CHEM 308 Lab Final Exam Boise State
1/134
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
135 Terms
Thin-Layer Chromatography (TLC) is used to...
1. Determine the purity of a sample
2. Identify a compound (this experiment)
3. Monitor the progress of a reaction
4. Check the effectiveness of a separation achieved by column chromatography (second part of experiment)
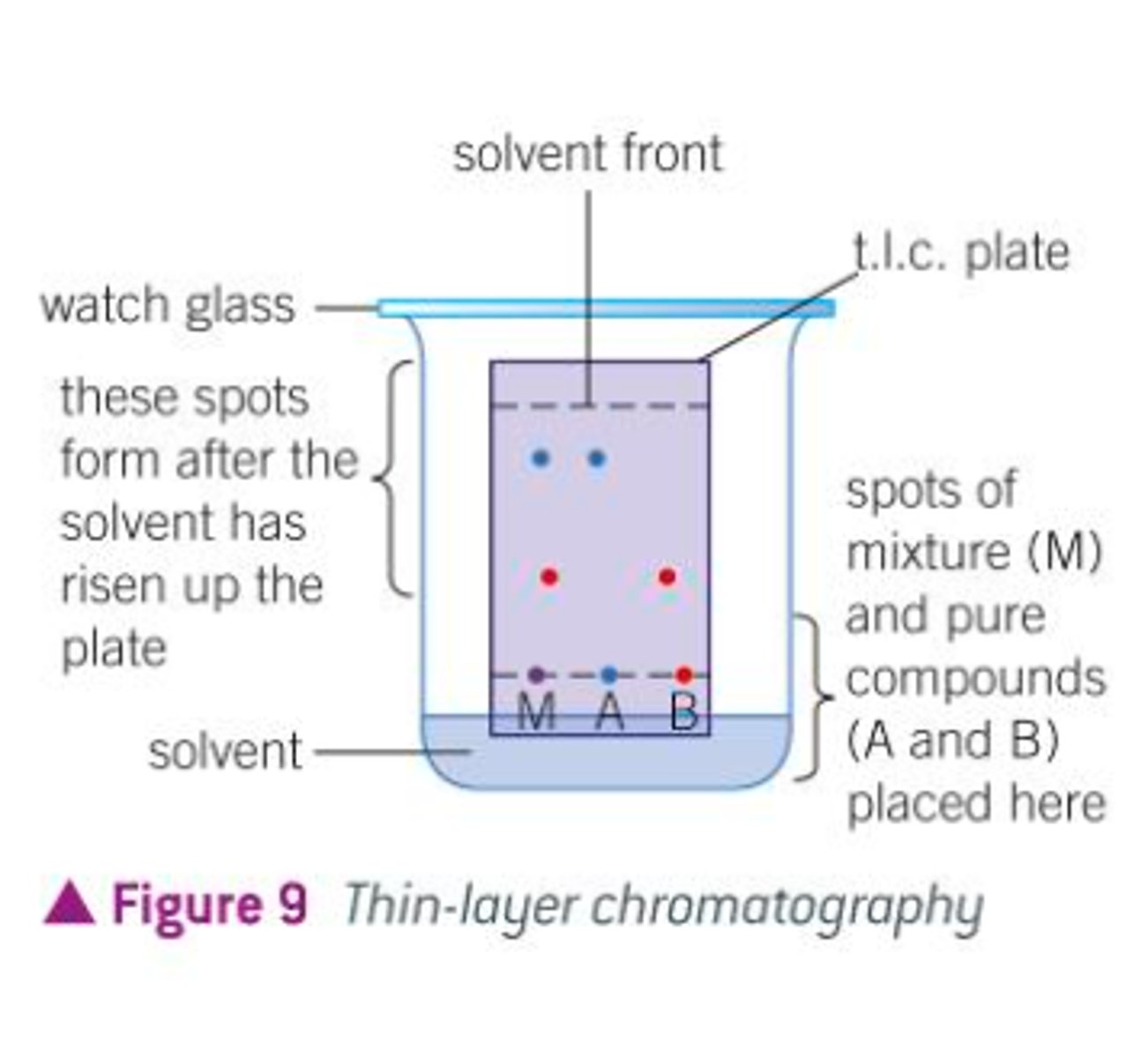
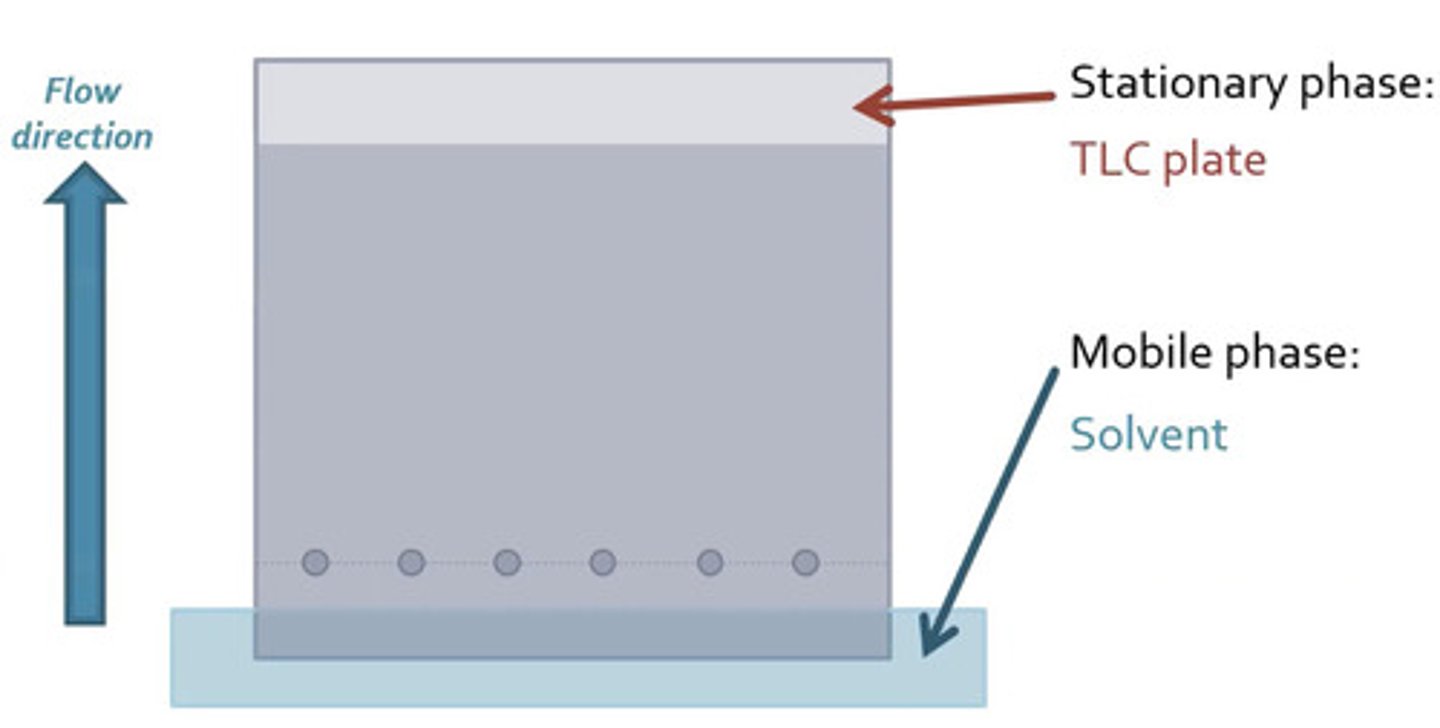
TLC stationary phase and mobile phase
silica gel or alumina; liquid
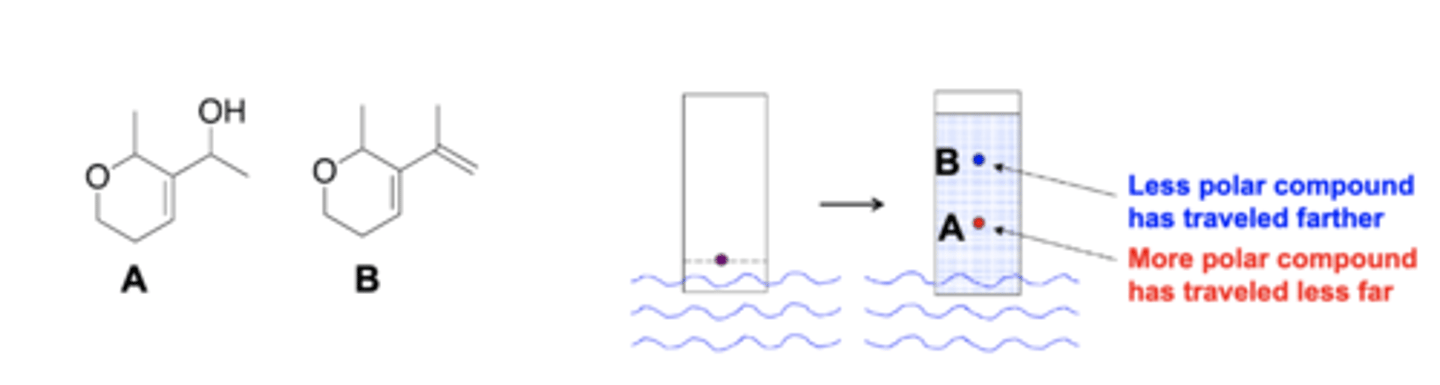
Which compound is the most hydrophilic?
B: O and OH on a molecule think polar
polar = hydrophilic (strong interactions with silica gel; wont move far up the plate)
Which compound will move up the plate the farthest, A or B?
B: less polar so it will travel farther; wont interact with silica gel
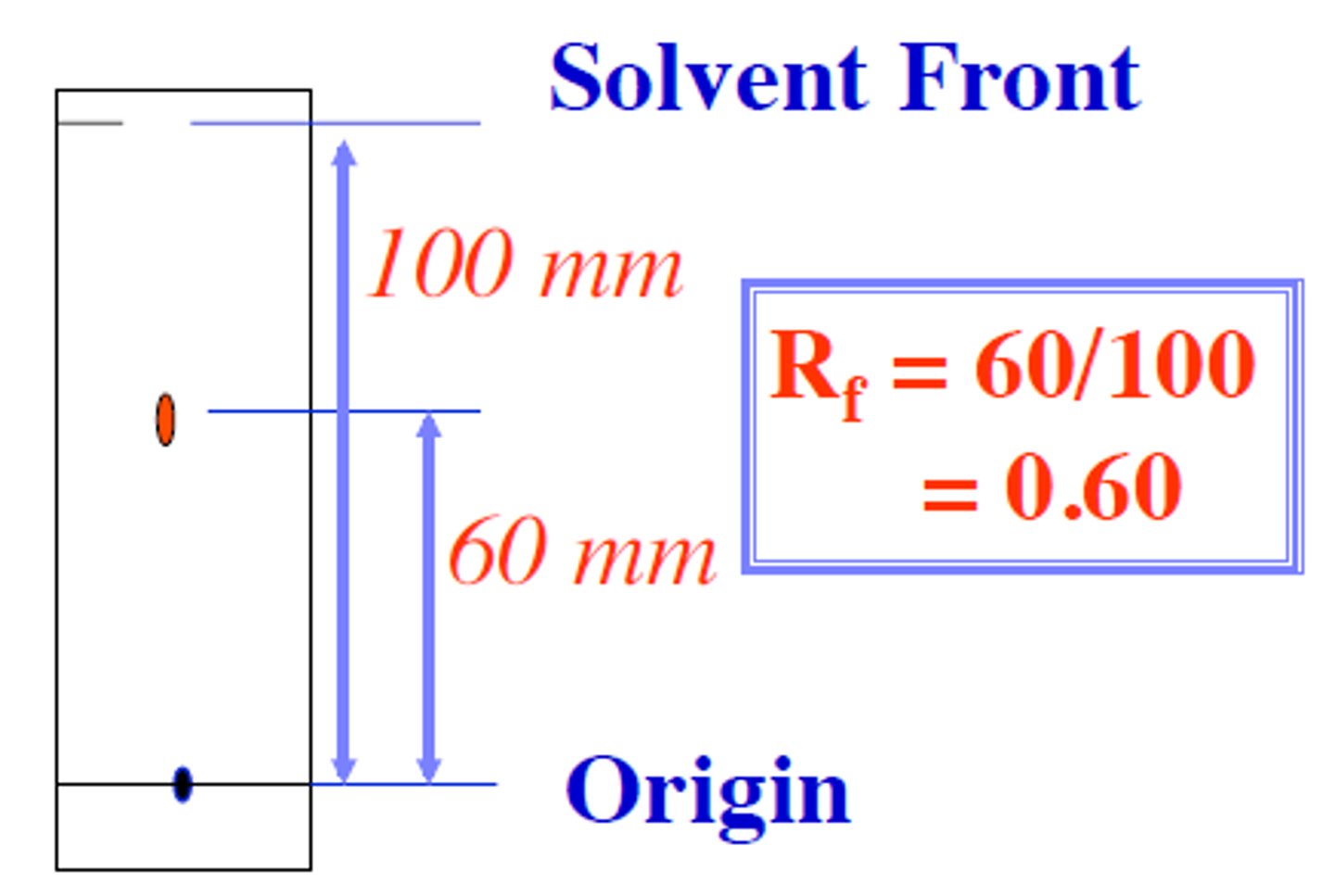
Rf factor
Rf = ratio of the distance traveled by a compound relative to the distance traveled by the solvent
Rf values depend on the stationary phase (usually silica gel), the polarity of the compounds,and the polarity of the solvent(s)
MORE POLAR THE COMPOUND THE SMALLER THE RF
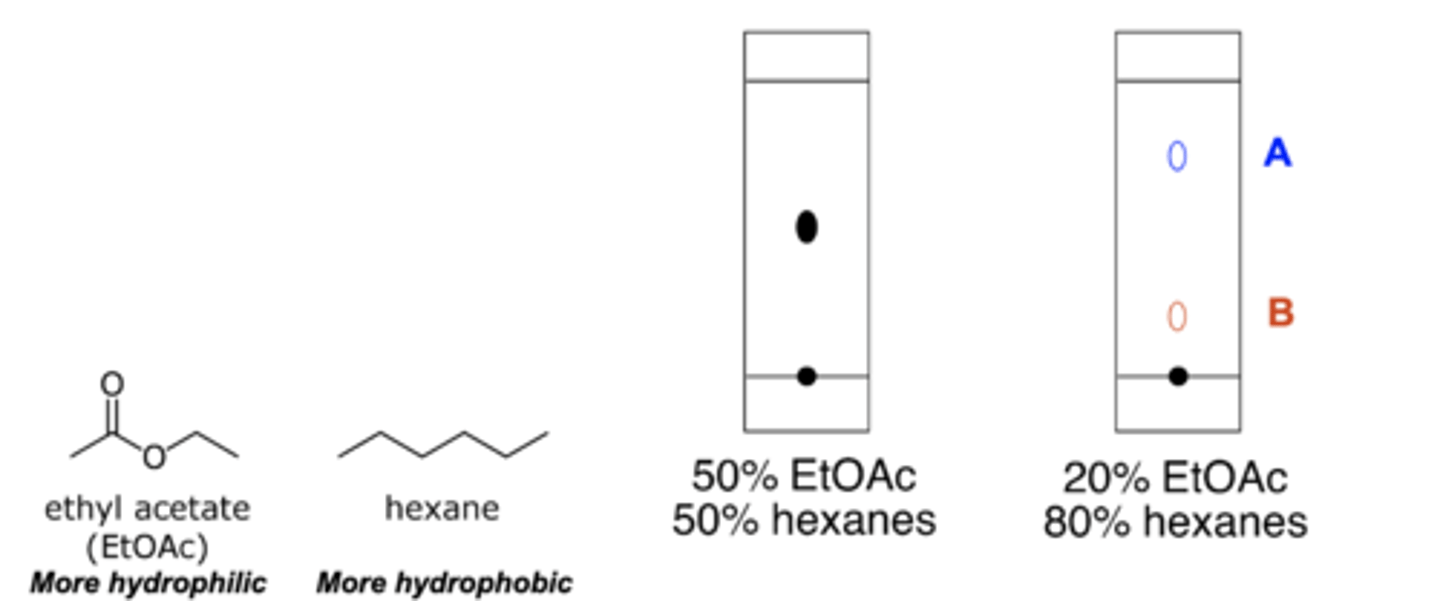
The TLC plate on the left was run in 50% ethyl acetate and 50% hexanes. If a TLC plate with the same compound was run in 20% ethyl acetate and 80% hexanes, will the spot appear at location A or location B?
B: a more nonpolar solution so the compound will appear lower
Common mistakes with TLC
Letting solvent level cover the baseline
Touching plates with bare hands (this leaves fingerprints from the oils on your hands)
Using pen and not pencil (why might this be a problem?)
Spotting too much sample
Which of the following is an incorrect way to set up a TLC experiment?
a. Carefully lay the plate down flat in the solvent on the bottom of the eluting chamber.
b. Limit the amount of eluting solvent so its level is below the origin line of the plate.
c. Seal the chromatography chamber so solvent vapors cannot escape.
d. The sample should be applied sparingly about 1 cm from the bottom of the plate.
A
Which would move further on a silica gel TLC plate, a polar compound or a nonpolar compound?
a. A polar compound
b. A nonpolar compound
c. They would move the same distance.
d. One is unable to tell without knowledge of the solvent used.
B
In TLC on silica gel or alumina,
a. A nonpolar solvent will move a polar compound further than a polar solvent will.
b. A nonpolar compound will move further up the plate than a polar compound.
c. A polar compound will move further up the plate than a nonpolar compound.
d. The plate is typically coated with nonpolar material.
B
What does the term 'elution' mean in TLC?
a. Elution is the movement of sample and mobile phase on the adsorbent.
b. Elution refers to the increase in the size of spots as they travel along a TLC plate.
c. Elution is the feeling of joy one feels when an experiment works.
d. Elution is the separation of a mixture into separate components (or spots)
A
Which of the following results indicates that a different developing solvent should be used?
a. All of the above
b. The spots cannot be clearly differentiated.
c. Spots don't move.
d. All samples move with the solvent front.
A
Which one of the following statements is always true of an Rf value?
a. A compound will have the same Rf value in all solvent systems.
b. A compound will have the same Rf value with all stationary phases.
c. A compound's Rf value depends on the amount of interaction it has with the stationary phase.
d. An Rf value will always range between 1 and 2.
e. If two compounds have the same Rf value, then they are the same compound.
C
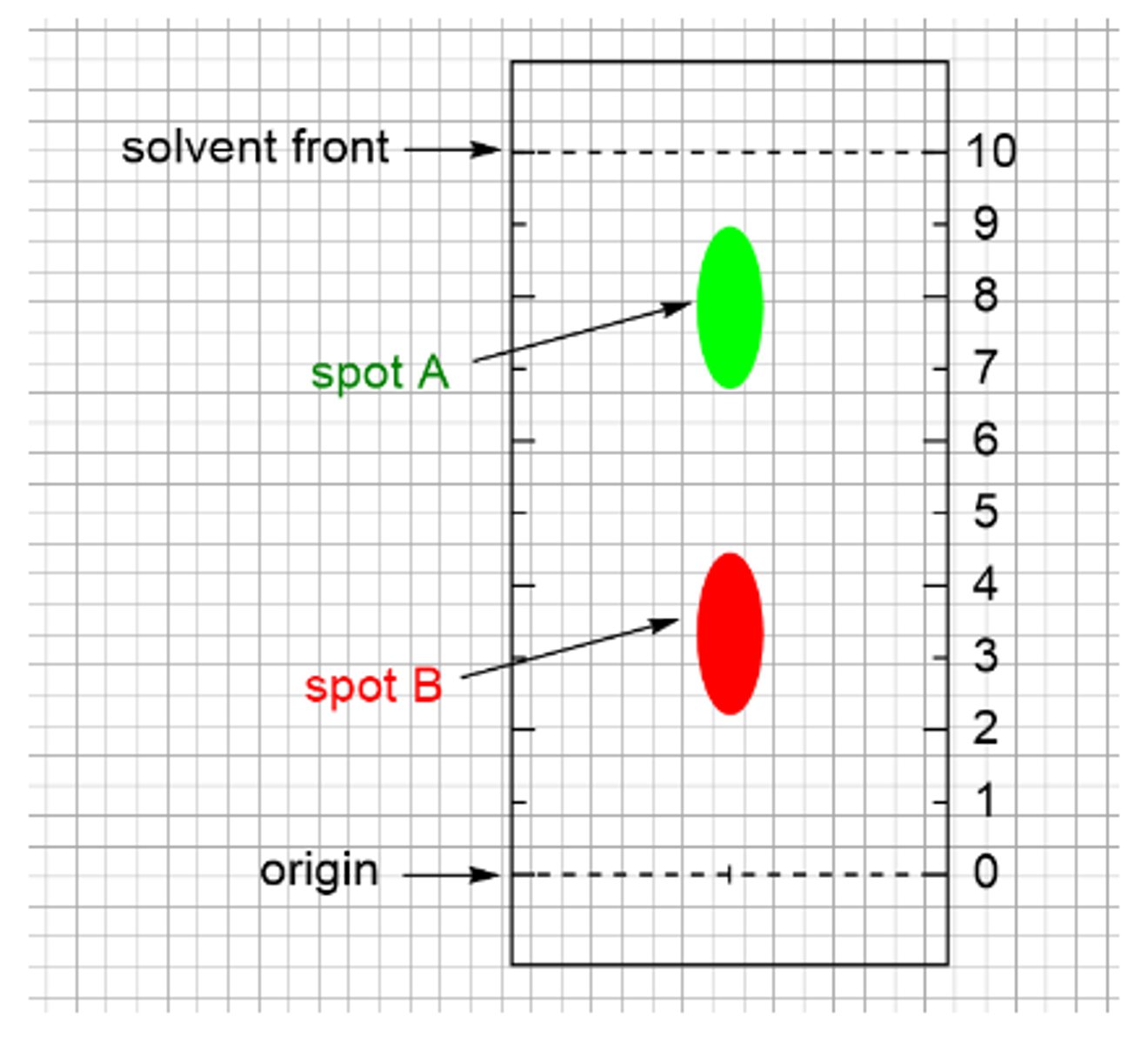
Determine the retention factor, Rf, for the red spot B on the chromatogram. Pay close attention to the scale.
0.33
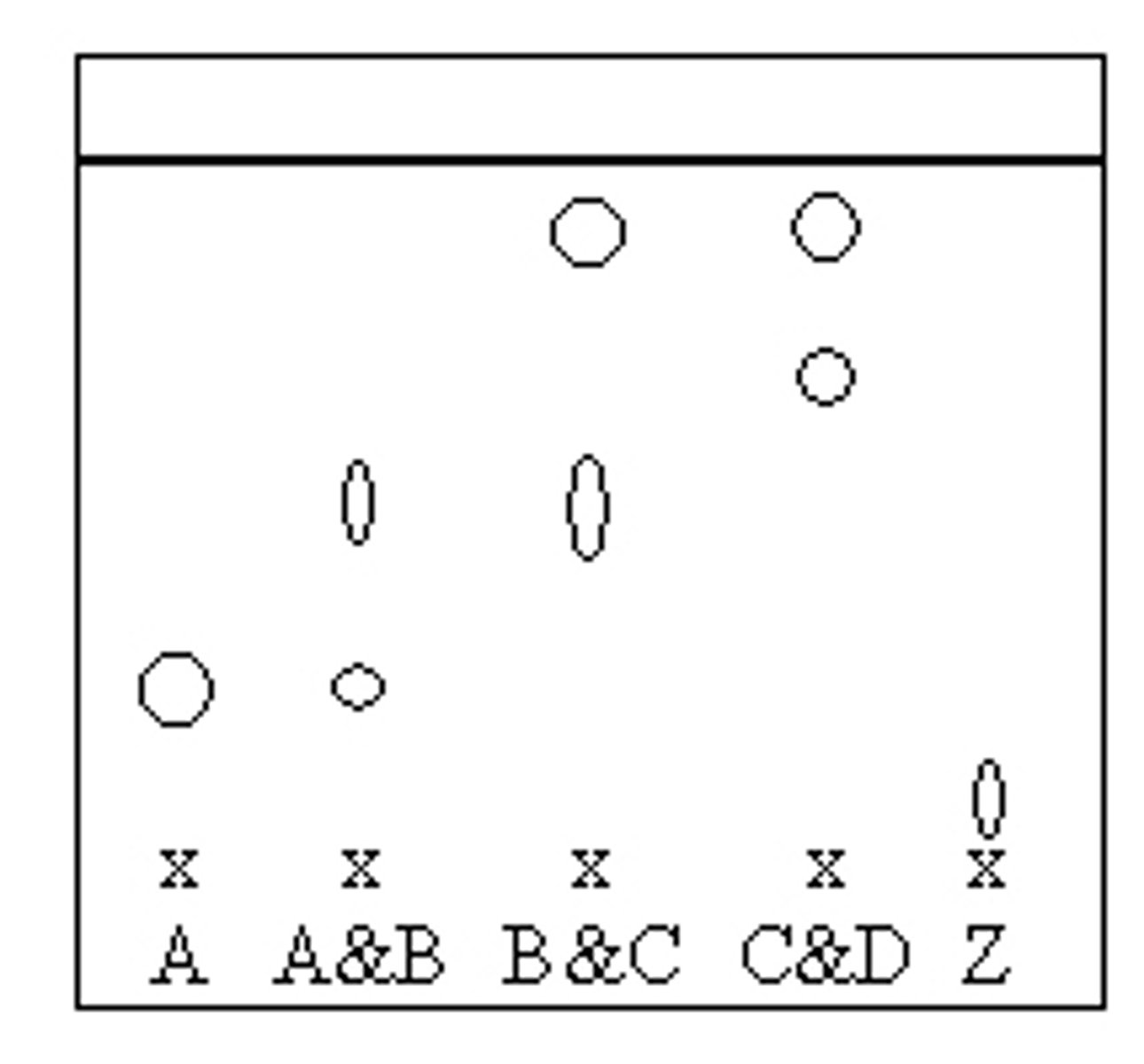
Use the information on the chromatogram to determine the identity of unknown compound Z.
a. Unknown Z is Compound D.
b. Unknown Z is not Compound A, B, C, or D.
c. Unknown Z is Compound B.
d. Unknown Z is Compound C.
e. Unknown Z is Compound A.
B
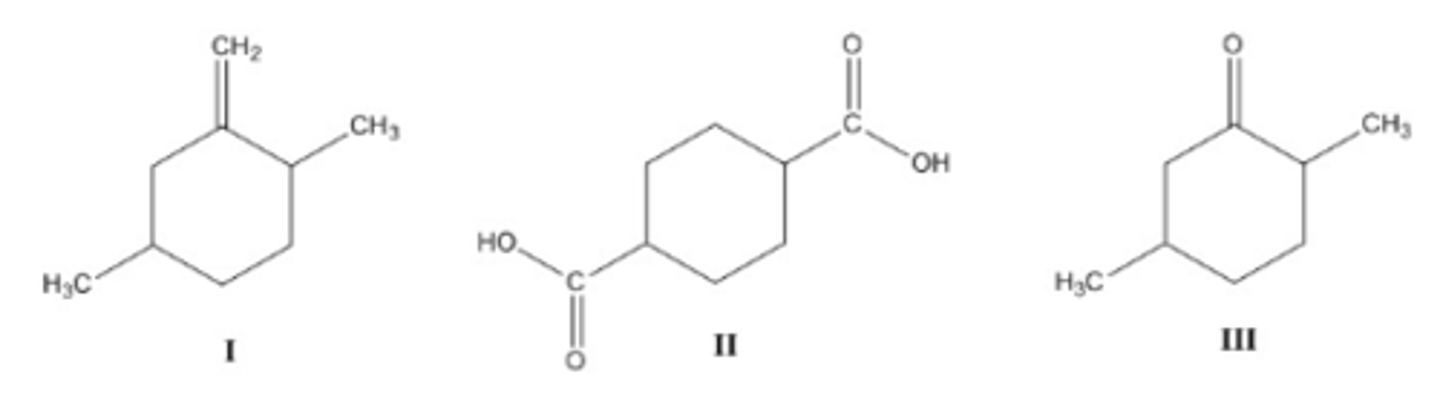
Rank the following molecules in order of increasing Rf value on a silica gel TLC plate, that is start with the molecule with the lowest Rf value.
a. III < I < II
b. I < II < III
c. II < III < I
d. II < I < III
C
Relatively polar compound X will have the highest Rf value when eluted on a silica gel TLC plate in which of the following solvents?
a. 25:75 ethyl acetate-hexanes
b. hexanes
c. 75:25 ethyl acetate-hexanes
d. 50:50 ethyl acetate-hexanes
C
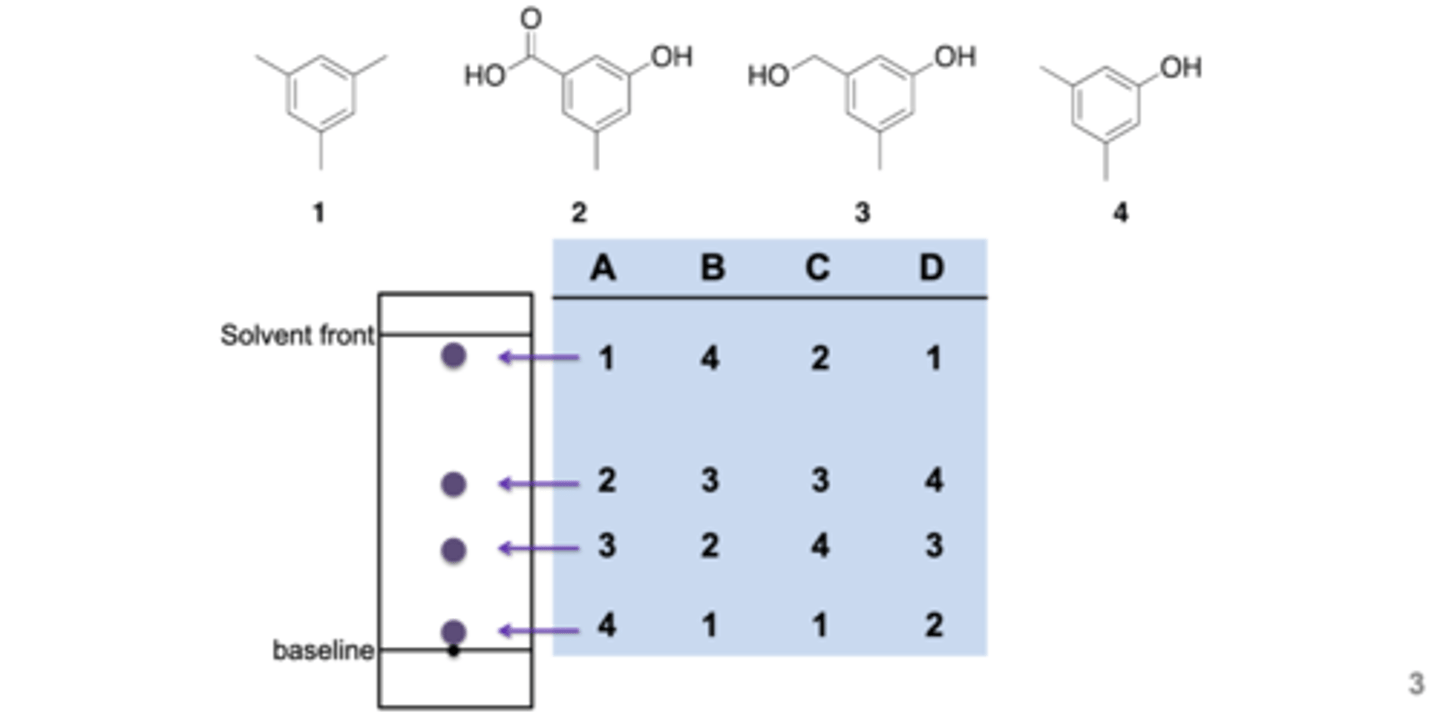
Match the compounds with their spots
D: 1 is the most hydrophobic (non-polar) cant stick to the plate. 2 is the most hydrophilic (polar) and is the stickiest
column chromatography
aka liquid chromatography: used to separate a mixture into its individual components
the collections are termed "fractions"
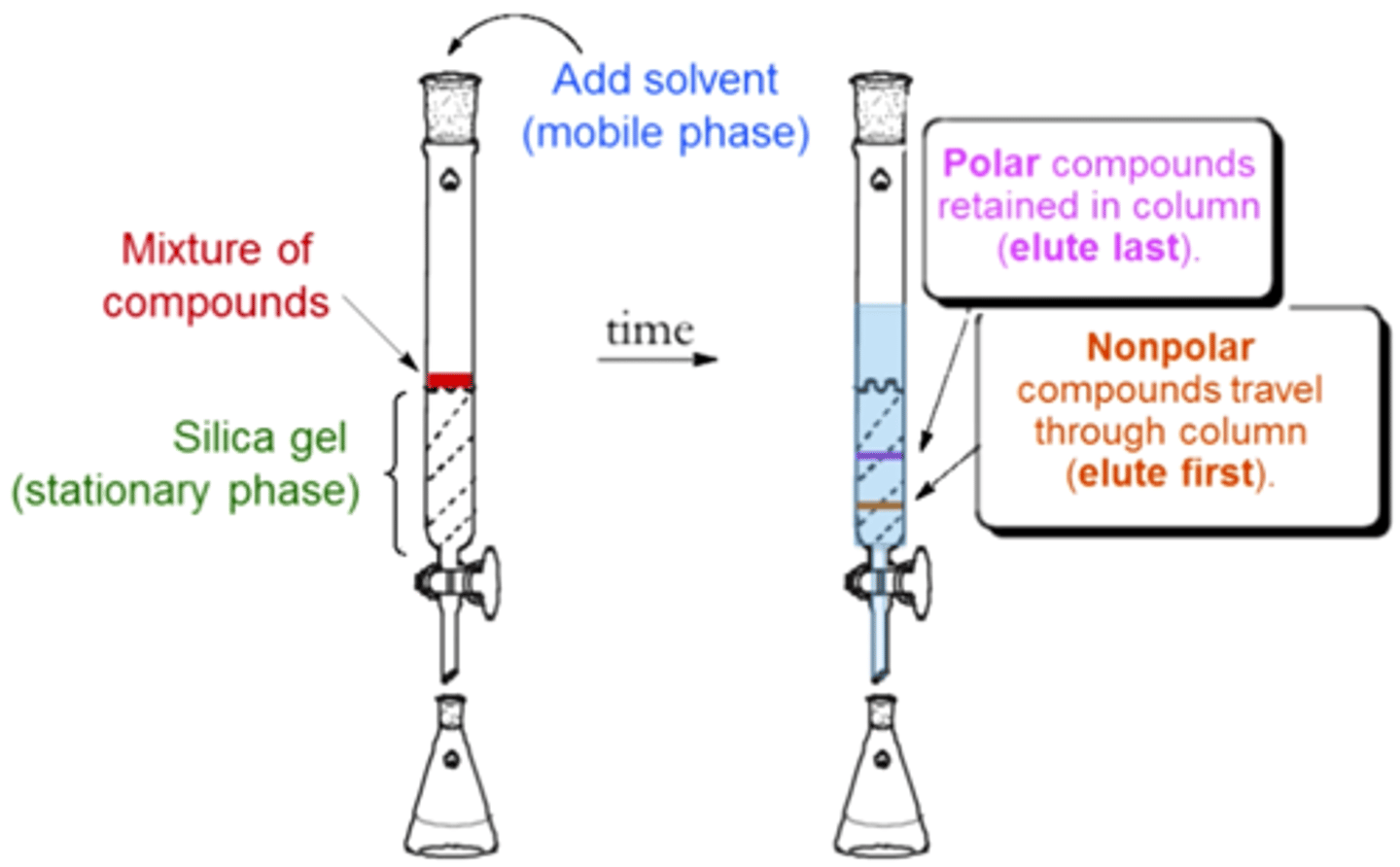
Column chromatography stationary phase and mobile phase
silica gel or alumina; liquid
Column chromatography set-up
1. Stationary phase (Silica gel)
2. Mixture to be separated
3. Sand on top to allow sample to stabilize packing material
Elutent (mobile phase) poured over sand
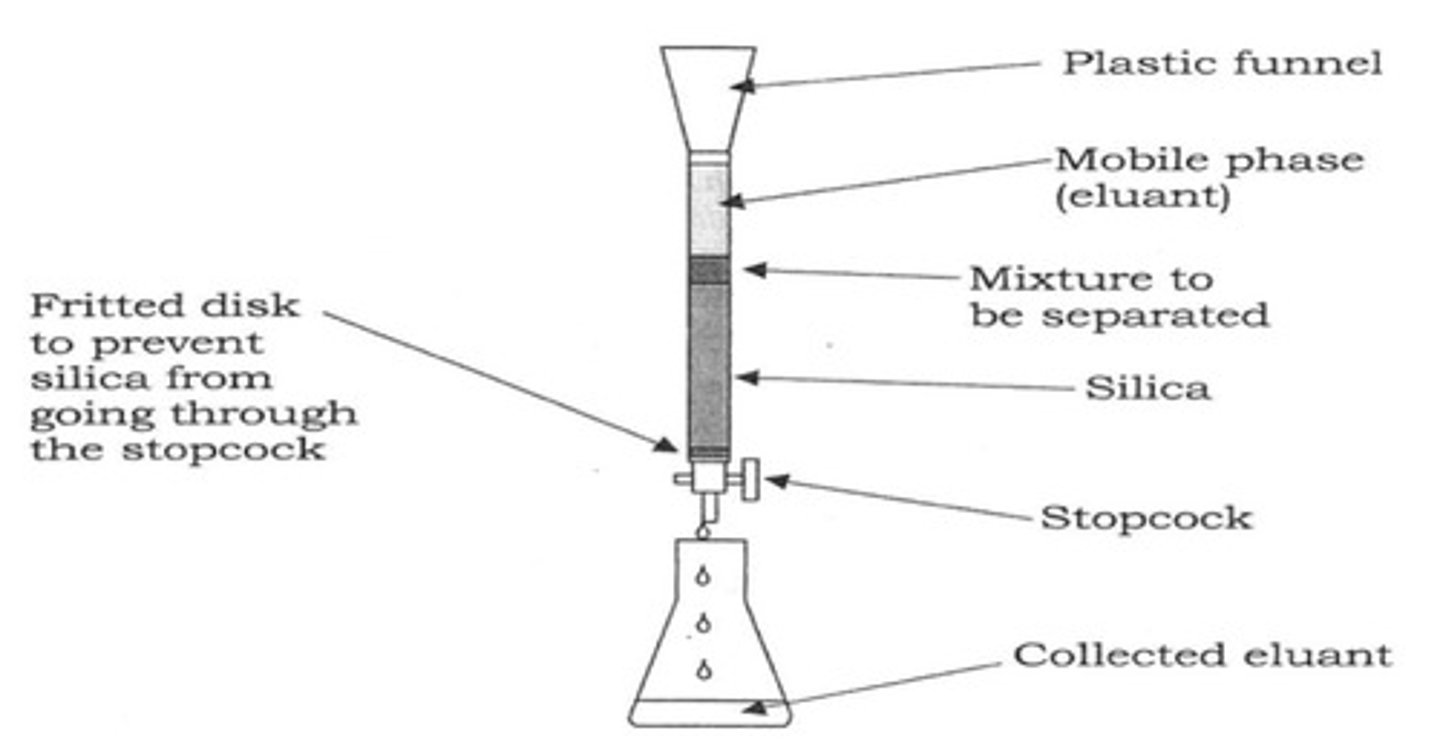
in Column chromatography, NEVER...
let the column run dry
heres why:
- cracks will form, allowing compounds to fall rather than interact with the alumina
- there should always be solvent above the alumina
Which will elute first during column chromatography?
the nonpolar compounds and then the polar compounds.
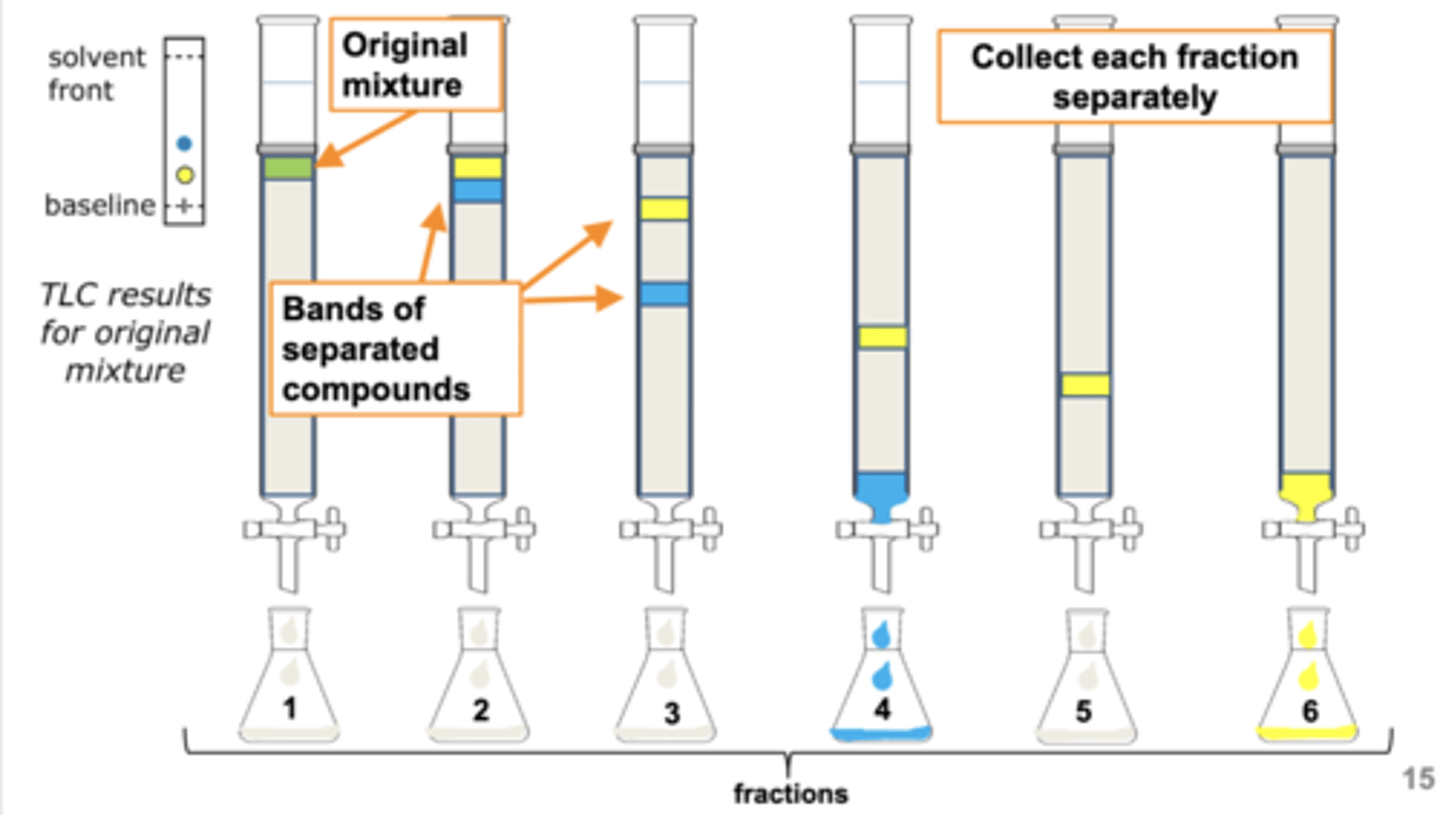
Select all statements below that apply to Column Chromatography.
a. column chromatography is often used to purify large (10 g) sample mixtures.
b. The compound in a mixture with the lowest Rf value will exit the column first.
c. Column chromatography is used mainly as an analytical tool.
d. column chromatography can be used to purify small (< 0.1 g) sample mixtures.
A, D
To prepare a column for chromatography
a. solvent and dry alumina are added in alternating portions and mixed into a slurry by agitating the column.
b. the column is filled with dry solid alumina then solvent is allowed to flow through, thus making a slurry.
c. the column is filled with solvent, then a slurry of alumina and solvent is swirled together and poured quickly into the column.
d. the column is filled with solvent, then dry solid alumina is slowly added while swirling the column to make a slurry in the column.
C
What order of solvents is typically used when the mobile phase is varied in column chromatography?
a. Typically one would start with one solvent and not change.
b. Solvents can be interchanged back and forth throughout the elution process.
c. Start with a nonpolar solvent and change to a more polar one.
d. Start with a polar solvent and change to a more nonpolar one.
C
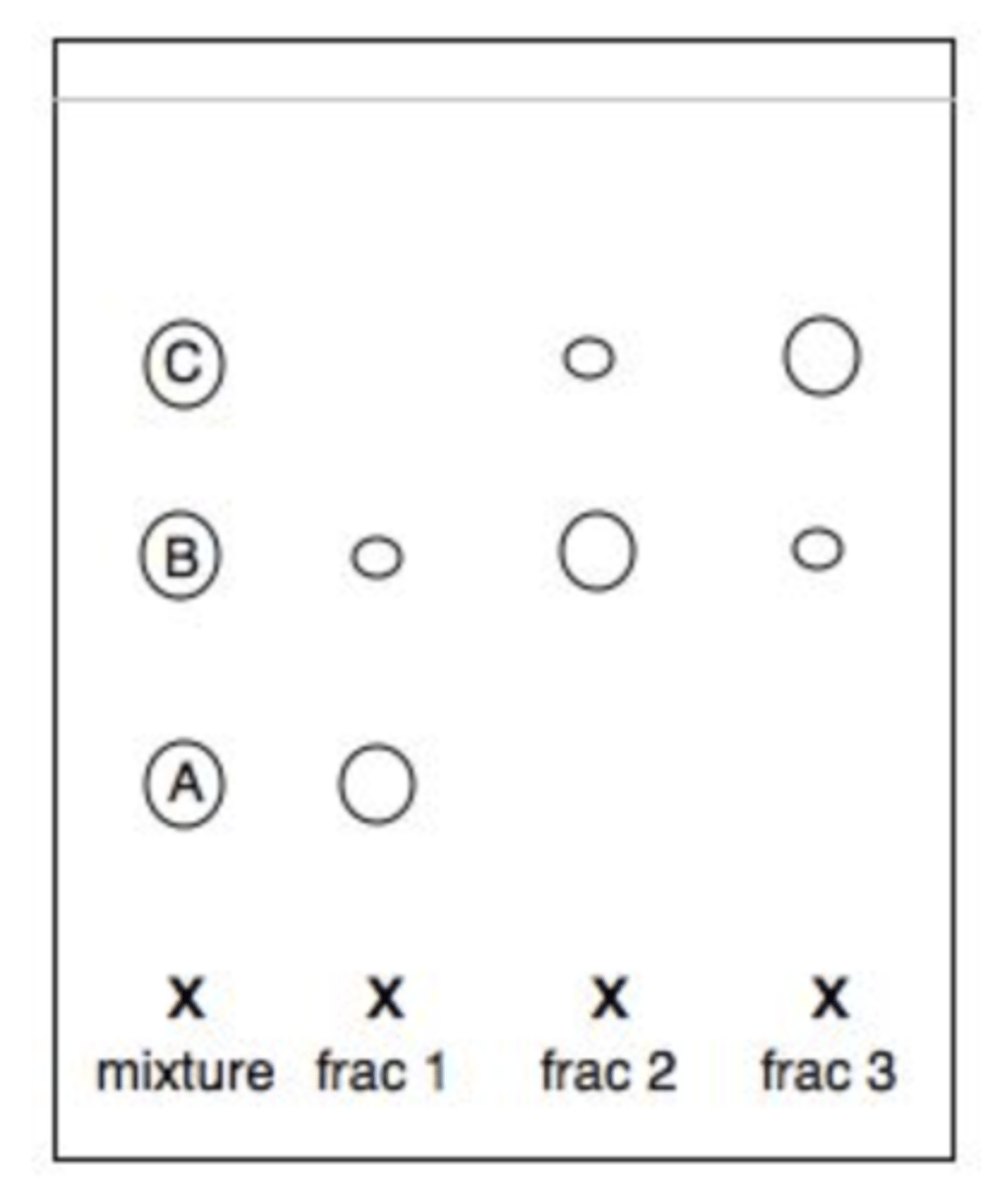
A mixture of three compounds (A, B and C) was chromatographed using a column packed with silica gel and appropriate solvents to collect three fractions. Based upon the silica gel TLC analysis displayed, how successful was the separation?
a. The separation was successful.
b. The separation was not very successful. Compounds B and C were not completely separated.
c. The separation was not completely successful. Compound B was not completely separated from compound A.
d. The separation was not successful at all. None of the compounds were completely separated.
D
If you get ferrocene/acetylferrocene mixture on your skin, what should you do?
a. Immediately begin flushing the skin with water while consulting with your instructor for the best course of action.
b. Wipe off the ferrocene/acetylferrocene using a tissue moistened with methylene chloride.
c. Check the ferrocene and acetylferrocene MSDS for skin contact hazard information.
d. Brush it off, since both acetylferrocene and ferrocene are solids. No further action is necessary.
A
During the column chromatography procedure, what eluting solvent(s) should the 90:10 hexanes/ethyl acetate be changed to, and when should the change take place?
a. Change the solvent to 80:20 hexanes/ethyl acetate when the first band has traveled about halfway down the column.
b. Change the solvent to 80:20 hexanes/ethyl acetate when you begin collecting the first fraction.
c. Change the solvent to 80:20 hexanes/ethyl acetate after the first fraction is almost completely collected.
d. Change the solvent to 80:20 hexanes/ethyl acetate when you can see two separate bands on the column.
C
When the TLC plate is set up to evaluate the column chromatography separation, what samples will be spotted on the plate?
a. The separated fractions and authentic samples of pure ferrocene and acetylferrocene.
b. The original mixture, the two separated fractions, and authentic samples of pure ferrocene and acetylferrocene.
c. The original mixture and the two separated fractions.
d. The original mixture before separation and authentic samples of pure ferrocene and acetylferrocene.
B
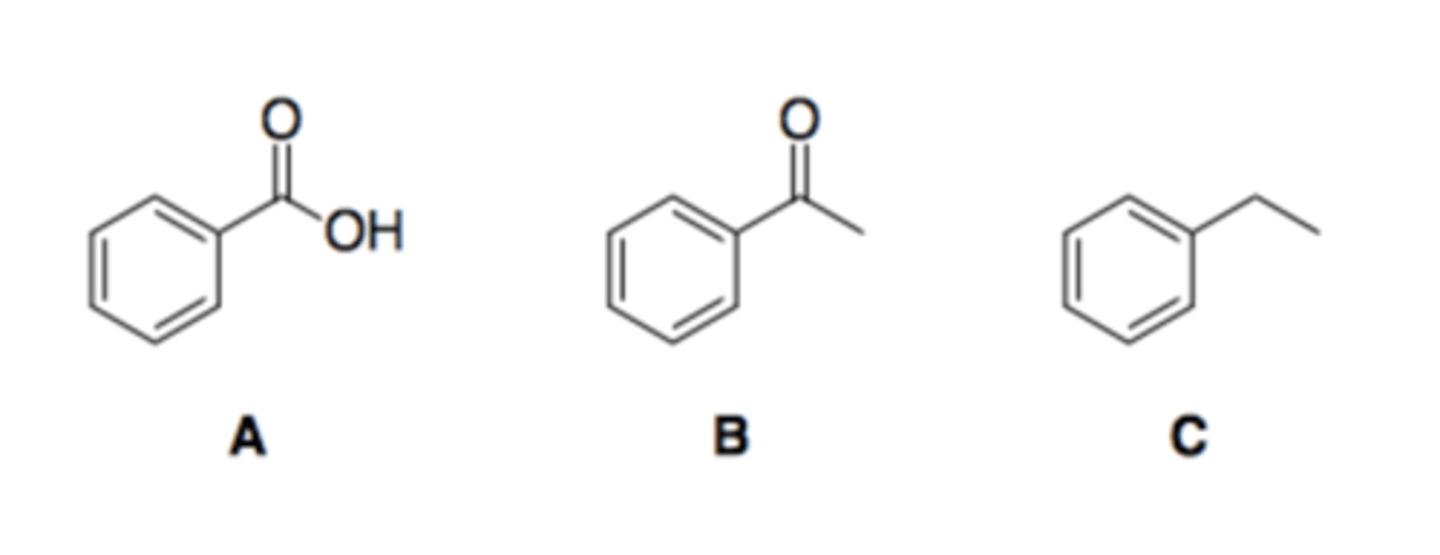
Arrange the following compounds in order of elution from a silica gel chromatography column (i.e., 1 = first compound to be collected).
C,B,A
nonpolar first then polar
Order the solvents from most to least polar.
a. ethanol
b. dichloromethane
c. toluene
d. hexane
A, B, C, D
Distillation is used for separating compounds based on their:
A. molecular weight
B. boiling points
C. polarities
D. melting points
E. solubility
B
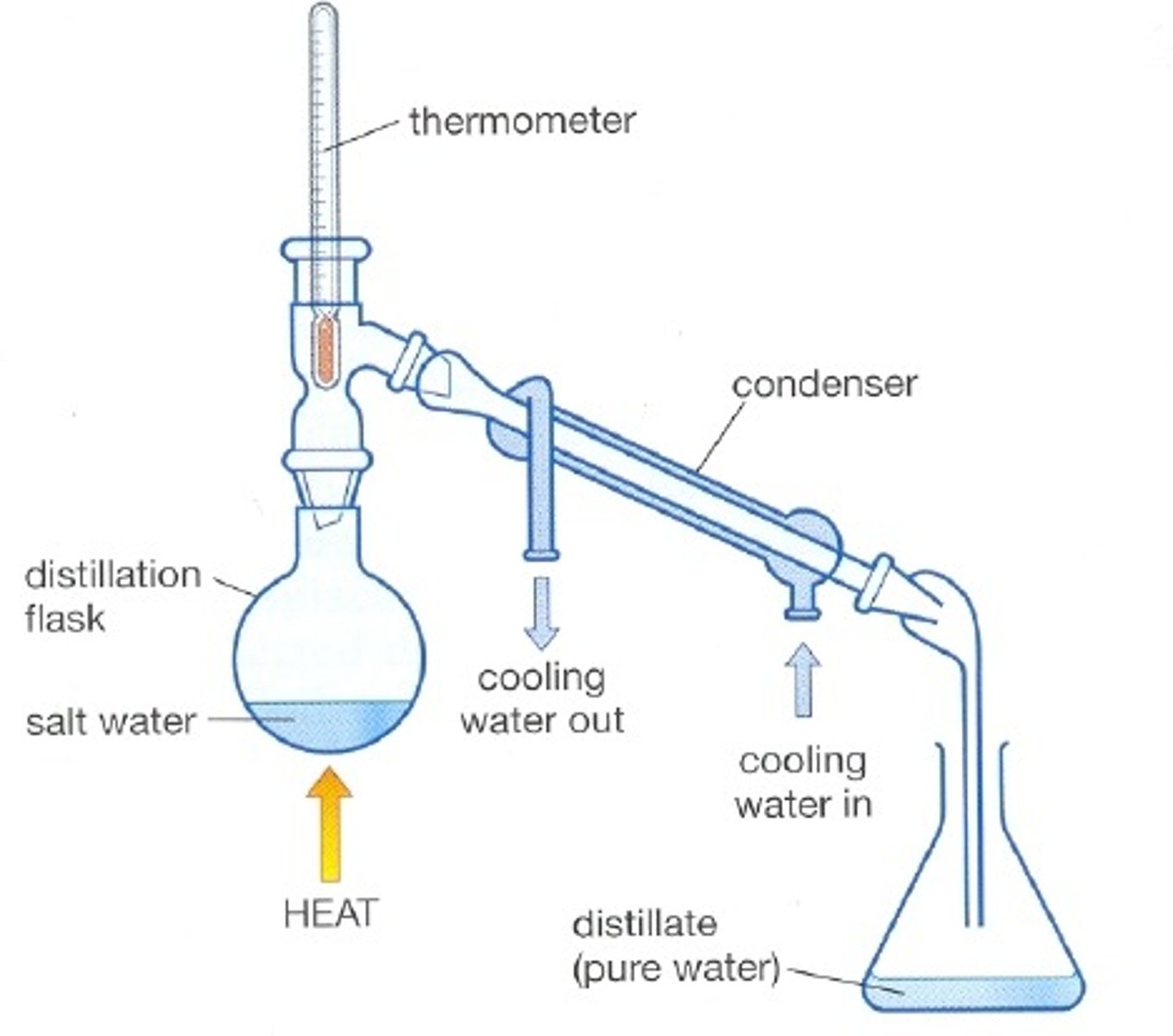
Simple distillation
effective when boiling points of liquids differ by more than 100 C
Used to separate a liquid from a solution and purify an impure liquid
Distillation heating is adjusted to control the
rate of distillation: 1 drop per 1-2 seconds
Never distill to
dryness; can result in explosions
If liquid A is more volatile than liquid B, will the distillate contain more A or B?
A, has lower bp so goes through first
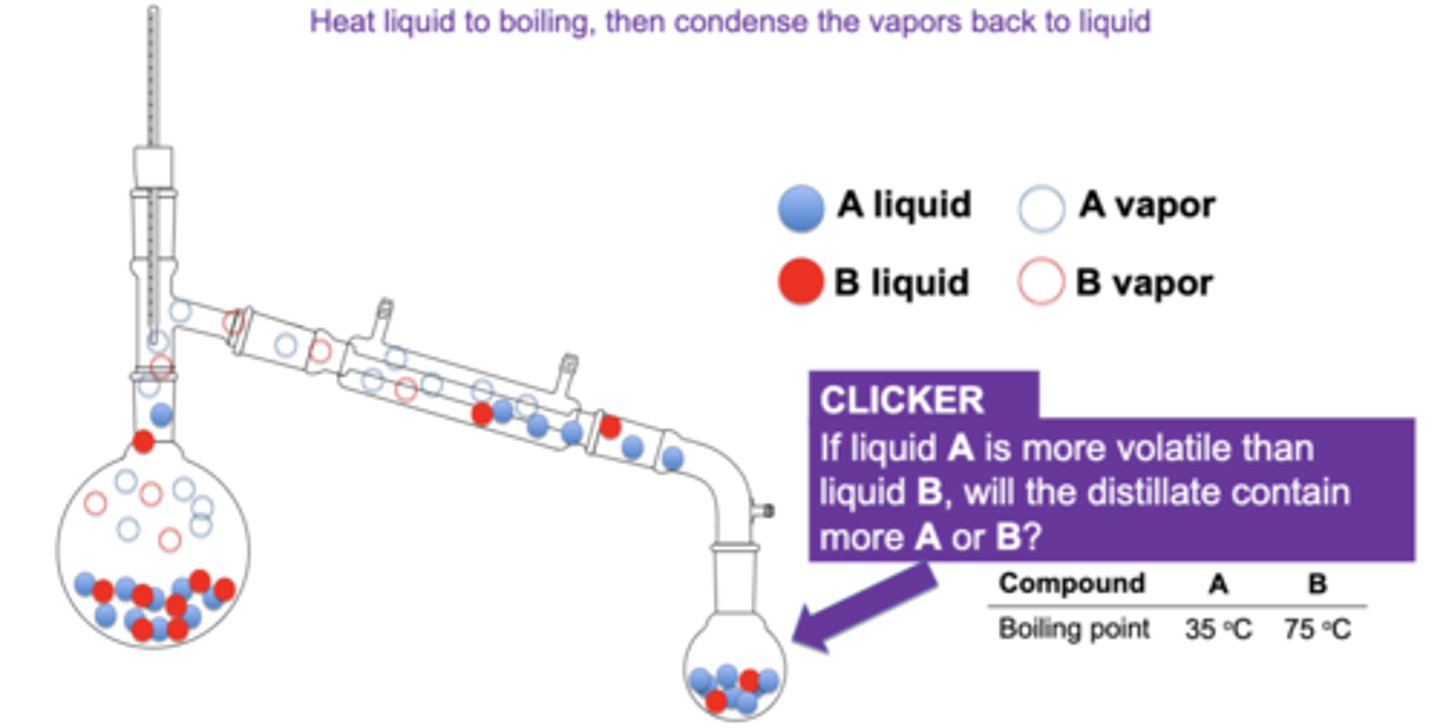
lower boiling point =
higher vapor pressure
boiling point
the temp at which the vapor pressure of the liquid equals the total pressure exerted on the liquid surface (atmospheric pressure)
Boiling point composition curve
look at image
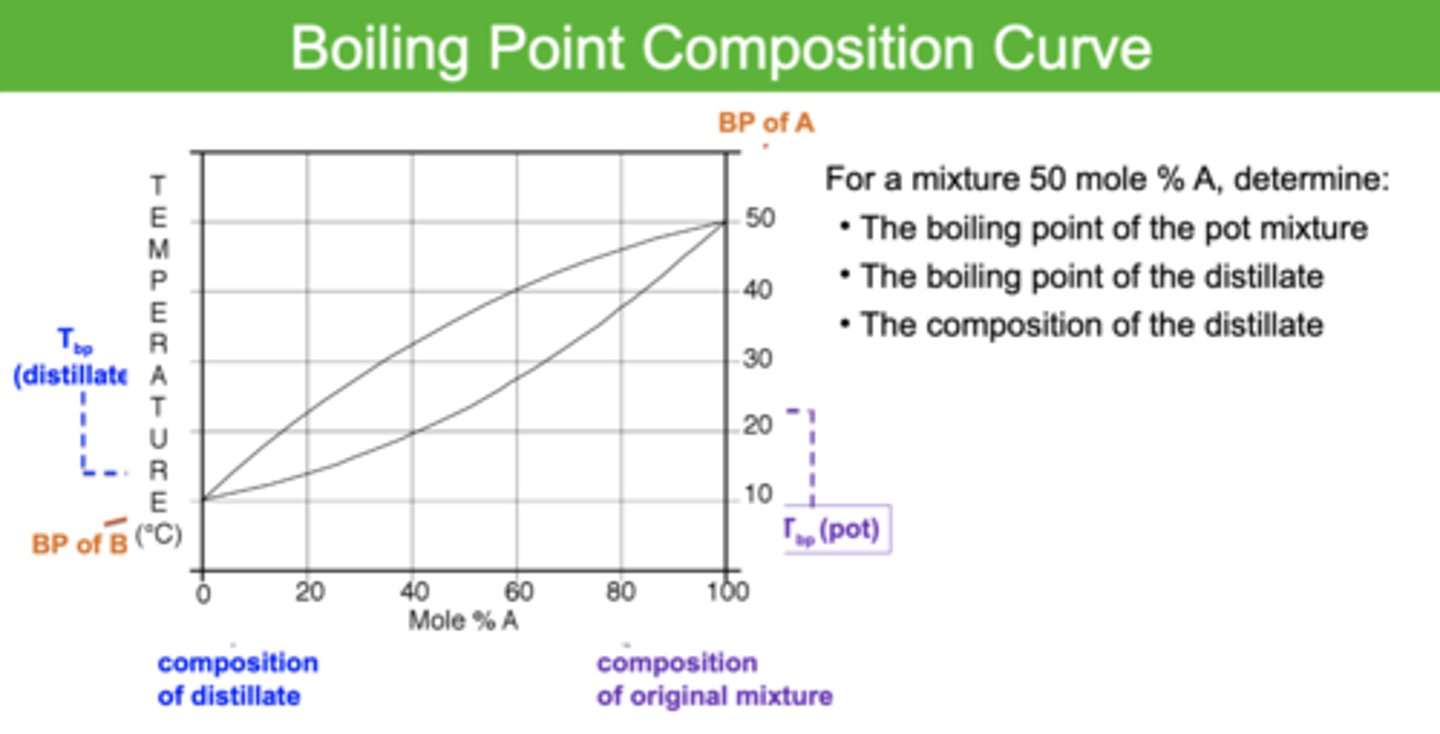
One "Vaporization-Condensation Cycle" is defined as a
theoretical plate
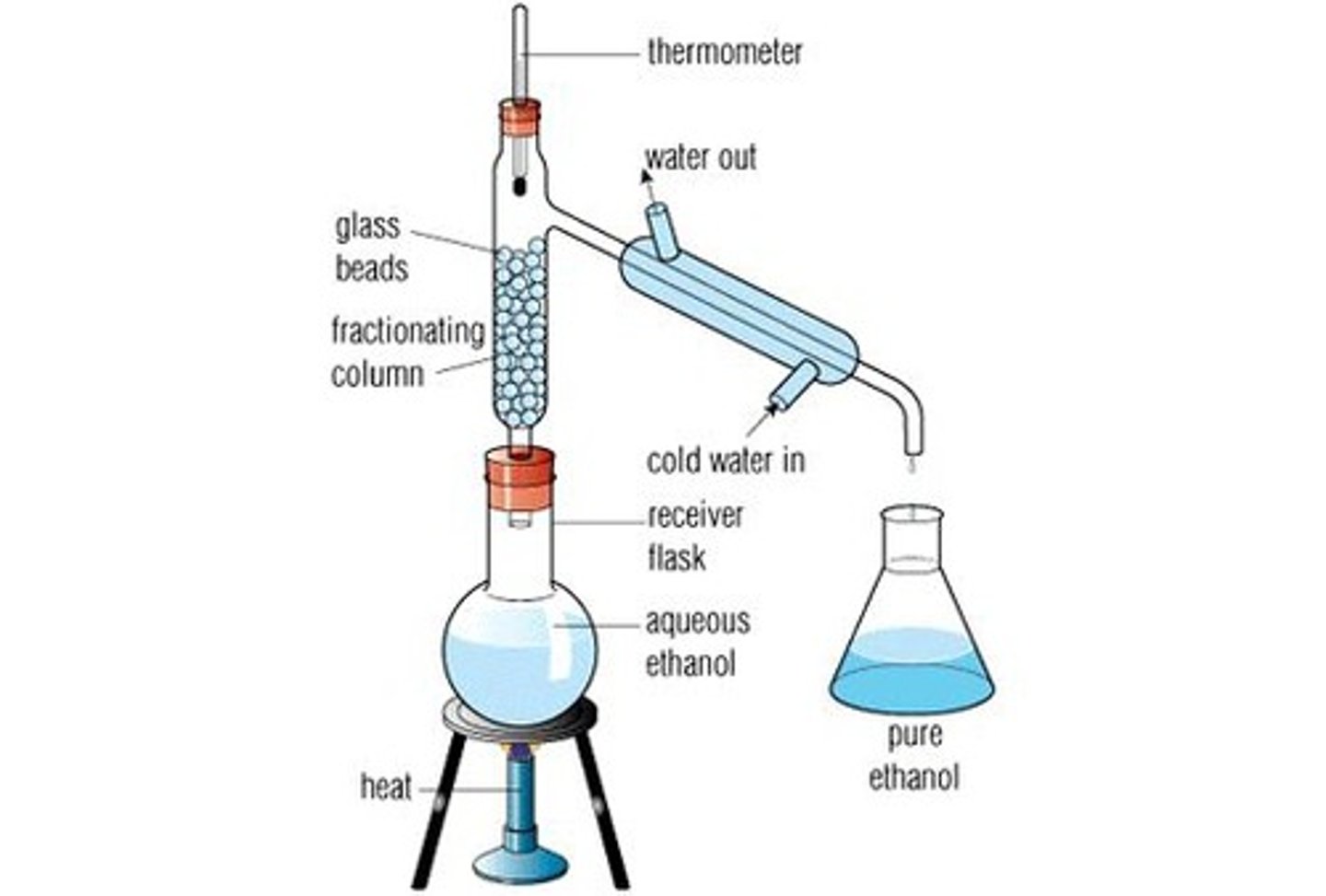
Fractional distillation
just like simple but with a large surface area before vapor gets to condenser
- used to separate two liquids whose boiling points are close (less than 100 C)
- vapor condenses on steel wool
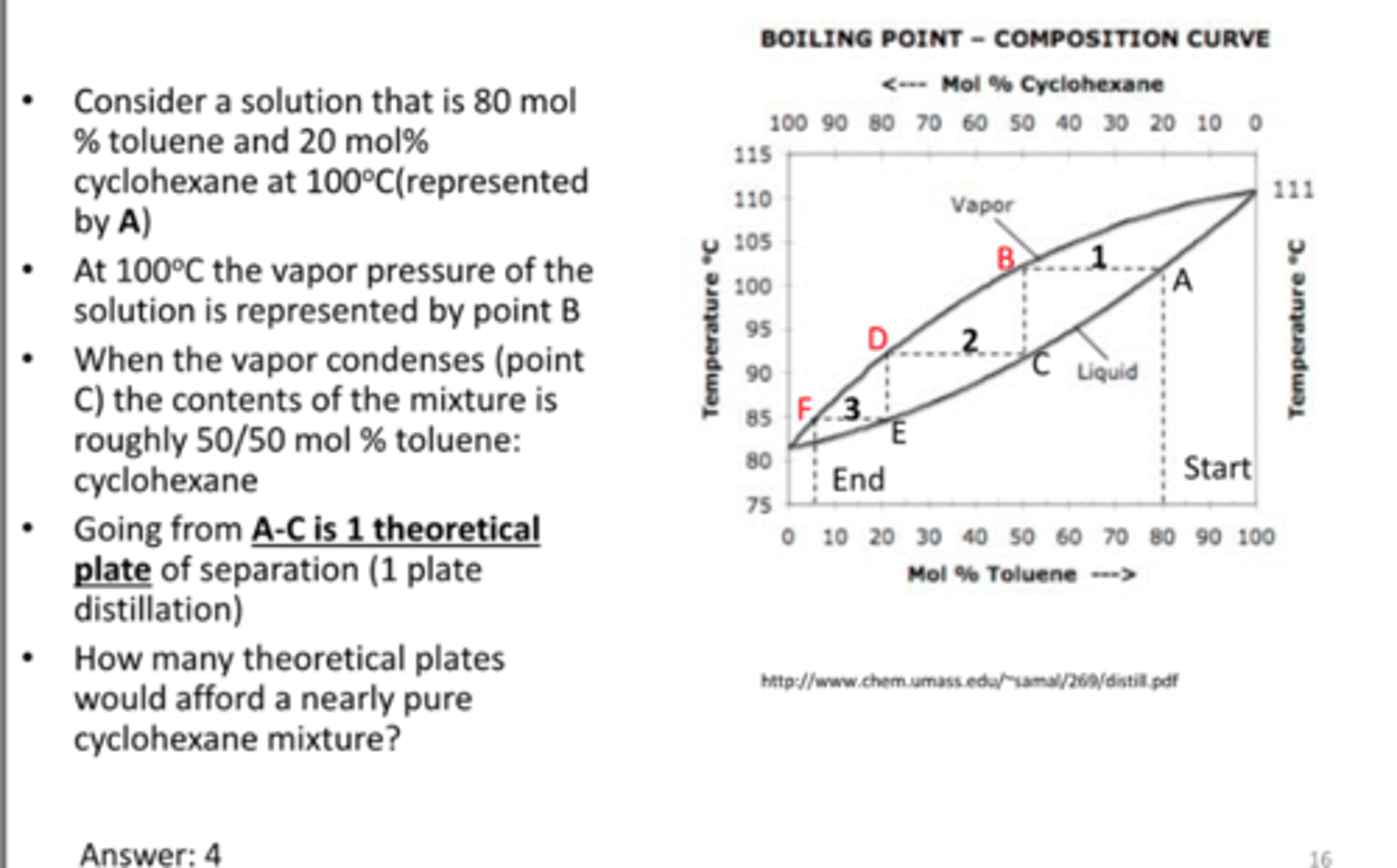
theoretical plates
-the point in a distillation at which liquid-vapor equilibrium is reached
-is a measurement of efficiency of a distillation apparatus
1 simple distillation=1 theoretical plate
1 fractional distillation= >1 theoretical plate
Azeotropes
-A mixture of liquids with a definite composition that distills at a constant temperature without a change in composition•
-Behaves like a pure substance
-the two liquids that comprise an azeotrope cannot be separated by distillation
The proper position for a thermometer in a distillation apparatus is ...
B
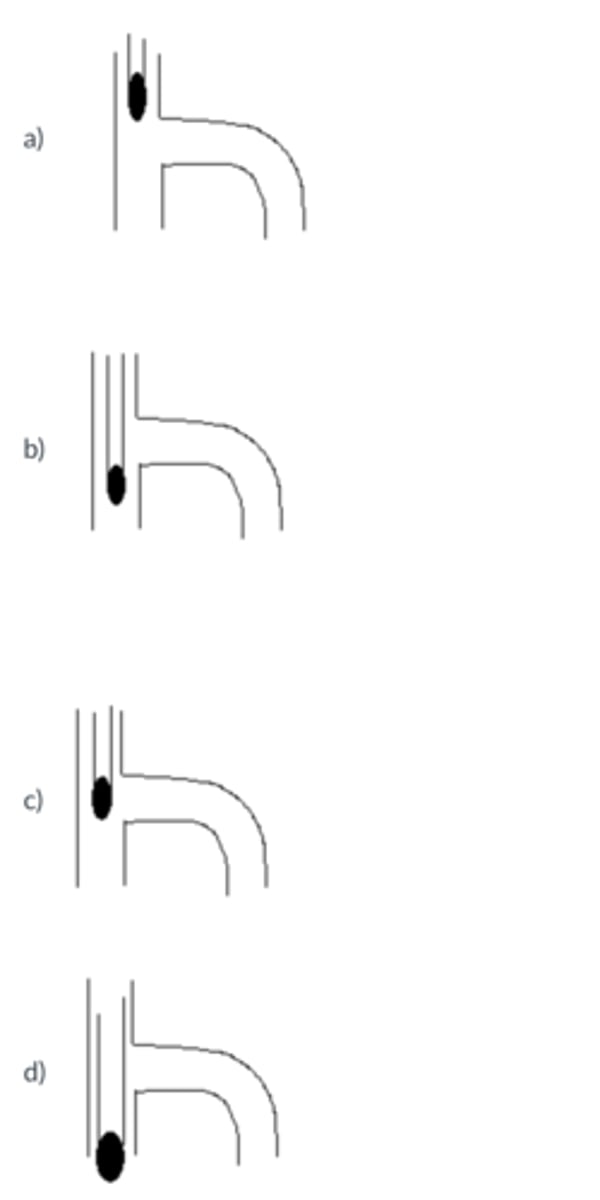
In a distillation:
a. It is more important to maintain a steady temperature than to maintain a steady distillation rate.
b. The placement of the thermometer bulb is critical in a fractional distillation, but not in a simple distillation.
c. The most important clamp is the one holding the receiver.
d. The distillation range is typically representative of the boiling point of the compound being distilled.
D
The proper rate for a fractional distillation...
a. 1 drop about every 1 second.
b. can be several times faster than that for a simple distillation due to the increased surface area.
c. is 1 drop every 1-2 minutes.
d. should be several times slower than that for a simple distillation to achieve better separation.
A
The partial vapor pressure of a liquid in a mixture depends upon two factors (besides temperature). What are the two factors?
a. The hotplate setting and atmospheric pressure.
b. The relative amount (or mole fraction) and vapor pressure of the liquid.
c. The size of the vessel containing the mixture and the volume of the mixture.
d. The partial vapor pressure only depends upon the temperature
B
In our laboratory, the distillation range of a pure compound would generally be expected to be __?__ the literature BP760 of the liquid (assuming the thermometer is correctly placed and accurate).
a. exactly the same as
b. higher than
c. lower than
d. nowhere near
C
Using the boiling point-composition diagram below, starting with a mixture of 10 mole % A and 90 mole % B, which compound is the most volatile?
a. A
b. B
c. they are equal in volatility
A
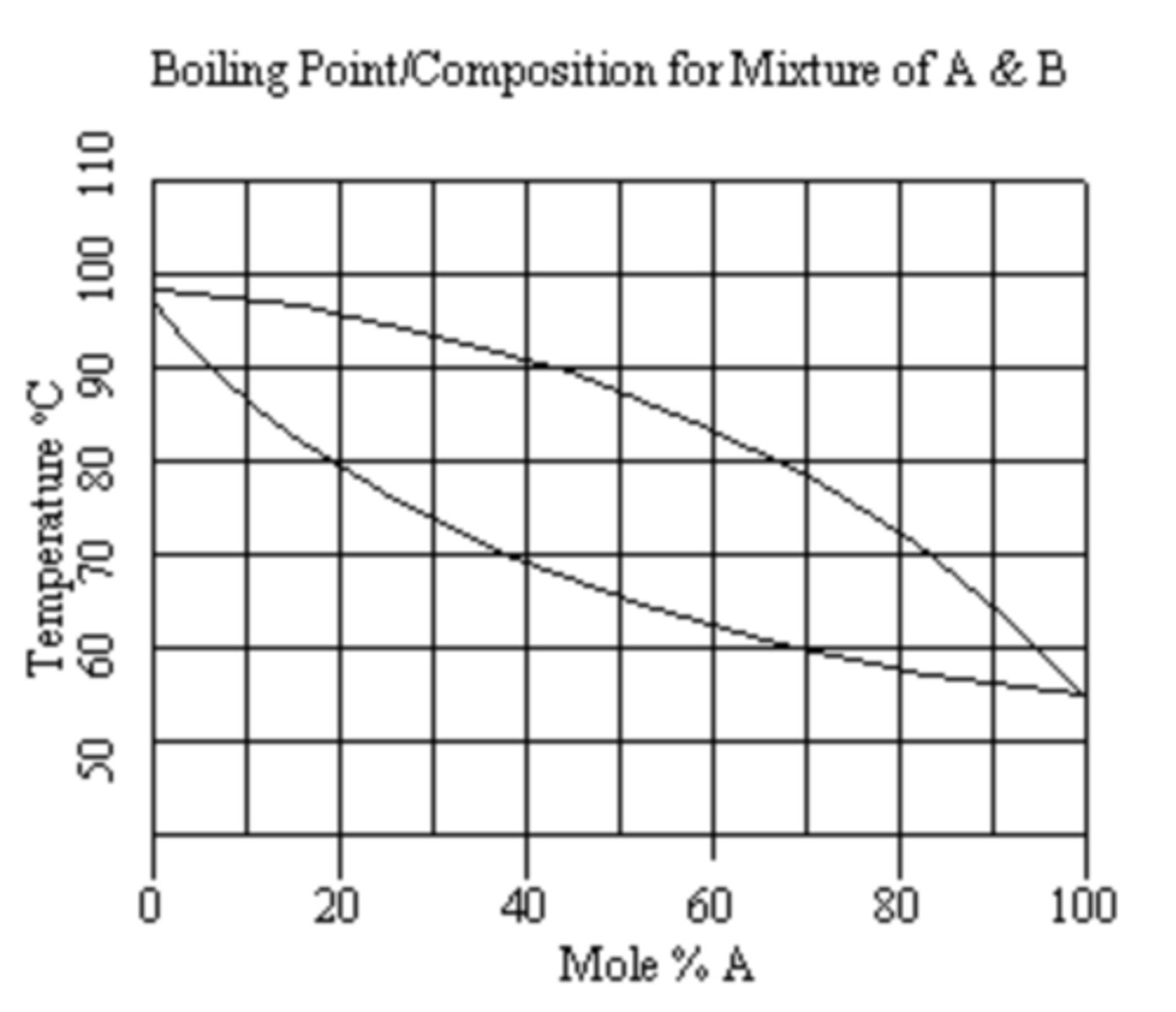
Using the boiling point-composition diagram below, starting with a mixture of 10 mole % A and 90 mole % B, in a distillation with two theoretical plates (i.e. two vaporization-condensation steps), what would be the boiling point of the distillate?
a. 65oC
b. 87oC
c. 56oC
d. 50oC
C
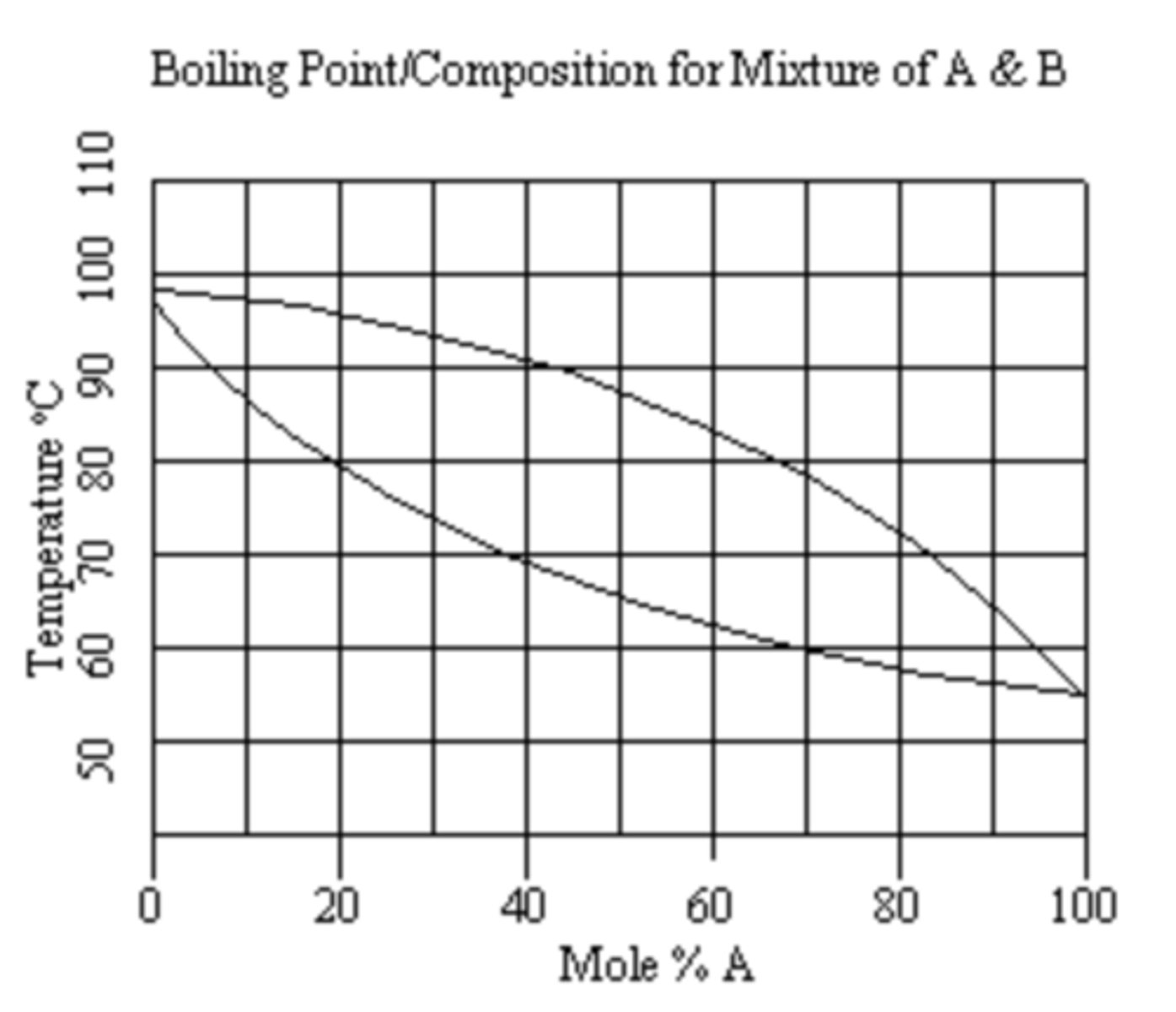
Using the boiling-composition diagram, determine the composition and boiling point of the distillate obtained from a 30 mole % A sample distilled with two vaporization-condensation steps (i.e., two theoretical plates)?
a. 58 mol % A and 33oC
b. 83 mol % A and 13oC
c. 58 mol % A and 21oC
d. 83 mol % A and 21oC
B
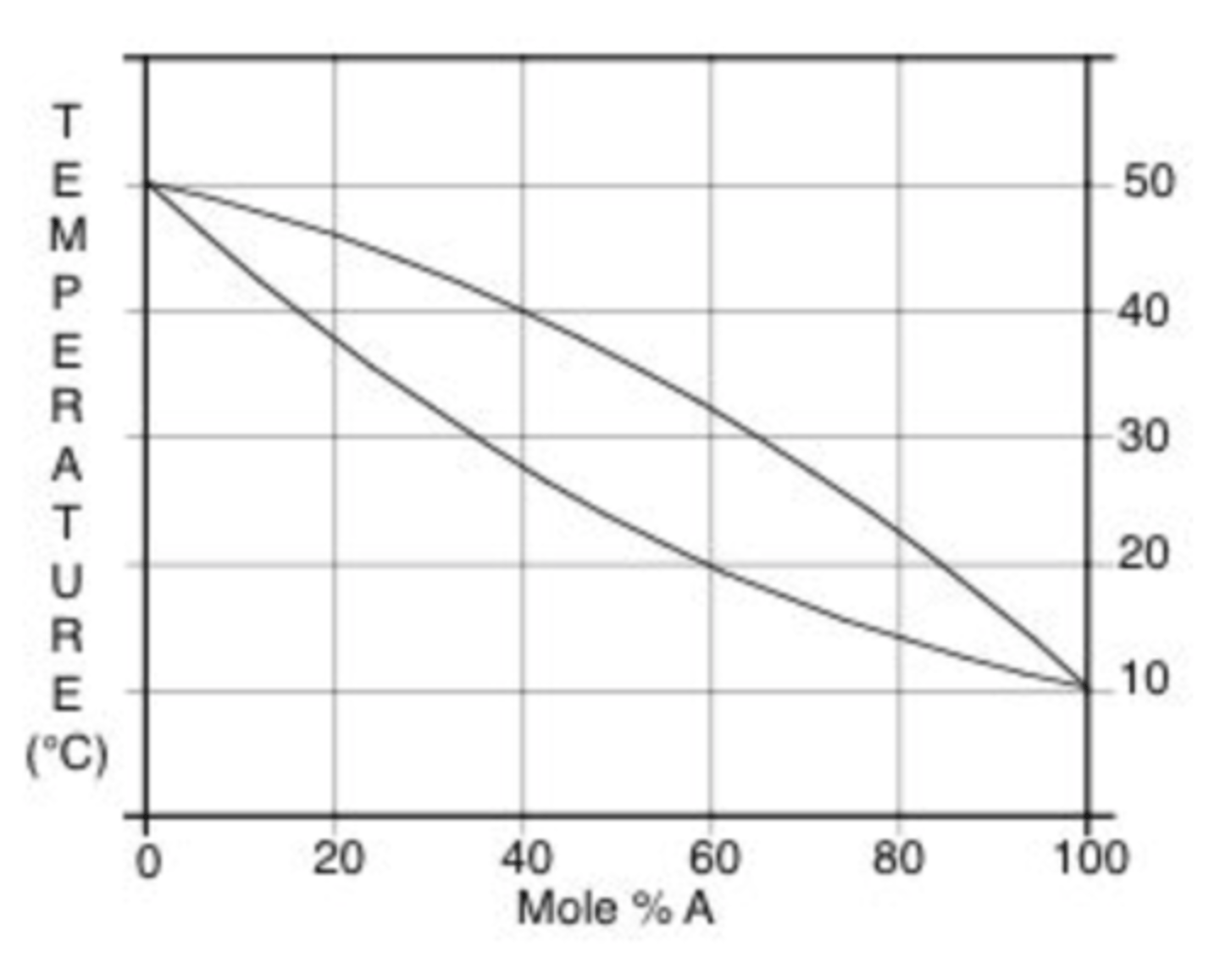
Which of the following statements is true for a distillation?
a. An azeotropic mixture can be separated by distillation if there is a sufficient number of theoretical plates.
b. If the difference in boiling points between two liquids is >100 °C a fractional distillation is needed to separate them completely.
c. Distilling to dryness is recommended to increase the yield.
d. A mixture of liquid A (bp 84) and liquid B (bp 101) can be easily separated by a simple distillation.
e. A simple distillation can be used to separate nonvolatile impurities from a liquid.
E
Extraction
Separates compounds based on solubility in polar/nonpolar solvents.
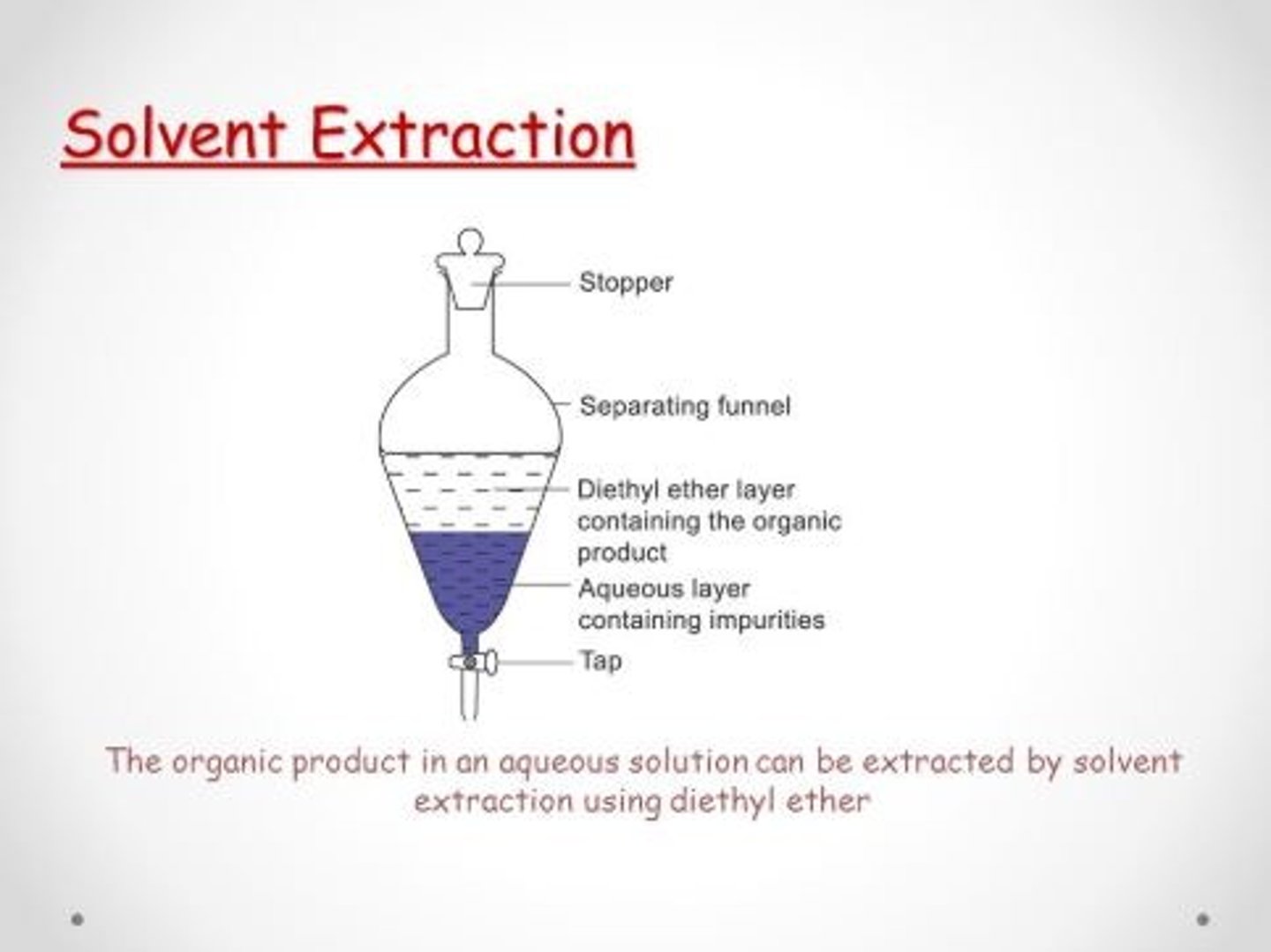
When two solvents are immiscible, which solvent forms the bottom layer?
A. the most polar solvent
B. the least polar solvent
C. the most dense solvent
D. the most volatile solvent
C
When two immiscible solvents are combined, they separate into layers based on their densities. The solvent with the greater density will form the bottom layer, regardless of polarity or volatility. For example, water (a polar solvent) is more dense than most organic solvents like hexane, so water would typically form the bottom layer.
Extraction relies on
differences in solubility between water and organic solvents
- Used to isolate a specific compound from a mixture
• Used to purify a compound
• Based on density differences
• Types: Liquid/liquid and solid/liquid
If you have a mixture of water anddichloromethane, which layer will be on the bottom?
A. Water
B. Dichloromethane
B
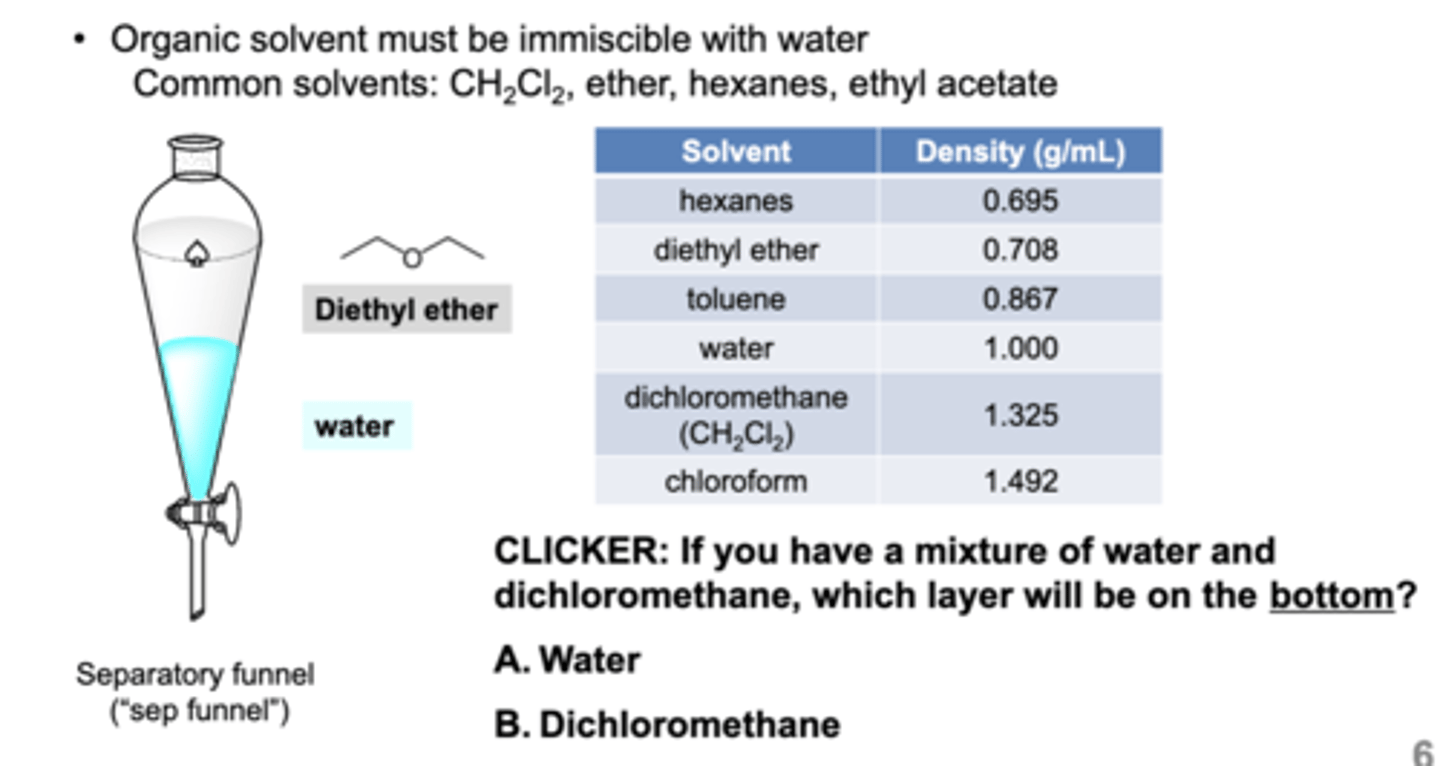
what do you do to identify layers in the seporatory funnel during an extraction?
drop test: add a drop or two of water
extraction set up
just another visual
Brine
saturated aqueous NaCl solution that is used to finish the extraction process
- removes water from the organic phase
- can be used to disrupt emulsions
Drying Agent
used to remove INVISIBLE traces of water with a drying agent
- drying agent binds with water to form a hydrate
rotary evaporator (rotovap)
Used to evaporate solvent
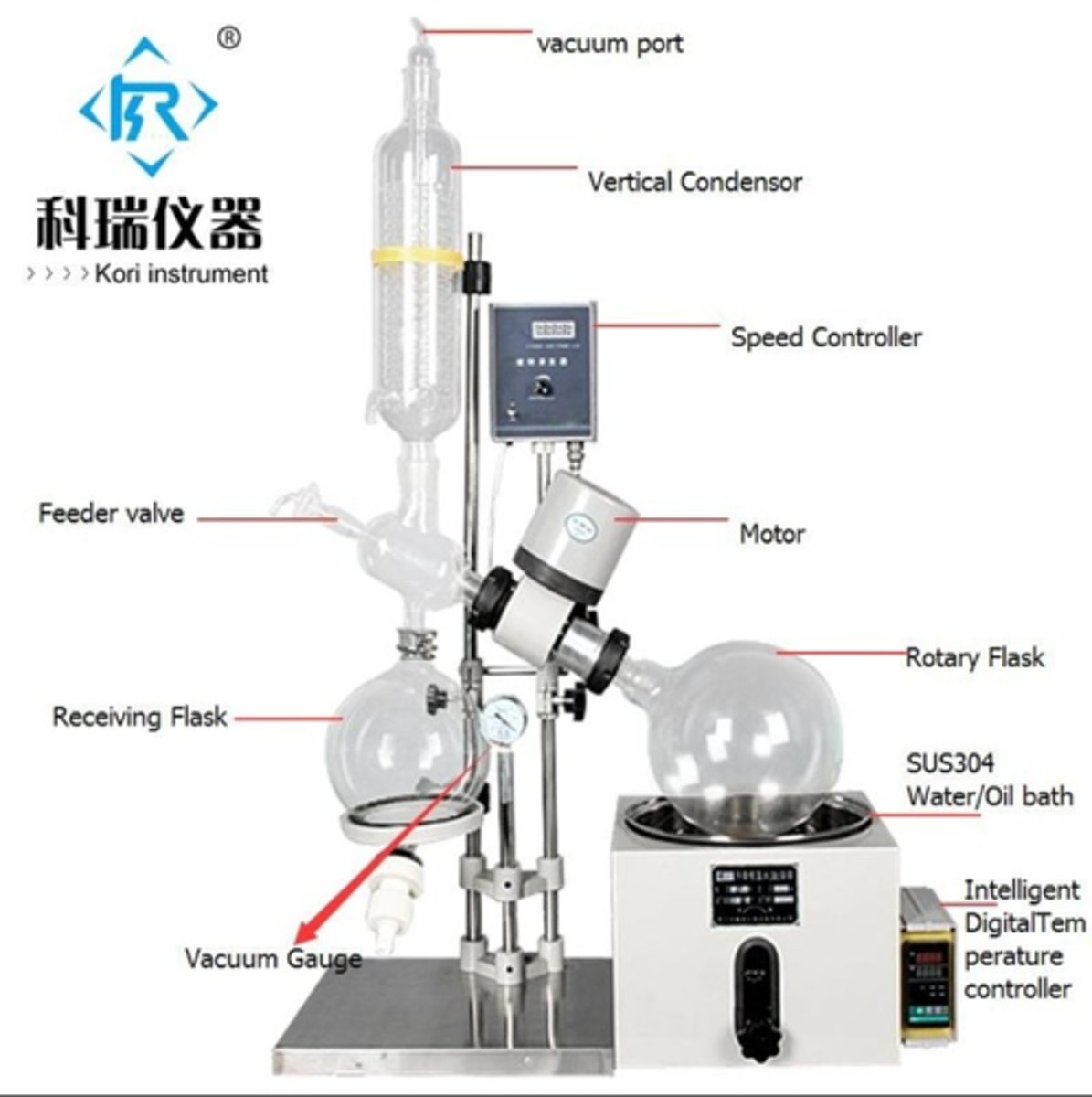
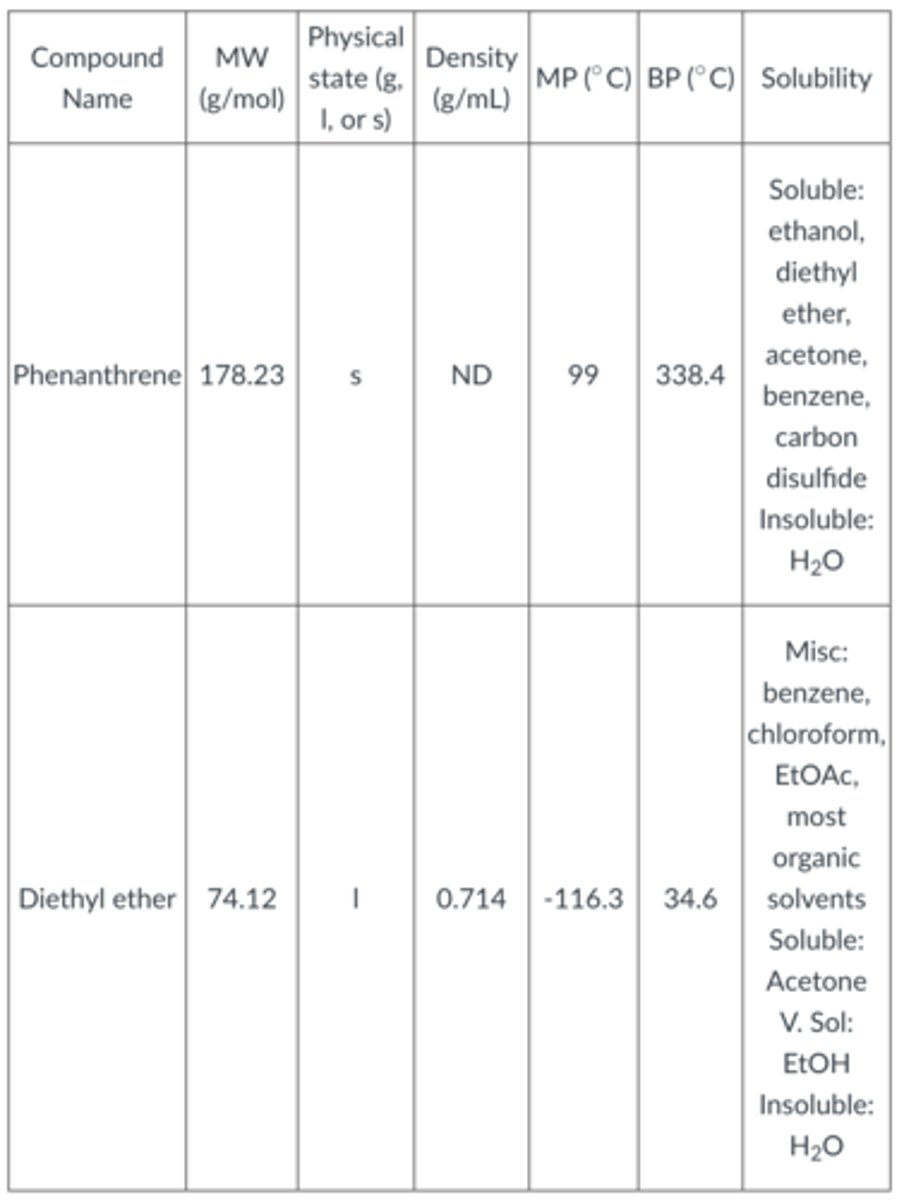
Assume the organic compound you are isolating is phenanthrene. The physical constants for phenanthrene and ether are below. After you remove the ether by rotovap, what would you expect in your round bottom flask?
a. A mixture of diethyl ether and phenanthrene in the flask.
b. A solid in the flask that looks like phenanthrene
c. A liquid in the flask that looks like phenanthrene
d. Nothing. It would not be isolated in the organic layer
e. Nothing. It would be removed by the rotovap
B
When setting up a separatory funnel apparatus, what do you always want to be sure to do before adding any liquid to the separatory funnel? (select all that apply)
a. Place a beaker below the separatory funnel to drain your layer into
b. Close the stopcock to prevent the liquid from falling out
c. Add any solid compound before adding the liquid
d. Add grease to the inside of the separatory funnel so nothing sticks to the walls
e. Open the stopcock so the gas can escape
f. Place an Erlenmeyer flask below the separatory funnel to catch any spills
B,F
In a solvent extraction, a straightforward way to predict the identity of each solvent layer is by evaluating:
a. The density of each solvent.
b. The volatility of each solvent.
c. The ability of each solvent to absorb drying agent.
d. The solubility of the solute in each solvent.
A
A precise, "clean" separation of the two layers is important in an extraction because...
D. Both B and C.
A. Dirty solutions are unsightly.
B. Otherwise, an excessive amount of chemical drying agent will be required.
C. Otherwise, some of the sample will be lost in the process.
D
Solvent extractions depend on a difference in the ____________________ of a compound and its impurities between the two solvents used.
a. Melting points
b. Boiling points
c. Solubilities
d. Densities
C
Given the following information table of solubility and other data, which of the statements below is false?
a. The water will be in the bottom layer.
b. The ABA will be dissolved in water.
c. The water and hexane will form two layers.
d. There will be three layers.
D

In addition to solubility, what other method may be utilized to achieve separation/purification by solvent extraction?
a. Boiling points
b. Size of molecule
c. Chemical reactivity
d. Color
C
Enough drying agent has been added when:
a. It stops clumping together and stays free flowing.
b. It starts clumping together, rather than staying free flowing.
c. It changes color.
d. When water can no longer be observed.
A
The purpose of a chemical drying agent is ...
a. to dry the isolated product after the solvent is removed.
b. to remove water gobules carried over during separations.
c. to remove water that cannot be seen in organic solvents.
d. to remove water from aqueous layers.
C
If Kether/water = 3, how much compound would be extracted by 20 mL of ether from 20 mL of a water solution containing 4 g of the compound?
a. 1 g
b. 3 g
c. 5 g
d. 1.33 g
B
Acid-base extraction
changes solubility via protonation/deprotonation.
ACIDs donate a proton (H+)
BASEs accept a proton
lower pka= stronger acid
Clicker question: Which species is the stronger acid?
A) H2O pKa = 14
B) H2S pKa = 7.2
C) H2 pKa = 35
B bc it has the lowest pka
What direction is the following reaction favored?
H2S + HO- [-> <-] HS- + H2O
A) Left, toward reactants
B) Right, toward products
C) At equilibrium (some reactants and some products)
H2O pKa = 14
H2S pKa = 7.2
B
CARDI! or just look at pka
Steps to Acid Base Reactions
1. Write out the acid-base reaction
2. Label the acid & base; conjugate acid & conjugate base
3. Determine the pKa of the acids
4. Determine direction of equilibrium (reactants or products)
5. Is the organic species charged or uncharged in that direction? Charged = aqueous; uncharged = organic
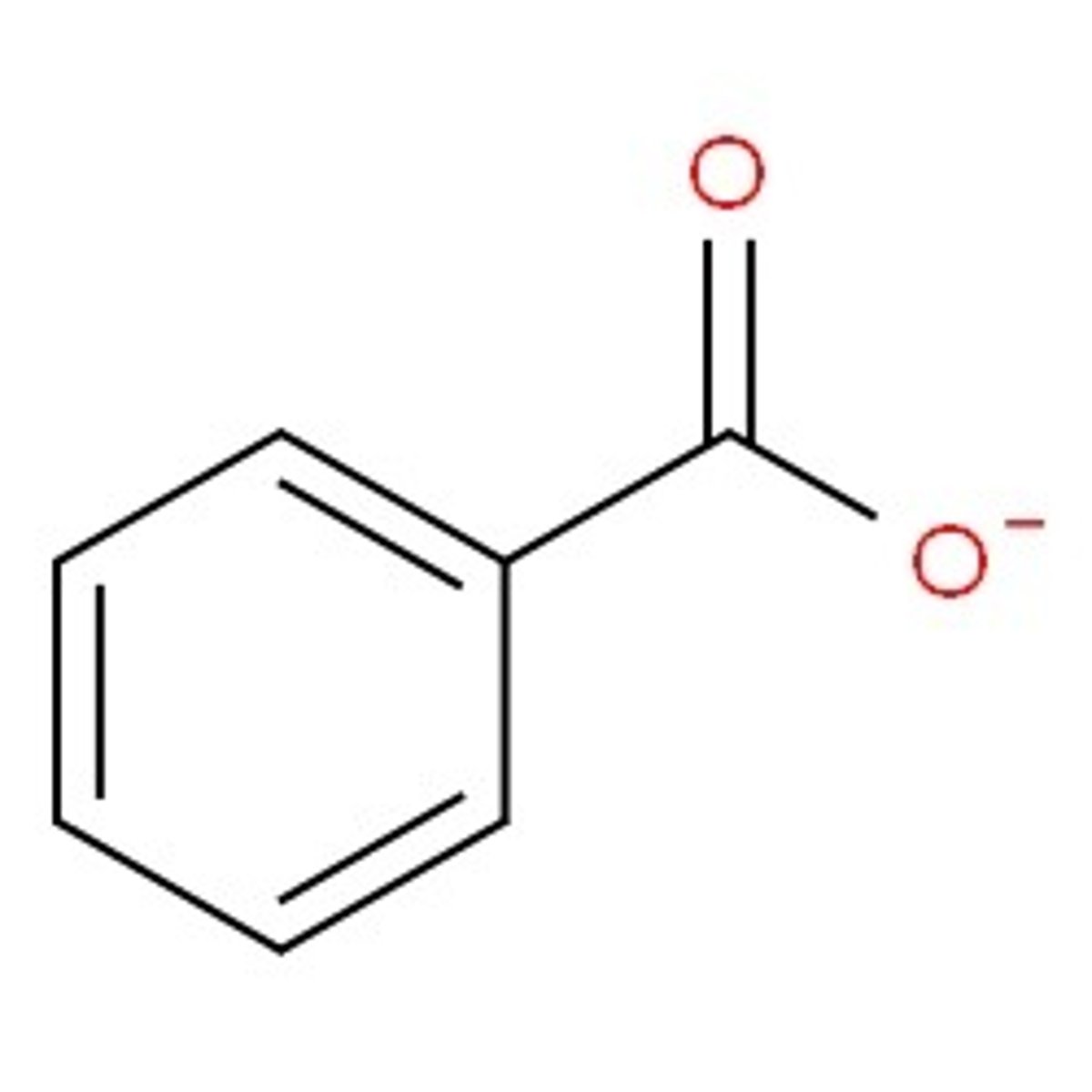
If I were to complete an extraction withdiethyl ether and water, which layerwould I find the benzoate ion in?
A. the top layer, aqueous
B. the bottom layer, aqueous
C. the top layer, ether
D. the bottom layer, ether
B bc it is charged so aq layer and aq is on the bottom
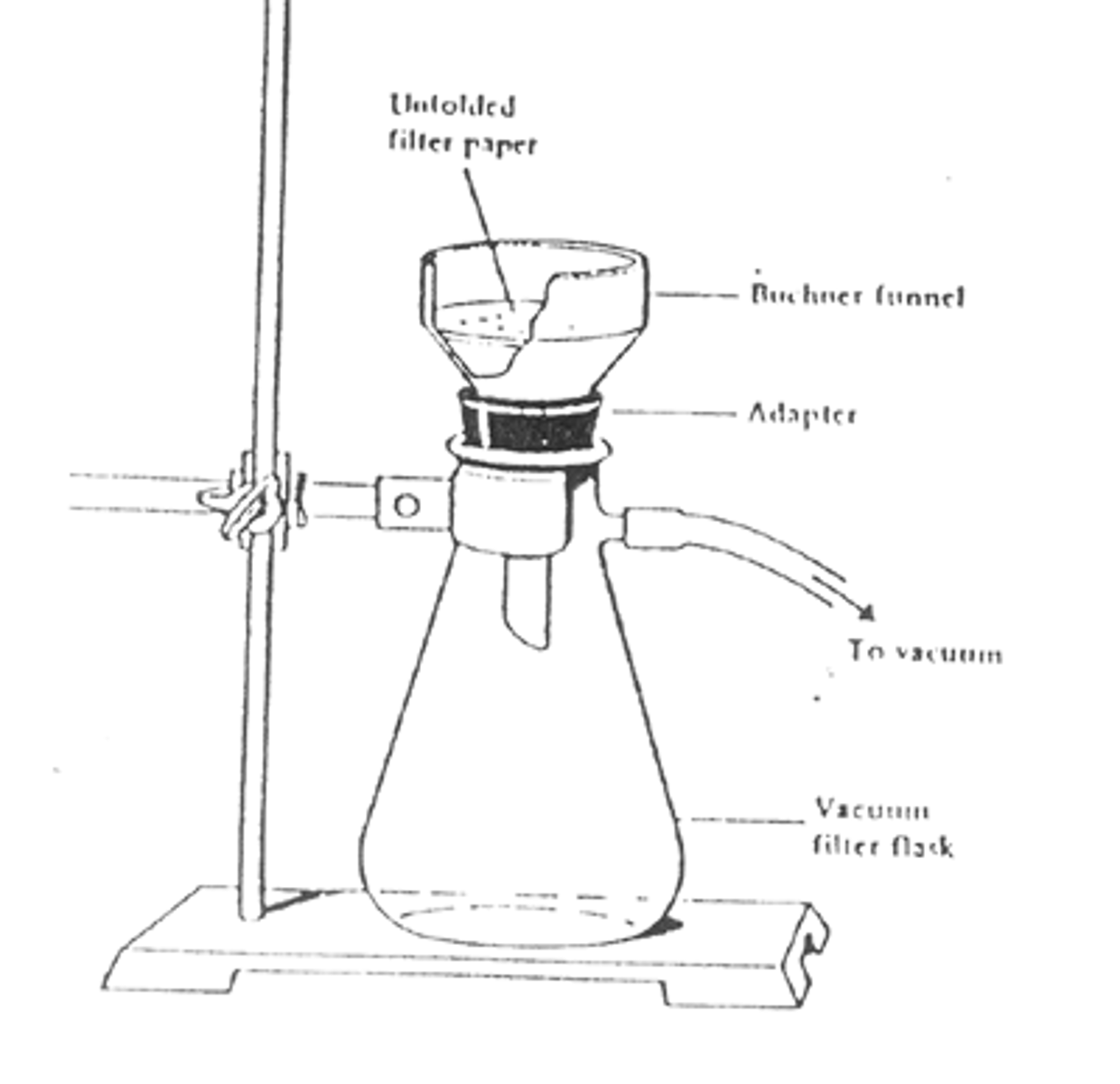
Vacuum Filtration
separate a precipitate from the solvent
Vacuum filtration is performed with a Hirsch or Buchner funnel.
A vacuum is created inside of the flask which creates suction to pull the filtrate through the filter paper.
The filter usually has holes in it which are covered by the filter paper.
The primary advantage is that it is faster than gravity filtration.
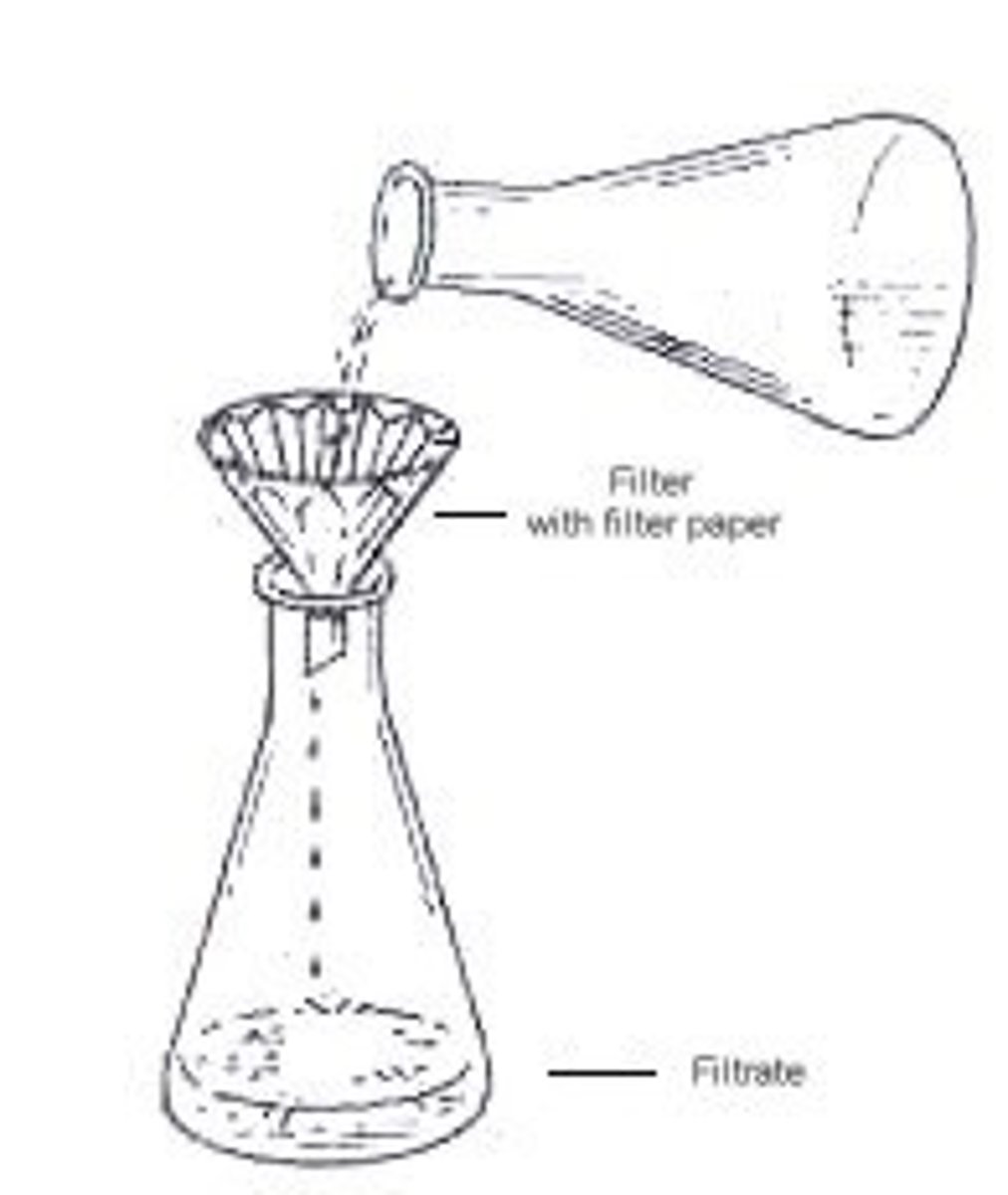
Gravity Filtration
remove drying agent from organic phase
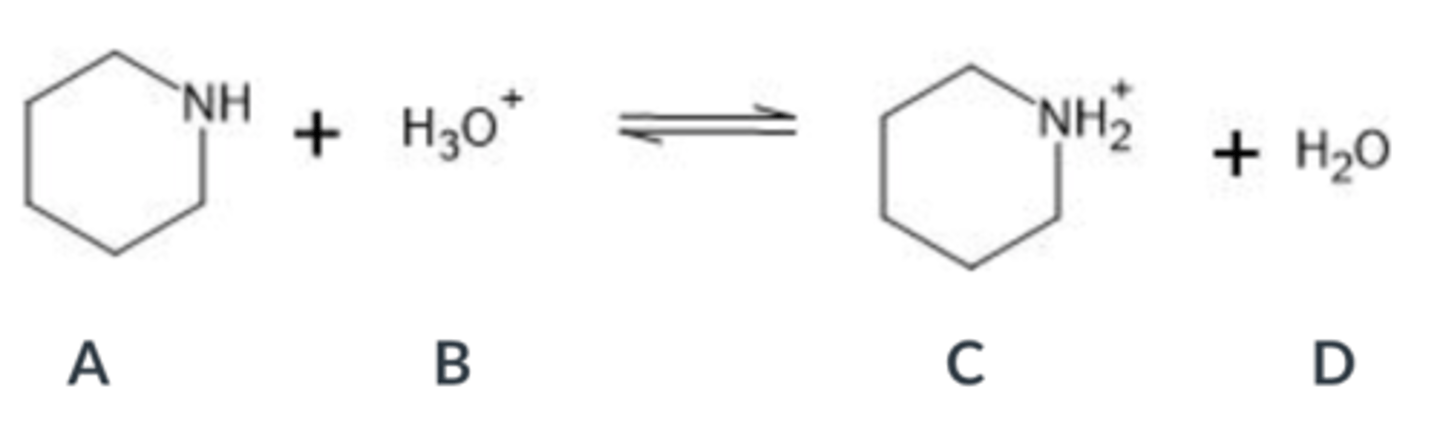
Select the base in the following acid-base equation
A
B
C
D
A
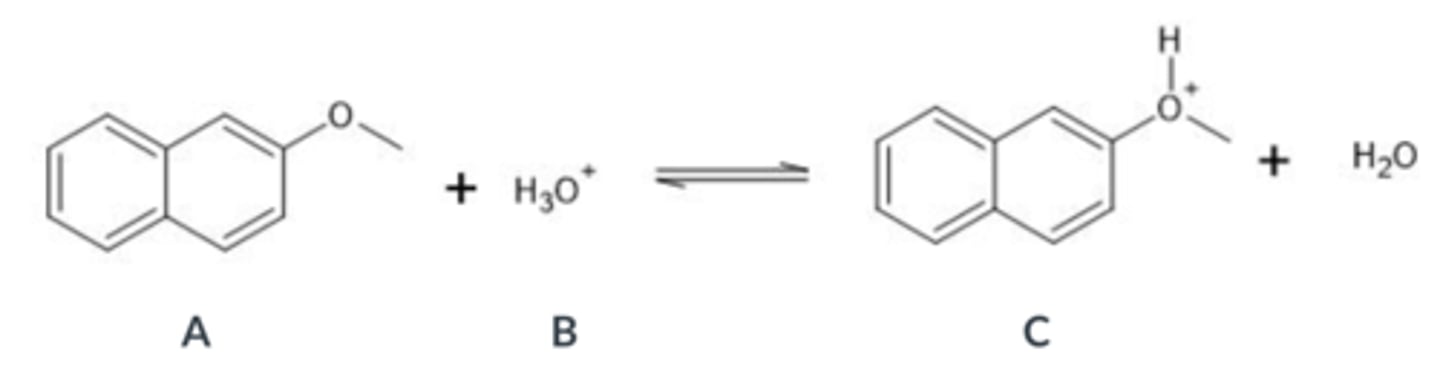
Select the conjugate acid in the following acid-base equation
A
B
C
D
C
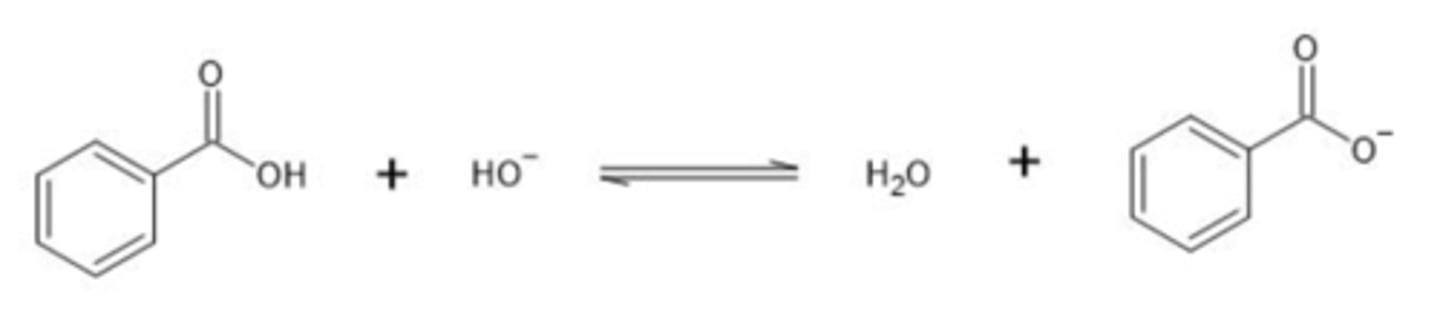
What direction is equilibrium favored in the following reaction?
a. Toward left (reactants)
b. Toward right (products)
c. It will be at equilibirum
B
What would you add to a separatory funnel to separate these compounds via an acid-base extraction? (They are already dissolved in ether)
a. HCl
b. NaOH
c. Ethanol
d. Water
A
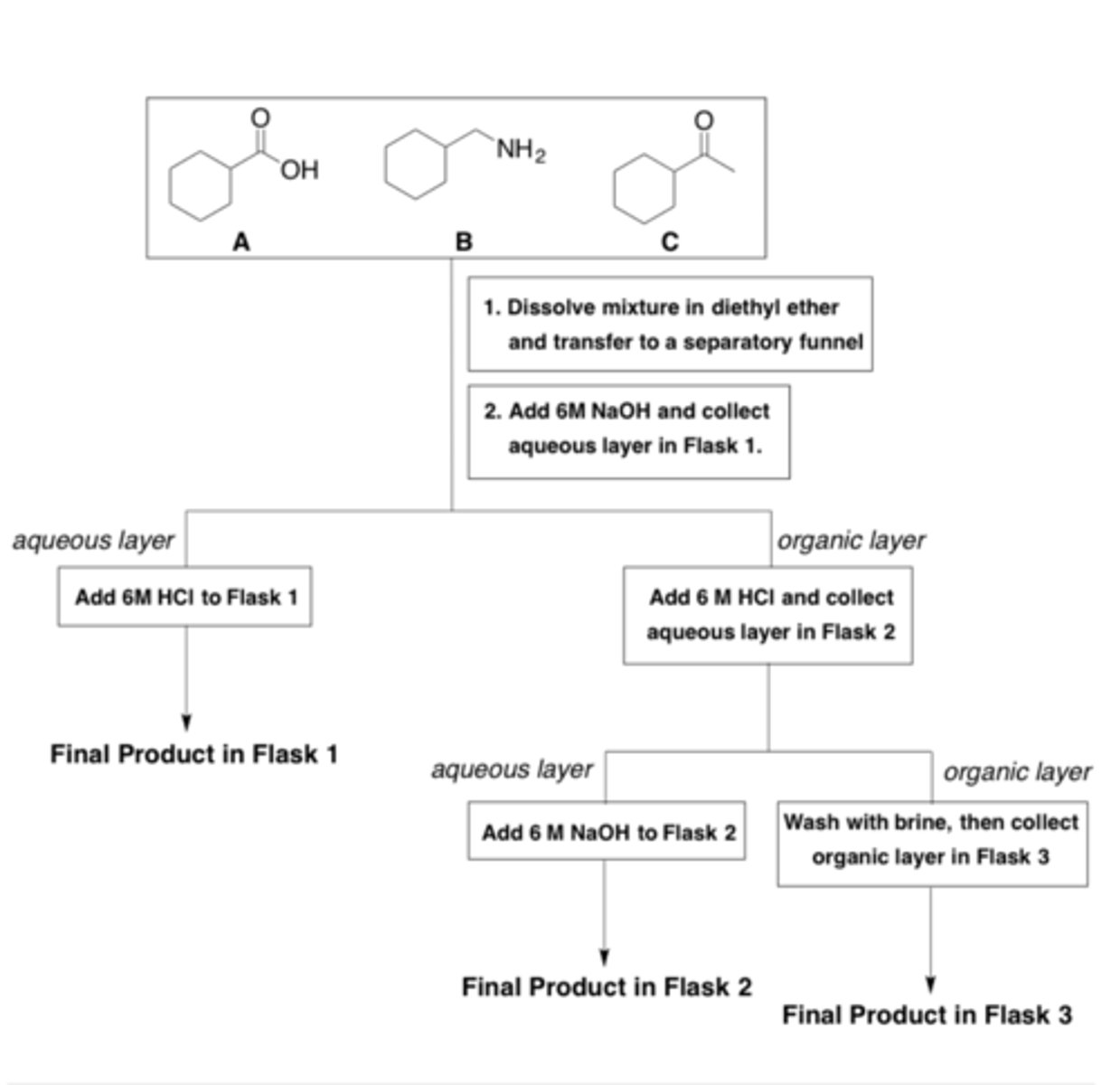
An acid-base extraction is performed to separate compounds A, B, and C. The flow chart for the procedure is shown below.
Which compound (A, B, or C) will be present in Flask 2 at the end of this purification step?
B
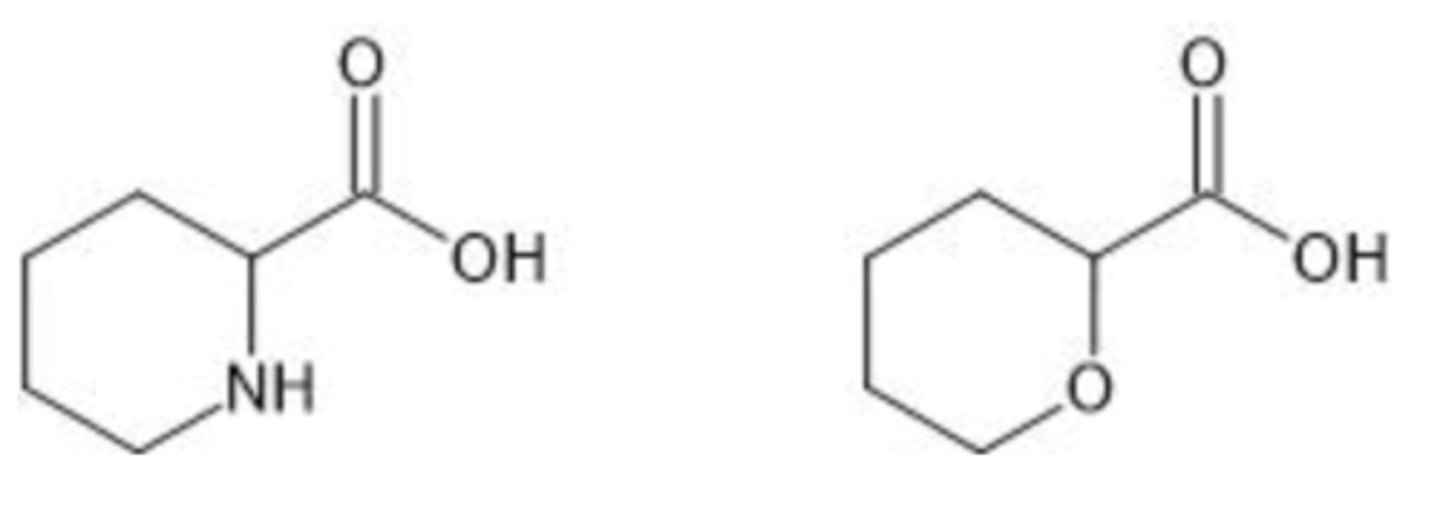
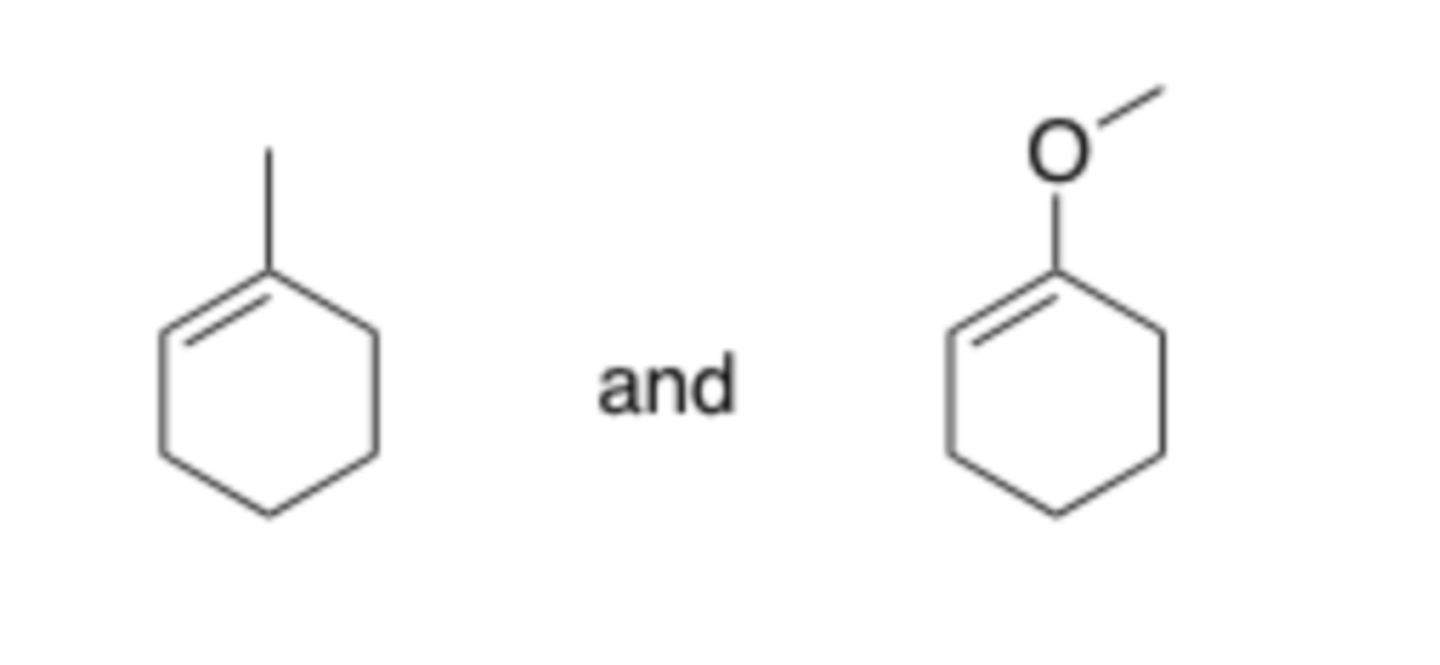
Could extraction be used to separate a mixture of the two compounds shown below? (Think ahead to reactivity)
a. Yes
b. No
B
What would be Ktoluene/water be for a compound A if, at equilibrium after extraction, it was found that 6.1g of A were dissolved in 10mL of toluene and 21.2g of A was dissolved in 20mL of water?
a. 0.575
b. 1.738
c. 0.610
d. 1.060
A
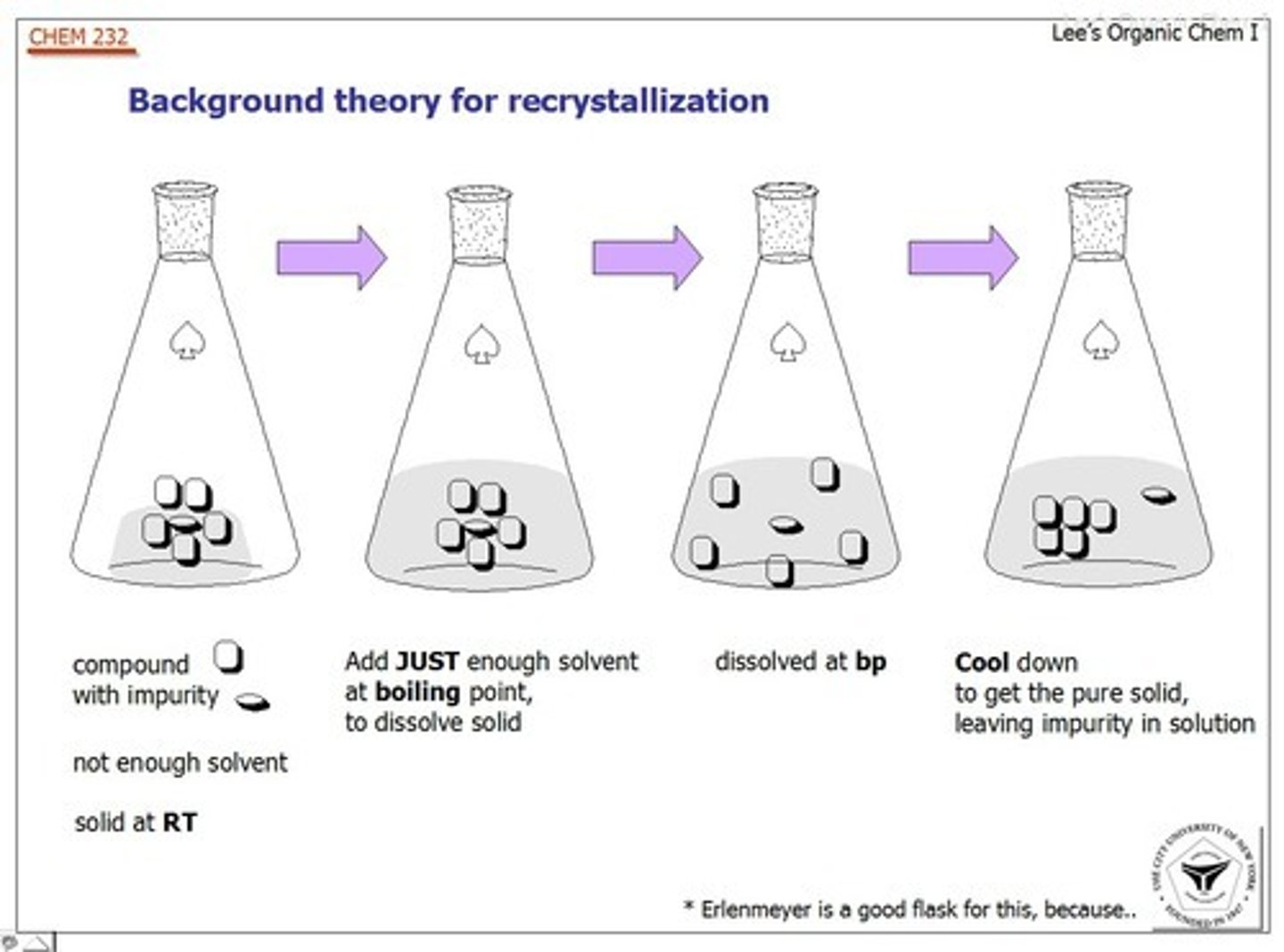
Recrystallization
- Purifies solids by dissolving them in hot solvent, cooling to precipitate pure crystals.
- Slow cooling = purer crystals.
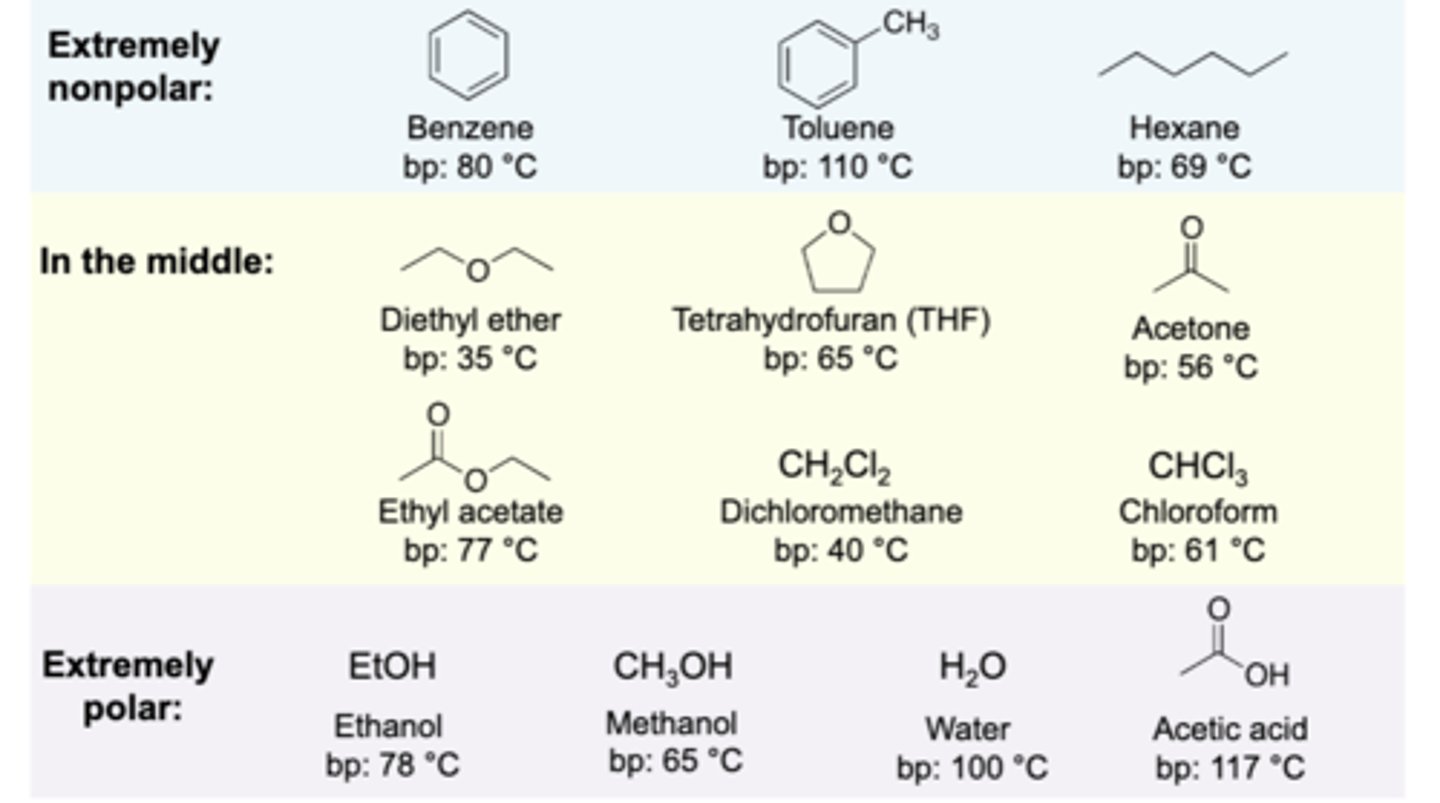
Common Solvents Table
helpful visual.
Mixed Solvent - a pair of miscible solvents, where a solute is very soluble in one solvent (aka "good" solvent), and poorly soluble in another (aka "bad" solvent).
Recrystallization steps
1. Dissolve the solid in a minimal amount of hot solvent.
2. Allow the solution to cool to room temperature.
3. Cool further in an ice bath.
4. Filter the mixture through a Buchner funnel to collect the solid.
5. Weigh the solid to get a mass of compound recovered
6. Measure the melting point
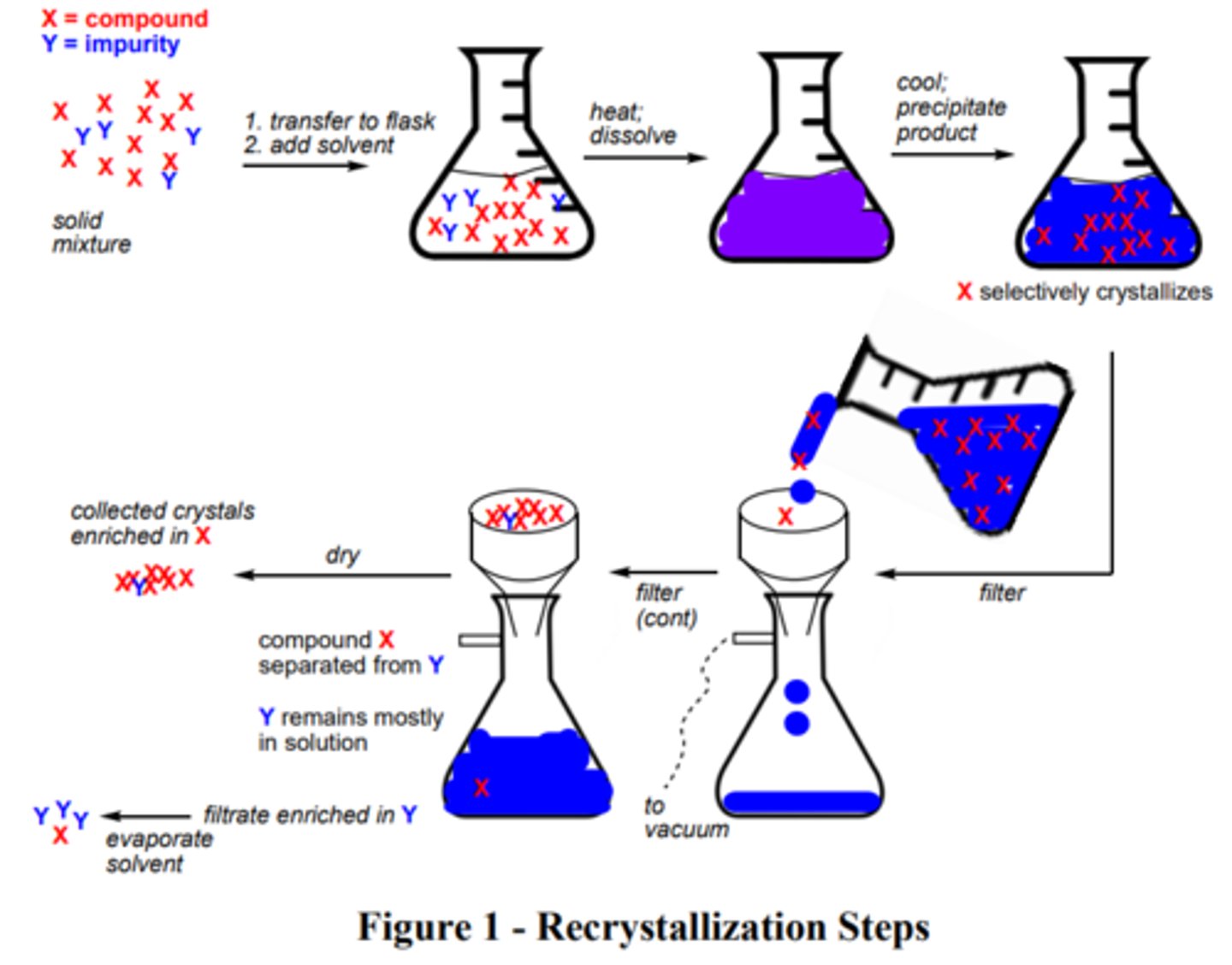
Melting point
- Pure compounds = sharp melting point.
- Impurities lower/broaden melting point.
Typically reported as a range (e.g., 102-104 °C): this indicates the temperature at which it begins to melt and the temperature at which it is entirely liquid
• Some compounds decompose rather than melt [e.g., 102 d or 102 °C (dec)]
If soluble impurities are present, melting point is lowered (mp depression) and range is broadened (sharp mp generally indicates a pure sample)
• Disrupt crystal lattice ® less energy is required to overcome attractive forces inthe solid
• Even a 1% impurity can cause mp depression
Insoluble impurities do not affect the melting point (can't be incorporated into thecrystal lattice)
IR Spectroscopy
IR (infrared) spectroscopy deals with the infrared region of the electromagnetic spectrum.
- Used to confirm the presence of functional groups
- IR is used to say "yes, a group is there" but cannot be used to say "no, a group is not there" without additional data
• When analyzing IR spectra, work from left to right. The left side of the spectrum tends to give us the most information
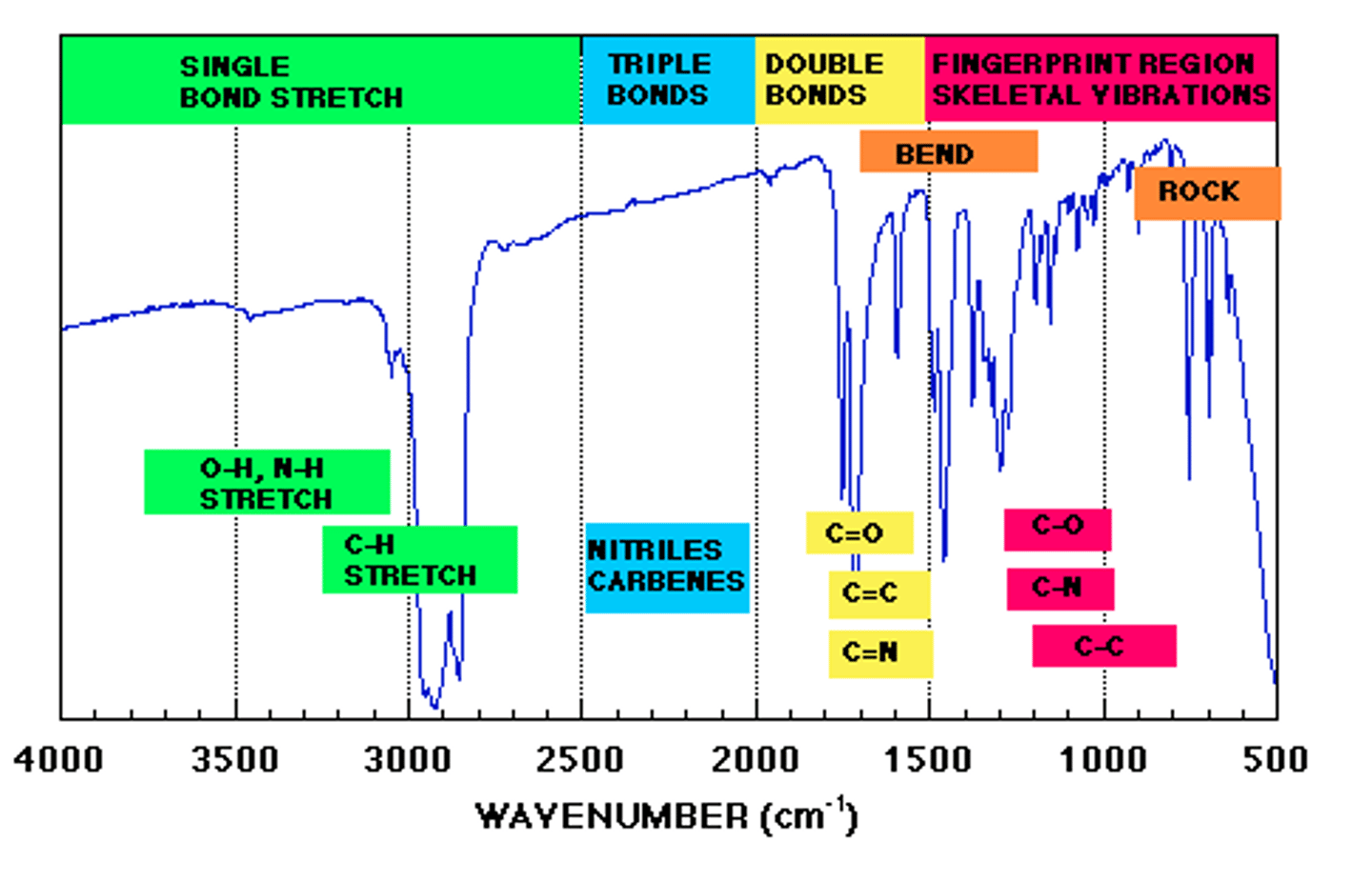
Major regions of absorption
look at graph
Mechanical losses can occur during the recrystallization process because...
a. All of the above.
b. too much solvent is added to dissolve the compound.
c. the product is filtered before crystallization is complete.
d. crystals form in the pipette during a microscale gravity filtration.
A
Often during recrystallization, a boiling stick is used in the heated solvent or sample, rather than boiling stones. Which of the following statements concerning boiling sticks is true?
a. All of the above are true.
b. A boiling stick is used rather than boiling stones because the stick is easier to remove.
c. Without the boiling stick, the solution being heated will most likely superheat and boil over.
d. Boiling sticks promote even boiling by providing a porous surface on which small bubbles can nucleate.
A
Given the following information, which solvent would be best for the recrystallization of compound Y?
A
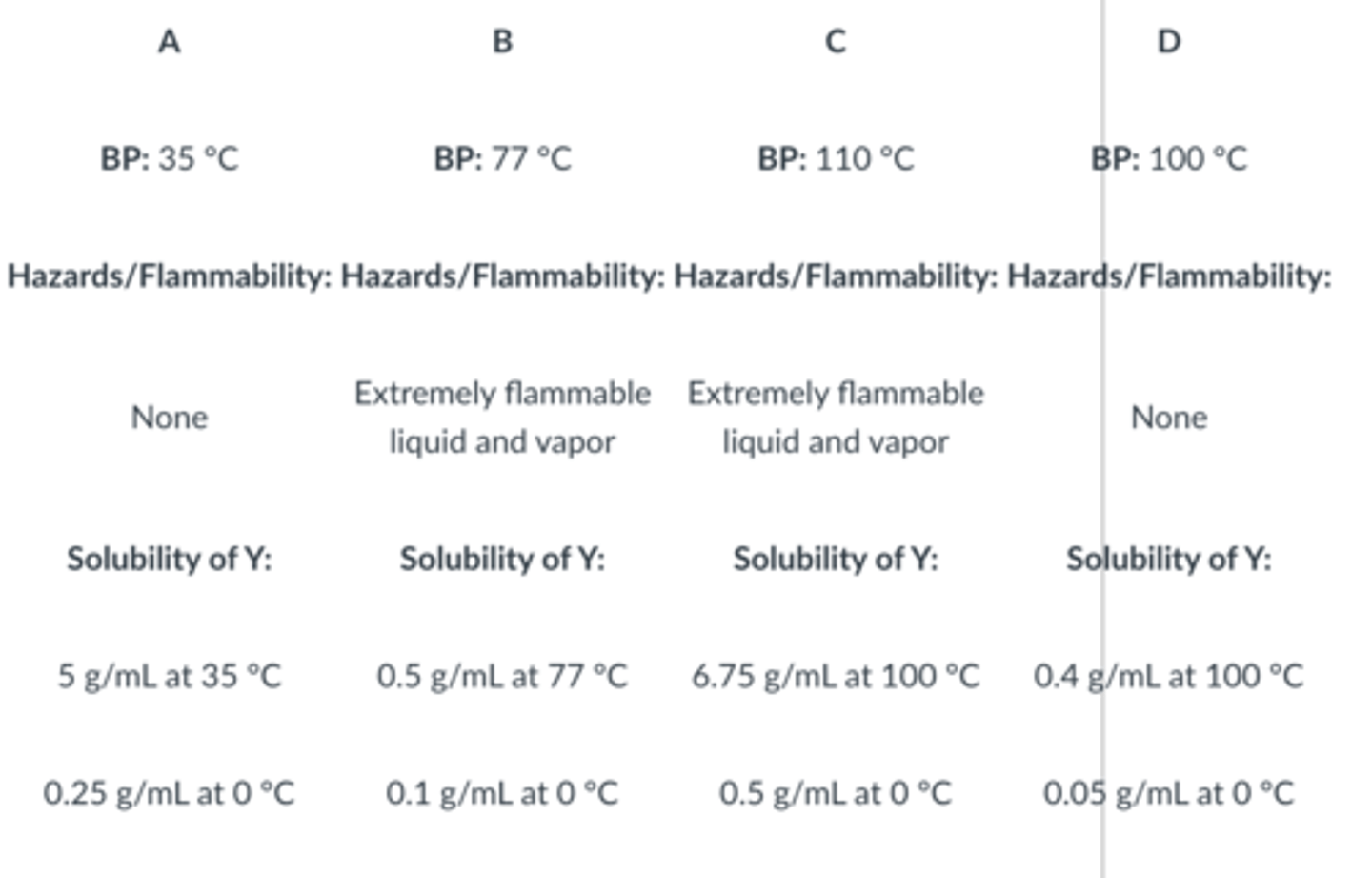
Which of the following is not true of a good recrystallizing solvent?
a. It is reactive.
b. It dissolves the desired compound better at high temperatures.
c. It is non-toxic.
d. It is volatile.
A
Theoretically, how much water would it take to dissolve a 150 mg sample of a compound whose solubility in water at 98 oC is 3.0 g/100 mL water?
a. 5.0 mL
b. 2.0 mL
c. 10. mL
d. 4.5 mL
A
If the melting point of a sample is unknown, which of the following can be done in order to save time while obtaining a value?
a. The unknown sample can be inserted into a preheated apparatus at 100 oC. If the sample melts, the apparatus is cooled and an accurate mp is recorded. If the sample doesn't melt, the temperature is quickly raised to 200 oC and the process above is repeated.
b. There is no way to save time when determining a melting point. The only way to obtain an accurate melting point is by increasing the temperature at a rate of 1-2 oC per minute.
c. A rough melting point can be obtained by increasing the temperature at a rate of about 5-10 oC per minute and then repeating more accurately once the apparatus has cooled.
d. The best way to save time is to find a neighbor that has already obtained the melting point and ask them their values, which are then recorded in your notebook.
C
While obtaining a melting point temperature, what is a proper rate of temperature change during melting?
a. 1 oC/sec
b. 1 oC/min
c. 5 oC/min
d. 1 oC/5 sec
B