Physics
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
Conservatives Forces:
are path independent and do not dissipate energy (gravitational, elastic spring, etc)
Work:
the process by which a quantity of energy is moved from one systetm to another
Work in references to gases
if gas expanded, gas did the work
if gas compressed, work was done on the gas
Power:
the rate at which energy is transferred from one system to another
Work-Energy Theorem:
calculates work via adding all the kinetic energies
W = Delta K
Units for Force, Work/Energy, and Power
Newton, Joule, and Watt
What is a vector?
Have both magnitude (an amount), and a direction.
Ex: displacement, velocity, acceleration, and force
What is a scalar?
Have only amount/magnitude
ex: distance, speed, energy, pressure, and mass
Split a vector into X and Y components

Dot Product Formula for scalar
A * B = |A| |B| cos(𝜃)
Cross Product Formula for vector
A x B = |A| |B| sin (𝜃)
Explain the Right Hand Rule to find resultants
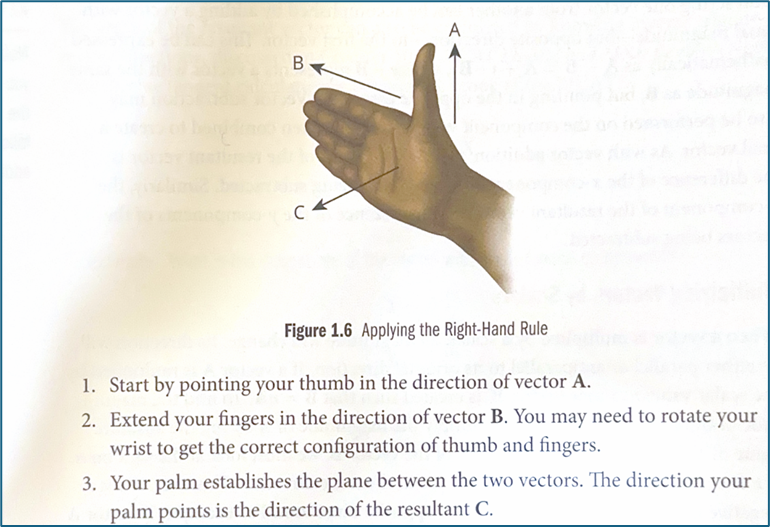
What are Newton’s Laws:

Torques that generate clockwise rotation are considered _______, while torques that generate counterclockwise rotation are _______.
negative … positive
Conservative forces
are those that are path independent and that do not dissipate energy
Work
the process by which a quantity of energy is moved from one system to another
positive vs. negative work on a gas
Positive work = gas expanded = gas did work
Negative work = gas compressed = work was done on the gas
Power
the rate at which energy is transferred from one system to another
Zeroth Law of Thermodynamics
objects in thermal equilibrium do not cause any net heat transfer/flow across each other.
First Law of Thermodynamics
states that the change in the total internal energy of a system is equal to the amount of energy transferred in the form of heat to the system, minus the amount of energy transferred from the system in the form of work.
(add energy added, substract energy used for work (U = Q - W))
Second Law of Thermodynamics
objects in thermal contact and not in thermal equilibrium will exchange heat energy such that the object with a higher temperature will give off heat energy to the object with a lower temperature until both objects have the same temperature at thermal equilibrium.
Types of Systems (3)
o Closed: transfers energy, but no matter
o Isolated: doesn’t transfer anything
o Open: transfers both
Heat
the process by which a quantity of energy is transferred between two objects as a result of a difference in temperature
Conduction
heat transfer that is done via direct physical contact (pan)
Convection
heat transfer done by gases and liquids (oven)
Radiation
the transfer of energy by electromagnetic waves. Fun fact, can occur thru a vacuum (microwave)
Specific Heat of Water (in calories)
1 cal/g*k
Isothermal
constant temperature, and therefore no change in internal energy
(change in internal energy (delta U) = 0) … meaning Q (heat) = W (work)
Adiabatic
no heat exchange
Q = 0 (no heat) … so delta U = - W
Isovolumetric
no change in volume , and therefore no work accomplished
W = 0 …. so delta U = Q
Isobaric
constant pressure
Density of Water
1 kg/m3
Specific gravity for water
1 atm and 4 Celsius
Pressure Units
1.013x105 Pa = 760 mmHg = 760 torr = 1 atm
Pascal’s Principle
for fluids that are incompressible a change in pressure will be transmitted undiminished to every portion of the fluid and to the walls of the containing vessel
The fundamental Unit of Charge
e = 1.60 x 10-19 Coulombs
Insulators

will not easily distribute a charge over its surface and will not transfer that charge to another neutral object very well (nonmetals)
Conductors

when given a charge, the charges will distribute approximately evenly upon the surface of the conductor
Proton Field Lines
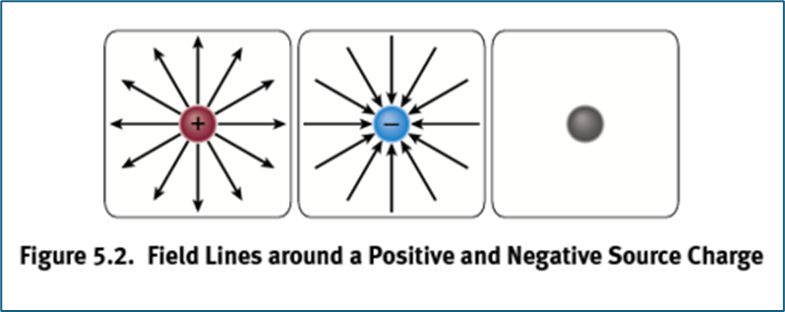
extend outwards
Electron Field Lines
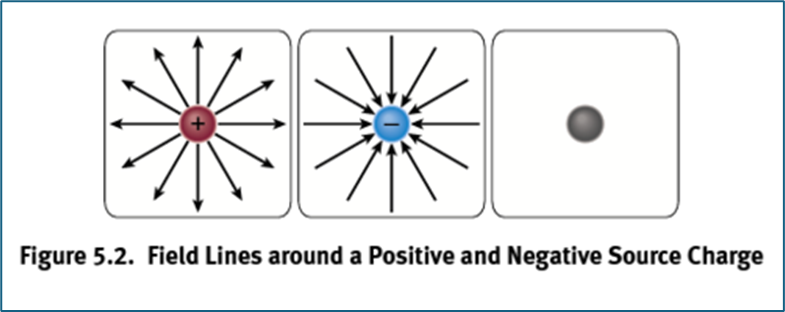
extend inwards
Electrical potential energy of two like particles
For like particles it increases as they get closer, and for opposite charges it decreases
Equipotential Lines
a line on which the potential at every point is the same. Three-dimensionally speaking, it isn’t an actual line, but rather a sphere. Hence, moving a charge across one of these lines would not need any work, only moving a charge from one line to another would.
(For negative charges, the electric potential energy increases as we “walk” away from a positive charge)
Electric Dipole / Moment
A dipole = separated + and − charges
A dipole moment = how strong that separation is + which way it points
Direction = negative → positive
Bigger charge or distance = stronger dipole
In fields → dipoles rotate and align
Ferromagnetic Materials
like paramagnetic materials, they have unpaired electrons and permanent atomic magnetic dipoles that are normally oriented randomly so that the material has no net magnetic dipole. However, unlike paramagnetic materials, they will become strongly magnetized when exposed to a magnetic field
What do moving charges create?
A magnetic field
Right-Hand Rule
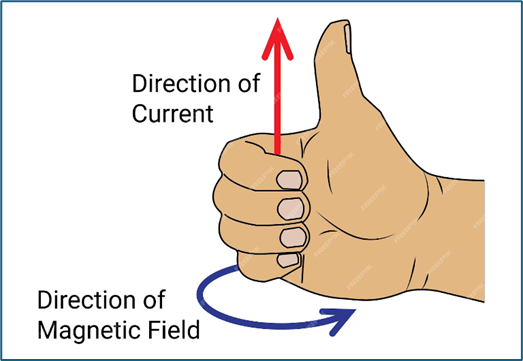
Point the thumb in the direction of I and curve fingers around the wire, that will give you the direction of the magnetic field.
Second Right-Hand Rule
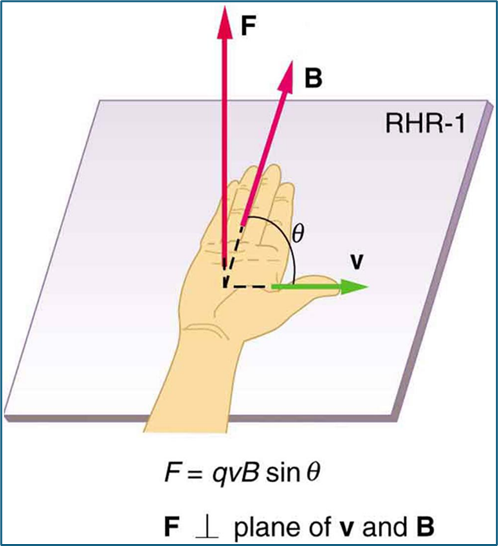
First, position your right thumb in the direction of the velocity vector. Then, put your fingers in the direction of the magnetic field lines. Your palm will point in the direction of the force vector for a positive charge, whereas the back of your hand will pint in the direction of the force vector for a negative charge.
What do you need to create a magnetic field vs. an electric field
magnetic field requires a moving charge
electric field requires only a charge
How to create a magnetic force?
one needs an external electric field acting on a charge moving any direction except parallel or antiparallel to the external field
Electromotive force (emf)
not an actual force but rather the potential difference of the voltage source for a circuit, usually a battery
Current
the movement of positive charge thru a conductive material over time
Voltage
a potential difference between two points
Conductivity
the reciprocal of resistance
Capacitor
have the ability to hold a charge at a particular voltage
Capacitance
the magnitude of the charge stored on one plate to the potential difference across the capacitor
Dielectric Material
another way of saying insulation. These materials increase the capacitance of a capacitor by a factor called the dielectric constant (k) …
o If capacitor is isolated, its voltage will decrease when dielectric material is introduced, and the stored charge will remain constant
o If the capacitor is in a circuit, its voltage will stay constant b/c it is dictated by the voltage source, and the stored charge will increase
Resistors in Series
o Are added … and so is the voltage ↓
o Vs = V1 + V2 + ….
Resistors in Parallel
o (1/R) = (1/R1) + (1/R2) ….
o Voltage is the same for each resistor in parallel
Capacitors in Series
o 1/C) = (1/C1) + (1/C2) ….
o Voltage is added just like for resistors in series
Capacitors in Parallel
o C = C1 + C2 + ….
o Voltage is the same for each capacitor in parallel
Rules for Voltage in resistors/capacitors
Voltage regardless of resistor/capacitor, in series is added, and in parallel is the same throughout
Transverse wave
wave oscillates perpendicular to the propagation
Longitudinal Wave
Wave oscillates parallel to the propagation
Frequency vs. wavelength vs. period
frequency: the number of waves passing through a fixed point per second
wavelength: length from crest to crest
period: number of seconds per wave (opposite of frequency)
propagation speed
equals the wavelength times the frequency
node
perfectly between a trough and a cresta
antinode
a crest (so the max point from the center of a wave)
Traveling Wave
Imagine a typical wave emitted from a cartoon character’s laser gun … a perpetually traveling wave (has constant amplitude)
Standing Wave
Appears to be fixed at both ends, and so it oscillates up and down, so as to have a maximum amplitude at one point and then it falls to equilibrium and then it just repeats.
Speed of light (c )
3×108 m/s
Visible light goes from
700 nm (red) to 400 nm (violet)
Law of Reflection
angle of incidence equals angle of reflection
In Snell’s Law, where is theta measured from
from the normal … not from the surface of the object reflecting the light
The photoelectric Effect
when a light of a sufficiently high frequency is incident on a metal in a vacuum, the metal atoms emit electrons, as seen above. Recall that the movement of electrons is known as a current
Value of Planck’s constant (h)
6.6×10-34 J.s
Strong Nuclear Force
provides the adhesive force between the nucleons within the nucleus
Mass Defect
the apparent loss of mass when nucleons come together as some of the mass is converted into energy … that energy is called the binding energy
What are the 4 Fundamental forces in nature from strongest to weakest
(1) Strong Nuclear Force
(2) Electromagnetic Forces
(3) Weak Nuclear Force
(4) Gravity
Fusion Nuclear Reaction
fusion occurs when small nuclei combine to form a larger nucleus, as the name implies. This produces energy and is how the sun functions
Fission Nuclear Force
kinda like the opposite of fusion, it’s when large nucleus splits into smaller nuclei
Alpha Decay
name like so b/c the atom releases an α-particle (He) … remember that Helium has 2 protons and 2 neutrons, so if some atom loses an α-particle it will become the element 2 protons before it and will have the molecular weight -4 of the current atom
Beta Decay
the atom will emit a ß-particle (an electron, ß-) …. But actually, it can also release a positron (ß+), which is a “positive electron” if you will. So, if we emit an electron our new atom will have a (+) charge, and if we emit a positron, it will have a (-)
Gamma Decay
we emit a photon, this is typically denoted by having the original atom that emits the photon have a next to it, so say, X è X … the (X) is just the same atom but with a removed photon
Neutron Decay
Neutron decay is the process by which a free “floating” neutron becomes a proton; when it decays it’ll emit an electron and a neutrino. All that I want you to gather from this is that going from neutron to proton forms an electron … thus if you perform the opposite of this “reaction” we would be losing a proton from the atom, as we make a neutron!
Electron Capture
when the atom captures an inner electron and combines it with a proton to form a neutron, our new atom will be the element before the original, since it’s missing a proton
Positive Controls
are those that ensure a change in the dependent variable when it is expected (something happens as expected)
Negative Controls
ensures no change in the dependent variable when no change is expected, usually related to the placebo effect. (ex. using water to grow a culture of bacteria against another culture that was given say sugar or something; we already expect that giving it water won’t fundamentally make it develop in an unexpected way)
Nothing happens, as expected
Validity is synonimous of
accuracy (picture a target, if you hit it dead on you are accurate)
Reliable is synonimous with
precision (picture a target, you don’t need to hit it dead on in this case, but if you hit the same area consistently you are precise (reliable), but multiple shots in random places are not)
Single-Blind Experiments
either the patient or the assessor may be “blinded” (not told) on certain information to help remove biases
Double-Blind Experiments
both the pt and assessor won’t know some information
Confounding Variable
an outside, unmeasured factor in a study that influences both the dependent (outcome) and independent (predictor) variables, creating a false or distorted relationship between them
Observational Studies
in medicine fit into 3 categories, which examine the connections between exposures and outcomes
Exposure = risk factors
Cohort Studies
we grab 2 groups, one that is connected to an exposure (risk factors, say smoking), and one that isn’t … then we monitor them for a certain amount of time (20 years), and see what happened to them (the outcome)
Cross-Sectional Studies
we look at a single point in time (for example, we look at how many smokers vs. nonsmokers have lung cancer right now)
Case-Control Studies
we start backwards, so we look for people with the expected outcome and track them back to assess what exposures they had contact with
Selection Bias
when we select the wrong ppl for a study, mainly b/c the makeup of our groups won’t match that of the general population
Detection Bias
when we know something already (like for example how hypertension and diabetes is higher in obese individuals, so a doctor might screen obese ppl for those two things a lot more than he would other people) … we detect a general trend and ride it
The FINER Method
o Assesses the value of a research question on the basis of whether or not it is
Feasible
Interesting
Novel
Ethical
Relevant
Equipoise
imagine before we finish a research, we learn that one of the treatments is better than the other, it would be unethical to continue the experiment, b/c by not giving the other group the better treatment you are causing harm