Copy of Bio 150 questions.docx
1/358
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
359 Terms
What are the key characteristics that define a living organism?
Living organisms grow, reproduce, respond to stimuli, metabolize, maintain homeostasis, adapt to their environment, and consist of one or more cells.
What are the six key elements in biology?
The six key elements are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S).
How do carbon and hydrogen primarily bond?
Carbon and hydrogen primarily form nonpolar covalent bonds.
How are oxygen, nitrogen, and sulfur different in their bonding?
Oxygen, nitrogen, and sulfur tend to form polar covalent bonds due to their higher electronegativity.
What is a covalent bond?
A covalent bond is a chemical bond formed by the sharing of electrons between atoms.
What distinguishes covalent bonds from non-covalent interactions?
Covalent bonds are strong connections due to shared electrons, while non-covalent interactions are weaker, including hydrogen bonds, ionic interactions, van der Waals forces, and hydrophobic effects.
What is a polar covalent bond?
A polar covalent bond occurs when electrons are shared unequally, resulting in partial charges on the atoms.
What defines a nonpolar molecule?
A nonpolar molecule has a nonpolar covalent bond, where electrons are shared equally, resulting in no partial charges.
Why is water considered essential for life?
Water is essential due to its polarity, high specific heat, cohesive and adhesive properties, ability to dissolve many substances, and role in temperature regulation.
What are the four main types of non-covalent interactions in biology?
The four non-covalent interactions in biology are hydrogen bonds, ionic interactions, van der Waals forces, and hydrophobic interactions.
How do functional groups enhance non-covalent interactions in macromolecules?
Functional groups interact through various non-covalent interactions: hydroxyl groups form hydrogen bonds, charged groups participate in ionic interactions, and nonpolar groups engage in van der Waals forces.
What is a macromolecule?
A macromolecule is a large biological molecule such as proteins, nucleic acids, carbohydrates, or lipids.
What is a polymer?
A polymer is a chain of repeating monomers, which are smaller subunits that compose macromolecules.
What are the four types of macromolecules in biology?
Proteins (amino acids), nucleic acids (nucleotides), carbohydrates (monosaccharides), and lipids (fatty acids).
How are polymers synthesized?
Polymers are synthesized through dehydration synthesis, where monomers are linked by covalent bonds with the release of water.
What process breaks down polymers?
Polymers are broken down through hydrolysis, which adds water to the bonds, breaking them.
How do nucleic acids utilize non-covalent bonding?
Nucleic acids use complementary base pairing (A-T/U, G-C) through hydrogen bonding, forming the backbone structure.
What is DNA replication?
DNA replication is the process of copying DNA, facilitated by the double-helix structure and complementary base pairing.
What is gene expression?
Gene expression is the process by which information from DNA is used to produce functional products, typically proteins.
What happens during transcription?
Transcription is the process where a cell copies a specific sequence of DNA (a gene) into a complementary RNA molecule, essentially creating a "message" that can be used to build a protein; this process involves an enzyme called RNA polymerase reading the DNA template strand and assembling a new RNA strand by matching nucleotides according to base pairing rules, with the resulting RNA molecule called messenger RNA (mRNA) carrying the genetic information to the ribosomes for protein synthesis. RNA polymerase is the main enzyme used. (Initiation (RNA polymerase binds to the DNA), elongation (RNA nucleotides are added to the growing RNA chain), and termination (RNA polymerase releases the DNA and the newly formed RNA molecule). )
What happens during translation?
a cell uses the information carried by messenger RNA (mRNA) to build a protein by linking together amino acids into a polypeptide chain, essentially converting the genetic code from nucleotides to a sequence of amino acids; this occurs at the ribosome within the cell cytoplasm, where the mRNA is "read" three bases at a time (codons) to match with corresponding transfer RNA (tRNA) molecules carrying specific amino acids, ultimately forming a protein
What defines the structure of an amino acid?
An amino acid consists of an amino group, a carboxyl group, and a unique R group (side chain).
What are the four structural levels of proteins?
The four levels are primary (amino acid sequence), secondary (alpha helices and beta sheets), tertiary (3D folding), and quaternary (assembly of polypeptides).
How do proteins fold into functional structures?
Proteins fold through the formation of secondary, tertiary, and quaternary structures driven by various non-covalent interactions.
What constitutes secondary structure in proteins?
Secondary structure includes alpha helices and beta sheets stabilized by hydrogen bonds within the backbone.
What is tertiary structure in proteins?
Tertiary structure refers to the 3D arrangement of a polypeptide, stabilized by various non-covalent interactions.
What is quaternary structure in proteins?
Quaternary structure involves the arrangement of multiple polypeptides; not all proteins have this level.
How does information flow from DNA to protein?
Information flows from DNA to RNA via transcription, then from RNA to protein via translation.
What is the general formula for carbohydrates?
The general formula for carbohydrates is (CH2O)n.
Why is sugar molecule shape important?
Shape determines function; specific glycosidic bonds affect properties and digestibility.
Why are sugars soluble in water?
Sugars like glucose form hydrogen bonds with water due to their hydroxyl groups, making them highly soluble.
What gives polysaccharides their properties?
Cellulose ropes are rigid from extensive hydrogen bonding, while polysaccharide jellies form gels by trapping water.
What is the general structure of a hydrocarbon?
The general formula of a hydrocarbon is CxHy. Usually hexagonal shaped
What defines lipids?
Lipids are hydrophobic or amphipathic molecules that do not mix well with water.
What is the structure of a fatty acid?
A fatty acid contains a carboxyl group and a hydrocarbon chain, which varies in length and saturation.
What defines triacylglycerols?
Triacylglycerols consist of three fatty acids linked to a glycerol molecule, serving as energy storage.
What is a phospholipid's role in membranes?
A phospholipid has a hydrophilic head and hydrophobic tails, forming lipid bilayers essential for membranes.
What does amphipathic mean?
Amphipathic means possessing both hydrophilic and hydrophobic regions, vital for membrane structure.
What is the hydrophobic effect?
The hydrophobic effect causes nonpolar molecules to cluster in aqueous environments, promoting membrane assembly.
How do hydrocarbons interact?
Hydrocarbons interact through van der Waals forces due to induced dipoles.
What is potential energy?
Potential energy is the stored energy, including chemical energy in bonds.
How does diffusion relate to entropy?
Diffusion increases entropy as systems naturally tend toward higher disorder.
What distinguishes endergonic from exergonic reactions?
Endergonic reactions require energy input (positive ΔG), while exergonic reactions release energy (negative ΔG).
Why do cells maintain steady-state non-equilibrium?
Cells should not be at equilibrium because reaching equilibrium would mean a complete cessation of chemical reactions and therefore no ability to perform work, essentially leading to cell death; living organisms require a constant flow of energy to maintain their functions
What is chemical equilibrium?
Chemical equilibrium occurs when the rates of forward and reverse reactions are equal, with no net change.
What is steady-state non-equilibrium?
Steady-state non-equilibrium involves continuous energy and matter input-output, differing from equilibrium.
What is a kinetically stable molecule?
A kinetically stable molecule resists reaction due to high activation energy, despite being thermodynamically favorable.
Why is ATP considered the cell's energy currency?
ATP hydrolysis releases energy essential to power other reactions.
What is energetic coupling?
Energetic coupling involves using ATP hydrolysis to drive non-spontaneous reactions forward.
How do cells propel endergonic reactions?
Cells drive endergonic reactions using enzymes, ATP, and other molecules.
What is the transition state in a reaction?
The transition state is a high-energy intermediate; activation energy is needed to reach this state.
How do enzymes catalyze reactions?
Enzymes lower activation energy by stabilizing transition states during reactions.
What is the active site of an enzyme?
The active site is where substrates bind and catalysis occurs, often orienting substrates for efficiency.
How do cells control enzyme activity?
Cells manage enzyme activity by altering enzyme or substrate concentrations through various mechanisms.
What is metabolism?
Metabolism includes all chemical reactions in a cell, encompassing catabolism and anabolism.
What is feedback inhibition?
Feedback inhibition limits overproduction by using the end product to inhibit early steps, maintaining homeostasis.
What is the function of kinases and phosphatases?
Kinases add phosphate groups to molecules; phosphatases remove them, both regulating enzyme activity.
What is the basic structure of membrane lipids?
Membrane lipids consist of hydrophilic heads and hydrophobic tails, allowing for bilayer formation.
What role does the hydrophobic effect play in membranes?
The hydrophobic effect stabilizes membranes by excluding nonpolar molecules from water.
What is the function of triacylglycerols?
Triacylglycerols store energy but do not contribute to membrane structure.
What determines membrane fluidity?
Cell membrane fluidity is the property of the cell membrane that allows it to adapt its shape and movement to different conditions. Three key factors influence cell membrane fluidity: temperature, cholesterol, and the kind of fatty acids in the phospholipids that form the cell membrane.
How do cells regulate membrane fluidity?
Cells adjust lipid composition, increasing unsaturated fatty acids at lower temperatures.
What are the types of membrane proteins?
Integral membrane proteins-These proteins are embedded in the lipid bilayer, and can cross the membrane entirely. They have at least one stretch of uncharged amino acids that spans the membrane, and their hydrophobic nature keeps them embedded. Integral proteins can act as receptors, channels, or transporters to move molecules across the membrane.
Peripheral membrane proteins-These proteins don't cross the membrane, but can attach to either side of the membrane or to other proteins. They can function in cell signaling, anchoring intracellular components, and acting as enzymes.
Lipid-anchored membrane proteins-These proteins are covalently attached to lipid molecules in the lipid bilayer. They can function in cell signaling, enzyme activity, and molecular recognition.
What is the function of alpha helices in transmembrane proteins?
Alpha helices span the hydrophobic membrane, forming transport channels.
How do proteins associate with membranes?
Proteins associate with membranes through hydrophobic regions or covalent lipid attachment.
What role does the plasma membrane serve in homeostasis?
The plasma membrane controls substance exchange, regulating internal conditions.
What is selective permeability?
Selective permeability allows certain molecules to pass, crucial for cell function.
What is diffusion?
Diffusion is the passive movement of molecules from high to low concentration.
What distinguishes facilitated diffusion from diffusion?
Facilitated diffusion requires transport proteins, while diffusion does not.
What defines osmosis?
Osmosis is the diffusion of water across a selectively permeable membrane.
What is active transport?
Active transport moves molecules against concentration gradients using energy.
Give an example of facilitated diffusion.
GLUT transporter is an example that transports glucose without energy input.
How do prokaryotes utilize transport systems?
Prokaryotes use transporters and pumps for nutrient uptake powered by gradients or ATP.
What is the basic structure of a prokaryotic cell?
A prokaryotic cell has a cell membrane, cytoplasm, cell wall, singular DNA chromosome in the nucleoid, and ribosomes.
How can organisms be classified based on energy and carbon sources?
Organisms can be classified as phototrophs or chemotrophs for energy, and autotrophs or heterotrophs for carbon sources.
What is the equation for oxidative metabolism?
6O2 + C6H12O6 → 6CO2 + 6H2O + ATP.
What is the equation for photosynthesis?
6CO2 + 6H2O + light energy → C6H12O6 + 6O2.
Why is ATP a good energy currency?
ATP's phosphate bonds are intermediate in strength, allowing controlled energy release.
Differentiate substrate-level and oxidative phosphorylation.
Substrate-level phosphorylation involves direct phosphorylation by a substrate, while oxidative phosphorylation uses energy from redox reactions.
What is fermentation?
Fermentation is an anaerobic metabolic process converting nutrients like sugars into ATP, recycling electron carriers.
How does diffusion limit cell size in prokaryotes?
Diffusion limits cell size as larger volumes take longer to transport resources via passive diffusion.
Compare prokaryotic and eukaryotic cell organization.
Prokaryotic cells are unicellular and lack a nucleus, while eukaryotic cells are multicellular with membrane-bound organelles and a nucleus.
What constitutes the endomembrane system?
The endomembrane system includes the rough ER, Golgi apparatus, plasma membrane, lysosomes, and vesicles.
Which organelles are not part of the endomembrane system?
Mitochondria, chloroplasts, and peroxisomes are not part of the endomembrane system.
What is the nuclear envelope's structure?
The nuclear envelope is a double membrane with large protein channels called nuclear pores.
What are the rough and smooth ER's roles?
The rough ER is involved in protein synthesis, while the smooth ER is involved in lipid synthesis.
What are vesicles, and which organelles do they connect?
Vesicles transport materials and interconnect organelles like the Golgi apparatus and the plasma membrane.
What occurs in the Golgi apparatus?
The Golgi apparatus modifies, sorts, and packages proteins and lipids for their final destinations.
What happens in lysosomes?
Lysosomes contain enzymes that degrade macromolecules like proteins, nucleic acids, and lipids.
Why are cells limited in size?
Diffusion limits cell size due to the time needed to transport nutrients over larger volumes.
How do organelles support larger cell sizes?
Organelles allow compartmentalization of functions within cells, enhancing efficiency.
How do we think organelles evolved?
Organelles might have evolved from free-living prokaryotes that were engulfed by other cells.
What does 'repurposed plasma membrane' refer to?
Repurposed plasma membrane refers to membranes from previous cells used to create new organelles.
What is endosymbiosis?
Endosymbiosis is the theory suggesting that some organelles were once free-living bacteria engulfed by a host cell.
How does diffusion limit prokaryote cell size?
Prokaryote size is limited as nutrients diffuse slowly over larger volumes, restricting cellular function.
What are the stages of cellular respiration?
The stages are glycolysis, pyruvate oxidation, the Krebs cycle, and oxidative phosphorylation.
How do inputs and outputs differ for cellular respiration steps?
Glycolysis inputs glucose, producing pyruvate; pyruvate oxidation inputs pyruvate to yield acetyl CoA; the Krebs cycle processes acetyl CoA to generate electron carriers; oxidative phosphorylation inputs electrons to create ATP.
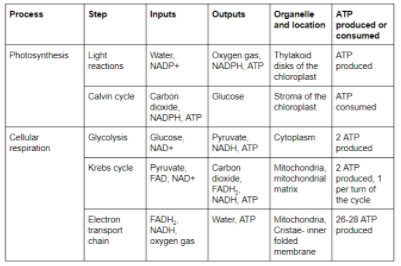
Describe inputs and outputs of glycolysis. What occurs in glycolysis?
Inputs: Glucose, 2 NAD+, 2 ATP. Outputs: 2 pyruvate, 2 NADH, 4 ATP, 2 H2O. In glycolysis, glucose is broken down to generate energy.
Describe pyruvate oxidation. What are the inputs and outputs?
Inputs: Pyruvate, NAD+, CoA. Outputs: Acetyl CoA, CO2, and NADH. Pyruvate is oxidized to enter the Krebs cycle.
What happens in the citric acid cycle?
Inputs: Acetyl CoA; Outputs: CO2, NADH, FADH2, ATP. The cycle fully oxidizes fuel molecules to harvest energy.