Kidney Unit
1/41
Earn XP
Description and Tags
Lecture 19, 20, 21
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Major functions of kidneys
1) Regulate water and ion balance in plasma/body
2) Remove metabolic waste products (e.g. urea, the end product of protein breakdown)
3) Remove foreign chemicals from the body
4) Secrete hormones: Erythropoietin (long-term feedback regulation of blood oxygen carrying capacity, EPO), 1,25 dihydroxy vitamin D (insulin pathway)
In and Out of Kidney
1 input: renal arter
2 outputs: renal vein (back to circulatory system) and ureter (to be excreted as urine)
Basic processes affecting what is retained
1) Glomerular filtration
2) Tubular filtration
3) Tubular reabsorption
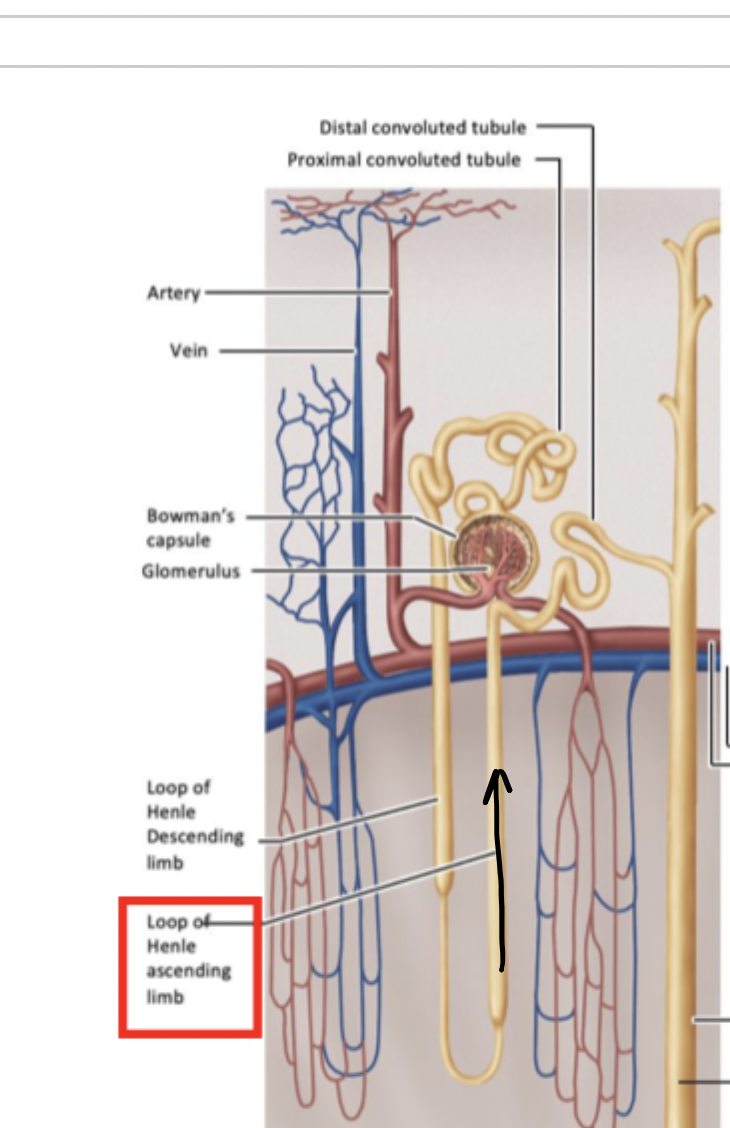
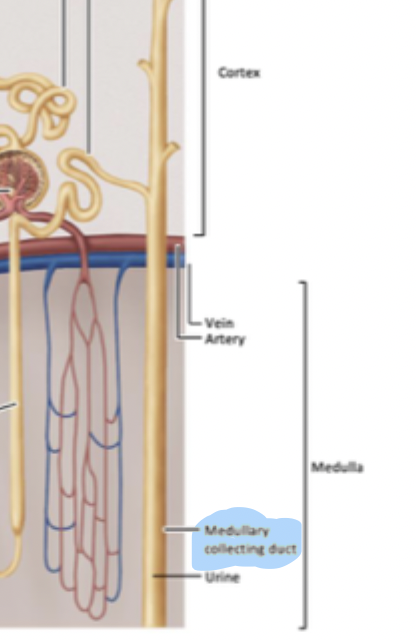
Nephrons
Functional units of the kidney
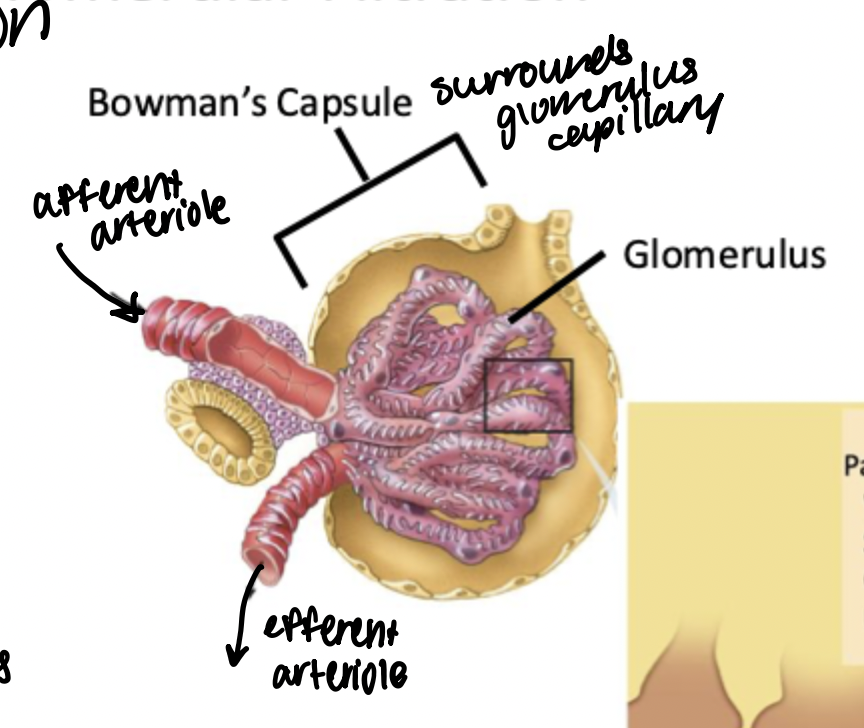
1) Glomerulus and Bowman’s capsule
2) Proximal tubule
3) Descending limb of loop of henle
4) Ascending limb of loop of henle
5) Distal tubule
6) Medullary collecting duct
-all tubules and parts of loop of henle are surrounded by capillaries that allow for exchange across epithelial cells with circulatory system
Glomerulal Filtration
Pressure driven filtration. This is the first point of filtration, between the afferent and efferent capillaries from the renal artery.
What enters: 20% of plasma, glucose, ions water (large amount enters which will then go through reabsorption later)
What doesn’t enter: cells, proteins (too large to cross the capillary membrane)
Step in kidney processing where the largest amount of substance moves into the filtrate. Following steps focus on reabsorption of salts, water, and nutrients from this initial filtrate.
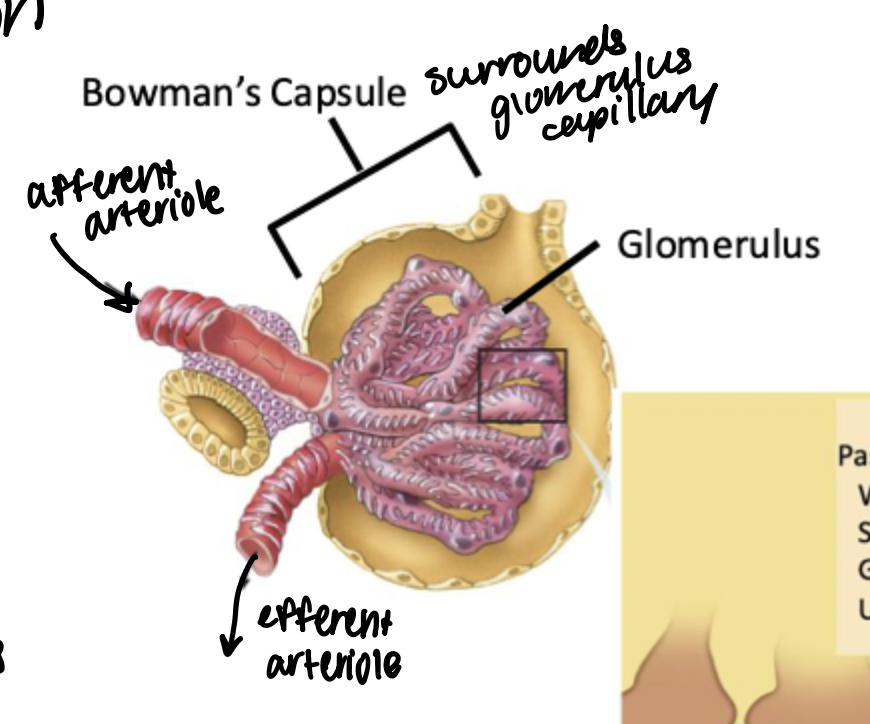
Bowman’s capsule
Surrounds the bundle of capillaries that make up the glomerulus
Glomerulus
Bundle of capillaries that make up the first site of absorption from circulation into the kidneys. Surrounded by Bowman’s capsule.
Water percentage reabsorbed
99
Sodium percentage reabsorbed
99.5
Glucose percentage reabsorbed
100
Urea percentage reabsorbed
44
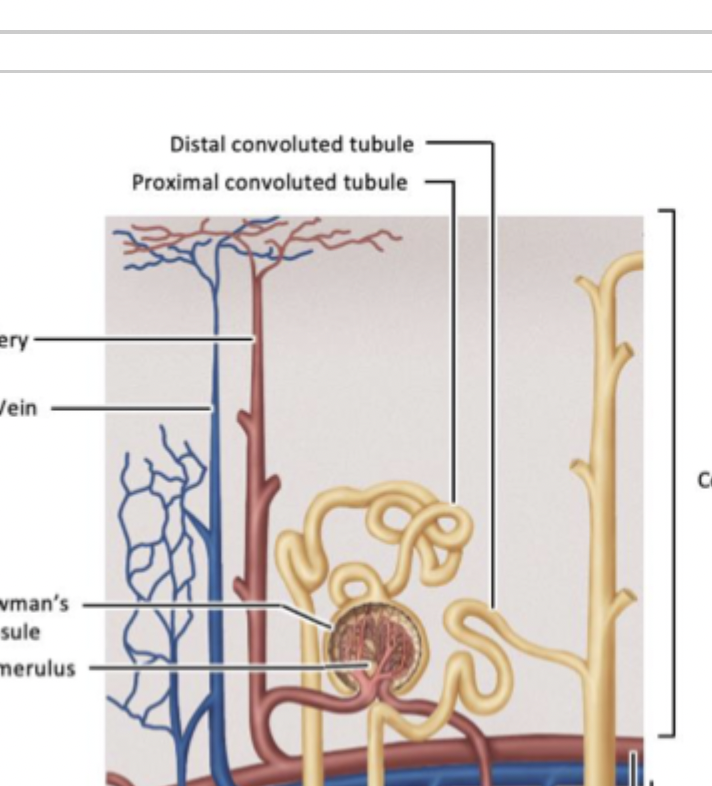
Proximal tubule
Nephron component between the glomerulus and bowman’s capsule, and the descending loop of henle. Located in the kidney cortex, surrounded by capillaries.
Site of about 65% of reabsorption, including 100% of glucose reabsorption in healthy kidneys.
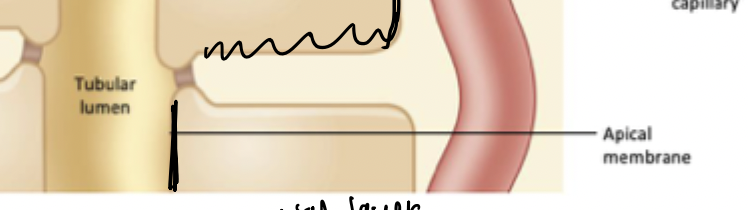
Apical membrane
Membrane of epithelial kidney cells that face the tubular lumen → where there are membrane proteins that facilitate transport of key nutrients from tubular lumen into epithelial cell.
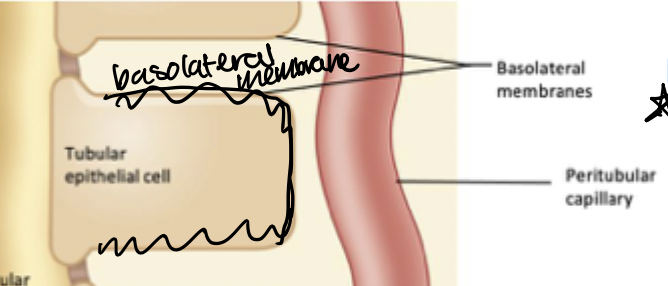
Basolateral membrane
Membrane of epithelial kidney cells that faces the interstitial space (DOES NOT face the tubular lumen). Transport proteins here facilitate transport from epithelial cell into interstitial space or into circulation (peritubular capillary)
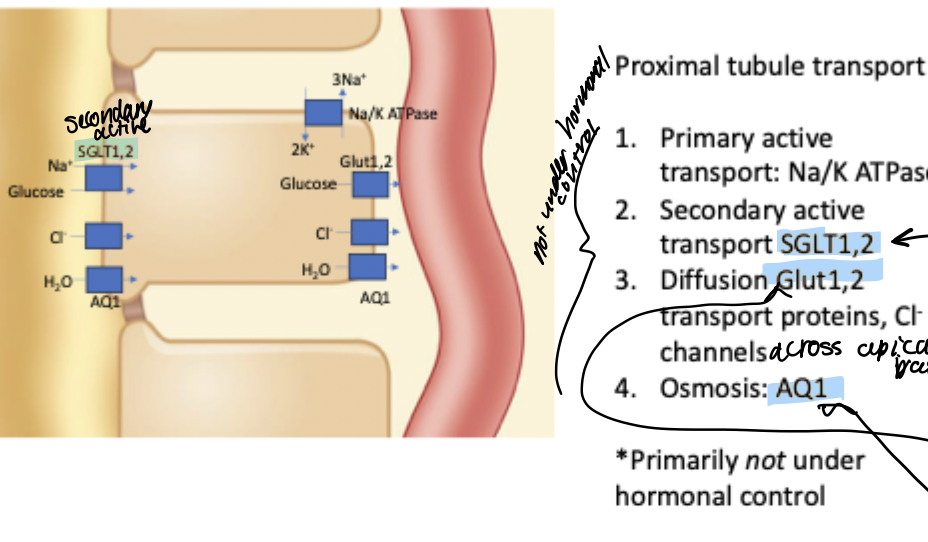
Proximal tubule transport
65% of reabsorption happens here, including 100% of glucose reabsorption in healthy kidneys.
Transport:
1) Na/K ATPase to establish key gradients
2) Secondary active transport of glucose through SGLT1, 2 on apical membrane- paired with transport of Na+ down gradient established. by Na/K ATPase (allows for full reabsorption of glucose beyond just balancing concentration gradient)
3) Diffusion of glucose through Glut1,2 transport proteins on basolateral membrane → allow glucose in epithelial cell to enter capillary
4) Osmosis (some amount of water reabsorption happens here) via AQ1
Transport here primarily not under hormonal control
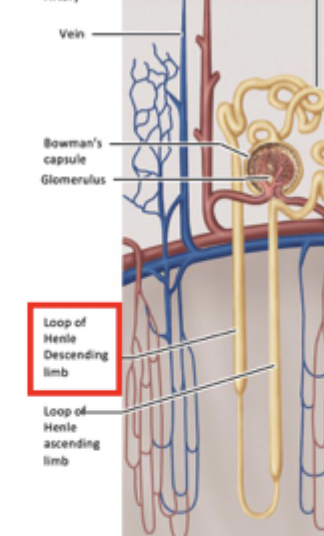
Descending limb of loop of henle
Extends from the proximal tubule in the cortex of the kidney down into the medulla.
Site of most water reabsorption.
Defined by high AQ1 expression (not under hromonal control). And also low conductance of ions (Cl-, Na+, K+, aside from Na/K pump)
Uptake of water also driven by osmotic gradient. In kidney medulla due to salt reuptake occuring in ascending limb of loop of henle, there is high salt concentration in the interstitial space.
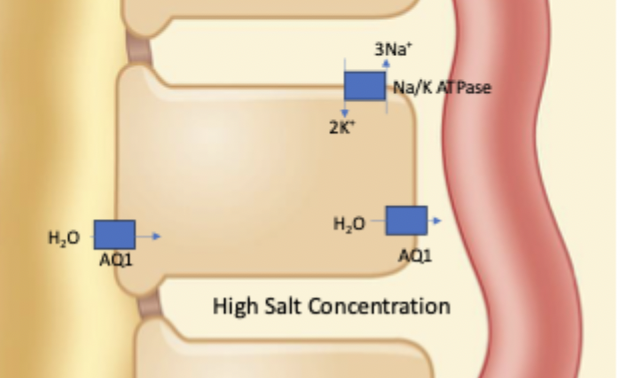
Ascending limb of loop of henle
Tubule component in the kidney medulla. Between the descending limb of the loop of henle and the distal tubule.
Primary site of reabsorption of salt. Also serves to create high salt concentrations in the medulla that are key to driving water reabsorption in the descending limb of loop of henle.
Descending limb of loop of henle transport
Na/K ATPase, high expression of AQ1 (not under hormonal control), and low ion conductivity. Reuptake of water also largely driven by high salt concentration in medulla interstitial space.
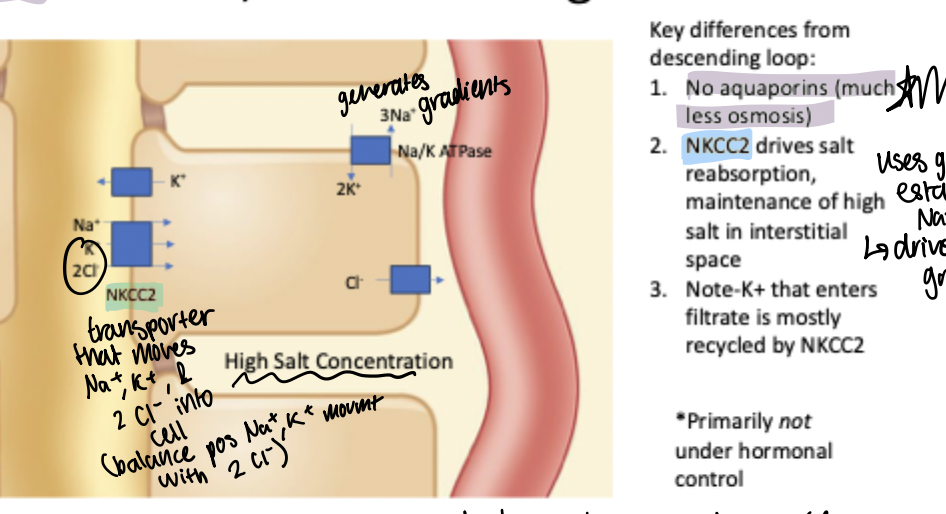
Ascending limb of loop of henle transport
Primary function is to reabsorb salt.
Key differences:
1) No aquaporins (much less movement of water) - allows for movement of ions without significantly altering water reuptake at this step of the tubule
2) NKCC2: drives salt reabsorption and maintenance of high salt in the interstitial space. Transporter on apical membrane that moves one Na+, 1 K+, and 2 Cl- into the epithelial cell for a net neutral charge movement of ions into the cell driven by the Na+ gradient established by Na/K ATPase
Note: there are K+ leak channels on apical membrane so that some K+ moves back into the filtrate down its gradient, but this is mostly recycled by the NKCC2 as movement of K+ into cell is paired with movement of Na+ down gradient.
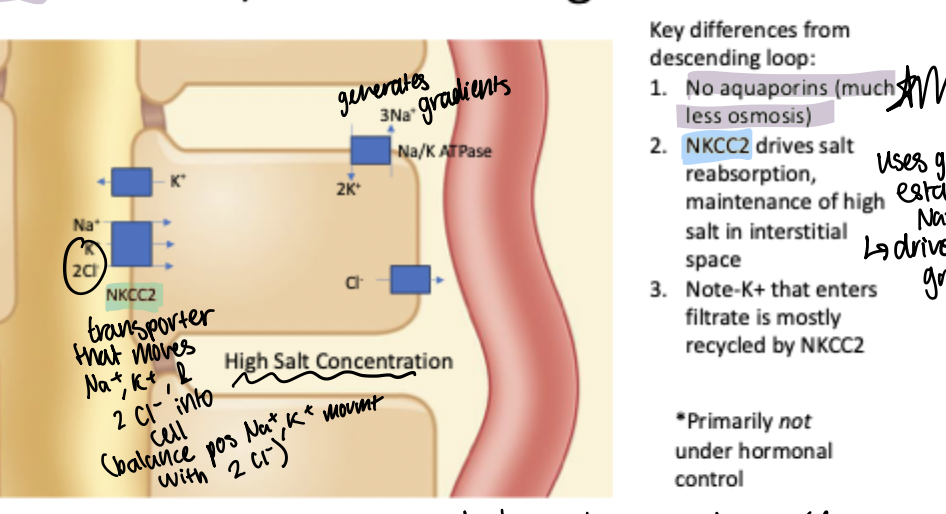
NKCC2
Drives salt reabsorption and maintenance of high salt in the interstitial medulla space. High expression on apical membrane of ascending limb of loop of henle.
Transporter on apical membrane that moves one Na+, 1 K+, and 2 Cl- into the epithelial cell for a net neutral charge movement of ions into the cell driven by the Na+ gradient established by Na/K ATPase
Note: there are K+ leak channels on apical membrane so that some K+ moves back into the filtrate down its gradient, but this is mostly recycled by this transport protein as movement of K+ into cell is paired with movement of Na+ down gradient (proper function relies on enough K+ in tubule/lumen to function)
SGLT1,2
Key transport proteins on apical membrane of epitelial cells lining the proximal tubule. Secondary active transport of glucose into cells from the lumen paired with the movement of Na+ down gradient. Allow for full glucose reabsorption in healthy kidneys.
GLUT1,2
Passive transport of glucose from epithelial cells into capillaries. Located on basolateral membrane of kidney epithelial cells lining the proximal tubule.
Effect on urine formation formation if Na/K ATPase in ascending limb of loop of henle is inhibited
Less salt reasorption in ascending limb of loop of henle. Transport of ions into epithelial cells through NKCC2 driven by movement of Na+ down gradient established by Na/K pump.
This means lower salt concentration in the kidney medulla, which then will decrease the osmotic gradient driving water reabsorption in the descending limb of loop of henle, so less water reabsorption.
So more water in filtrate and more salt in filtrate too.
Distal tubule
Between loop of henle and the medullary collecting duct. Site of Na and Cl reabsorption under hormonal control.
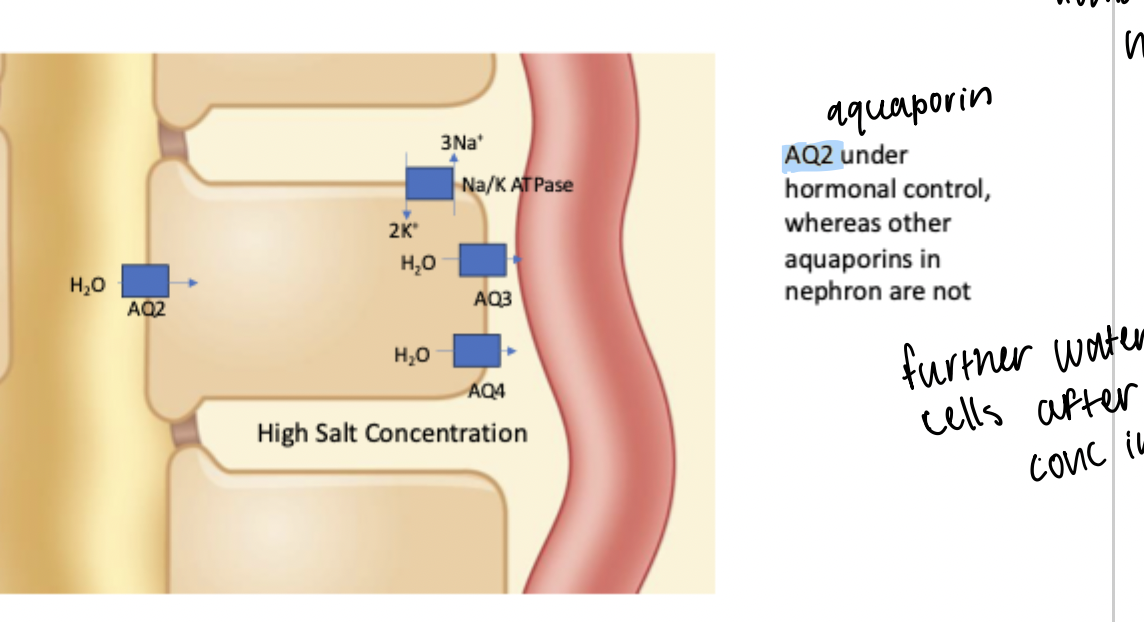
Medullary Collecting Duct
Between distal tubule and urine. Site of water reabsorption under hormonal control via AQ2 expression regulation on apical membrane. Also has AQ3 and AQ4 expressed on basolateral membrane but not under hormonal control.
Water reabsorption primarily occurs in the descending limb of loop of henle through AQ1 + high interstitial salt concentration. Further reuptake occurs here.
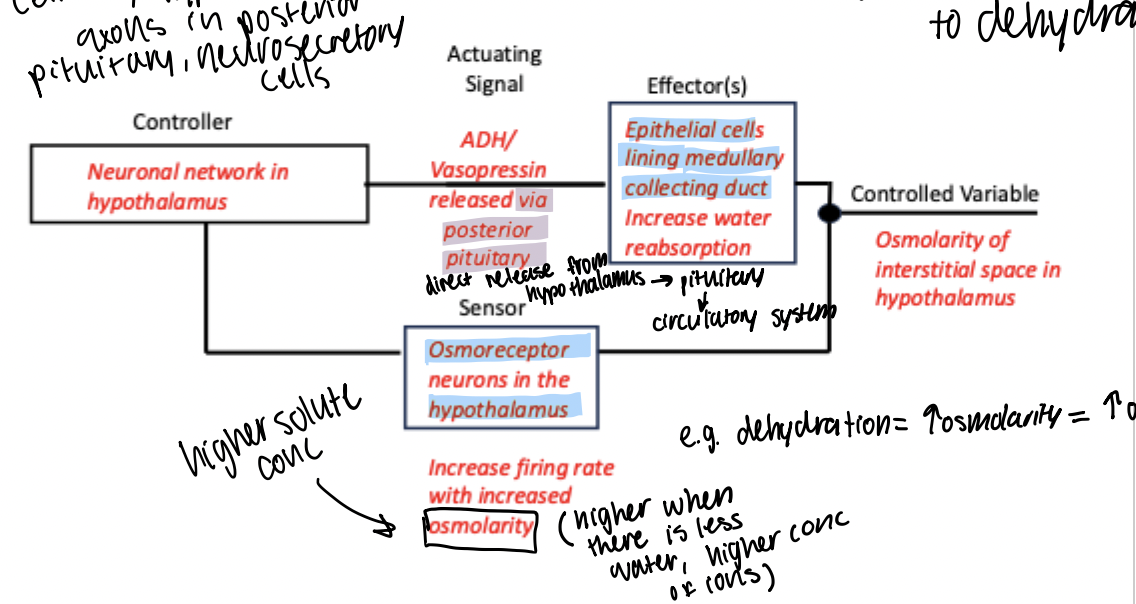
Control of water retention/excretion by ADH/Vasopressin
Controlled variable: osmolarity of interstitial space of hypothalamus (high osmolarity means high solute concentration i.e. dehydration)
Sensor: osmoreceptor neurons in the hypothalamus. Increase AP frequency with high osmolarity.
Controller: neuronal networks in the hypothalamus
Actuating signal: ADH (antidiuretic hormone)/Vasopressin is released via neurosecretory cells with axons in the posterior pituitary → released directly into circulation
Effectors: epithelial cells lining the medullary collecting duct. Causes the exocytosis of AQ2 proteins into the cell membrane to increase water reabsorption
Allows us to regulate water reabsorption in response to dehydration.
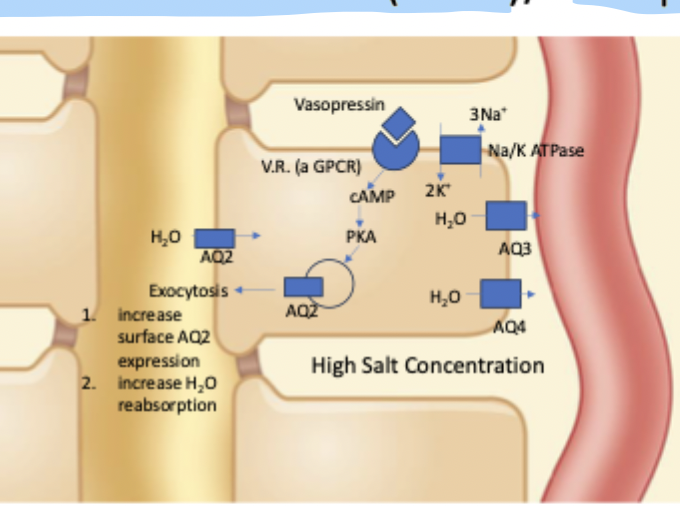
Vasopressin
Also known as Antidiuretic hormone (ADH). A peptide hormone released by the posterior pituitary that interacts with a membrane GPCR on epithelial cells lining the medullary collecting duct. Leads to signaling pathway that increases cAMP and PKA, which triggers exocytosis of AQ2 which is stored intracellularly across vesicle membranes.
Exocytosis leads to higher AQ2 presence in apical membrane → higher water reuptake
Key hormone that allows for regulation of water reabsorption (to a small extent) in response to dehydration)
Dehydration pathway
1) Dehydration means an increased osmolarity in the body, including in the hypothalamus interstitial space
2) Osmoreceptors in the hypothalamus sense the high osmolarity and increase AP frequency
3) Error signal from high osmolarity causes the release of vasopressin/ADH into the circulatory system
4) Vasopressin interacts with GPCR on basolateral membrane of epithelial kidney cells lining the medullary collecting duct
5) Increase in cAMP and PKA from GPCR pathway drives exocytosis of AQ2 proteins
6) More AQ2 proteins on apical cell membrane allows for increased water reabsorption from the filtrate

Diabetes insipidus
Rare genetic disorder that impairs the vasopressin/ADH feedback loop, inhibiting the ability of the body to adjust water reabsorption in response to dehydration.
2 types
Neureogenic: loss of function mutation affecting hypothalamus so that ADH/vasporessin is not released
Nephrogenic: mutation affecting collecting duct cells so that the receptor is not able to respond to vasopressin
Neurogenic diabetes insipidus
Loss of function mutation affecting the hypothalamus, impairing ability to produce/release ADH/vasopressin in response to high hypothalamus osmolarity (dehydration)
Could treat person with vasopressin.
Nephrogenic diabetes insipidus
Loss of funciton mutation affecting the vasopressin receptors on the membranes of epithelial kidney cells in the medullary collecting duct in feedback loop to increase water reuptake during dehydration (high hypothalamus osmolarity).
Leads to higher than normal levels of ADH in circulation. Treating person with vasopressin would not fix problem.
Beginning of the distal tubule
Site of transition from the ascending limb of loop of henle to the distal tubule.
Comes next to the afferent arteriole next to the glomerulus.
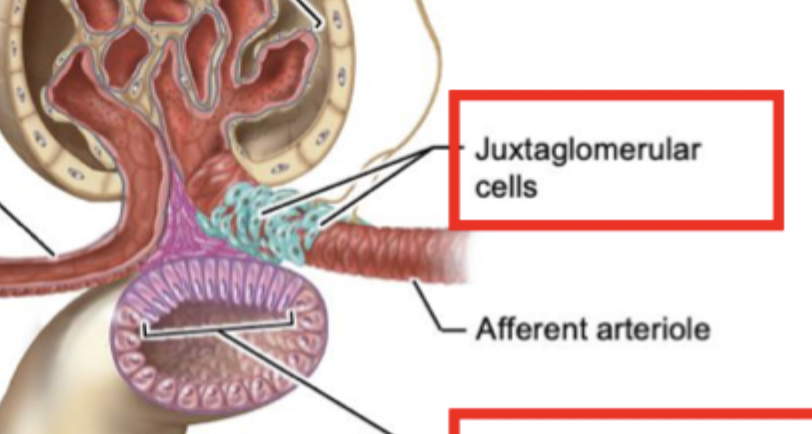
The afferent arteriole that comes next to it is surrounded by juxtaglomerual cells. Site of hormonal control of salt reuptake.
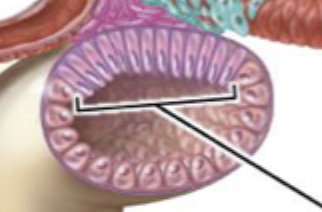
Contains macula densa cells lining the lumen.
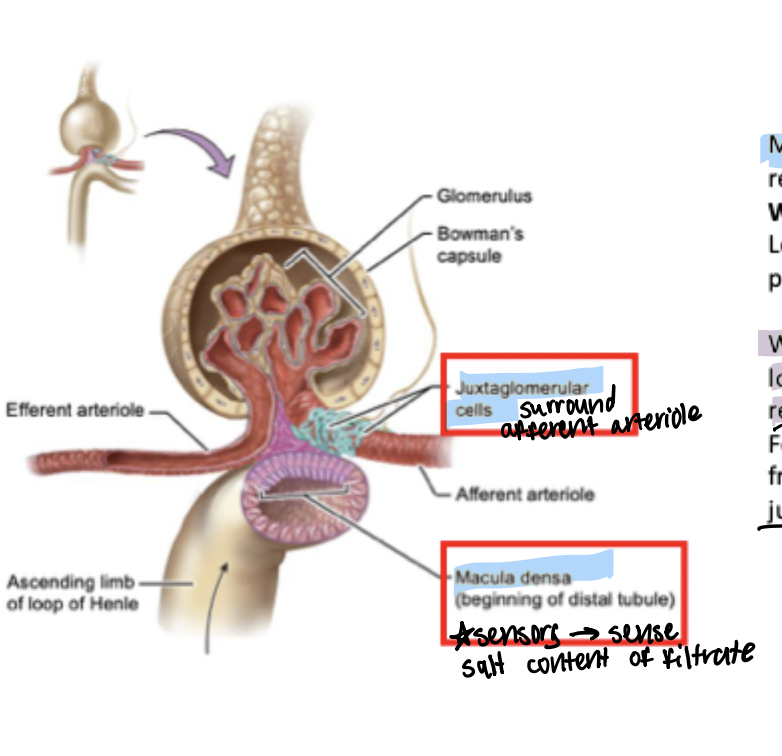
Macula Densa
Epithelial cells lining the beginning of the distal tubule. Serve as sensors to sense the salt content of the filtrate. Key to the feedback loop regulating salt reabsorption (component that is under hormonal control).
Salt reabsorption feedback loop
Controlled variable: salt content of the beginning of the distal tubule (proxy for salt content within the blood since original source of salt in filtrate is from blood)
Sensor: macula densa cells (epithelial cells at the lumen of the beginning of the distal tubule)
Controller: macula densa cells - serve as comparator of lumen salt content of beginning of distal tubule and begin actuating signal of PG (prostoglandin
Actuating Signal: prostglandin interacts with juxtaglomerual cells on the afferent arteriole next to the glomerulus to trigger the release of renin. Renin then interacts with a precursor angiotensinogen produced by the liver to form inactive angiotensin I and then angiotensin II;
Angiotensin II is an active signaling molecule that interacts with a receptor on the adrenal cortex that then causes the release of aldotestosterone.
Effector: aldotestosterone is a steroid hormone released by the adrenal gland that interacts with an intracellular receptor in the cortical collecting duct and distal tubule.
Prostoglandin (PG)
Hormone released by macula densa cells at the beginning of the distal tubule in response to low filtrate salt content. Interacts with juxtaglomerular cells on the afferent arteriole of the glomerulus. Causes juxtaglomerual cells to release renin into the bloodstream.
Juxtaglomerual cells
Found along the afferent arteriole next to the glomerulus. Responsible for receiving the signal from prostglandin from macula densa cells in response to low salt filtrate content and then releasing renin to eventually become angiotensin II. Key part of actuating signal for hormonal feedback regulation of salt reuptake.
Renin-angiotensin pathway
Renin is released by Juxtaglomerular cells in the kidney. Interacts with inactive angiotensinogen produced by the liver to form the inactive intermediate angiotensin I. Then forms angiotensin II which is an active signaling molecule that is received by a receptor in the adrenal cortex to trigger the release of aldotestosterone.
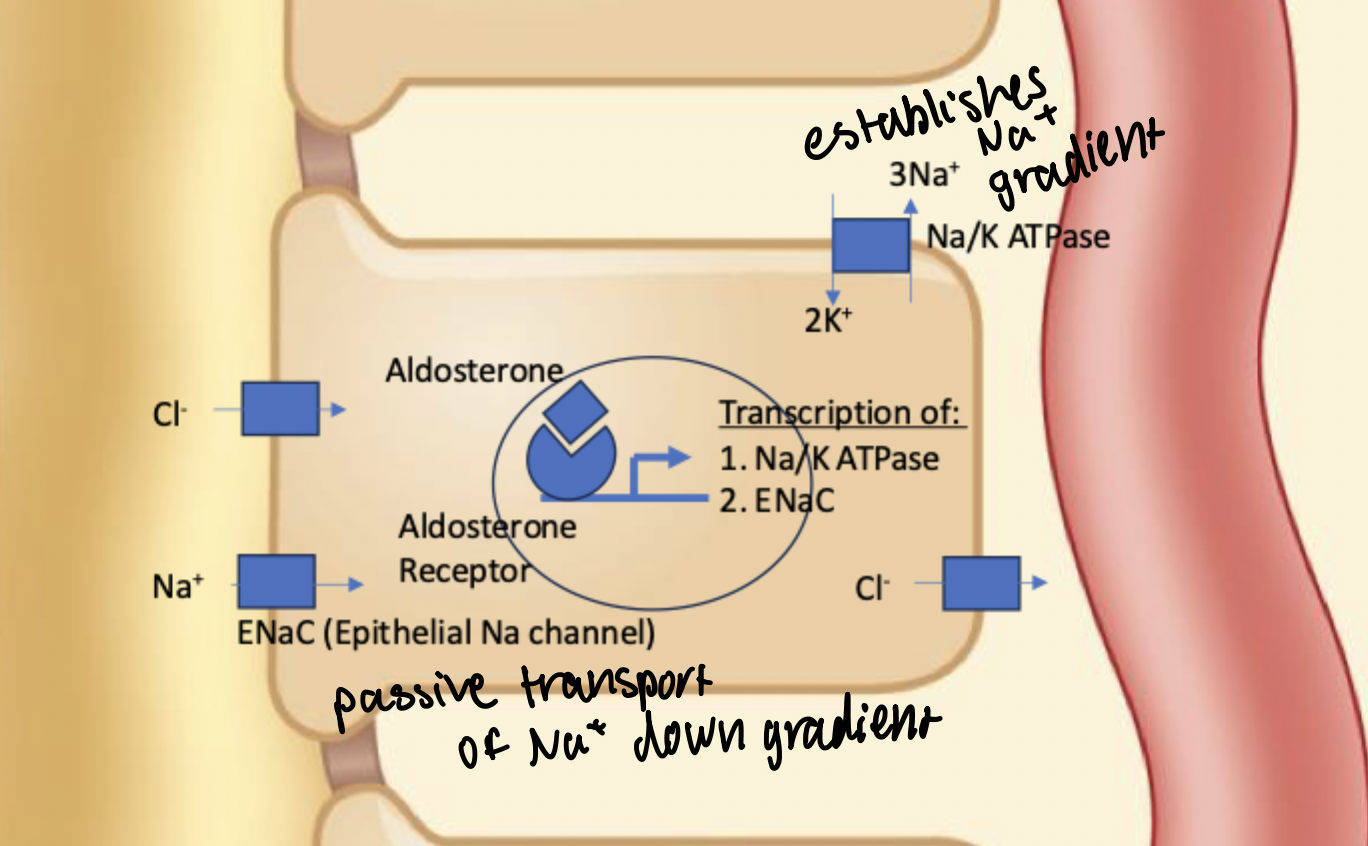
Aldotestosterone
Steroid hormone released by the adrenal cortex in response to the signaling molecule angiotensin II released in the feedback loop regulating hormonal salt reuptake. This is a steroid hormone that can cross the cell membrane and therefore interacts with an intracellular receptor inside epithelial kidney cells lining the cortical collecting duct and distal tubule.
The receptor complex can bind to DNA in the nucleus to increase transcription of Na/K ATPase and ENaC which are epithelial sodium channels that allow for the passive diffusion of sodium into the epithelial cell (exocytosed to the apical membrane)
Aldotestosterone receptor
This is an intracellular receptor that binds to aldotestosterone and can bind to DNA to regulate transcription. When bound to aldotestosterone, triggers increased expression of Na/K ATPase to establish and maintain an Na gradient, as well as increased expression of ENaC to allow diffusion of Na+ from the filtrate into the cell driven by the continuous Na+ gradient created by the active transport.
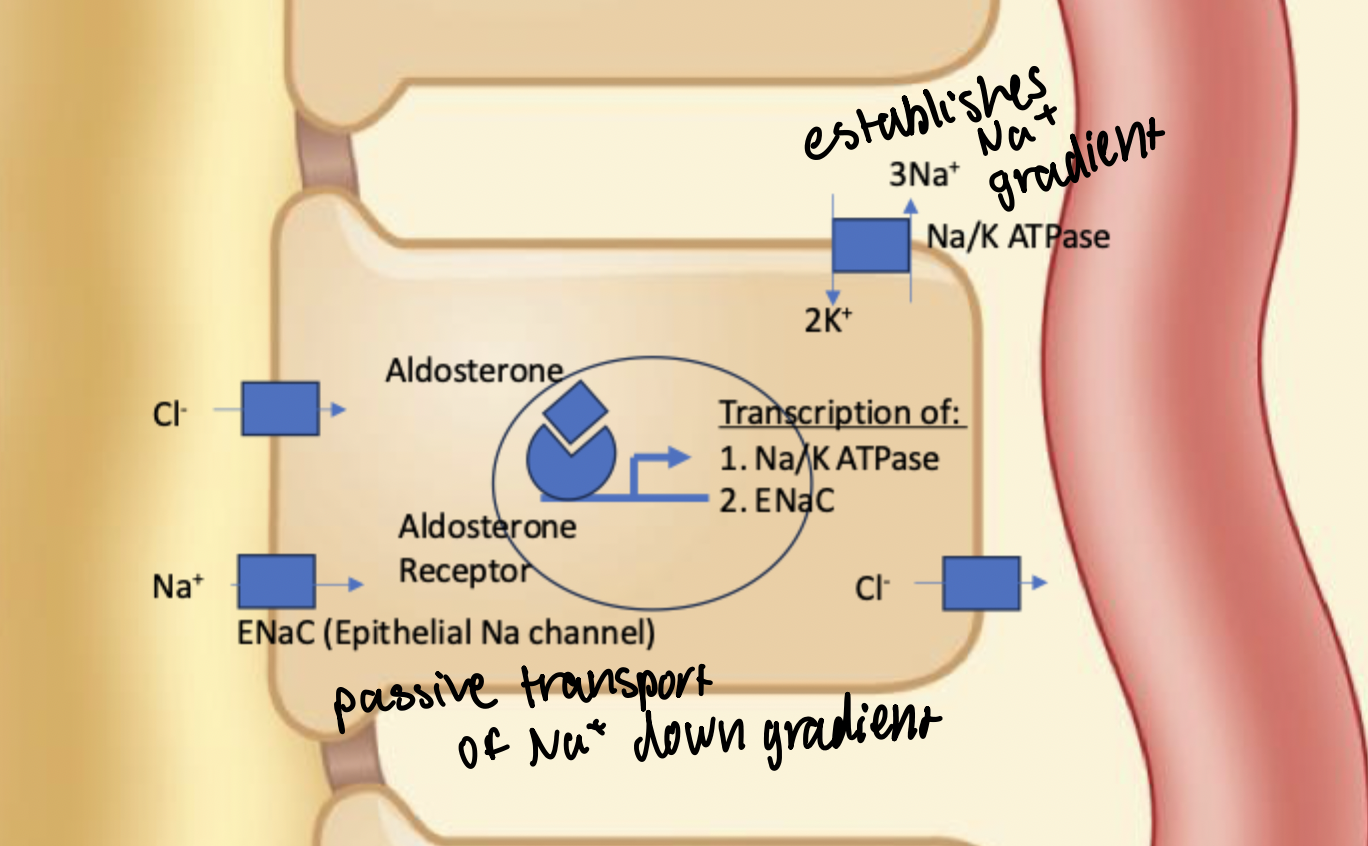
ENaC
Epithelial Sodium Channel. Expression regulated by aldotestosterone. Aldotestosterone/receptor complex causes increased expression and exocytosis to apical membrane to allow increased movement of Na+ into the epithelial cell at the cortical collecting duct and distal tubule driven by the sodium gradient created by the Na/K ATPase.
Range of sodium excretion
Due to influence of aldotestosterone. Can range from 0-2%.
Effect of variable salt reuptake
When more salt is taken up through the renin/aldotestosterone pathway, this also causes increased water reuptake. As ions move, so does water following the osmotic gradient.