1b - Proteins, enzymes, nucleic acids
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
What are amino acids made up of?
carbon, hydrogen, oxygen, nitrogen
(also contain sulphur and phosphorus)
What are proteins made up of?
amino acids
What is the general structure of an amino acid?
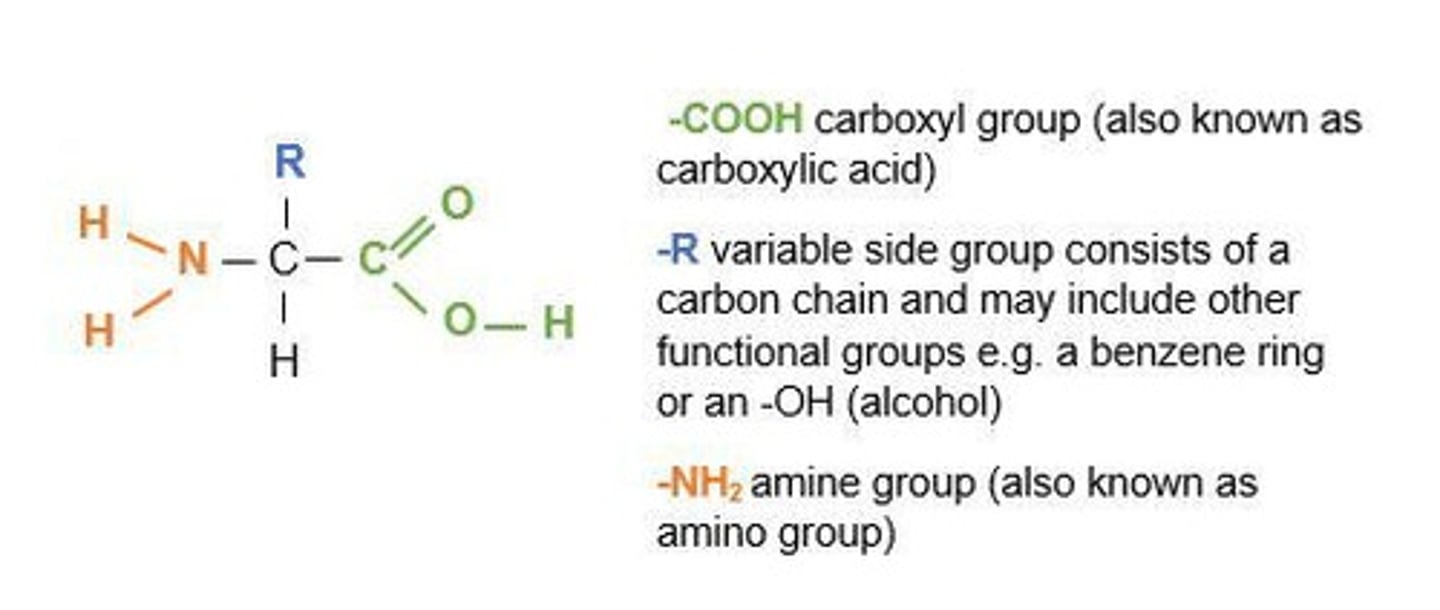
What's the name of the group containing amino acids?
Amine group (NH2)
What's the name of the carboxylic acid group?
carboxyl group (COOH)
what's the other name for proteins?
polypeptides
how many amino acids are there that occur naturally in proteins?
20
what two groups do amino acids have?
amine group(NH2) and carboxylic group(COOH)
what chemical test is used to detect the presence of peptide bonds?
the biuret test (add biuret solution)
write out a method to test for proteins
1 - place a sample of the solution to be tested in a test tube and add an equal volume of sodium hydroxide solution at room temp.
2 - add a few drops of very dilute (0.05%) copper (II) sulfate solution and mix gently
3 - a purple colouration indicates the presence of peptide bonds and hence a protein. if not protein is present, the solution remains blue
what are some safety precautions for the protein test?
- wear goggles
- wear a lab coat
- tie your hair up
- be cautious with glass
- be cautious with spilling and clean up carefully
what equipment is needed for a test for proteins?
- test tube
- pipette
- measuring cylinder
- solutions (sodium hydroxide, copper (II))
whats a dipeptide?
two amino acids joined together by a single peptide bond
how are dipeptides formed?
condensation reaction between two amino acids with the loss of a water molecule forming a peptide bond
how are polypeptides formed?
through the repeated condensation of many amino acids with the loss of water molecules , forming a peptide bond
what is formed between the condensation of two amino acids?
a peptide bond
what reaction can split a dipeptide back into two amino acids?
hydrolysis reaction
What is the sequence (order) of amino acids called in a polypeptide chain?
the primary structure
What does a condensation reaction look like?
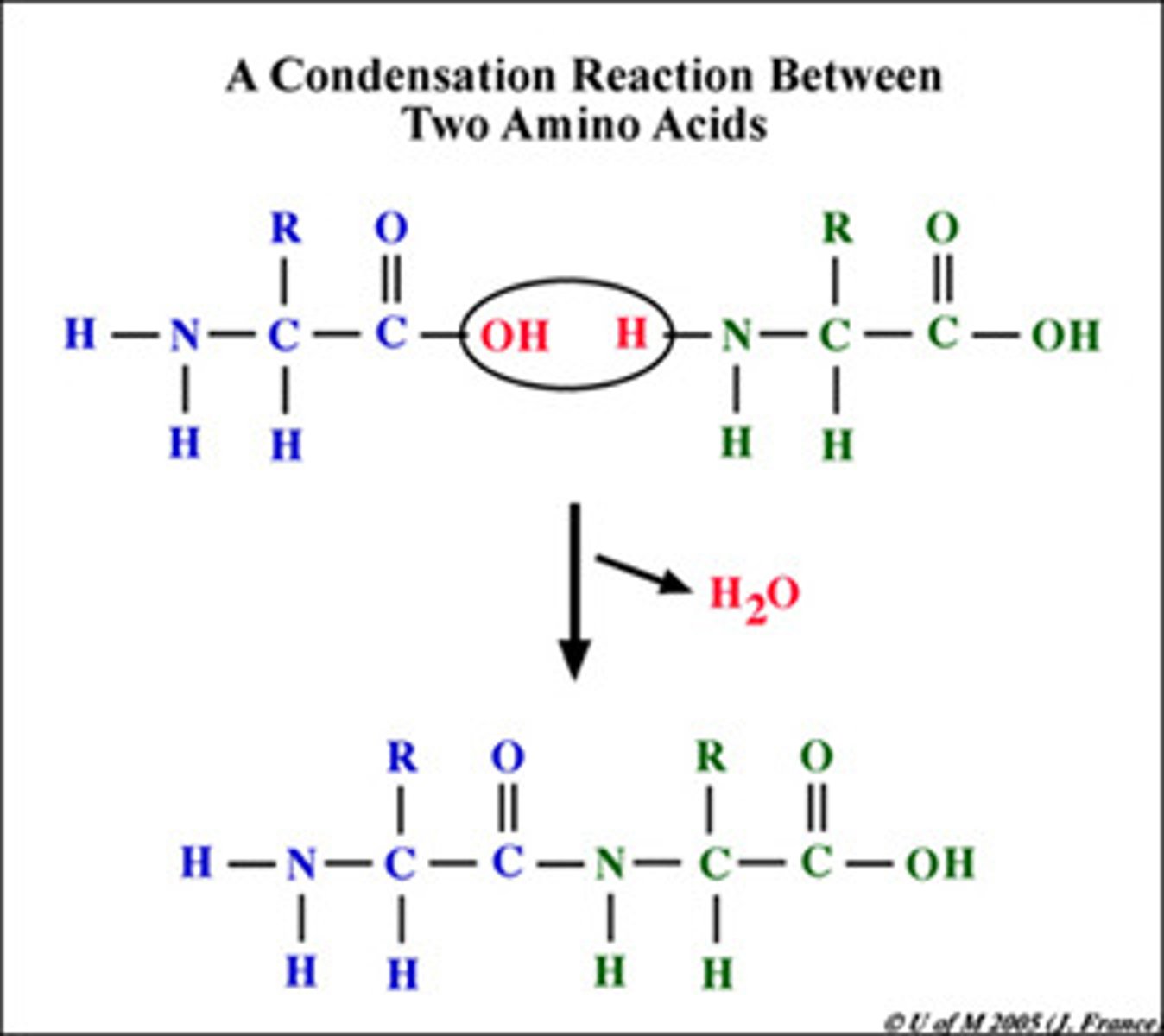
what's the primary structure protein?
- the order (primary sequence) of the amino acids joined by peptide bonds
What's the secondary structure of a protein?
- the specific 3D shape that the protein takes due to hydrogen bonding between the amino acids
- alpha helix - hydrogen bonds form between every 4th amino acid because the O is negative and the H is positive meaning they are attracted
- beta pleated sheet - 2or more segments of a polypeptide chain line up next to each other and form hydrogen bonds between the amino acids
- can have both types of structures in different regions of the protein depending on the primary structure
when does a polypeptide become a protein?
after its folded into its correct shape
What's the tertiary structure of a protein?
- A unique, finished 3D shape made of folding of the secondary structure, it's held in place by ionic, hydrogen and disulphide bridges.
- Ionic and disulphide bonds form between the R groups
- disulphide bridges only between cysteine amino acids
- ionic bonds btween charged R groups
- hydrogen bonds between R groups
- Weak hydrophobic interactions between non-polar R groups
What's the quaternary structure of a protein?
- made of 2 or more tertiary proteins joined together by the same bonding in tertiary e.g. haemoglobin consists of 4 polypeptide chains
what are globular proteins + features?
- molecules which have a chemical function in living organisms and take part in specific reactions
- compact, spherical
- soluble in water allowing them to take part in metabollic reactions
- examples = enzymes, hormones, antibodies and transport proteins
what are fibrous proteins + features?
- insoluble protein molecules which have a structural function
in living organisms e.g. may give strength or elasticity to tissue
- less folded than globular appearing like long ropes or filaments
- examples = keratin in hair, collagen in cartilage and bone, elastin in skin
enzyme structure
- proteins - a chain of amino acids joines by peptide bonds
- globular tertiary structure held together by hydrogen bonds, ionic bonds, disulphide bridges and hydrophobic interactions
- the amino acids (and their R groups) that are present determine which bonds form and where, giving
an enzyme a specific active site
- substrate bonds to active site
what's a biological catalyst?
- Biological catalysts are produced by
organisms that speed up a chemical
reaction without being used up in the
reaction
- all enzymes are biological catalysts
what's activation energy?
- The energy needed for a reaction to
occur by causing bonds to break or form by increasing the number of collisions.
- all enzymes lower activtion energy
how do enzymes lower activation energy?
If 2 substrate molecules need to be joined:
Attaching to an enzyme holds them close together reducing any repulsion - therefore bond more easily.
If the enzyme is catalysing a break down
reaction:
The substrate fitting into the active site puts a strain on the bonds in the substrate so it breaks apart more easily.
enzyme action process
1. Random movement causes the enzyme and substrate to collide, and the substrate enters the active site
2. Enzyme-substrate complex forms.
Charged groups attract, distorting the
substrate and aiding bond breakage
(catabolic) or formation (anabolic)
3. Products are released from the
active site leaving the enzyme unchanged and ready to accept another substrate molecule.
lock and key theory
- Only the correctly sized key
(substrate) fits into the key hole
(active site) of the lock (enzyme).
- The enzyme is rigid
induced fit theory
- The active site can be partially flexible
- A substrate can bind to the active site even if it isn't a perfect fit
- The active site changes to fit the substrate
how does temperature affect enzyme action?
- As the temperature increases, so does the rate of reaction due to the particles having more kinetic energy.
- This means more successful collisions occur between the particles, forming more enzyme- substrate complexes.
- At temperatures around 60°C denaturing occurs- a permanent change in the enzymes active site shape - causing the rate of reaction to fall to 0.
How does pH affect enzyme action?
- Changes in pH also alter an enzyme's shape.
- The optimum pH for an enzyme depends on where it normally works
- A change from the optimum pH alters the charges on the amino acids that make up the active site which can cause bonds in the active site to break.
- As a result- the active site changes shape and no enzyme-substrate complexes can be formed.
How does enzyme concentration affect enzyme action?
- Increasing the enzyme
concentration increases the rate
of reaction because there are
now more active sites available to
the substrates so more reactions
can occur so more product can be
produced.
- If the substrate is not in excess
the graph will level off instead of
continue to increase- as substrate
concentration is limiting the rate
of reaction.
How does substrate concentration affect enzyme action?
1. Increasing the substrate increases the rate of reaction because there will be more successful collisions between substrate and enzyme
2. At high substrate concentrations the rate cannot increase any further, even though there is more substrate,
excess substrate is unable to be used:
at this point the enzymes are saturated (all active sites are occupied). Known as the saturation point
how is rate measured?
- Rate can be measured as the amount of substrate broken down or amount of product produced in a set time
Rate is initially high because: lots of substrate rate slows over time as amount of substrate decreases
how to calculate initial rate of reaction
Calculating the initial rate of reaction by drawing a tangent (straight line)
Gradient = change in y axis / change in x axis
what's an inhibitor?
substances that prevent enzymes
reaching there maximum working rate by interfering with the function of the active site.
what are the two main types of inhibitors?
Competitive inhibitors: bind directly to the active site of the enzyme
Non-competitive inhibitors: bind to the enzyme at a position other than the active site.
competitive inhibitors
- Competitive inhibitors have a similar molecular shape to the substrate.
- The inhibitor occupies the active site (not permanently), meaning the substrate must compete for it. The reduces the number of enzyme-substrate complexes formed
- Increasing the substrate or enzyme
concentration reduces the effect of the inhibitor.
non-competitive inhibitors
- Non-competitive inhibitors do not resemble the substrate
shape and bind to the enzyme elsewhere altering the active site
shape. The place where they bind is called the allosteric site
- No enzyme- substrate complexes can be formed- rate of reaction falls quickly as the active site is changes irreversibly. Increasing substrate or enzyme concentration does not over come this.
method for rp 1 - Investigation into the effect of a named variable on the rate of an enzyme-controlled reaction.
1. Make two control samples: Take two flat bottomed tubes. Add 5cm3 of milk suspension to each tube. Add 5cm3 of distilled water to one tube- this control will indicate the absence of enzyme activity. Add 5cm3 of hydrochloric acid to the other- this control indicates the colour of a completely hydrolysed sample.
2. Take three test tubes and measure 5cm3 milk into each. Place in water bath at 10°C for 5 minutes to equilibrate.
3. Add 5cm3 trypsin to each test tube simultaneously and start the timer immediately.
4. Record how long it takes for the milk samples to completely hydrolyse and become colourless.
5. Repeat steps 2-3 at temperatures of 20°C, 30°C, 40°C and 50°C.
6. Find the mean time for the milk to be hydrolysed at each temperature and use this to work out the rate of reaction
gene definition
a sequence of bases on a DNA
molecule coding for a sequence of amino acids in a polypeptide chain.
Genome definition
all the genes in a cell or an organism
structure of a nucleotide
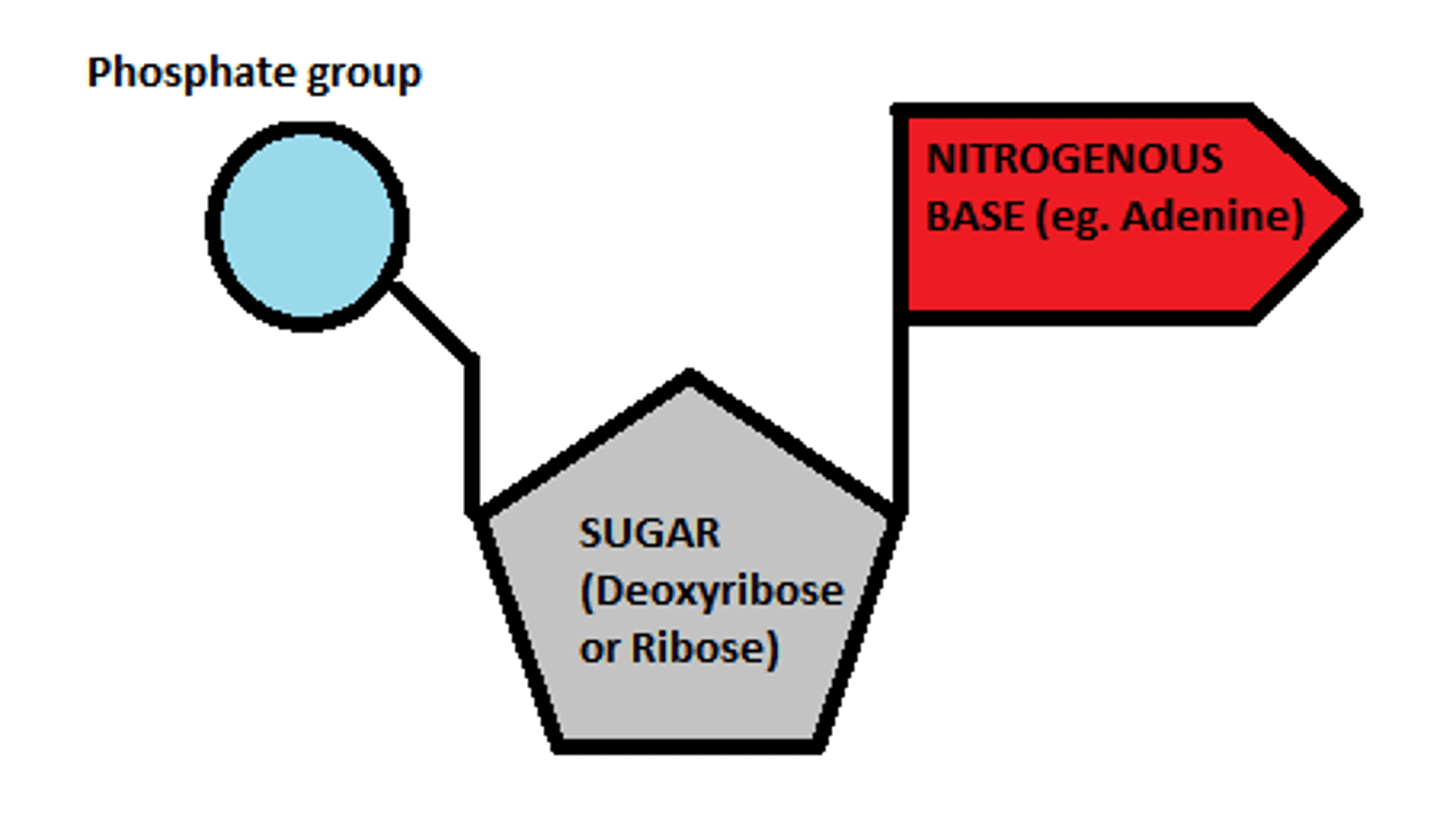
what bond joins the phosphate and sugar together in a nucleotide
phosphodiester bond - forms
the sugar phosphate backbone
what bond joins the sugar and base together
glycosidic bond
complementary base pairing
- Adenine joins to Thymine (2 hydrogen bonds)
- Cytosine joins to Guanine (3 hydrogen bonds)
what does DNA and RNA stand for?
deoxyribonucleic acid
ribonucleic acid
what is DNA's function?
stores genetic information
who discovered the structure of DNA?
Watson and Crick
who discovered that DNA follows a semi-conservative replication process?
meselson and stahl
what is RNA's function?
- transfers genetic information from DNA to the ribosomes
- can also be combined with proteins to create ribosomes
how are ribosomes formed?
formed from RNA and proteins
what are the components of DNA nucleotides?
- deoxyribose
- phosphate group
- a base (adenine, cytosine, guanine, thymine)
what are the components of RNA nucleotides?
- ribose
- phosphate group
- base (adenine, cytosine, guanine, uracil)
what bond is formed after a condensation reaction between two nucleotides?
phosphodiester bond
What is the structure of a DNA molecule?
- double helix with 2 polynucleotide chains held together by hydrogen bonds between specific complementary base pairs
RNA nucleotide structure
- single stranded nucleic acid
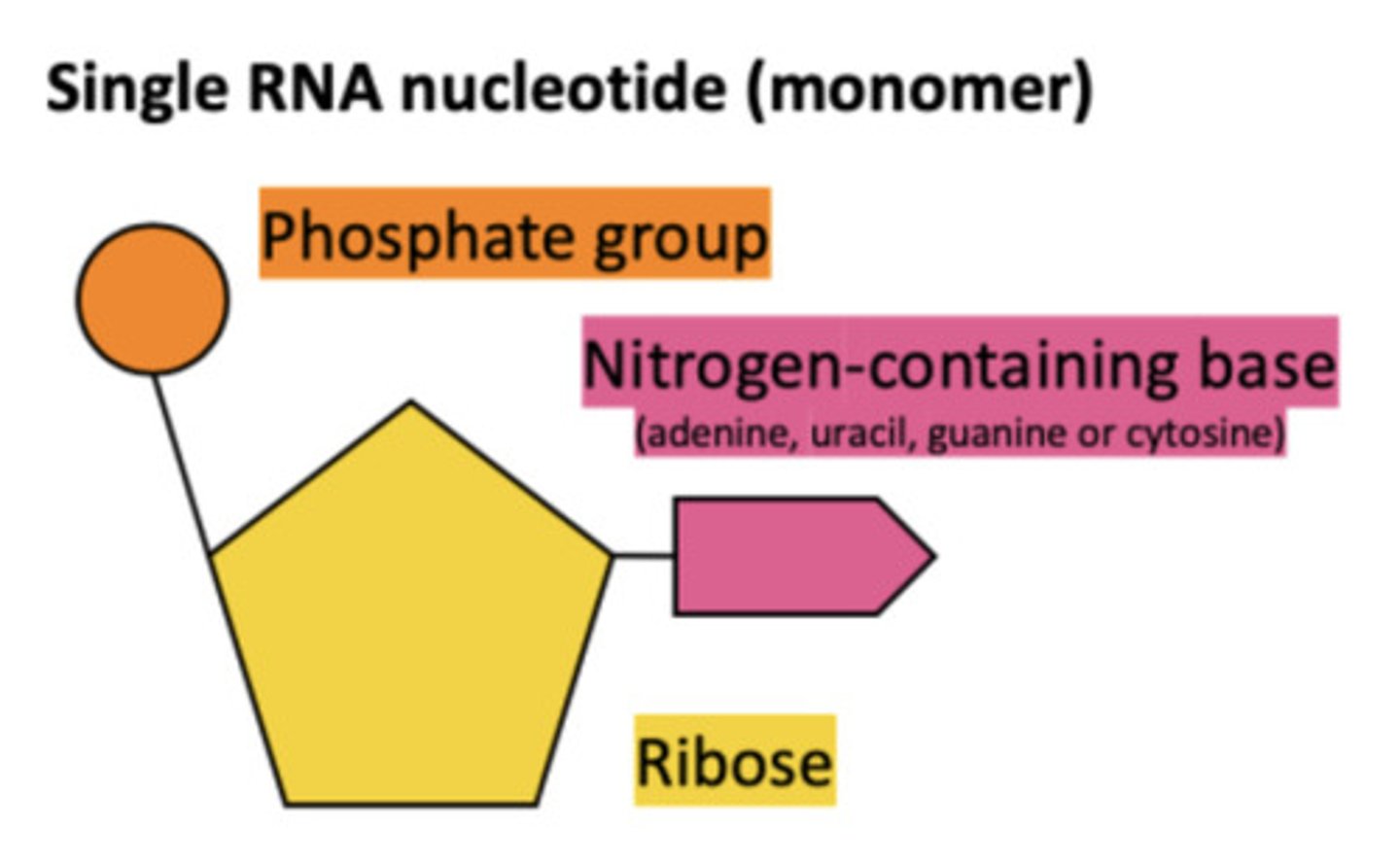
what are the types of RNA?
Messenger RNA (mRNA)
Ribosomal RNA (rRNA)
Transfer RNA (tRNA)
what does 3' prime end and 5' prime end mean?
3' prime end - end without phosphate sticking out
5' prime end - end with phosphate sticking out
why is DNA described as running in anti-parallel strands?
the strands run in opposite directions
differences between DNA and RNA?
- DNA has thymine base RNA has uracil base
- DNA uses deoxyribose sugar RNA uses ribose sugar
- DNA is much longer (whole genome)
RNA is much shorter (1 gene)
- DNA is double stranded RNA is single stranded
- DNA has hydrogen bonds (between bases) RNA has no hydrogen bonds
what is mRNAs function?
- messenger
- copy of a gene from DNA
- goes to ribosome to make proteins
what's the structure of tRNA?
- single stranded
- folds in on itself - clover leaf shape
- held in place by hydrogen bonds
- has an amino acid binding site to attach to amino acids
what's the function of tRNA?
- transfer RNA
- attaches to one of the amino acids and transfers it to the ribosome creating a polypeptide chain
what's the function of rRNA?
- ribosomal DNA
- combines with a protein to create ribosomes
when does a DNA molecule need to replicate?
- before a cell divides (mitosis or meiosis) DNA needs to be replicated
- this occurs during interphase
- essential in ensuring that the daughter cells contain the correct amount of genetic material
DNA replication process
1) DNA molecule unwinds
2) DNA helicase breaks hydrogen bonds and causes the 2 strands of DNA to separate
3) free nucleotides in the nucleoplasm are attracted to their complementary base pairs on the exposed strands (which act as templates)
4) the strong sugar phosphate back bone is re-joined by DNA polymerase (happens in condensation reactions, forming phosphodiester bonds). DNA polymerase catalyses the condensation reaction between the sugars and phosphates
5) forms 2 identical strands of DNA because the strands each contain half of the original material. It is called the semi-conservative method of replication
what are the models of DNA replication?
- conservative
- semi-conservative
- dispersive
what is the conservative model of DNA replication?
- one new strand made, one old one
what is the semi-conservative model of DNA replication?
- both strands contain old and new DNA strand
what is the dispersive model of DNA replication?
- cut at various points and reattach
- contain old and new DNA
what experiment proved the semi-conservative model was the most accurate?
the Meselson and Stahl experiment
What was the Meselson-Stahl experiment?
By separating DNA by density using
a centrifuge, they observed that each round of replication produced DNA molecules of intermediate density (one heavy and one light
strand), confirming that each new DNA double helix conserves one original strand and synthesizes a new one.
how did the Meselson-Stahl experiment prove that the semi-conservative method was the most accurate?
1st test - intermediate band formed meaning it contained heavy and light nitrogen. This disproved conservative method as their would have been a light and heavy band
2nd test - formed intermediate and light band which disproved dispersive as it would have been intermediate and thicker
what's a conjugated protein?
- a type of protein that has other chemical groups, such as carbohydrates, lipids, or metal ions, attached to it, distinguishing it from simple proteins which consist solely of amino acids
- such as glycoproteins