2026 OC Regional Science Olympiad: Materials Science
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
Metal nanomaterials
Composed of pure metals at nanoscale (ex: gold, silver, platinum nanoparticles)
Metal nanomaterial applications
Catalysis, medical imaging, electronics

Metal-oxide nanomaterials
Composed of metal oxides (ex: titanium dioxide, zinc oxide)

Metal-oxide nanomaterial applications
Sunscreens, photocatalysts, sensors

Ceramic nanomaterials
Inorganic, non-metallic solids (high hardness, heat/corrosion resistant)
Ceramic nanomaterial applications
Aerospace, cutting tools, biomedical implants

Semiconductor nanomaterials
Quantum confinement affects electronic/optical properties (ex: cadmium sulfide, zinc selenide, silicon nanowires)
Semiconductor nanomaterial applications
Solar cells, LEDs, transistors

Carbon-based nanomaterials
Composed of carbon atoms at nanoscale
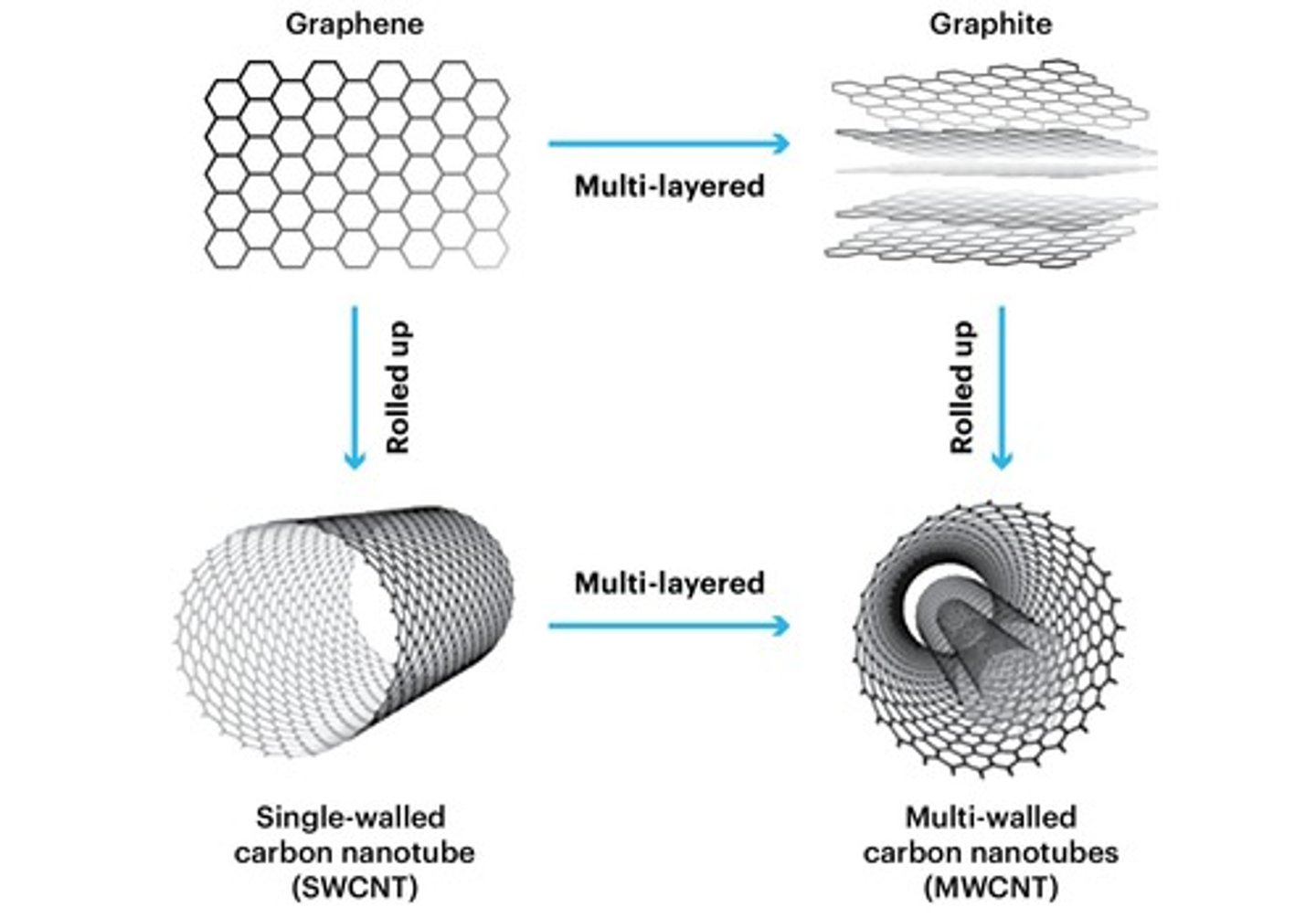
Graphene
Single layer of carbon atoms

Fullerenes
Spherical carbon cages
Carbon nanotubes
Cylindrical tubes of carbon
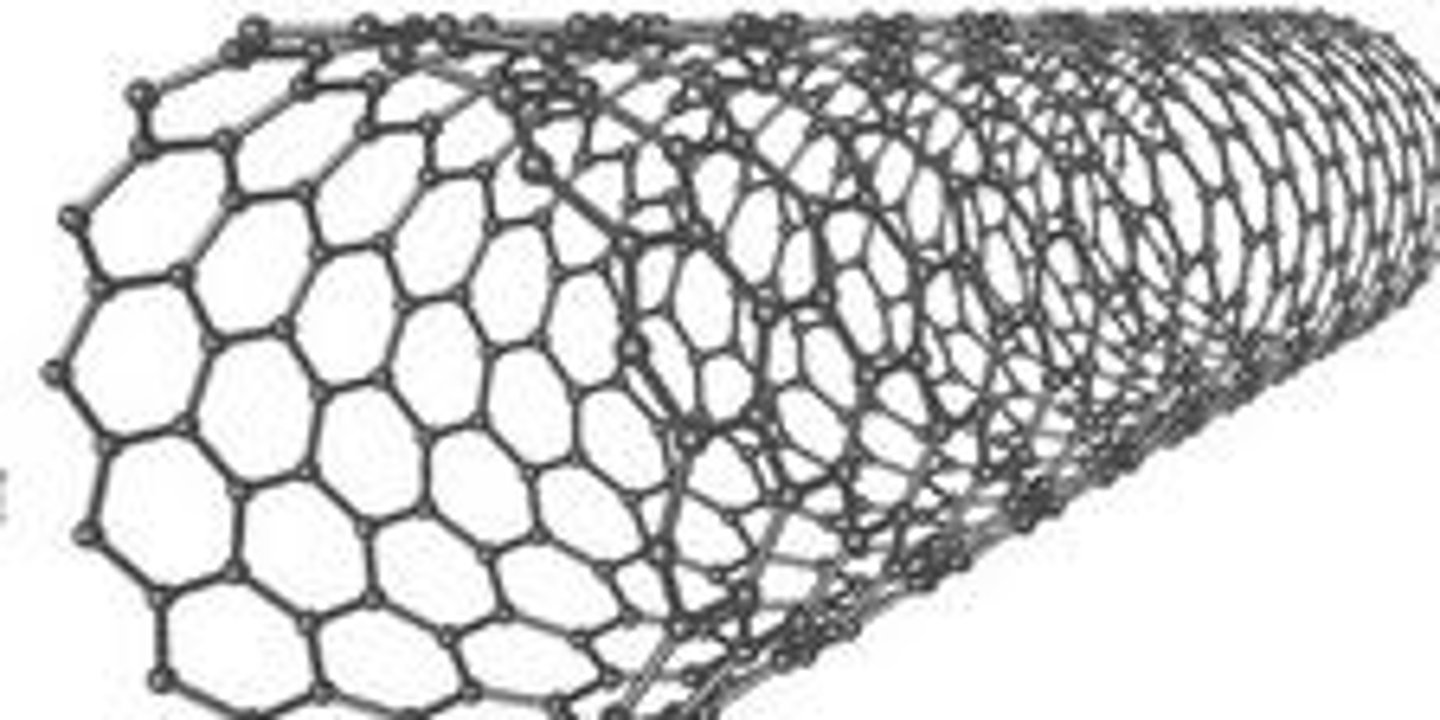
Carbon-based nanomaterial applications
Composites, electronics, energy storage
Nanomaterial dimensionality for application: 0D (zero dimensional)
Drug delivery, quantum dots in displays
Nanomaterial dimensionality for applications 1D (one dimensional)
Nanowires in sensors, nanotubes in composites
Nanomaterial dimensionality for applications 2D (two dimensional)
Graphene-based membranes, coatings
Nanomaterial dimensionality for applications 3D (three dimensional)
Nanoporous catalysts
Nanofluids
Fluids containing suspended nanoparticles
Nanofluid applications
Cooling systems, medical therapies
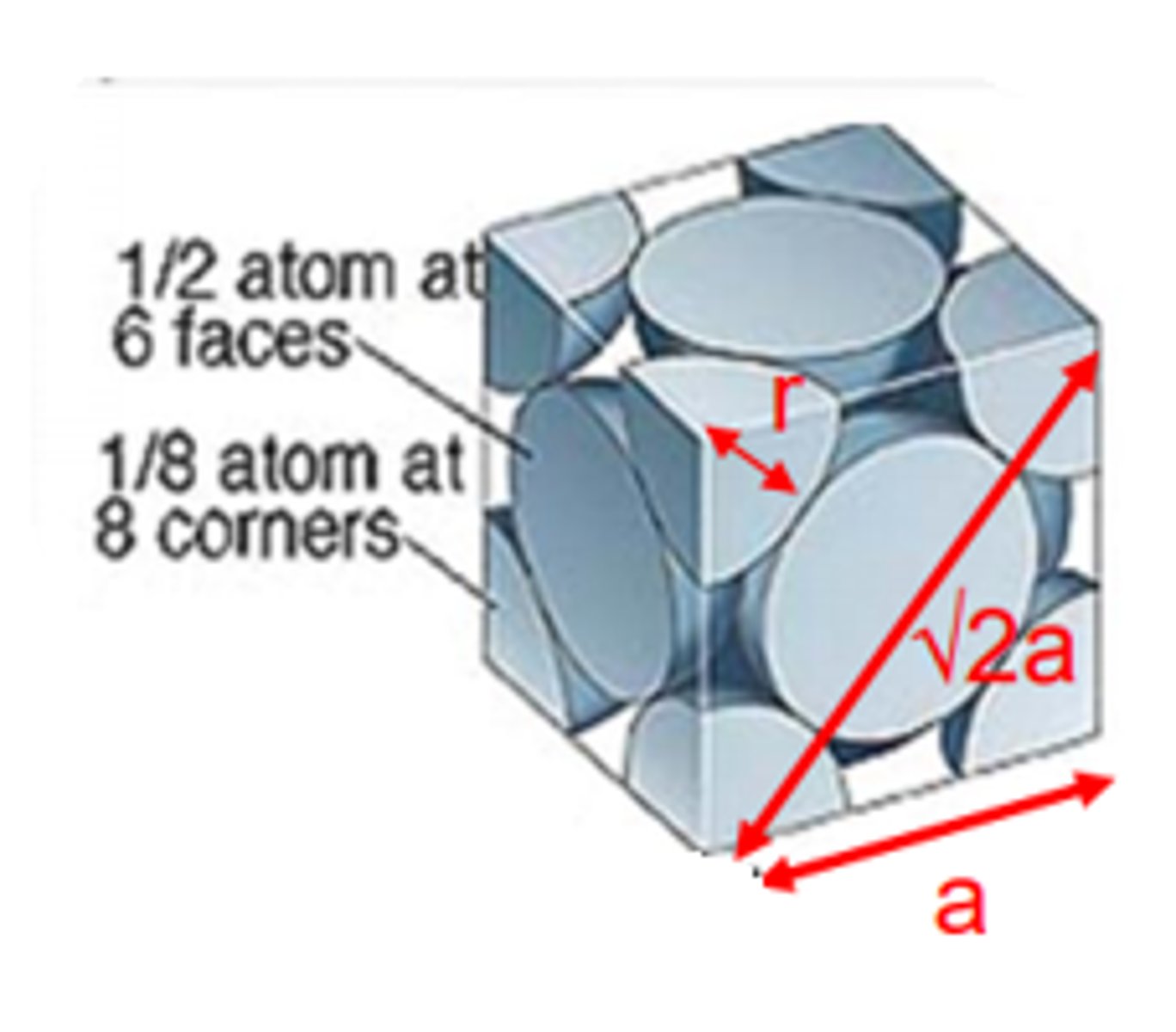
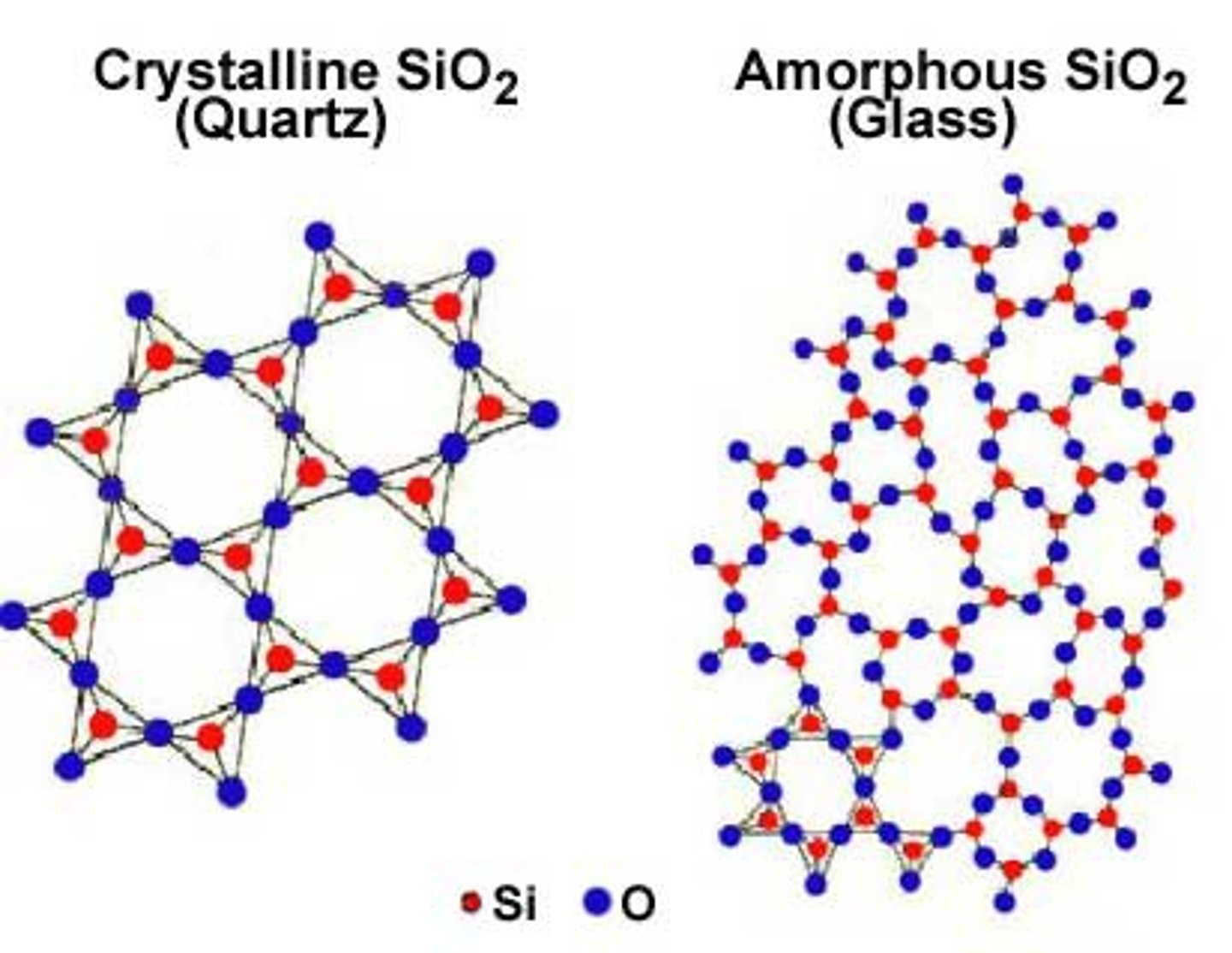
Crystalline structure
Atoms arranged in repeating 3D patterns
Common lattices
FCC, BCC, HCP
Amorphous nanomaterials
Atoms randomly arranged (not uniform)
Types of interatomic bonding
Ionic, covalent, metallic
Ionic bond
Nonmetal + Metal (gives)
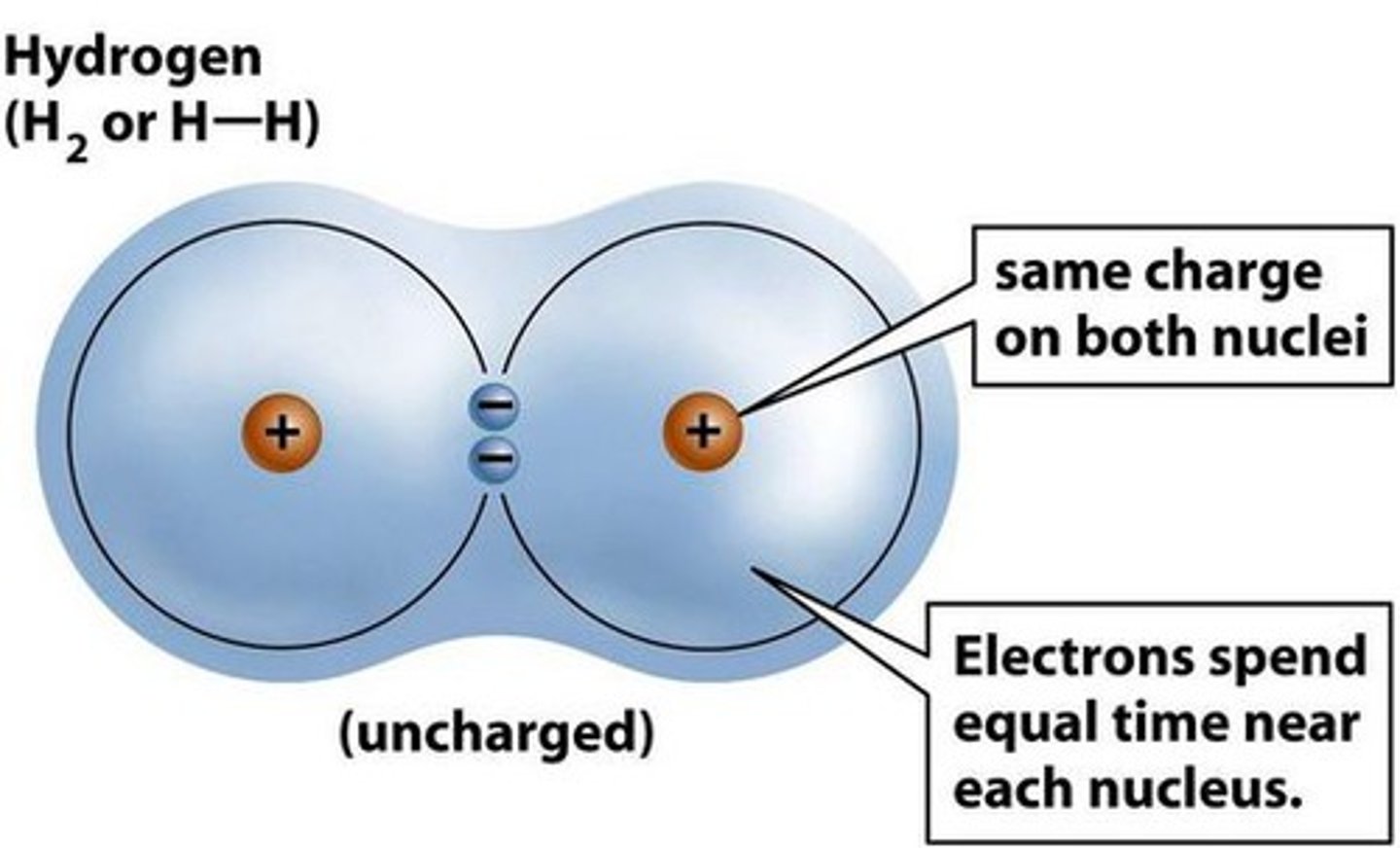
Covalent bond
Nonmetal + Nonmetal (shares)
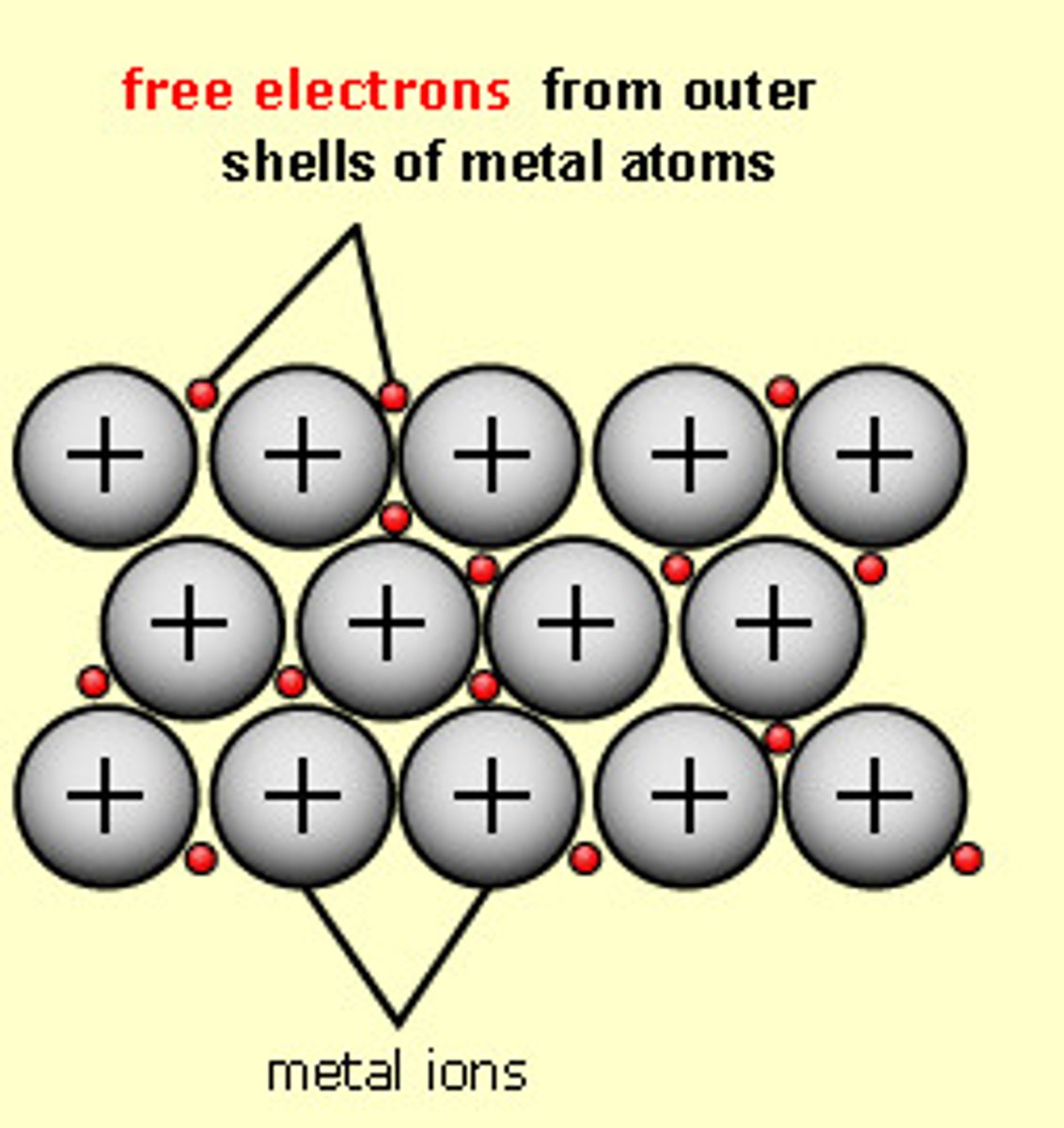
Metallic bond
Metal +Metal
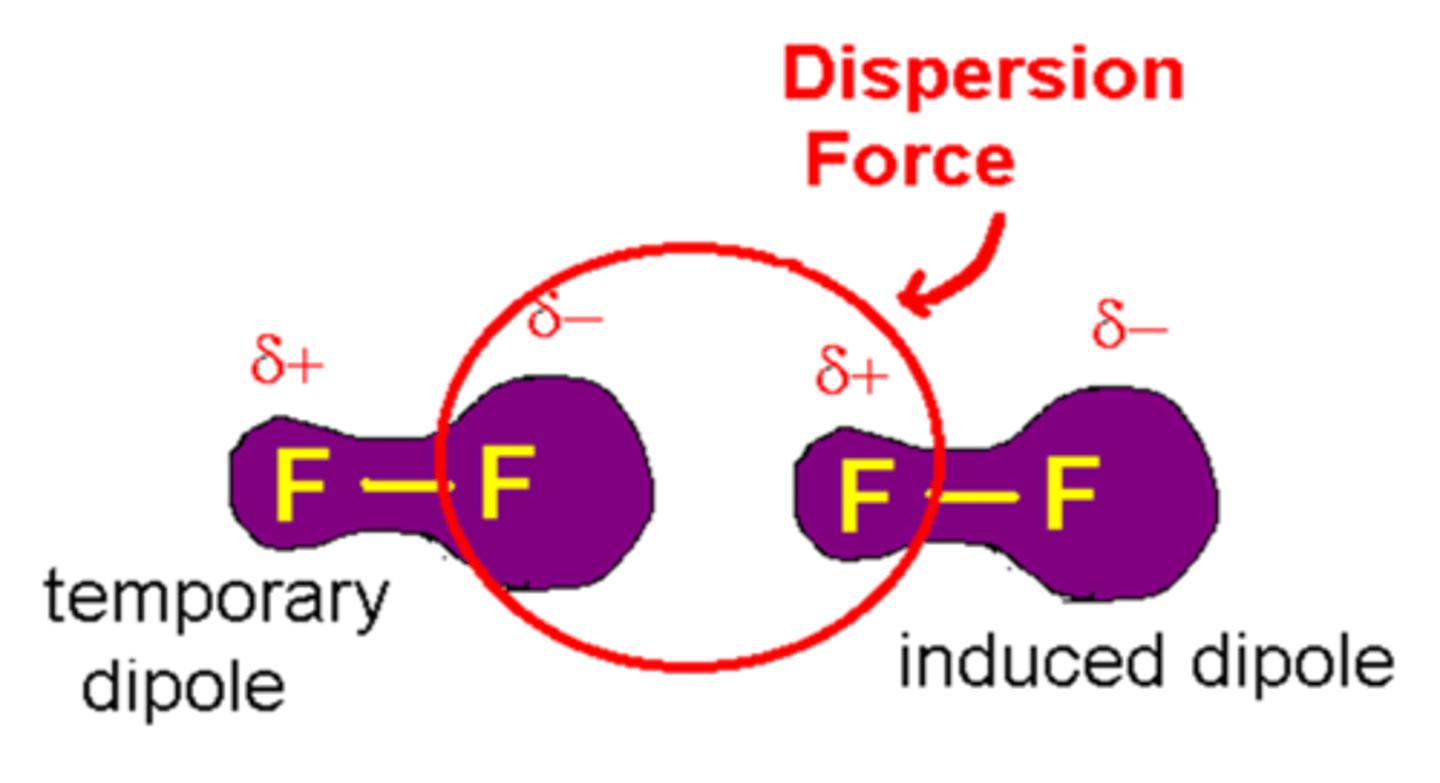
Van der Waals
Weak, short-range attractions between molecules caused by magnetic poles
Hydrogen bonding
Strong type of intermolecular dipole-dipole attraction. Occurs between hydrogen and F, O or N
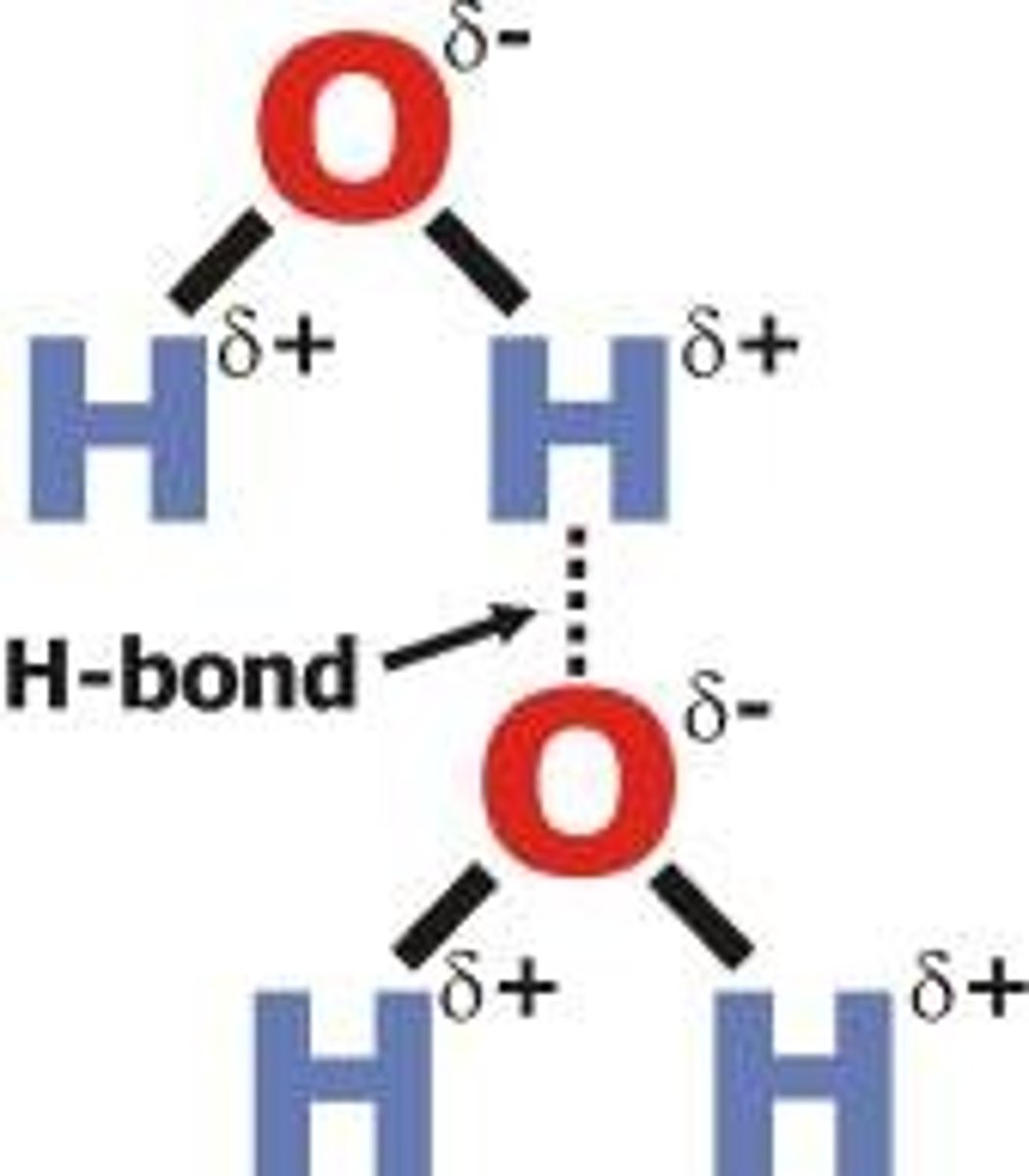
Dipole-dipole interactions
Attractive forces between polar molecules
Grain boundaries
The boundaries between crystals (grains) in a polycrystalline material
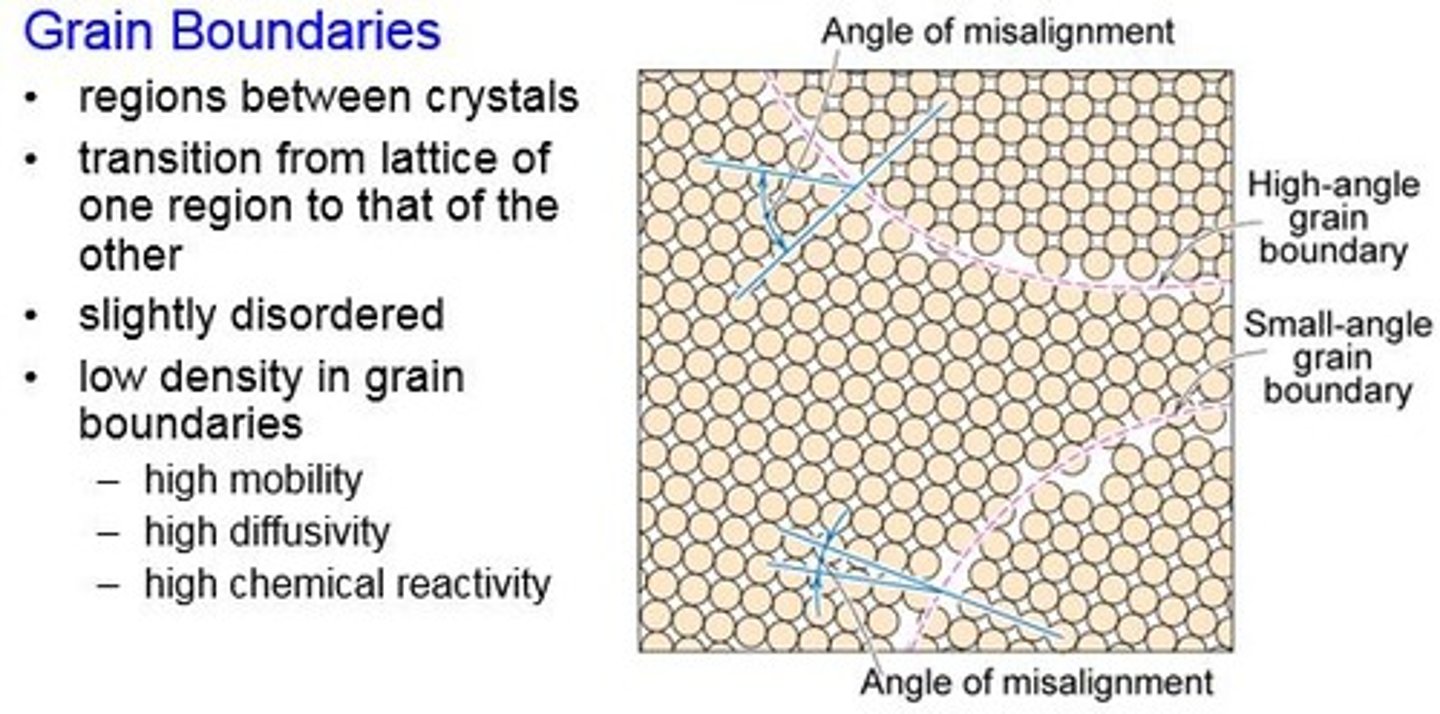
Hall-Petch relationship
Strength increases as grain size decreases

Surface area to volume ratio
As an object gets smaller, the surface area drastically increases compared to its volume

Surface energy
Energy required to create a unit of surface
Surface tension
For force per unit length at surface

Wetting angle
Measures how a liquid spreads on a surface

Hydrophobic
Large contact end, repels water
Hydrophilic
Small contact end, attracts water
Surfactants
Molecules with hydrophilic head and hydrophobic tail
Top-Down Synthesis
Breaking down bulk materials into nanomaterials (big -> small)
Top-Down: Mechanical Milling (4 steps)
1.) Bulk material placed into a heavy-duty container (mill)
2.) Heavy metal balls added
3.) The container spins/moves quickly, smashing the bulk material repetitively
4.) Result: nanomaterial
Top-Down: Lithography (4 steps)
1.) Solid flat slab of material
2.) Stencil is made using light, electron beams, or ions
3.) Chemicals or plasma remove the unmarked parts
4.) Smaller structure is the result
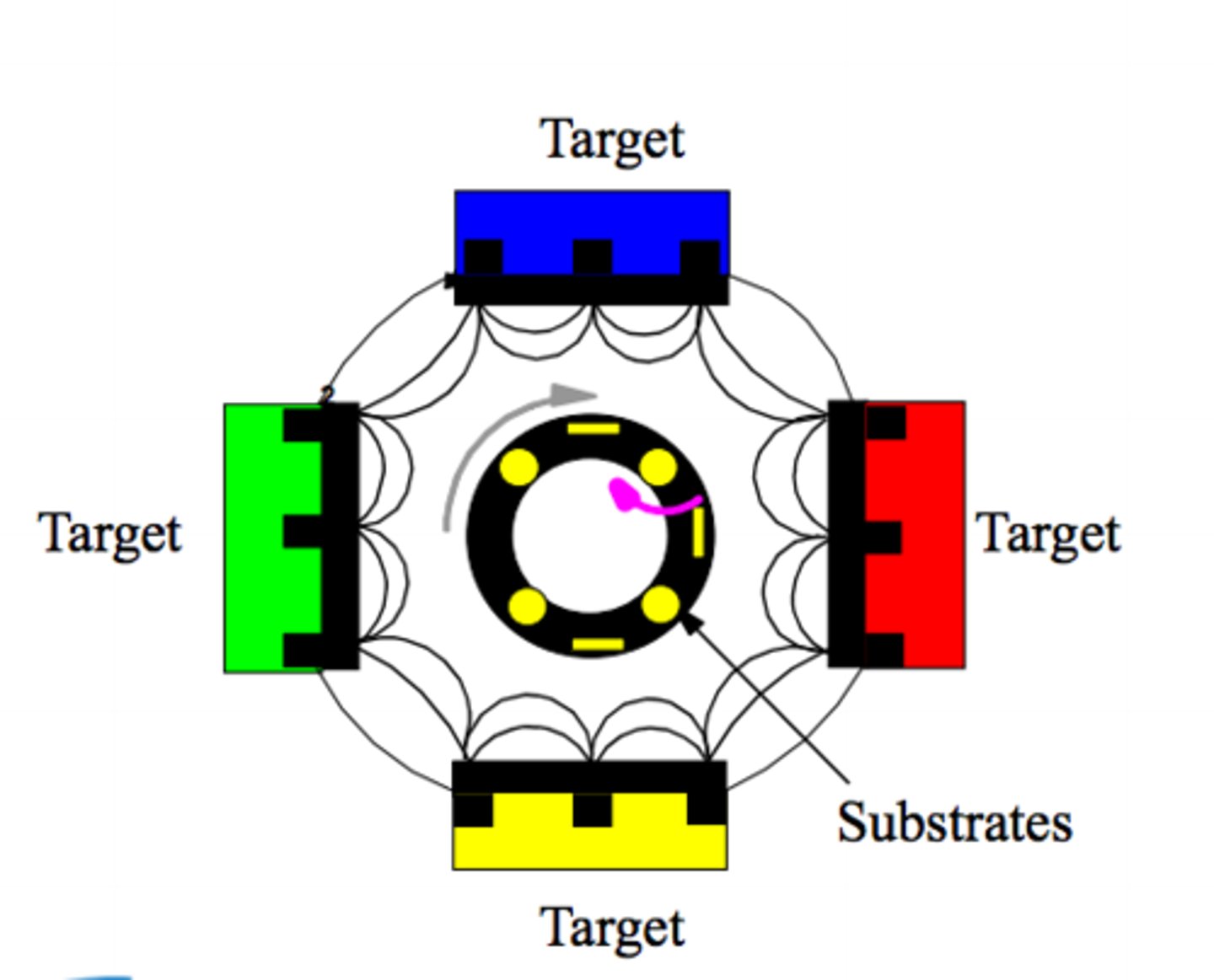
Top-Down: Sputtering (4 steps)
1.) Solid slab of material (the target) and object to coat (substrate) -> vacuum-sealed chamber (heavy gas)
2.) High voltage applied to gas -> plasma (fast ions attracted to target)
3.) Individual atoms removed from the bulk
4.) Loose atoms fly and land on the substrate
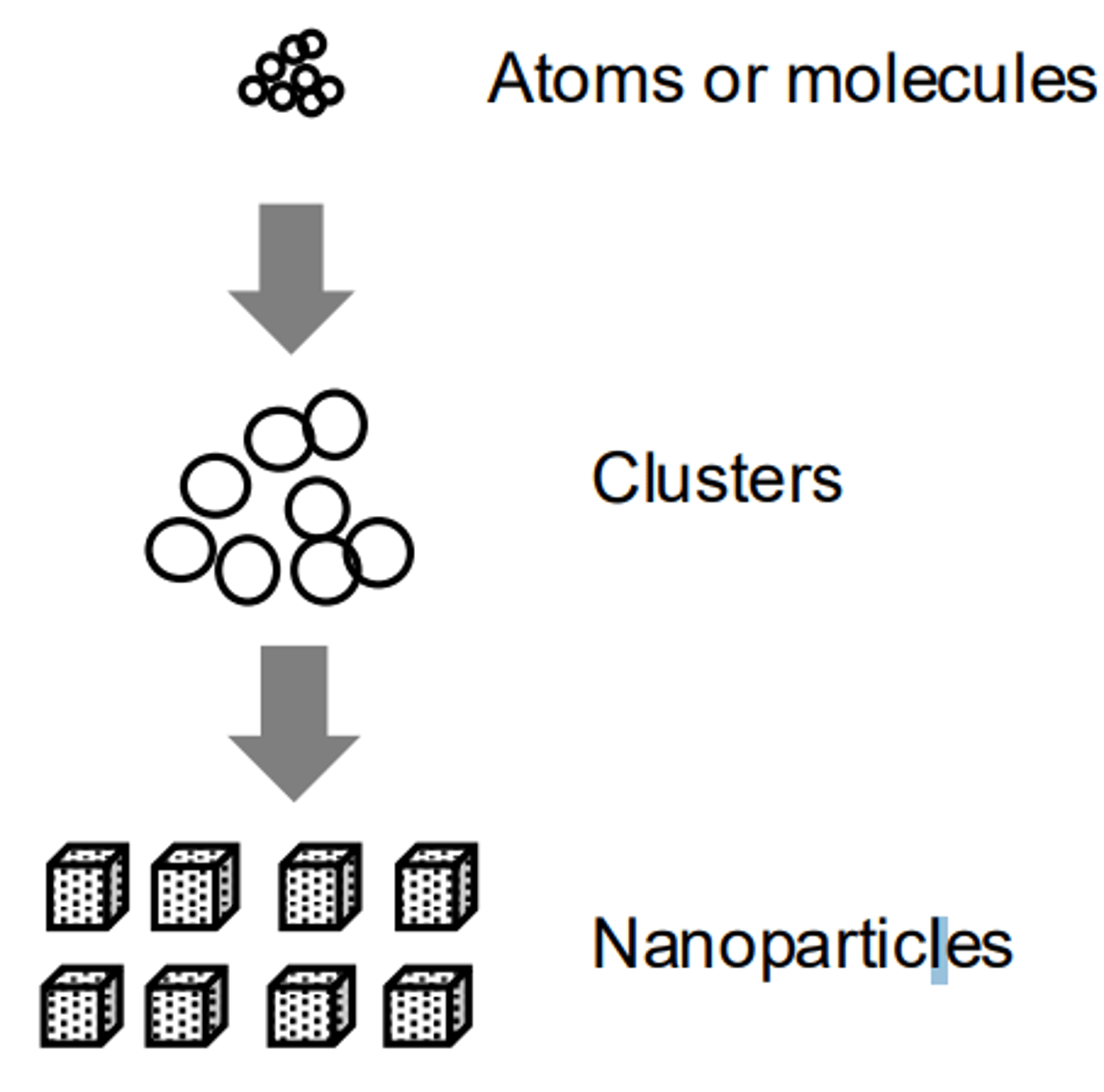
Bottom-Up Synthesis
Building nanostructures by stacking atoms or molecules on top of one another
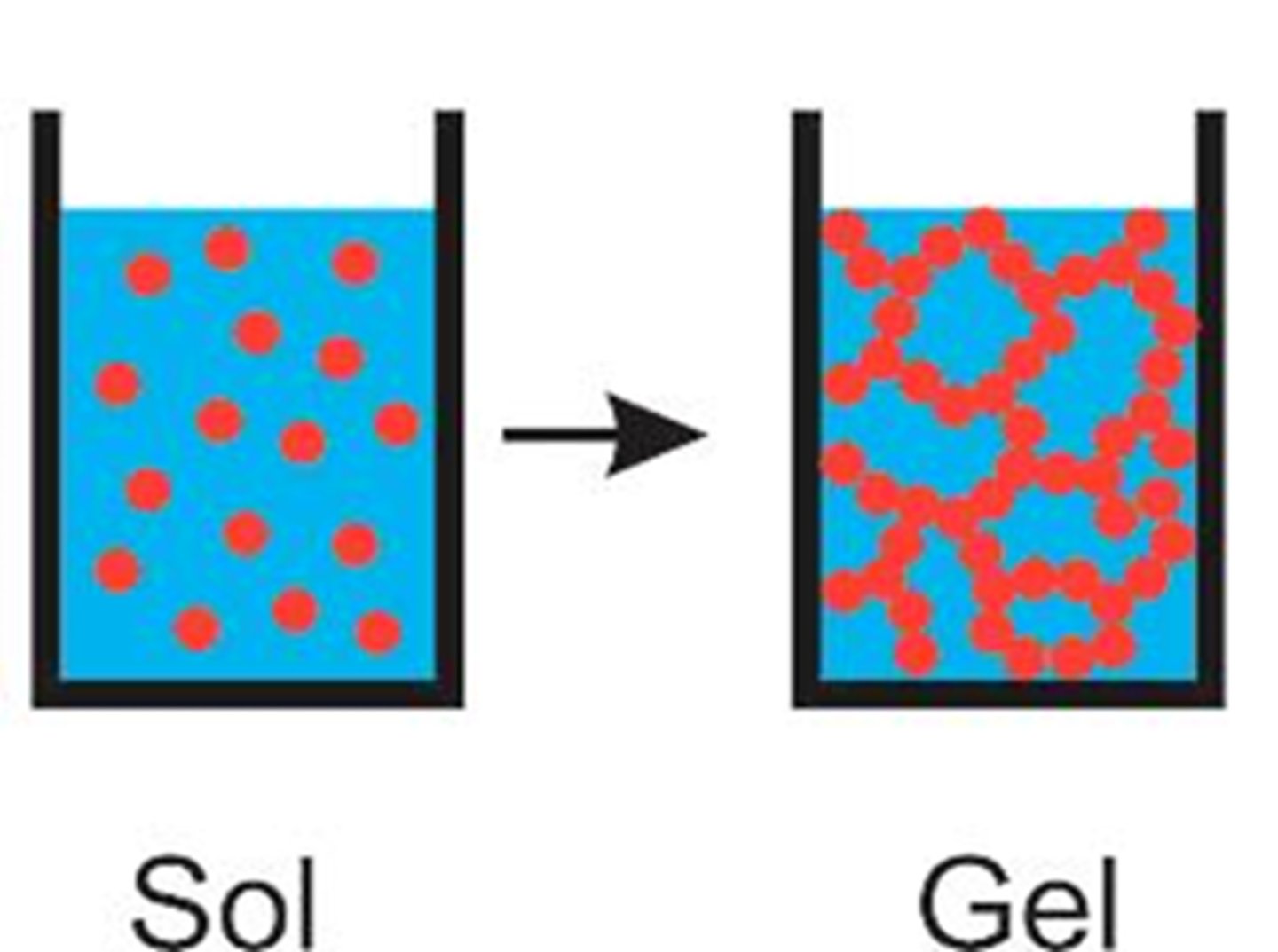
Bottom-Up: Sol-Gel Process (4 steps)
1.) Form a liquid from chemicals (sol)
2.) Chemicals link together -> wet solid web (gel)
3.) Remove surrounding liquid -> dry, stiff sponge left
4.) Heat in an oven -> hard glass/ceramic
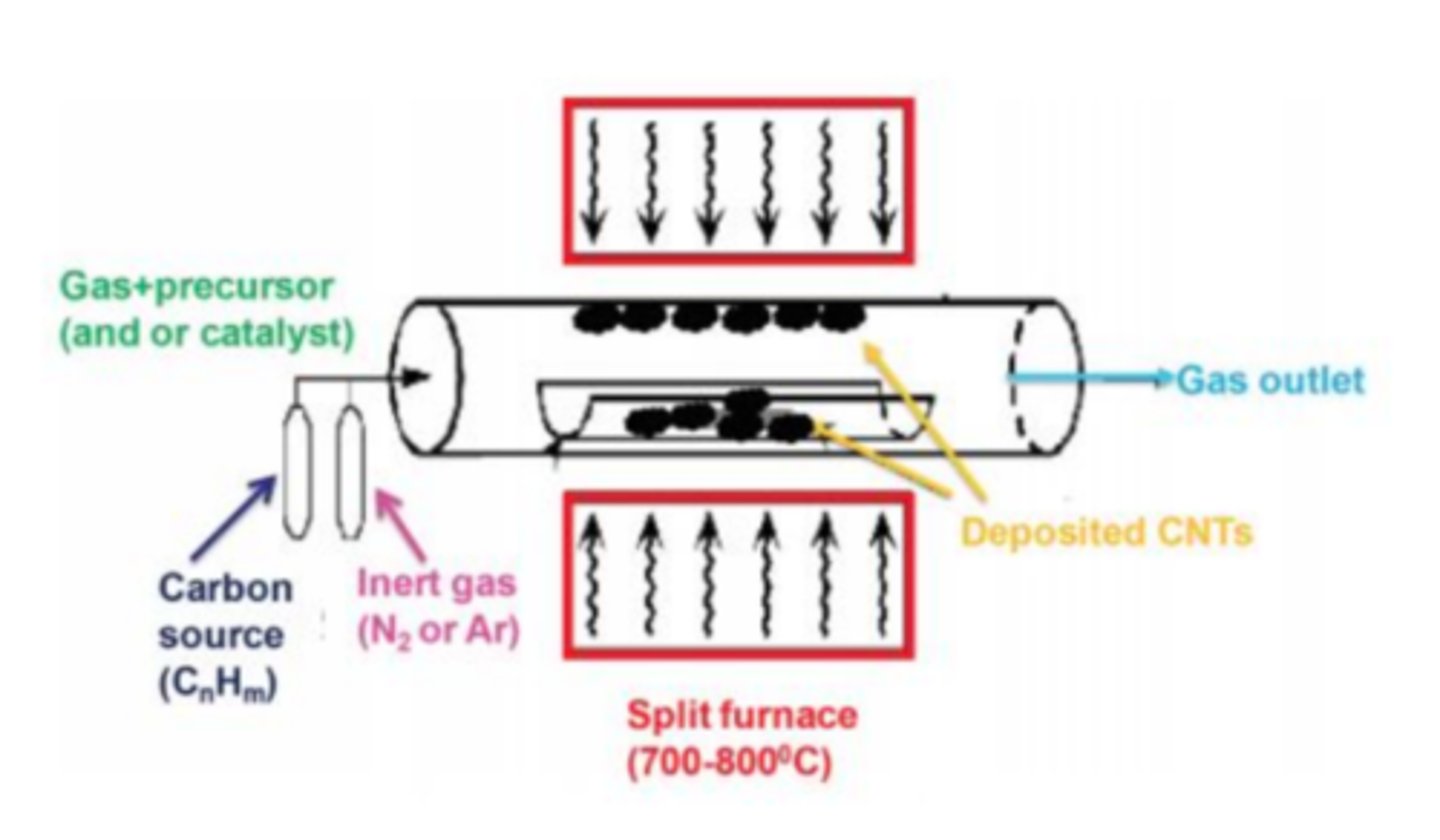
Bottom-Up: Chemical Vapor Deposition (CVD) (4 steps)
1.) Chemical gases pumped into vacuum chamber
2.) Heat/plasma breaks gas molecules -> individual atoms
3.) Atoms stick to a surface (substrate)
4.) Atoms link together -> ultra-thin layer
Bottom-Up: Self-Assembly (3 steps)
1.) Molecules w/ sticky ends put into a liquid or gas environment
2.) Molecules find each other
3.) Molecules snap together -> nanostructure
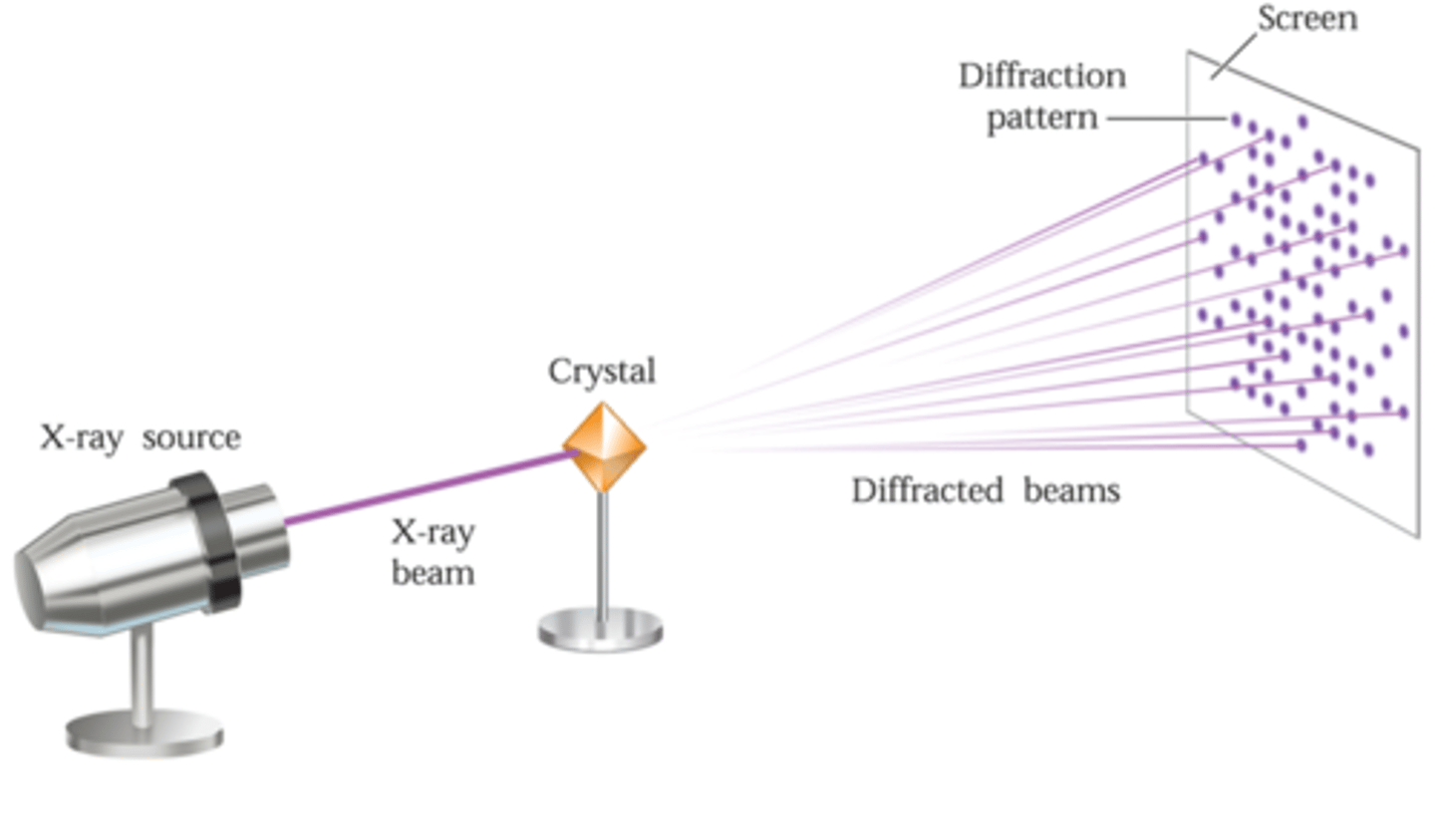
X-Ray Diffraction (XRD)
Non-destructive technique that uses X-rays to view the internal atomic structure
Light microscopy
Uses visible light to magnify samples
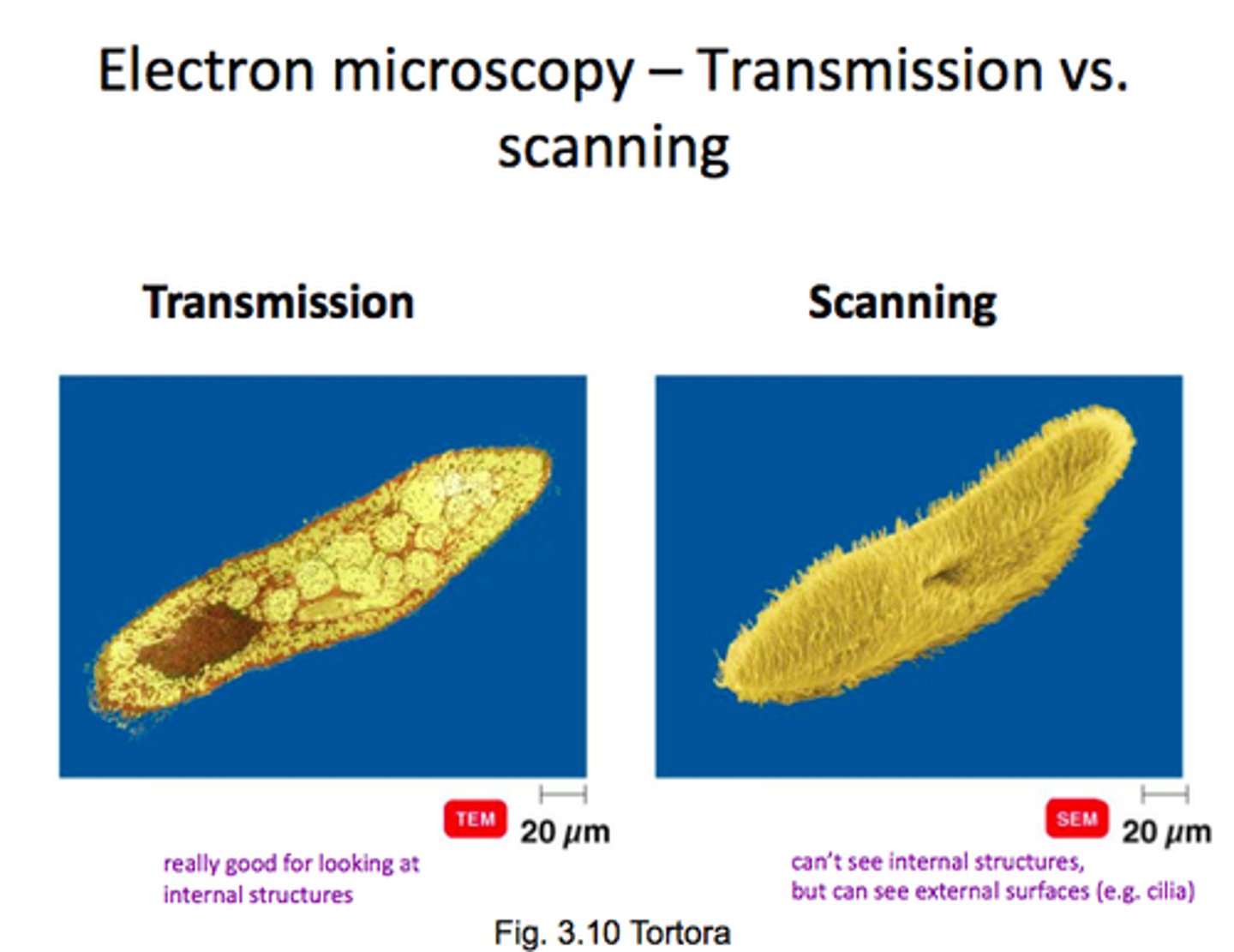
Electron microscopy (2 types/their functions)
1.) Scanning Electron Microscopy (SEM): Surface imaging
2.) Transmission Electron Microscopy (TEM): Internal structure
Scanning probe microscopy (2 types/their functions)
1.) Atomic force microscopy (AFM): Measures surface forces
2.) Scanning Tunnel Microscopy (STM): Maps electron density

UV-Vis
Measures light-absorption across UV-visible range
Photoluminescence
Emission of light after excitation
Energy dispersive spectrometry
Identifies elemental composition in electron microscopes
Mass spectrometry
Measures mass-to-charge ratios
Melting temperature
The temperature when a solid becomes a liquid
Density of nanomaterials
Mass per unit of volume of a material (decreases at nanoscale)
Hardness
A material's resistance to being scratched/dented

Strength
Material's ability to maintain shape under stress
Elastic modulus
A measure of the stiffness of a material
Adhesion
Force required to separate two forces
Wear
Material lost due to friction or contact
Scattering
Redirection of light by particles
Absorption
Conversion of light -> energy
Reflection
Light bouncing off of the surface
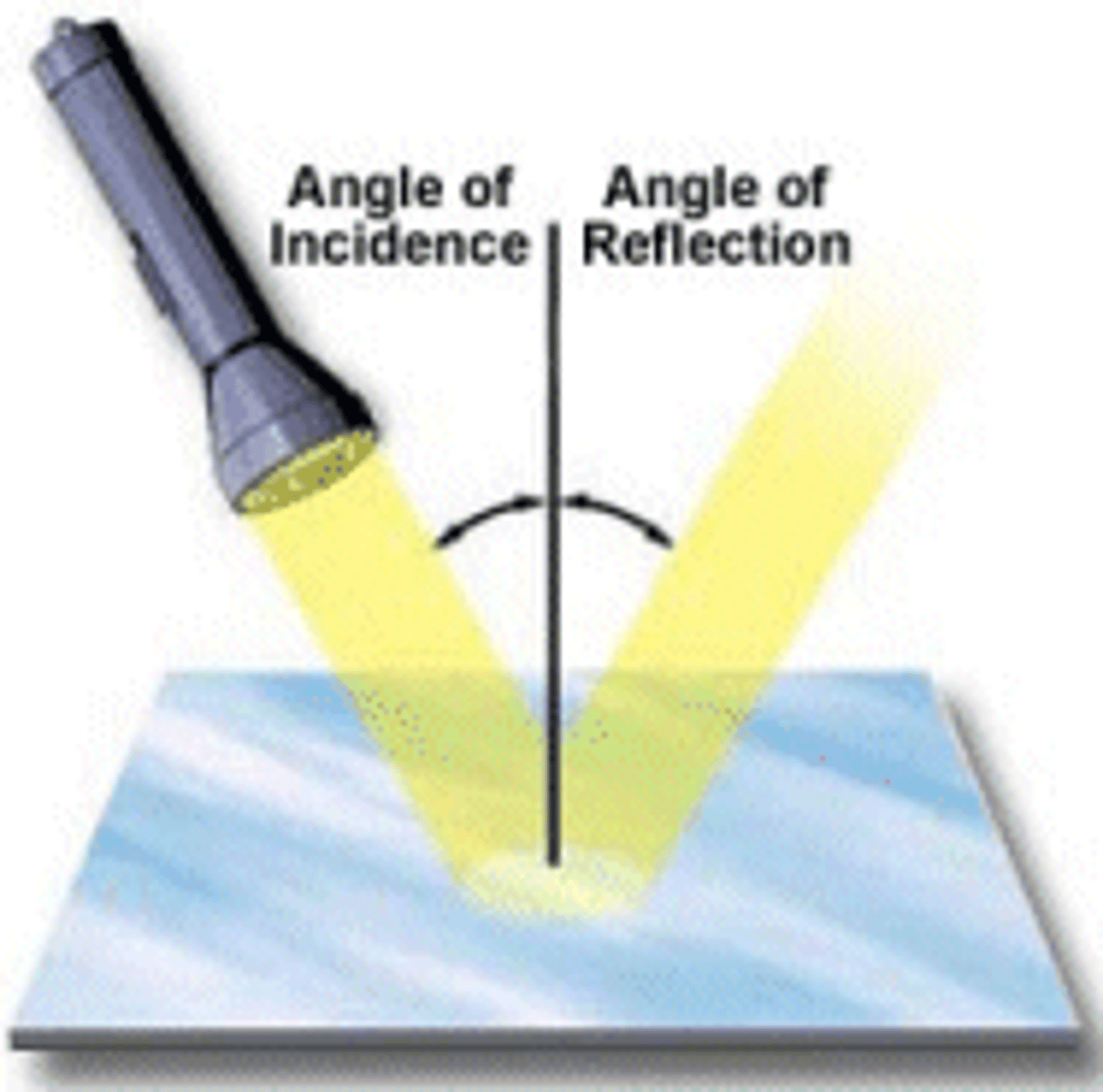
Refraction
Vending of light entering a material

Thermal conductivity
Ability of a material to conduct heat
Electrical conductivity
Movement of electrons through material
Dielectric properties
Material's ability to store electrical energy