Orgo Exam 3 Mechanisms
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
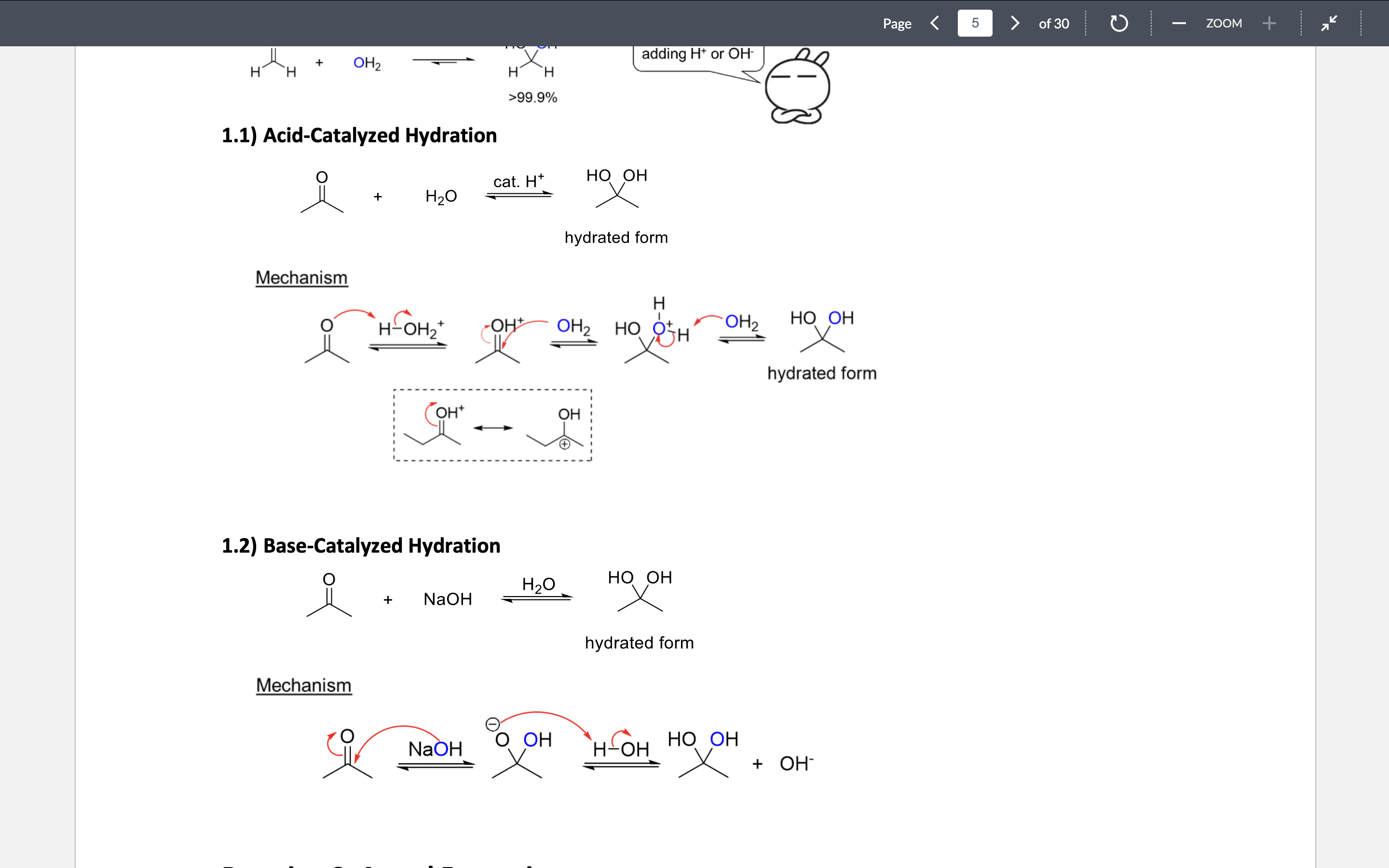
Hydration (basic & acidic)
Favor for ketone
Sped up by adding H+ or OH to the reaction
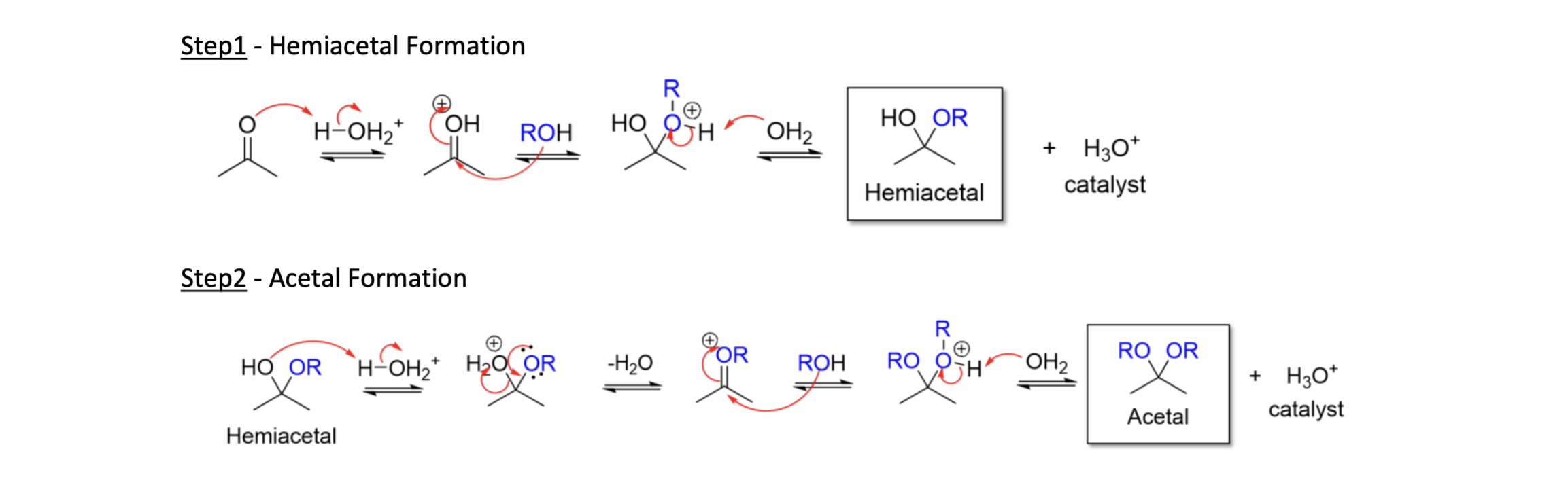
Acetal Formation
2 OH mol attack carbonyl
Hemiacetal intermed
TsOH & H2SO4 (expt = H3O+)
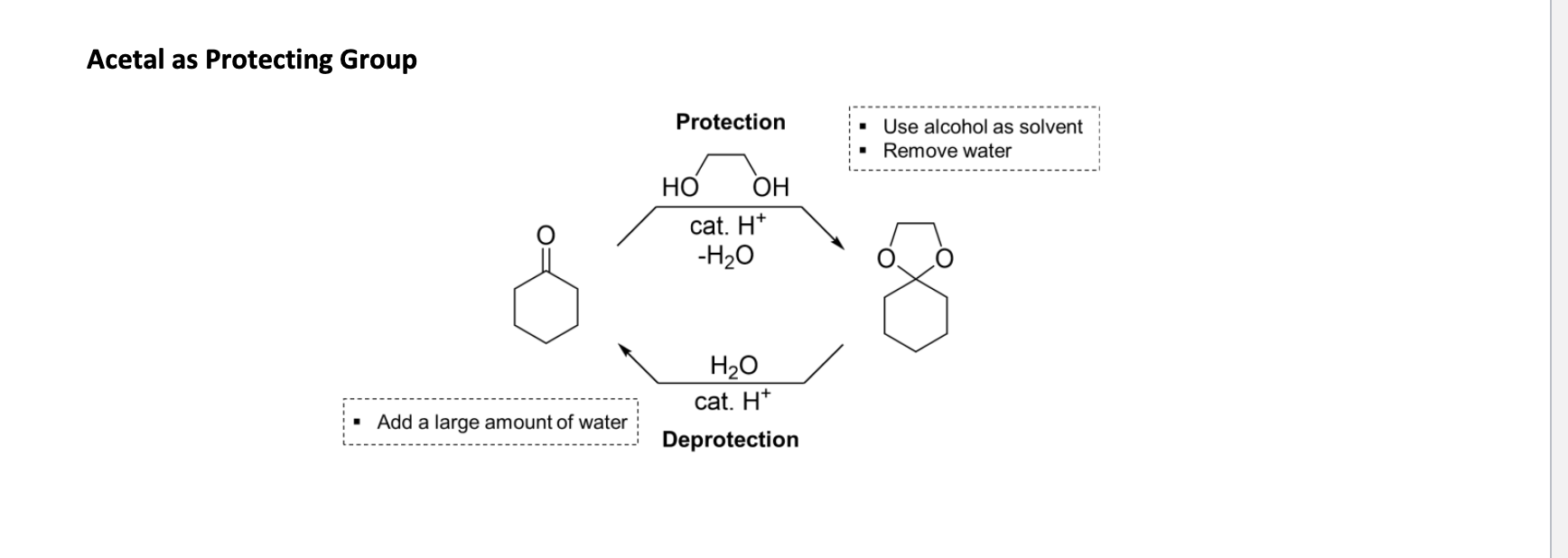
Acetal Formation + protecting group
Ald favors acetal
Eq favors reactant for ket
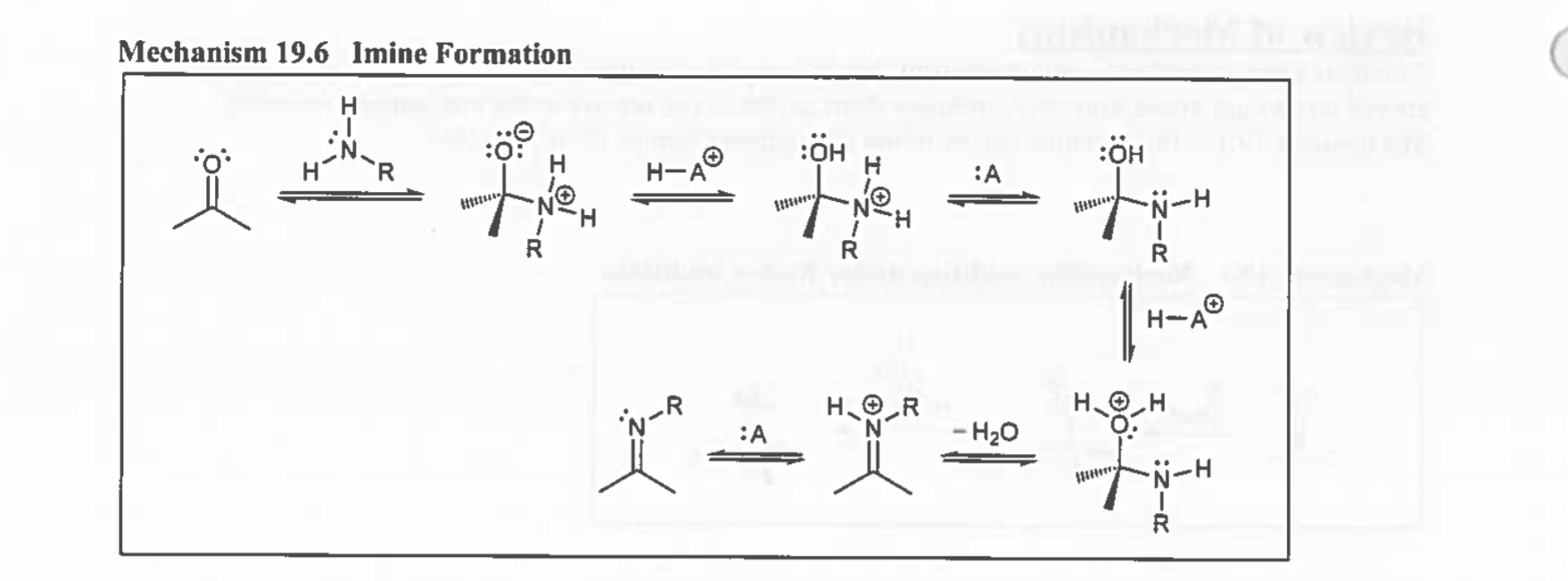
Imine Formation
C=N / cat.H+
Nitrile attacks H → N
Kicks H
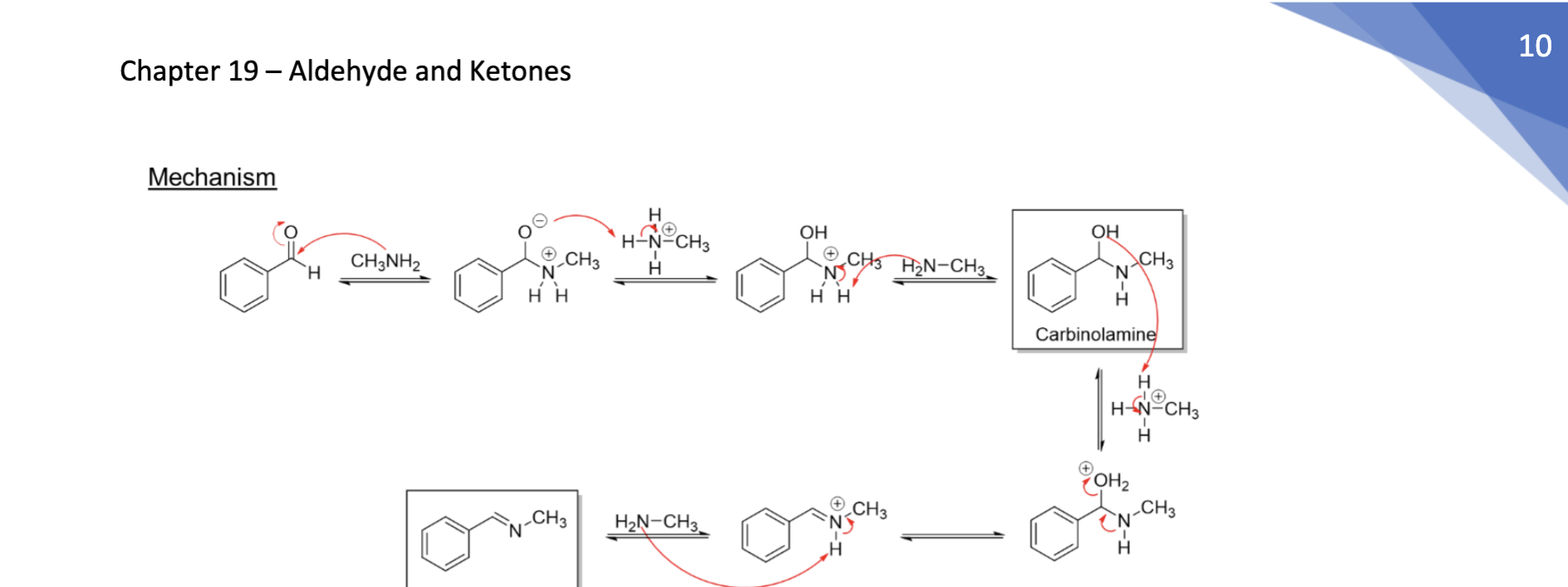
Enamine Formation
Nitrile attacks adj H → N → kicks + charge
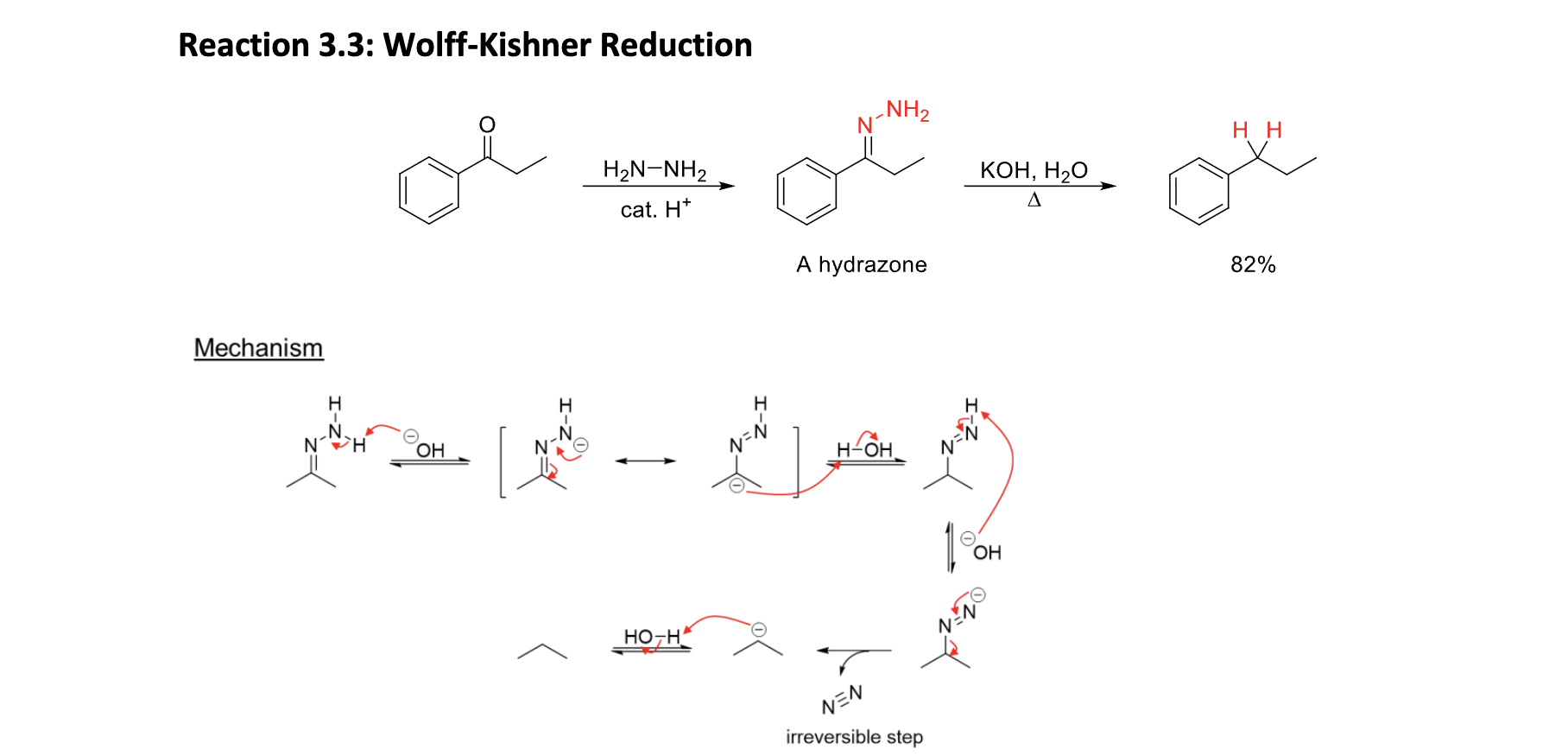
Wolf-Kishner Reduction
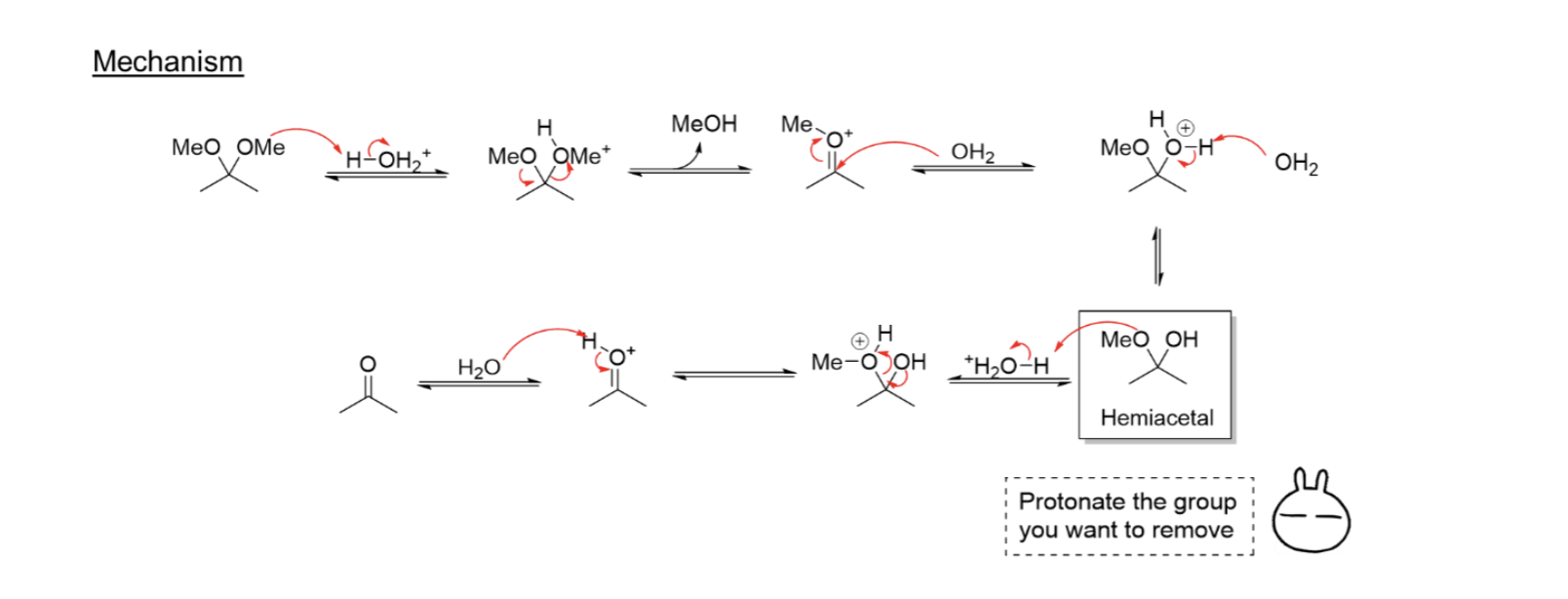
Acid Catalyzed Hydrolysis of Acetals
Acetal = Carbonyl + 2 ROH
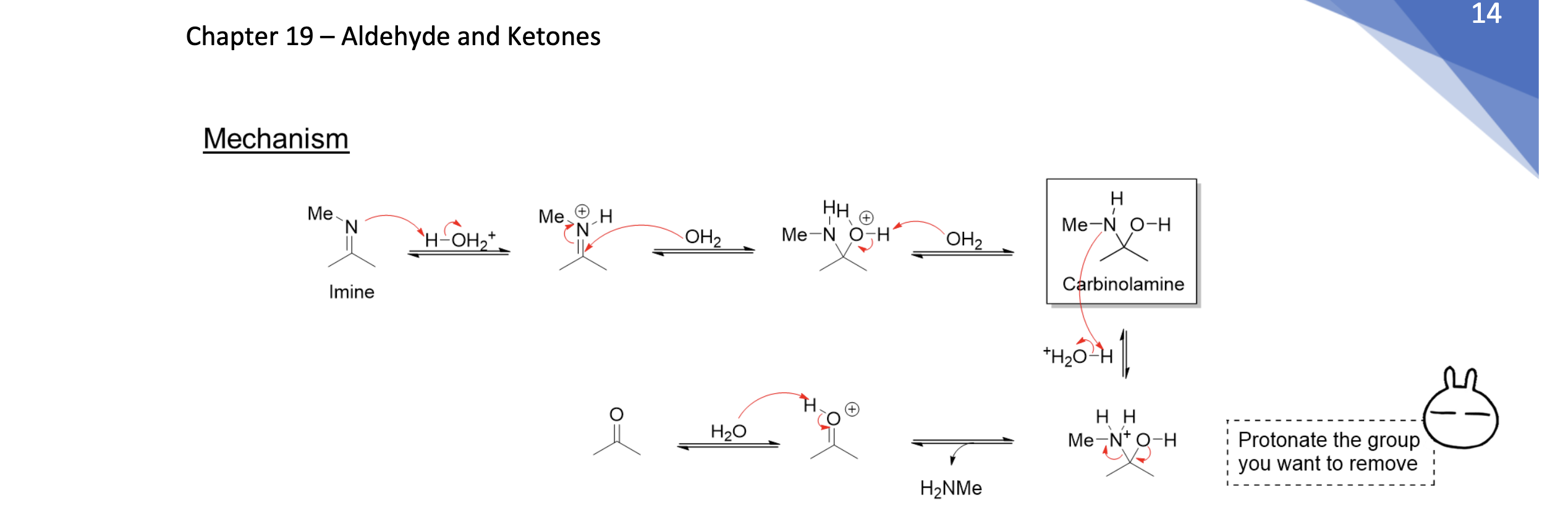
Acid Catalyzed Hydrolysis of Imines
Imine → H2O/Cat.H+ → Carbonyl + 2 RNH2
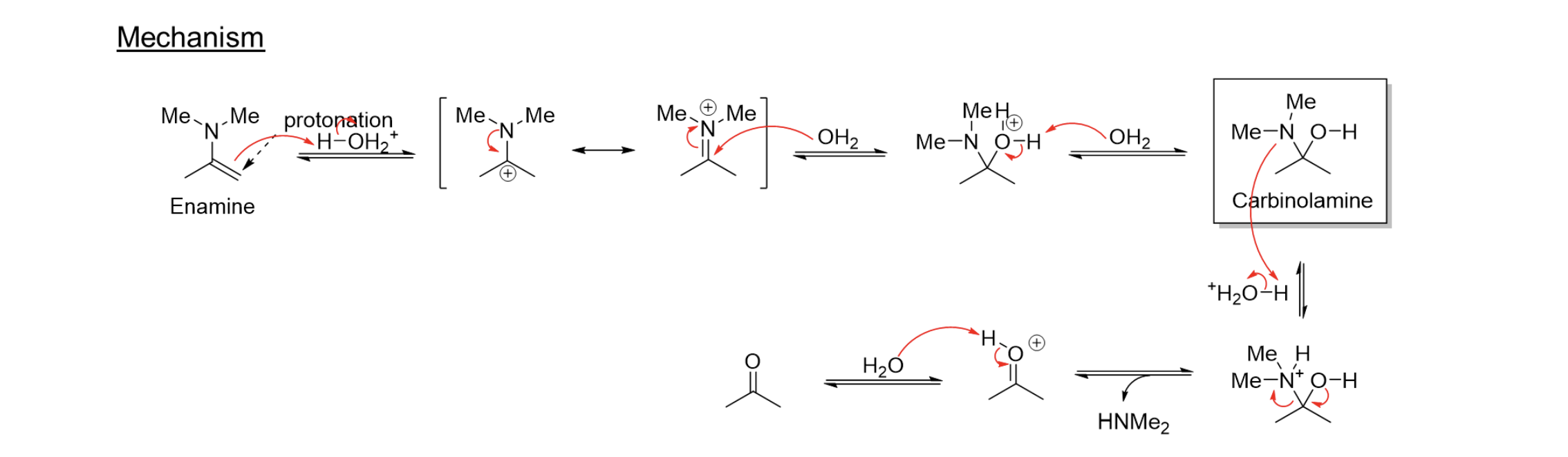
Acid Catalyzed Hydrolysis of Enamines
Enamine → H2O/Cat.H+ → Carbonyl 2 R2NH
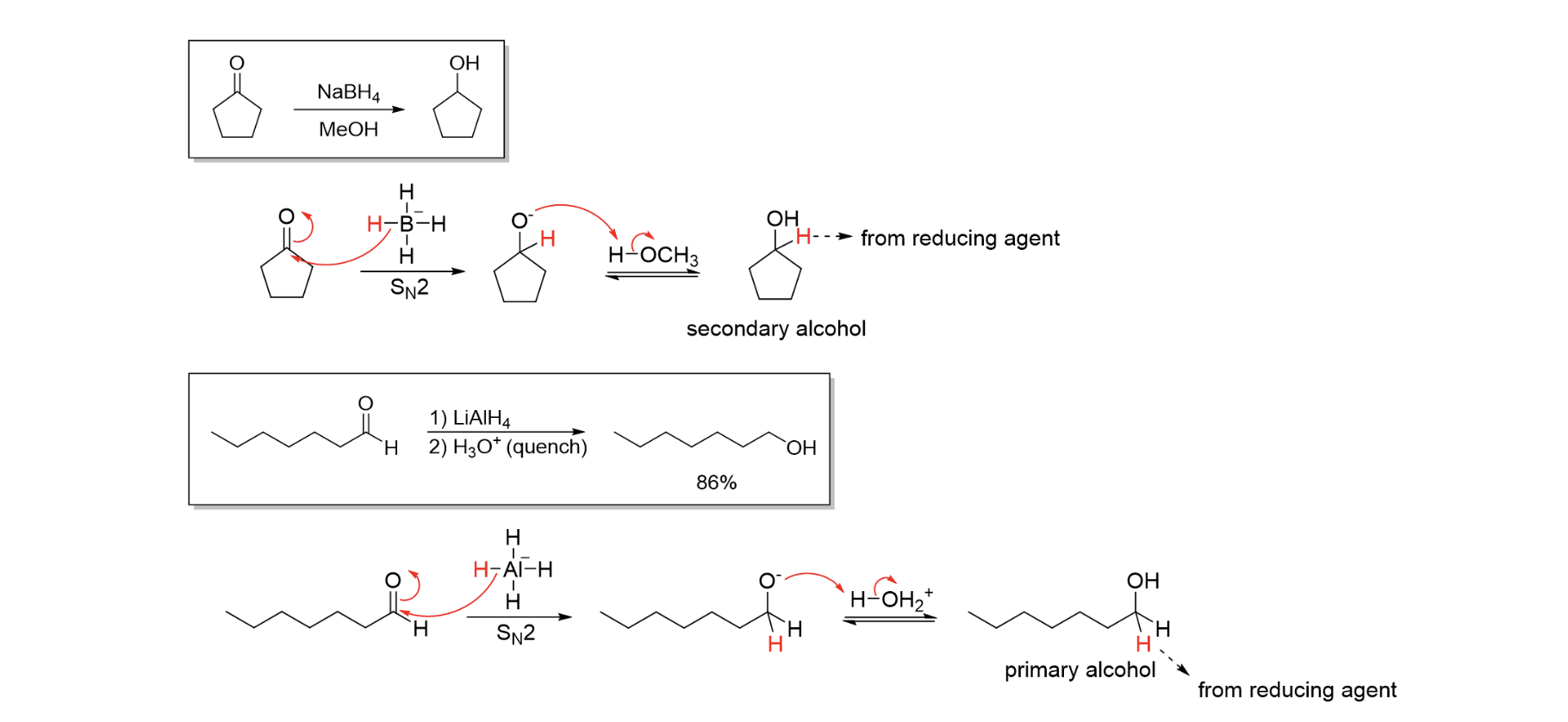
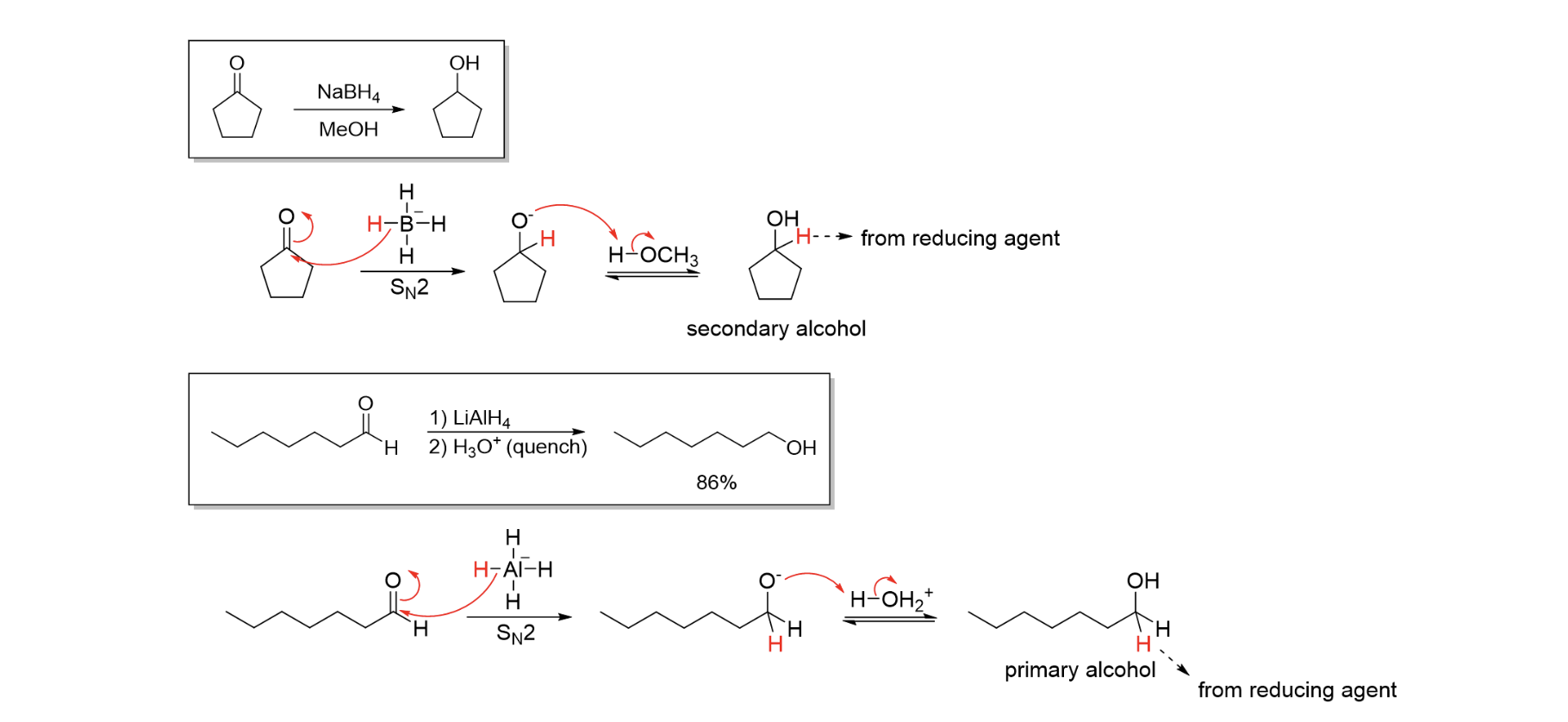
Red of Ald & Ket
H2 / Pd
Grignard Reagents
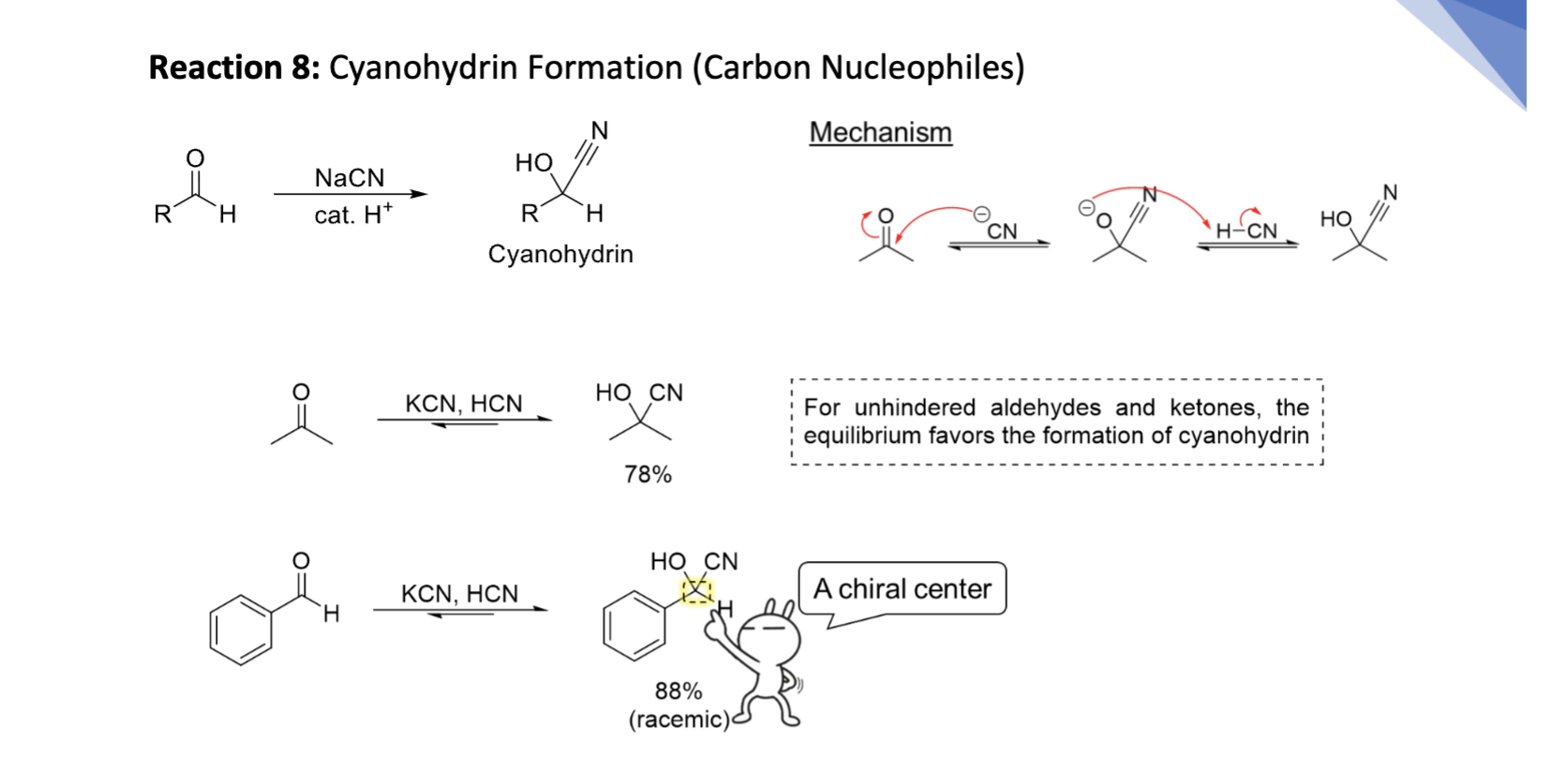
Cyanohydrin Hydration
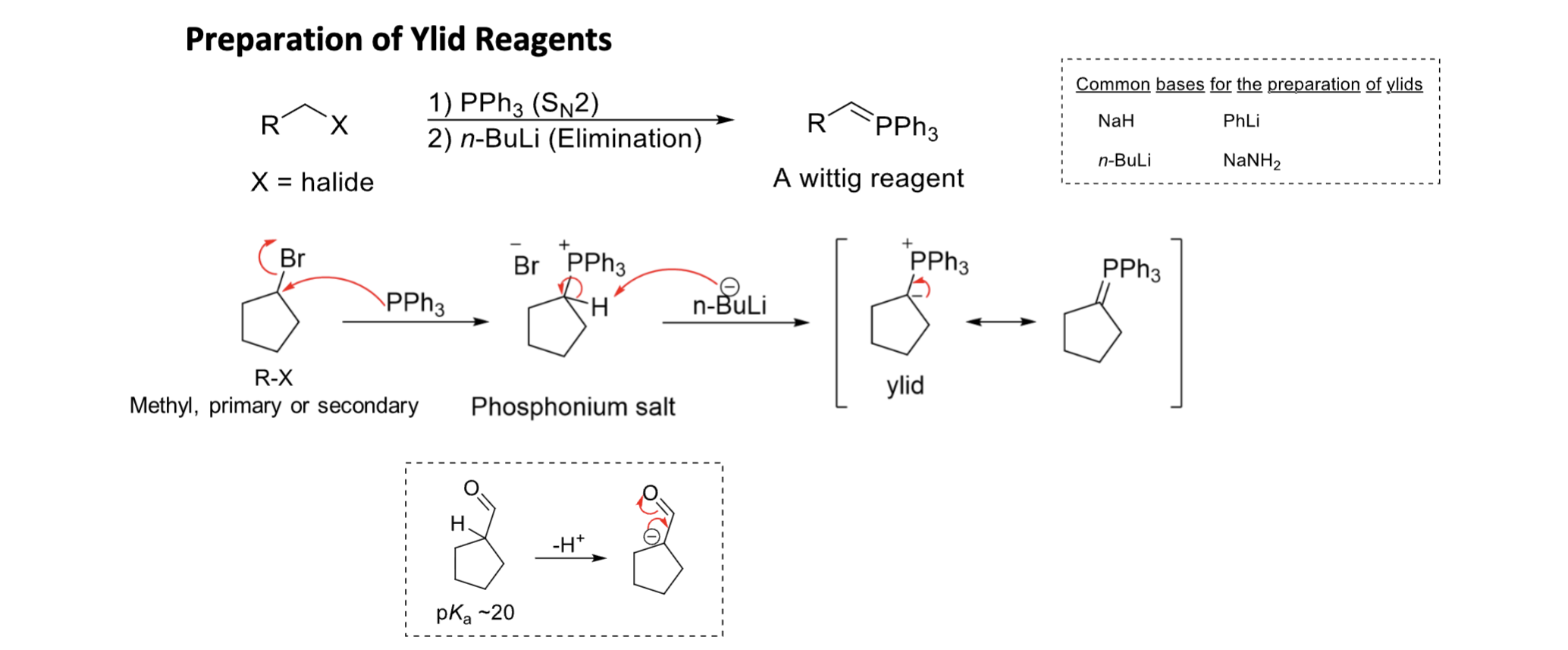
Wittig Reaction (O + ket + PPh3)
convr ald & ket = alkene
Ph3P = CHR
unstable ylid = Z alkene
stable ylid = E alkene
E more stable than Z
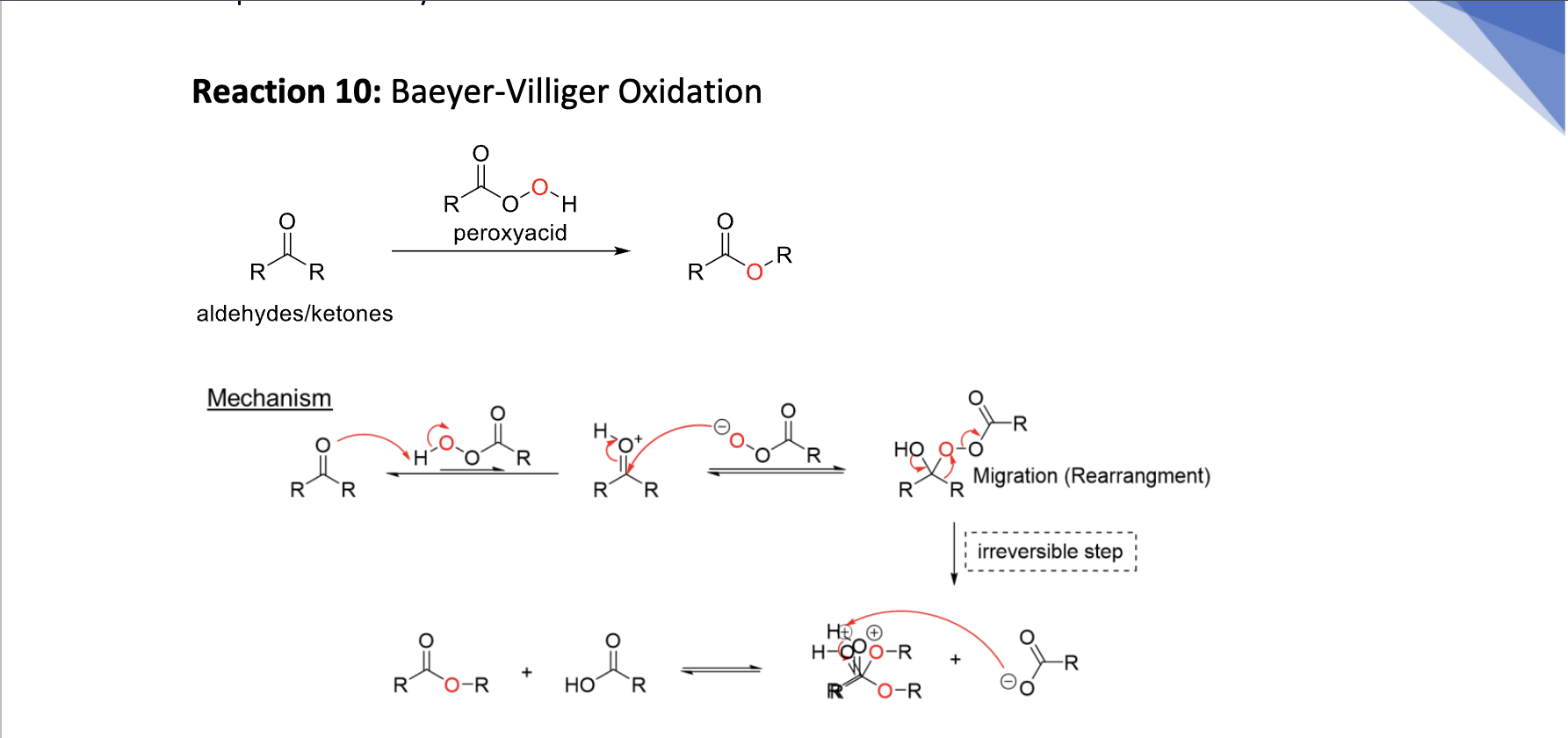
Baeyer-Village Oxidation
prot ke w acid → tert intermed → drives eq → proton transfer