Chemistry AS (09/03/26)
1/33
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is the mole calculation for gases?
What is the formula for density?
PV = nRT
P= pressure (pa), V= volume (m³), R= gas constant, T= temp (K)
density=mass/volume
What are the mole formulas for solids and liquids?
Moles=mass/mr, concentration=moles/volume(dm³)
Give the formulas for percentage yield and atom economy:
%yield= actual yield/theoretical yield x 100, atom economy= moles of useful products/mass of reactants x100
What is electronegativity?
relative tendency of an atom in a covalent bond in a molecule to attract electrons in a covalent bond to itself
How to work out calorimetry?
Q=mC∆T, use the mass of the solution to find energy change, find the number of moles, ∆H=q/no of moles
What is Hess’s Law?
Total enthalpy change is independent of route taken.
What is Le Chatelier’s principle?
If an external condition is changed, the equilibrium will shift to oppose the change.
Catalysts have no effect of the equilibrium, speeds rate that its achieved.
What is the equilibrium constant?
Kc= products^m/reactants^n, only temp has effect on Kc, not pressure or catalysts, smaller=favours reactants
Give oxidation numbers
Group 1 metals = +1, Group 2 metals = +2, Al = +3, H = +1 (except in metal hydrides = –1 eg NaH), F = -1, Cl, Br, I = –1 except in compounds with oxygen and fluorine, O = -2 except in peroxides (H2O2 ) and with fluorine where it is –1
Give the reactions of oxygen and water with Group 2 metals:
They all burn in oxygen, magnesium forms MgO (white powder), burns with bright white flame.
Magnesium with steam: MgO+ H2 (bright white flame), with warm water: Mg(OH)2 + H2 (no flame, slower), the other metals can react with cold water forming hydroxides, they fizz, dissolve and heat up (increases down group)
Group 2: metal hydroxides:
Mg(OH2)2 is mostly insoluable, slightly alkaline, can be used to treat acid reflux as suspension in milk of magnesia; Ca(OH2)2 is partially soluable, used to neutralize acidic soil, white precipitate; Ba(OH2)2 is very soluable, very strong alkaline; solubility increases down group.
Group 2: metal sulfate:
Less soluble down the group; Ba(SO4) toxic, insoluble so used as barium meal to absorb x-rays when imaging gut, reacts very slowly with sulfuric acid forming a Ba(SO4) layer preventing further reaction.
Group 2: how to test for sulfates?
Barium chloride (BaCl2) with HCl added to solution, forms white precipitate. HCl prevents false result formed with barium carbonate (also white)
Halogens: what is the trend of MP and BP?
What is the trend of electronegativity?
MP and BP increase down the group, more electrons leading to greater VDW, more energy to break intermolecular forces; electronegativity decreases down group
Halogens: what are the tests for the halides?
Silver nitrate + nitric acid (prevents carbonates forming), Fluorides produce no precipitate, Chlorides produce a white precipitate, Bromides produce a cream precipitate, Iodides produce a pale yellow precipitate, treat with ammonia if colours are similar.
Homolytic vs heterolytic fission
Homolytic fission is where a covalent bond breaks, and both atoms each get one electron each, forms free radicals; heterolytic fission is where one atom gets both electrons, forms an ion
What are the types of structural isomerism?
Chain: same mol formula, different carbon skeleton structure; Position: same mol formula, different position of func group; Functional group: same mol formula, different functional group
Stereoisomerism
If the biggest priority groups are on the same or opposite sides of a double bond
Alkanes: How to refine crude oil?
Fractional distillation: fraction is preheated, then passed into column, temperature decreases upwards, separation depends on boiling point, larger molecule leads to greater VDW forces so higher BP
Vacuum distillation: heavy residue from column distilled under vacuum, lower pressure = lower BP
Alkanes: What is cracking?
Converting larger hydrocarbons into smaller ones by breaking C-C bonds, smaller hydrocarbons more in demand
What are the two types of cracking?
Thermal cracking: high temp and pressure, produces mostly alkenes; Catalytic cracking: moderate pressure, high temp with zeolite catalyst
Alkanes: what are the products of complete and incomplete combustion?
Complete: H20 + CO2; incomplete: CO and/or C, with limited oxygen, less energy/mol
What pollutants are formed from combustion?
SO2: sulfur containing compounds from fractions produce SO2 when burned, leads to acid rain, can be removed from waste gas with flue gas desulfurisation; Nitrogen oxides: car engine has high enough temp and spark which breaks N2 bond, forms oxides, toxic and acid rain; Soot, unburnt hydrocarbons and CO
What do catalytic converters do?
Remove CO, NO2 and unburnt hydrocarbon from exhaust gas, honeycomb (high SA) ceramic coated with layer of catalysed metals (palladium, rhodium, platinum)
How are halogenoalkanes formed?
Free radical substitution, in UV light, alkanes react with halogens. Free radical: reactive species with an unpaired electron
Electrophilic addition with alkenes
What are the steps of free radical substitution to form chloromethane?
Initiation: UV light breaks the Cl2 bond, with homolytic fission forming 2Cl’
Propagation: CH4 + Cl’ → HCl + ‘CH3 (remove H, forms methyl free rad); ‘CH3 + Cl2 → CH3Cl + Cl’ (methyl free rad reacts with Cl2, forms main product with another Cl’ - leads to chain reaction)
Termination: left over free radicals react, terminates chain ‘CH3 + Cl’ → CH3Cl ; ‘CH3 + ‘CH3 → CH3CH3
What are the reactions of halogenoalkanes in aqueous conditions?
Nucleophilic substitution:
With aqueous hydroxide ions: halogenoalkane → alcohol, K/NaOH, warm aqueous
With cyanide ions: halogenoalkane → nitrile, KCN, dissolved in aqueous ethanol, heating under reflux
With ammonia: halogenoalkane → amine, NH3 with ethanol, heat under pressure.
Requires electron donator (nucleophile)
What is the reaction of halogenoalkanes in ethanol?
Elimination reaction: halogenoalkane → alkene, K/NaOH, ethanol, heat under reflux.
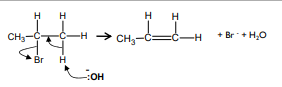
How to test for alkenes?
What reaction takes place?
Bromine water decolourises in contact with double bond
Electrophilic addition, requires electron acceptor (electrophile)
What are the oxidation reactions for alcohols?
(Partial) Primary alcohol → aldehyde (heat, limited potassium dichromate K2Cr2O, distil to remove)
(Full) Primary alcohol → carboxylic acid (heat under reflux, excess agent)
(Full) Secondary alcohol → ketone (heat under reflux, excess agent)
Tests and observations after oxidation of alcohol
Orange solution turns green
Sodium carbonate to test for carboxylic acid (forms CO2- effervescence)
Tollens reagent- aqueous ammonia + silver nitrate, aldehyde makes silver mirror)
Fehling’s reagent- blue Cu2+ solution → red precipitate with aldehyde)
Ethanol formation methods
Fermentation: glucose → ethanol + CO2, requires yeast, anaerobic (no air)
Hydration of ethene: high temp and pressure, strong acid catalyst
How does infrared spectroscopy work?
Certain groups in a molecule absorb infra-red radiation at characteristic frequencies
Above 1500cm^-1 = func group identification
Below 1500cm^-1 = fingerprinting region (unique to each compound, match against database)
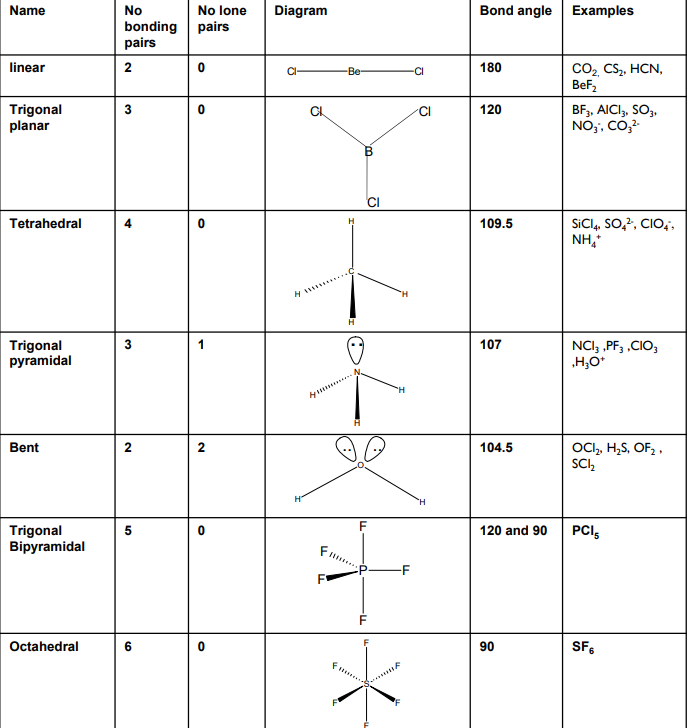
Bonding shapes