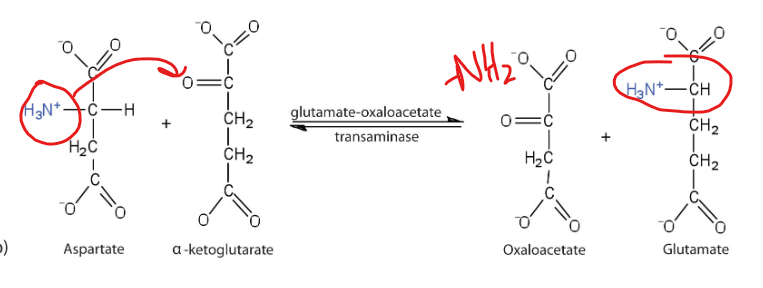
What is an example of this using aspartate?
Aspartate can be converted back to oxaloacetate
he structural difference between an alpha keto acid and the amino acid counterpart allows it to have profound metabolic implications, as it allows for the reversible interconversion between amino acids and TCA cycle intermediates.
What is an example of this using glutamate?
glutamate converting back to alpha ketoglutarate
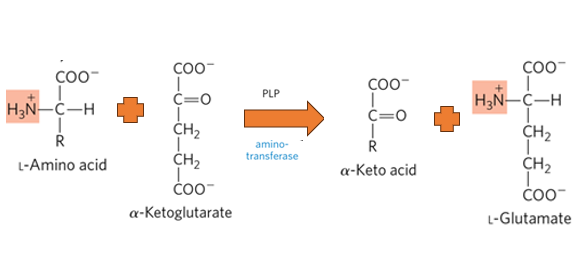
What is the primary biological mechism by which amino groups are transferred between molecules with the release of free ammonia.
Transamination
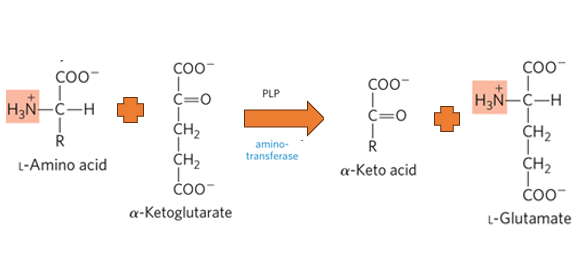
What happens during the process of Transamination
an amino group from an amino acid is transferred to an alpha-keto acid, typically alpha-ketoglutarate, resulting in the formation of a new amino acid and a new alpha-keto acid.
what enzymes catalyze transamination
aminotransferases
True or False: transamination is reversible
true
Since transamination is is reversible, what does this allow for
it allows for the dynamic redistribution of nitrogen depending on cellular needs
for example, if we dont need any more OAAC we just convert back into apsartate
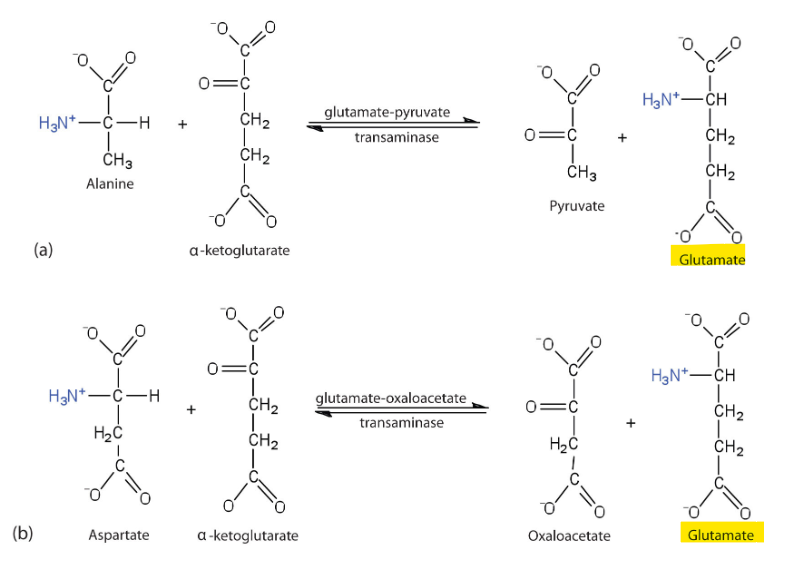
what is the most significant outcome of transamination
funneling of amino groups onto glutamate, effectively centralizing nitrogen in a single molecule
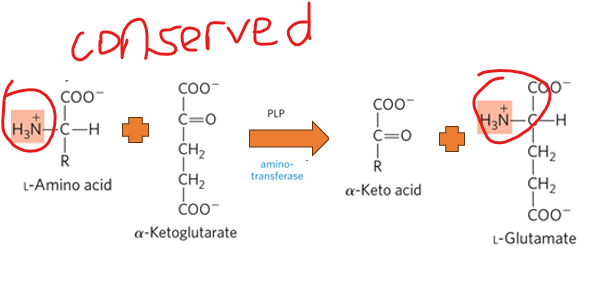
Transamination reactions do NOT generate ammonia, what do they do instead
they preserve the amino grouo in a bound, non-toxic form
Transamination reactions do not generate ammonia; instead, they preserve the amino group in a bound, non-toxic form, what makes this so essential
it prevents the accumulation of free ammonia in tissues, which could disrupt cellular function
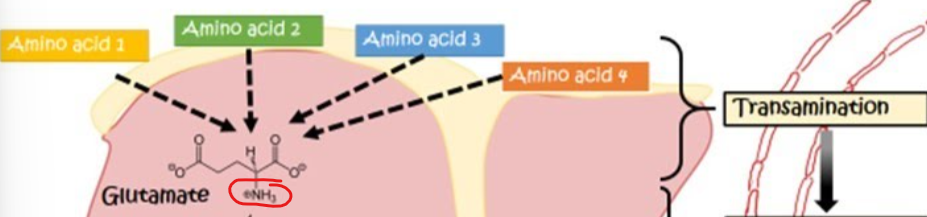
What type of mechanisms does transamination serve as
a protective mechanism, enabling nitrogen to be safely transported wihtin the cell
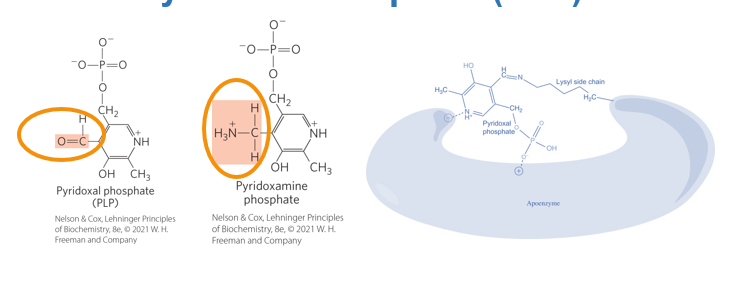
What does the transamination process critically depend on
the coenzyme pyridoxal phosphate (PLP)
what is the coenzyme pyridoxal phosphate (PLP) a derivative of
vitamin B6
What does Pyridoxal Phosphate (PLP) do?
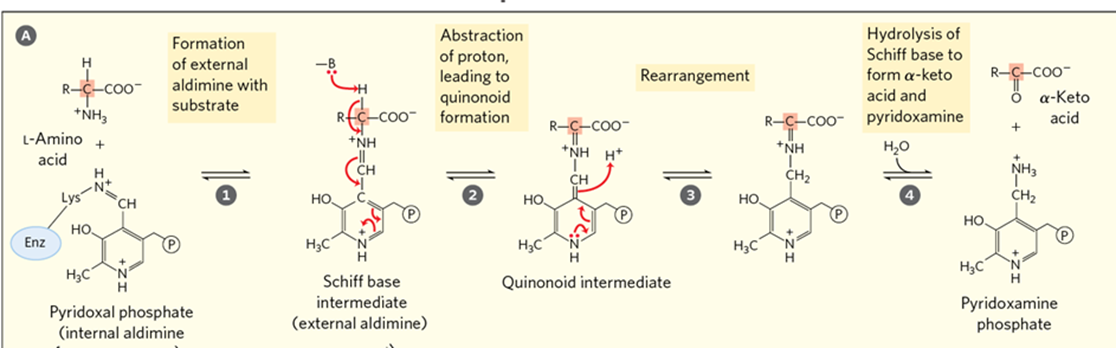
it acts as a transient carrier of amino groups during the transamination reaction
What is the key feature of PLP
its ability to stabilzie reaction intermediates and facilitate the movement of the amino group without releasing it into the surrounding environment
The key feature of PLP is its ability to stabilize reaction intermediates and facilitate the movement of the amino group without releasing it into the surrounding environment.
What does this ensure
nitrogen remains isolated throughout the reaction, preventing the formation of free ammonia
What does the dependence on PLP highlight
the importance of vitamin B6 in amino acid metabolism, as deficiencies in this vitamin can impair transamination reactions and disrupt nitrogen handling.
What are two clinically significant enzymes that catalyze transamination reactions involving alanine and aspartate
Alanine Aminotransferase (ALT)
Aspartate Aminotransferase (AST)
Where are the enzymes Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) mainly active
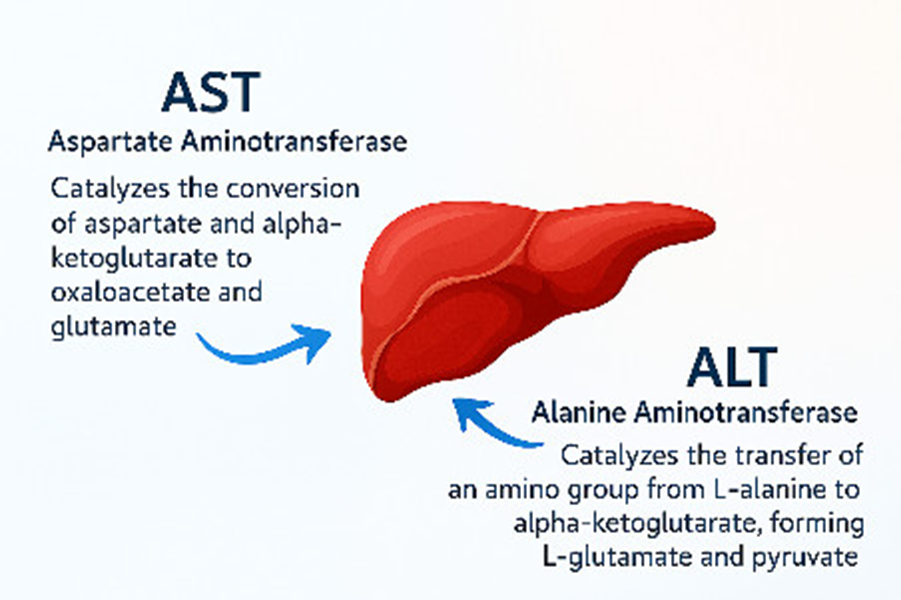
in tissues such as the liver and muscles
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) are two clinically significant enzymes that catalyze transamination reactions involving alanine and aspartate, respectively. These enzymes are highly active in tissues such as the liver and muscle
Why particularly here?
this is where amino acid metabolism is most prominent

what facilitates the interconversion of alanine and pyruvate- which plays a key role in the glucose alanine cycle
Alanine aminotransferase

What interconverts aspartate and oxaloacetate, linking nitrogen metabolism in the urea cycle
Aspartate aminotransferase (AST)
Where are ALT and AST primarily confined
within their cells
If there is a presence of AST or ALT in the bloodstyream aty elevated levels, what is this indicative of
cellular damage in the liver because they are mainly supposed to be confined within their cells
What conditions can cause the release of ALT ans AST into the serum albumin, making them diagnostic markers
hepatitis
cirrohsis
muscle injury
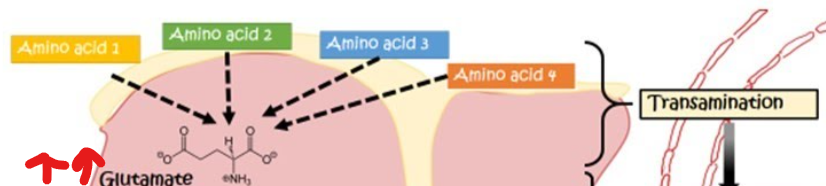
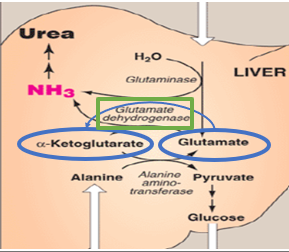
Following transamination , what happens
glutamate accumulates as the primary carrier of amino groups
what does increased glutamate necessitate
a mechanism for nitrogen disposal
How is nitrogen disposed
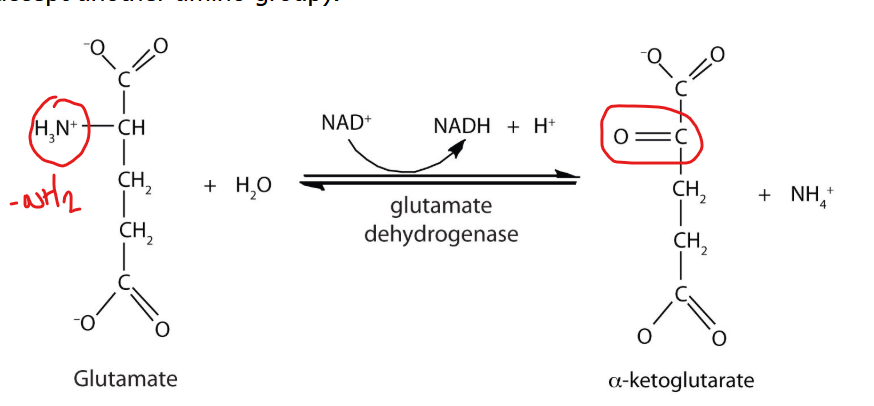
oxidative deamination
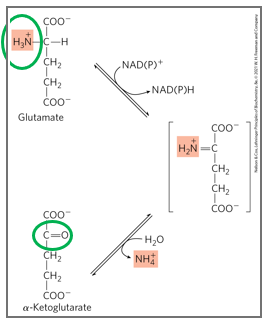
what catalyzes oxidative deamination
glutamate dehydrogenase
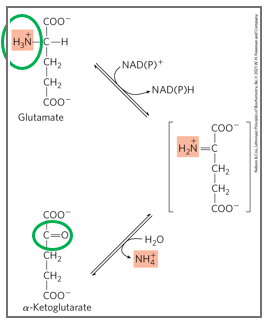
what is the mechanism off oxidative deamination
glutamate is converted back to alpha ketoglutarate while releasing free ammonia
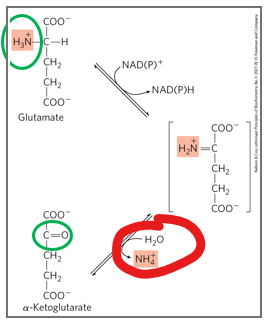
oxidative deamination is a reaction catalyzed by glutamate dehydrogenase (GDH), in which glutamate is converted back to alpha-ketoglutarate while releasing free ammonia.
What makes this step unique
because it is one of the few points in amino ac id metabolism where ammonia is actually generated
Given the toxicity of ammonia, oxidative deamination is tightly controlled and localized where
in the mitochondira of liver cells, where the urea cycle operates
By couplinjg the ammonia production directly with detoxification, what does the cell ensure
that ammonia does not accumulate or diffuse into the blood stream.
What does the regeneration of alpha ketoglutarate also allow
for continued transamination reactions, mantaining the cycle of nitrogen collection and disposal
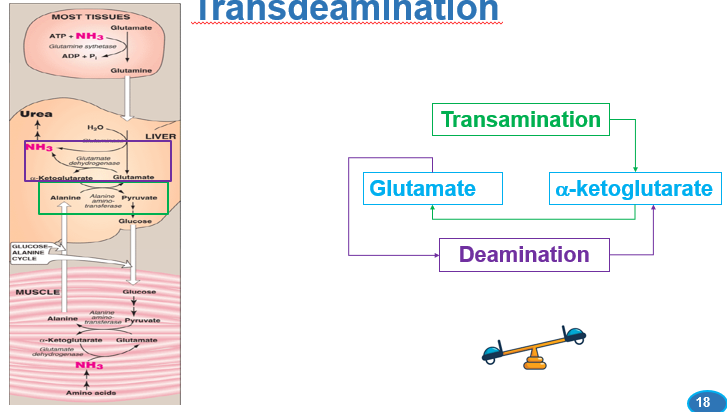
what process does the combination of transamination and oxidative deamination constitute
transdeamination
what does transdeamination represent
the primary pathway for amino acid nitrogen removal
What is the process of transdeamination
transamination reactions first transfer amino groups from various amino acids onto alpha-ketoglutarate, forming glutamate. Subsequently, glutamate undergoes oxidative deamination to release ammonia, which is then funneled into the urea cycle
What does the 2 step strategy of transdeamination ensure
that nitrogen from multiple sources is consolidated into a single molecule before being released in a controlled manner
What is the advantage of transdeamination
it minimizes the risk of ammonia toxicity while maximizing efficiency in nitrogen disposal
Transdeamination therefore serves as a central organizing principle in nitrogen metabolism by
integrating multiple pathways into a cohesice and regulated process
Glutamate dehydrogenase (GDH) is a key regulatory enzyme in amino acid metabolism, what is its activity closely tied tro
the energy status of the cell

Glutamate dehydrogenase is a key regulatory enzyme in amino acid metabolism, and its activity is closely tied to the energy status of the cell.
When is GDH inhibited
in high energy condition with elevated levels of ATP and GTP
This is because this is regulated through the breakdown of proteins. Breakdown occurs when there is not ample energy. If GDH is inhibited that means we have ample amount of energy that should be dedicated for protein synthesis instead of deamination
Glutamate dehydrogenase is a key regulatory enzyme in amino acid metabolism, and its activity is closely tied to the energy status of the cell.
In high-energy conditions, characterized by elevated levels of ATP and GTP, GDH is inhibited— why is this the case?
it prevents unnecessary breakdown of amino acids, thereby favoring protein synthesis and anabolic processes

Glutamate dehydrogenase is a key regulatory enzyme in amino acid metabolism, and its activity is closely tied to the energy status of the cell.
in low-energy states, where ADP and GDP levels are high, what happens to GDH
it is activated to promote amino acid degredation and generate carbon skeletons that can enter the citric acid cycle for energy production
Additionally, high concentrations of amino acids can stimulate GDH activity, what does this signal
that excess nitrogen and carbon skeletons need to be processed
WHat is one of the central challenges in nitrogen metabolism
the safe transport of ammonia between tissues, particularly from peripheral tissues such as muscle and brain to the liver, where detoxification via the urea cycle occurs.
Because free ammonia is highly toxic—especially to the central nervous system— what does the body avoid
transporting ammonia in its free form
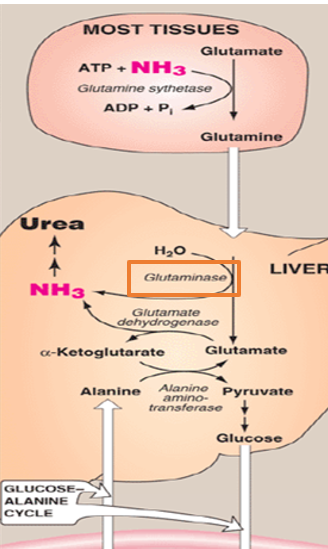
Because free ammonia is highly toxic—especially to the central nervous system—the body avoids transporting it in its free form.
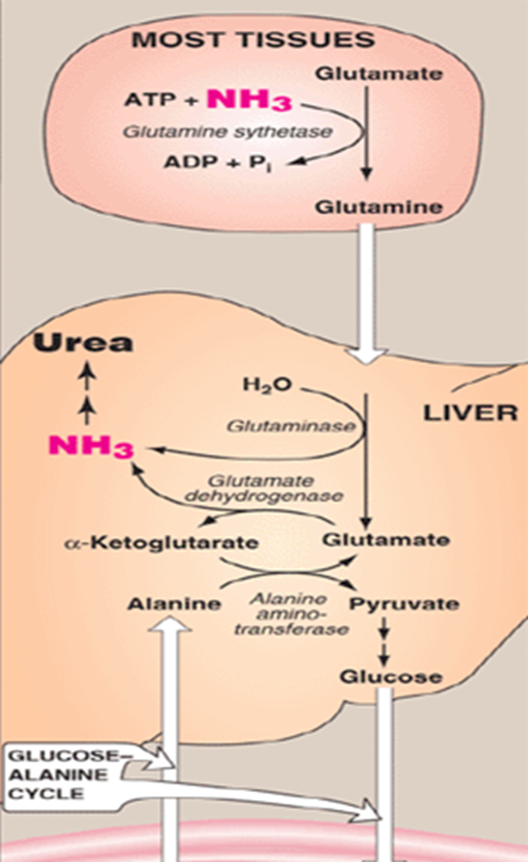
What is ammonia incorporated into instead when transporting from other tissues to the liver ( everything we discussed previously happened within the liver)
amino acid glutamine- a non toxic carrier of nitrogen
When ammonia is incorporated into the amino acid glutamine, which serves as a non-toxic carrier of nitrogen, what catalyzes this process
glutamine synthetase
what does the enzyme glutamine sythetase do
it combines glutamate with free ammonia in an ATP-dependent reaction to form glutamine
Ammonia is incorporated into the amino acid glutamine, which serves as a non-toxic carrier of nitrogen. This process is catalyzed by the enzyme glutamine synthetase, which combines glutamate with free ammonia in an ATP-dependent reaction to form glutamine.
What does glutamine effectively do
it “locks away” ammonia in a chemically stable form that can circulate safely in the bloodstream to the liver
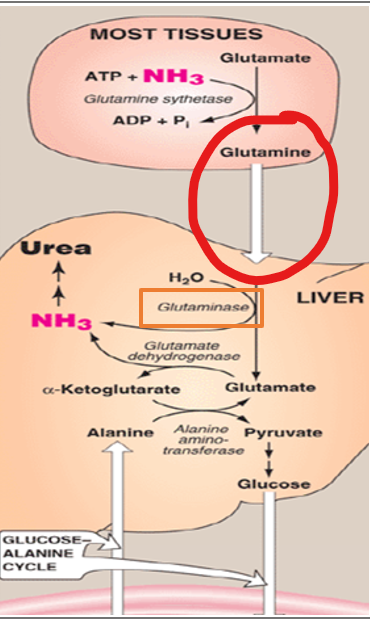
Once glutamine reaches the liver, it must be processed to release its nitrogen for entry into the urea cycle.
How is this accomplished?
by the enzyme glutaminase
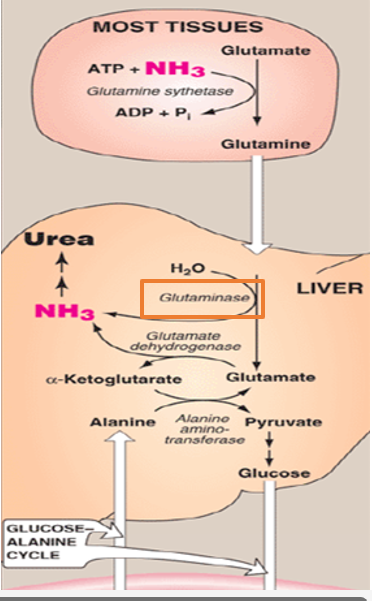
what does glutaminase do
catalyzes the hydrolytic deamination of glutamine to produce glutamate and free ammonia
Once glutamine reaches the liver, it must be processed to release its nitrogen for entry into the urea cycle. This is accomplished by the enzyme glutaminase, which catalyzes the hydrolytic deamination of glutamine to produce glutamate and free ammonia
Where does this reaction occur
within the mitochondra of heptocytes- the same compartment in which the urea cycle begins
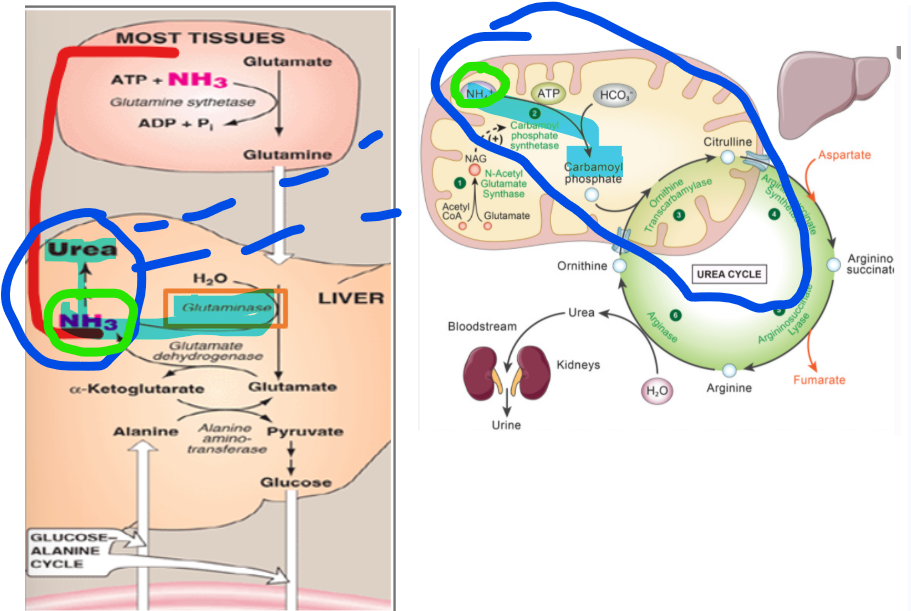
Once glutamine reaches the liver, it must be processed to release its nitrogen for entry into the urea cycle. This is accomplished by the enzyme glutaminase, which catalyzes the hydrolytic deamination of glutamine to produce glutamate and free ammonia.
Why is it important that this reaction occurs within the mitochondria of hepatocytes and being in the same compartment in which the urea cycle begins
it ensures that the ammonia released from glutamine is immediately available for incorporation into carbamoyl phosphate by carbamoyl phosphate synthetase I (CPS1), thereby minimizing the risk of ammonia diffusion into the bloodstream
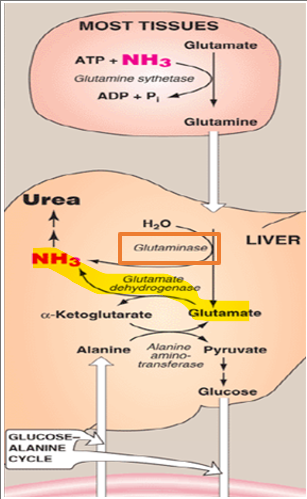
Once glutamine reaches the liver, it must be processed to release its nitrogen for entry into the urea cycle. This is accomplished by the enzyme glutaminase, which catalyzes the hydrolytic deamination of glutamine to produce glutamate and free ammonia.
What can the glutamate produced in this reaction do
it can undergo a second deamination step via GDH, generating an additional molecule of ammonia and regenerating alpha-ketoglutarate
The glutamate produced in this reaction can then undergo a second deamination step via glutamate dehydrogenase, generating an additional molecule of ammonia and regenerating alpha-ketoglutarate
What results from this
each molecule of glutamine effectively deliver 2 nitrogen atoms to the urea cycle
The glutamate produced in this reaction can then undergo a second deamination step via glutamate dehydrogenase, generating an additional molecule of ammonia and regenerating alpha-ketoglutarate. '
As a result, each molecule of glutamine effectively delivers two nitrogen atoms to the urea cycle
Where do each come from
amide group
amino group of glutamate
While glutamate dehydrogenase and glutaminase represent the primary mechanisms for deamination, other enzymes also contribute to nitrogen metabolism by processing specific amino acids. One such enzyme is…
asparaginase
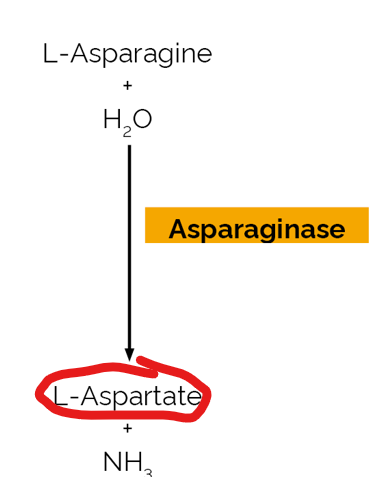
what does asparaginase do
it catalyzes the conversion of asparagine to aspartate while releasing ammonia
Aspartate serves as a
direct nitrogen donor, providing one of the two nitrgoen atoms that are ultimately incoprorated into urea
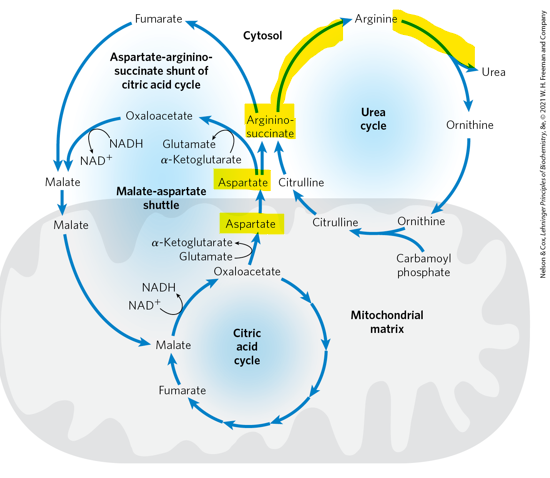
What represents a direct metabolic link between the urea cycle and the citric acid cycle
aspartate-arginiosuccinate shunt
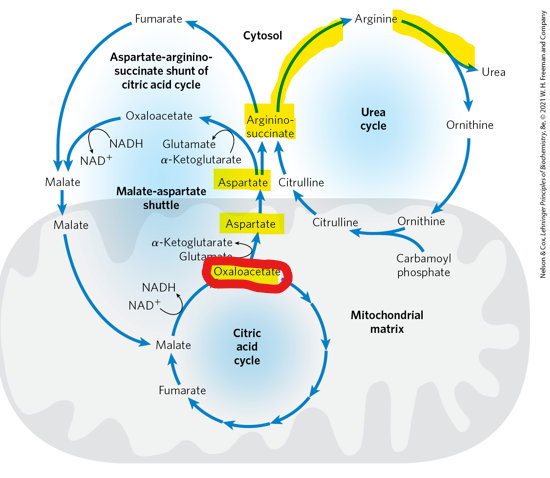
What TCA cycle intermediate does the aspartate–argininosuccinate shunt begin with
oxaloacetate
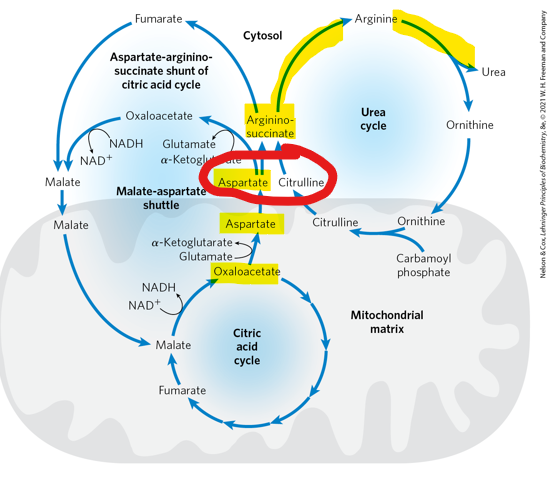
When aspartate enters the urea cycle, what does it combine with to form argininosuccinate
citrulline
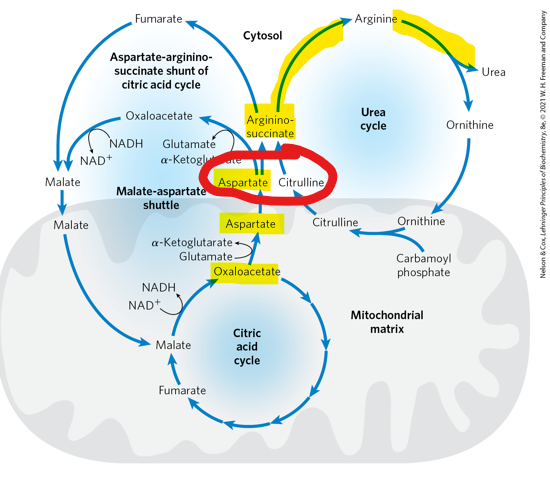
When aspartate enters the urea cycle cycle and combines with citrulline to form argininosuccinate- what does it contribute
one of the nitrogen atyoms that will be excreted as urea
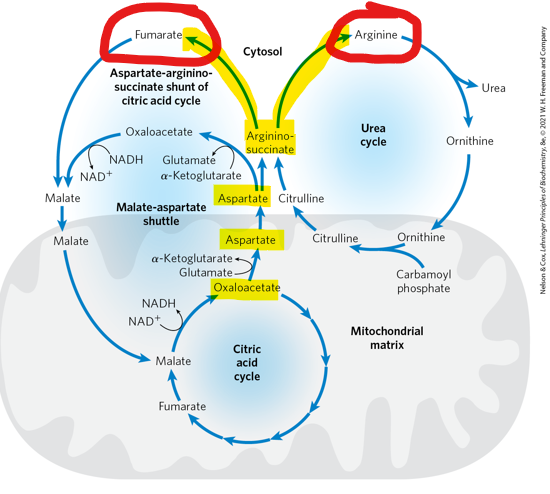
When argininosuccinate is subsequently cleaved, what does it produce
arginine and fumarate
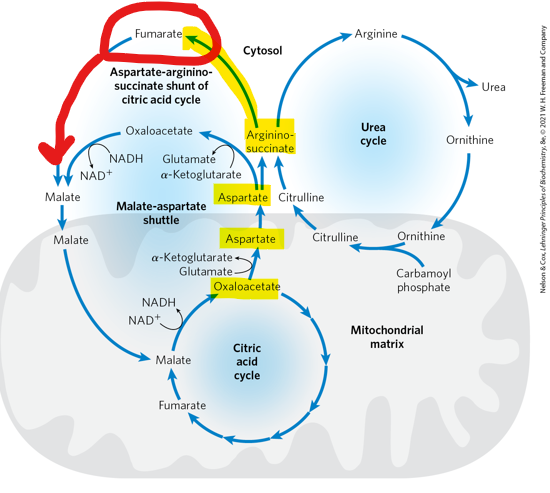
When argininosuccinate is subsequently cleaved, what happens to the fumarate
it is converted to malate tand then to oxaloacetate to re enter the TCA cycle - creating a continuous exchange f intermediates between the two cycles, and allowing them to fucntion in a coordinated manner
The integration of the urea cycle and the citric acid cycle through the aspartate–argininosuccinate shunt gives rise to what concept
the Krebs Bicycle
What does the Krebs Bicycle reflect
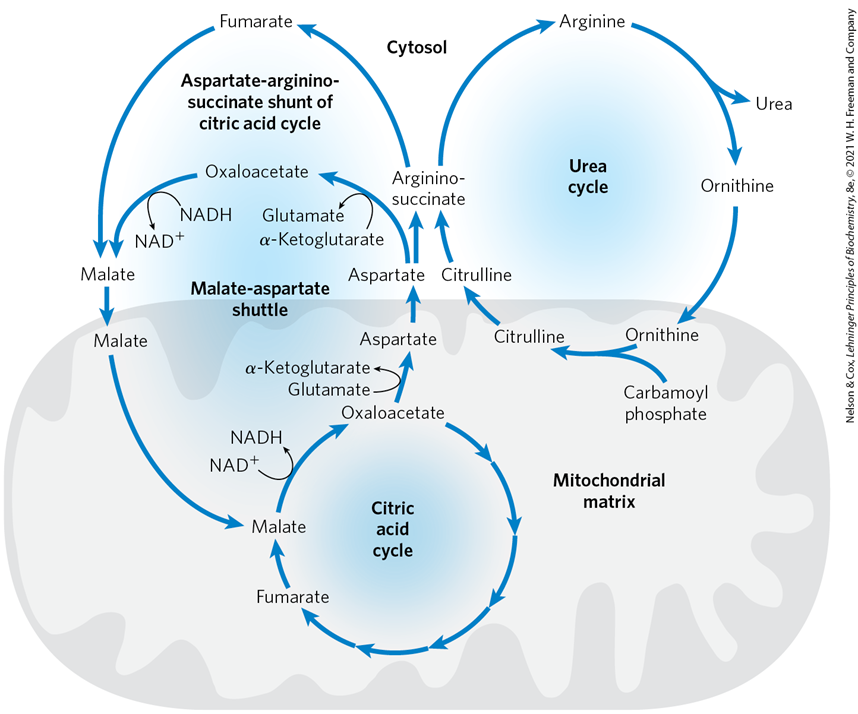
the cyclical and interconnected nature of these metabolic processes, in which intermediates are continuously exchanged to support both energy production and nitrogen excretion
Through the linkage of the Krebs cycle, the breakdown of amino acids contributes not only to the removal of nitrogen but also to the …..
replenishment of TCA cycle intermediates
what is the relenishment of TCA cycle intermediates called
anaplerosis
At the same time, what intermediate does the TCA cycle provide that is necessary for the continuation of the urea cycle
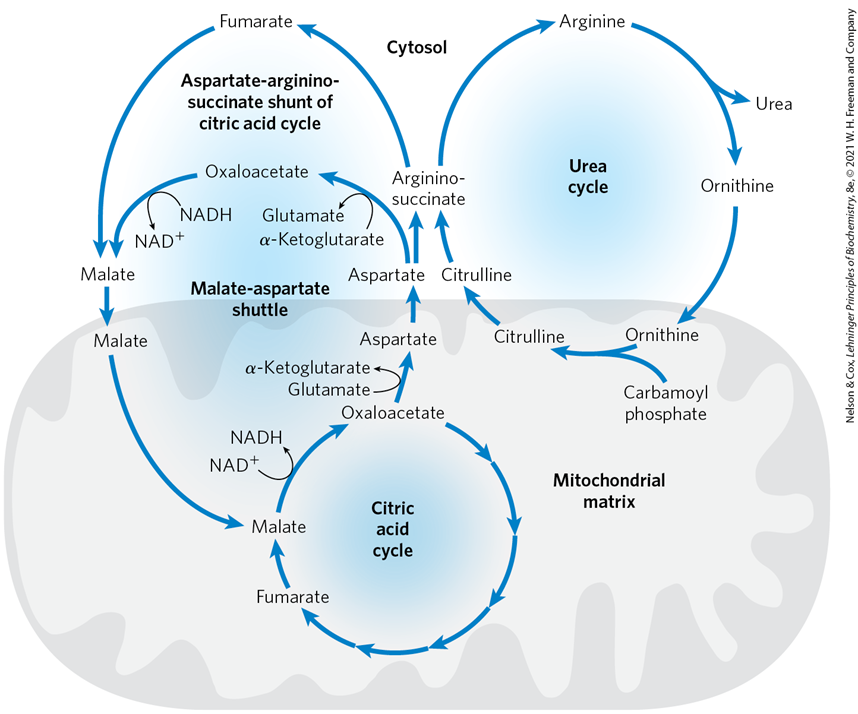
oxaloacetate
What are other deamination routes aside from the glutamate dehydrogenase route?
glutamine pathway
Aspaaragine to Aspartate Shunt