Biochem lec 22: Nitrogen fixation
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Nitrogen cycle
N2 to NH4+/amino acids to NO2-/NO3- to N2. (red arrows in anaerobic environments)
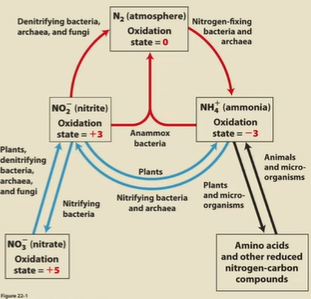
Nitrogen fixation
bacteria reduce N2 to NH3 (black arrows)
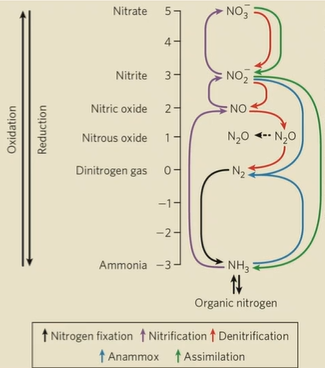
nitrification
conversion of ammonia to NO3- (purple arrows)
denitrification
conversion of NO3- to N2 (red arrows)
assimilation
plants and microorganisms reduce NO2- and NO3- to NH3 back into the soil (green arrows)
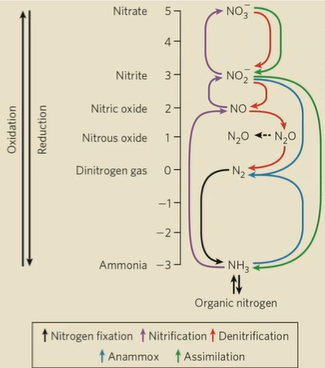
anammox (anaerobic ammonium oxidation)
specialized process where bacterial convert NH3 and NO2- directly into N2 under anaerobic conditions (blue arrows)
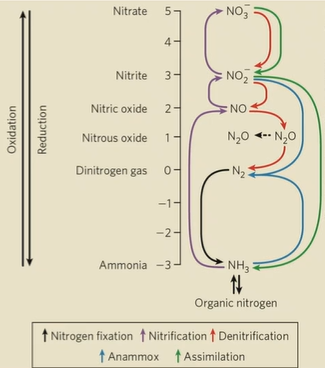
Oxidation of nitrogen compounds from oxidized to reduced
NO3-, NO2-, N2, NH3
Nitrogenase complex
N2 to NH3.
has 2 binding sites of ATP/ADP cofactors
Can be oxidized and reduced by 1 electron
central components: dinitrogenase reductase (iron-sulfur cluster), dinitrogenase (dimer)
in anaerobic baterica
dinitrogenase
heterodimer
P-cluster (Fe-S center) to transfer e-
FeMO cofactor to transfer e-
8 e- transferred from dinitrogenase reductase to dinitrogenase
each turn of the cycle requires 2 ATP
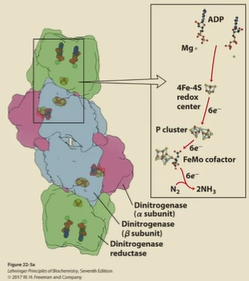
nitrogen fixation procedure
2 ADP bound to dinitrogenase reductase, electron is reduced and held by enzyme
2 ATP replace 2 ADP to induce conformational change
Dinitrogenase binds to nitrogenase reductase complex
electron transfer
ATP hydrolysis and Pi release
complex dissociation, repeat
redox reactions in nitrogenase stoichiometry
N2 + 8H+ + 8e- + 16 ATP = 2 NH3 + H2 + 16 ADP + 16 Pi
dinitrogenase reductase catalyzes
transfer of 8e- to dinitrogenase and hydrolysis of ATP with release of protons
dinitrogenase catalyzes
transfer of 6 e- to nitrogen to form NH3 and transfer of 2 e- to protons to form H2
why is nitrogenase only in bacteria
the enzyme is sensitize to oxygenation and requires anaerobic conditions
how do bacteria maintain anaerobic environment?
burn off O2 with partially uncouples electron transfer
specialized thick walled called to exclude O2
plants evolved to provide symbiotic niche for bacteria to fix N2 for them via rhizomes
gut bacteria’s ability to fix N2 to NH3
production of amino acids from ammonia
NH3 made into glutamate and glutamine
glutamine synthesis
enzyme: glutamine synthase converts glutamate to glutamine
glutamate + ATP (glutamate kinase) —> intermediate
intermediate + NH4+ —> glutamine + Pi + H+
rxn: glutamate + NH4+ + ATP —> glutamine + ADP + Pi + H+
glutamate synthesis
enzyme: glutamate synthase converts glutamine + alpha-KG to 2 glutamates
rxn: alpha-KG + glutamine + NAD(P)H + H+ —> 2 glutamate + NAD(P)+
net reaction of ammonia incorporation into biomolecules
alpha-KG + NH4+ + NAD(P)H + ATP —> glutamate + NAD(P)+ + ADP + Pi
glutamine synthase
in all organisms, primary regulatory point in N2 metabolism. Many things required to shut it off
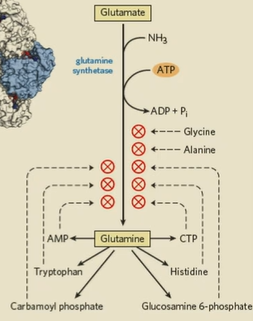
glutamate synthase
only in bacteria and plants, not animals
where does glutamate come from?
transamination of alpha-ketoglutarate and amino acid catabolism
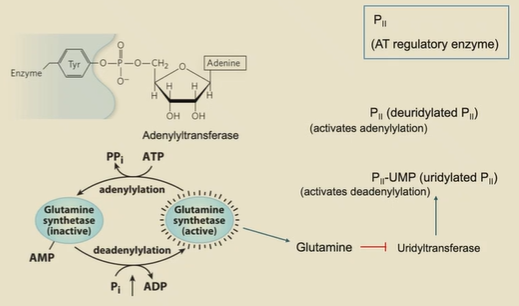
how is glutamine synthase covalently inhibited?
tryosine in active site needs to be phosphorylated to be active. If this tyrosine is adenylated (via adenylyltransferase and ATP), it won’t work.
P2 regulates adenylytransferase
uridylated P2 (via Uridyltransferase, which is inhibited by glutamine) activates deadenylation
P2 activates adenylation
what amino acids are made from glutamate?
proline, arginine, glutamine
how is proline made from glutamate?
glutamate kinase phosphorylates glutamate
NADPH + H+ cofactor used to reduce phosphorylated glutamate to a semialdehyde
nonenzymatic spontaneous cyclization to P5C
pyrroline carboxylate reductase reduces P5C using NADPH + H+ to get proline
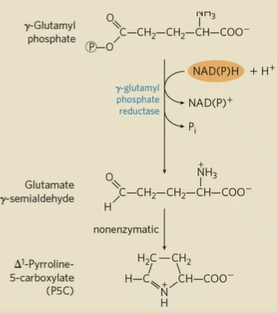
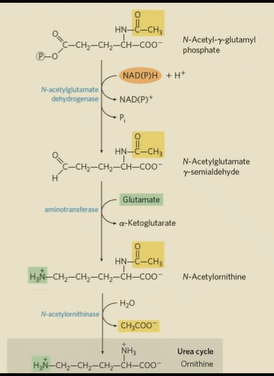
how is arginine made from glutamate?
gamma-glutamyl phosphate reduced by NADPH or NADH to glutamate gamma-semialdehyde
another glutamate is used for transamination
acetyl group is removed to make ornithine
Urea cycle to turn ornithine into Arginine
proline synthesis from arginine
reverse urea cycle reaction where ornithine is converted to proline