Equilibrium and Le Chatlier
1/16
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is equilibrium?
Reactions that occur in both directions (reversible reactions)
At equilibrium, do the forward and reverse reactions occur at the same rate?
Yes
At equilibrium, do the forward and reverse reactions produce equal amounts?
No, some amounts of reactants and products must be present, but generally not equal amounts of each.
What is Le Chatlier’s principle?
When a system at equilibrium is subjected to a stress, the equilibrium will shift in a direction that tends to reduce that stress
Describe the changes in concentration when something is added or removed at equilibrium.
When something is added, shift away from that thing
When something is removed, shift towards that thing
Only amounts of (aq) and (g) count; (s) and (l) have NO EFFECT
Adding a substance not in the reaction will have NO EFFECT
Describe the effect of a change in volume or pressure when a reaction is at equilibrium.
If volume is reduced (or pressure is increased), shift to side with fewer gas particles
If volume is increased (or pressure is decreased), shift to side with more gas particles
Describe the effect of a change in temperature when a reaction is at equilibrium.
If temperature is increased, shift to side without heat
If temperature is decreased, shift to side with heat
Describe the effect of other changes (not temperature, pressure, or volume) when a reaction is at equilibrium.
Other changes that may happen to the reaction at equilibrium, will NOT AFFECT it (for example, catalysts)
What is the equilibrium constant?
K (or Keq)
What does it mean if Keq is less than one?
The reactants are favored over the products
What does it mean if Keq is greater than one?
The products are favored over the reactants
What does it mean if Keq is equal to one?
Reactants and products are both present in significant concentrations at equilibrium
What are the types of stress a reaction can experience?
Changes in concentration
Catalyst
Particle Size
Changes in pressure
Changes in temperature
What does the graph of a reaction at equilibrium look like?
(could also be shown as two separate lines, one being the forward reaction and the other being the reverse reaction, converging into one at the equilibrium state)
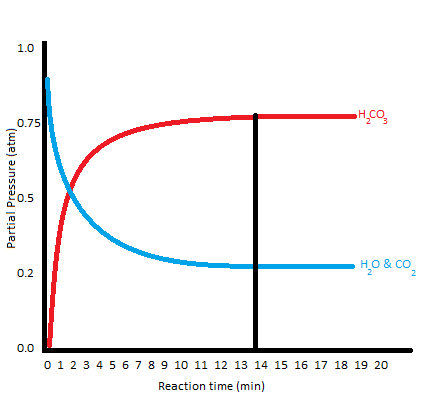
What does K measure?
The ratio of product concentrations in molarity (M), or pressures (in atm) to reactant concentrations or pressures
What is the formula for equilibrium expression in moles or liters?
In a reaction where aA+bB—>cC+dD
Keq= [C]^c[D]^d/[A]^a[B]^b
What is the formula for equilibrium expression in pressure?
In a reaction where aA+bB—>cC+dD
Kp=(P_D)^d(P_C)^c/(P_A)^a(P_B)^b