Bio Chapters 6,7,8 ,9
1/242
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
243 Terms
aerobic respiration
consumes organic molecules and O2 and yields ATP; uses the electron transport chain
C6 H12 6O2 —> 6 CO2 + 6 H2O + Energy (ATP + heat)
anaerobic respiration
consumes organic molecules (AND NOT O2) and also yields ATP; uses electron transport chain
fermentation
partial degradation of organic molecules that occurs without O2 yielding ATP (usally less than with cellular respiration); no electron transport chain used
cellular respiration
the catabolic pathways of aerobic and anaerobic respiration, which break down organic molecules and use an electron transport chain for the production of ATP
transport of electrons
transfer of electrons from one molecule to another = transfer of energy
a common activated carrier of energy is…
NAD+
(NAD+ ) + 2e- (high energy electrons from a fuel molecule) + (H+) —> NADH
doesn’t carry any phosphate groups
carries electrons
NAD+ is low in free energy
energy can be stored in electrons and
their interactions with atoms.
oxidation
loss of electrons
reduction
gain of electrons
Electrons transferred come from
free electrons that have been energized
Hydrogen atom (1 electron and one proton) bound as part of molecule [C-H], since [C-H] bonds are used in transfer of electrons a lot. More [C-H] bonds means more reduced
complete breakdown of glucose in eukaryotes:
glycolysis
energy investment phase
aerobic respiration (occurs in the mitochondria)
pyruvate oxidation
citric acid cycle
oxidative phosphorylation
Substrate-level phosphorylation
synthesis of ATP from direct transfer of phosphate group from another
glycolysis energy investment phase
2 ATP used
Glucose makes —> GAP and DHAP
only GAP continues on
glycloysis energy pay-off phase
2x GAP
GAP oxidized —> e- used to reduce NAD+ to form NADH (energized from the electron)
Another phosphate group added (NOT FROM ADP)
oxidative phosphorylation
the production of ATP using energy derived from the redox reactions of an electron transport chain; the third major stage of cellular respiration
glycolysis
a series of reactions that ultimately splits glucose (6-carbon molecule) into 2 pyruvate(3-carbon molecule) . Glycolysis occurs in almost all living cells, serving as the starting point for fermentation or cellular respiration.
starting reactant of glycolysis
glucose
final product of glycolysis
2 pyruvates
2 ATP’s
2 NADH
enzymes always have the suffix of
-ase
mitochondria
Double membraned, inner membrane folded to form cristae
site of aerobic respiration
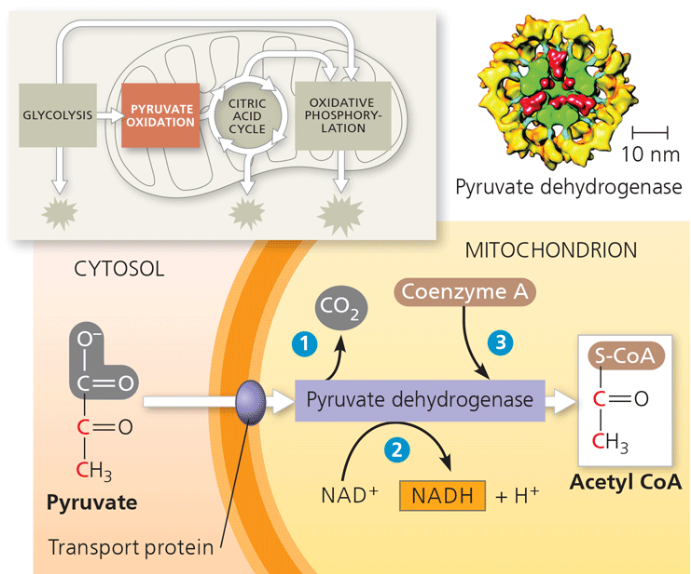
pyruvate oxidation
Pyruvate enters mitochondrial matrix.
Pyruvate undergoes oxidative decarboxylation (loses CO2 and electrons); the remainder gets attached to coenzyme to form A to form acetyl CoA.
NAD+ becomes reduced to form NADH.
oxidative decarboloyation
loss of CO2+ —> loss of electrons —> the electrons are added to NAD+ —> NADH
FAD —> FADH2 (citric cycle)
another activated carrier in electrons/ energy
For one Acetyl- CoA, you make…
3 NADH (carrier of electron)
1 FADH2 (carrier of electron)
1 ATP (GTP) (occurs via substrate-level phosphorylation)
Carbons from acetyl CoA are given off as CO2
For 1 glucose
(glycolysis) 2 ATP and 2 NADH
1 glucose makes 2 pyruvates,
(pyruvate oxidation) makes 2 NADH
2 pyruvates makes 2 acetyl COA
(citric acid cycle)
produces 2 CO2
6 NADH
2 FADH2
2 ATP
oxidative phosphorylation
synthesis of ATP through oxidation of NADH + FADH2 + electrons moving through a series of electron carriers
electron transport chain
chemiosmosis
Electron transport chain
Electrons (from NADH or FADH2) move from an electron carrier with a lower affinity for electrons to an electron carrier down the chain with a greater affinity for electrons, releasing free energy.
NADH or FADH2 oxidized
electrons transferred through oxidation/reduction reactions until reducing oxygen (O2)
chemiosmosis
an energy coupling mechanism that uses energy stored in the form of a hydrogen ion gradient across a membrane to drive cellular work, such as the synthesis of ATP. under aerobic conditions, most ATP synthesis in cells occurs by chemiosmosis.
production of ATP utilizing energy released from H+ moving down the concentration gradient.
Result of electron transport chain: proton gradient (CH+)
H+ accumulate in intermembranous space between inner and outer membrane
ATP synthase
a complex of several membrane proteins that functions in chemiosmosis with adjacent electron transport chains, using the energy of a hydrogen ion (proton) concentration gradient to make ATP.
ATP synthases are found in the inner mitochondrial membranes of eukaryotic cells and in the plasma membranes of prokaryotes.
fermentation
partial degradation of organic molecules that occurs without O2 yielding ATP (usually less than with cellular respiration); no electron transport chain used.
lactate fermentation (lactic acid fermentation)
pyruvate is reduced directly by NADH to form lactate as an end product, regenerating NAD+ with no release of CO2.
NAD+ regenerated
lactate produced
ethanol(alcohol) fermentation
pyruvate is converted to ethanol (ethyl alcohol).
(2 pyruvates use up 2 NADH)
step 1. CO2 is released from the pyruvate, which is converted to the two-carbon compound acetaldehyde.
step 2. acetaldehyde is reduced by NADH to ethanol. This regenerates the supply of NAD+ needed for the continuation of glycolysis.
for organisms without oxidation phosphorylation —> NADH accumulates
If NADH accumulates, you run out of NAD+
NAD+ is needed for glycolysis…
therefore, fermentation regenerates NAD+
If accumulated, ATP can act as…
a feedback inhibitor to glucose breakdown
catabolic pathways are regulated.
what metabolic processes can occur without oxygen?
citric acid cycle
glycolysis
fermentation
anaerobic respiration
during which process is the most ATP directly generated in the cell?
oxidative phosphorylation
the main purpose of cellular respiration is to
convert energy stored in the bonds of a fuel molecule to energy stored in ATP
products of glycolysis
NADH
ATP
pyruvate
H2O
products of citric acid cycle
NADH
ATP
FADH2
CO2
which of the following statements concerning the synthesis of ATP is true?
the establishment of a charge and concentration gradient across the mitochondrial membrane is potential energy used by ATP synthase to synthesize ATP from ADP and Pi
products of oxidative phosphorylation
ATP
H2O
products of alcohol fermentation
ATP
CO2
Plasma membrane
Passive Transport
transporting small molecules, it doesn’t require energy and can involve transport proteins.
Active transport
requires energy and transport proteins
Exocytosis
Large molecules are secreted when a vesicle fuses with the plasma membrane
Endocytosis
Large molecules are taken in wwhen the plasma membrane pinches inward, forming a vesicle.
Amphipathic
meaning it has a hydrophilic (water loving) and hydrophobic (water fearing) region.
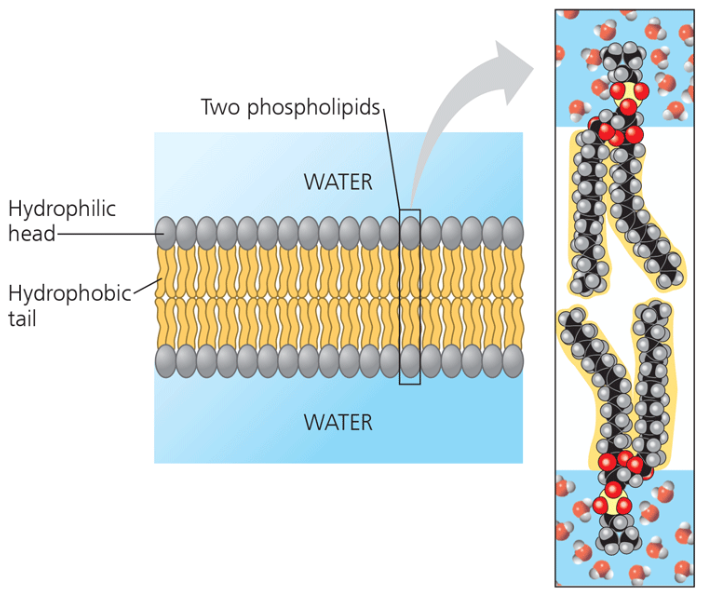
Fluid Mosaic model
The membrane is a mosaic of protein molecules bobbing in a fluid bilayer of phospholipids.
Membrane fluidity
Most of the lipids and some proteins can shift sideways. Lipids can also flip- flip across the membrane, switching from one phospholipid layer to the other.
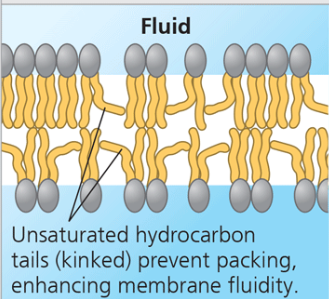
Unsaturated hydrocarbon tails (fluid)
Unsaturated hydrocarbon tails (kinks) prevent packing, enhancing membrane fluidity. (Remember! Unsaturated fatty acid means there is at least one double bond within that hydrocarbon chain. The double bond means sharing of two electrons. The double bond forms a bend in the fatty acid.)
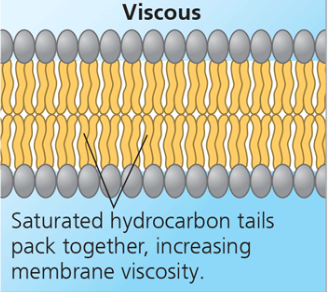
Saturated hydrocarbon tails (viscous)
Saturated hydrocarbon tails pack together, increasing membrane viscosity. (Remember! saturated fatty acid means no double bonds!)
The temperature at which a membrane solidifies depends on the types of lipids it is made of.
As the temperature decreases, the membrane remains fluid to a lower temperature if it has a lot of phospholipids with unsaturated hydrocarbon tails. Unsaturated hydrocarbon tails have kinks, meaning they cannot pack together as closely as saturated hydrocarbon tails, making the membrane more fluid.
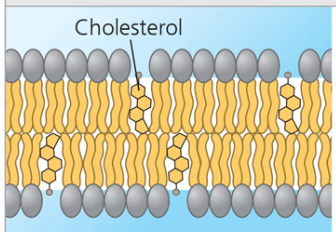
Cholesterol within the animal cell membrane.
Cholesterol reduces membrane fluidity at moderate temperatures by reducing phospholipid movement, but at low temperatures, it hinders solidification by disrupting the regular packing of phospholipids. Therefore, membranes are more fluid.
At normal/high temperatures, cholesterol decreases fluidity by packing in with unsaturated fatty acids, decreasing fluidity.
Integral proteins
Penetrate the hydrophobic interior of the lipid bilayer.
Peripheral proteins
not embedded in the lipid bilayer at all. They’re loosely bound to the surface of the membrane, often to exposed parts of integrap proteins.
transmembrane proteins
a type of integral protein that spans the entire length of a membrane. Pieces facing both sides of the membrane.
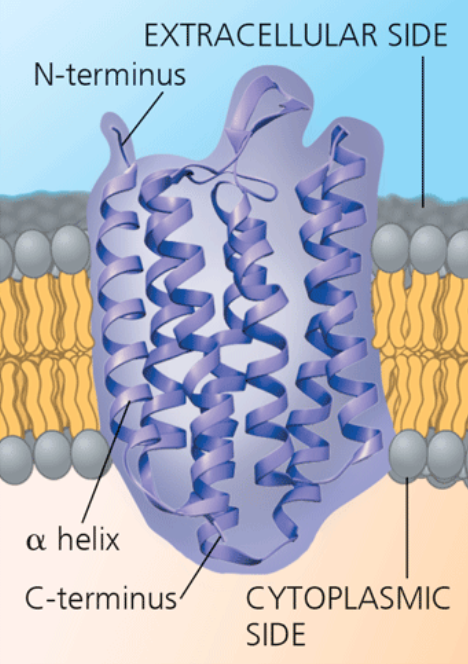
Bacteriorhodopsin (a bacterial transport protein)
It’s N-terminus side is outside the cell, and the C-terminus is inside. The ribbon model highlights the secondary structure of the hydrophobic parts. The nonhelical hydrophilic segments are in contact with the aqueous solutions on the extracellular and cytoplasmic sides of the membrane.
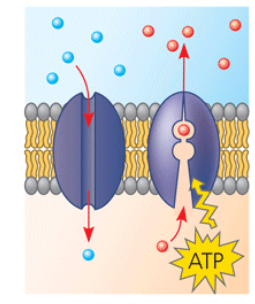
Transport proteins
Protein on the left provides a hydrophilic channel across the membrane that is selective for a particular solute.
Protein on the right: some transport proteins shuttle a substance from one side to the other by changing shape. Some proteins hydrolyze ATP as an energy source to actively pump substances across the membrane.
cell enzymatic acivity
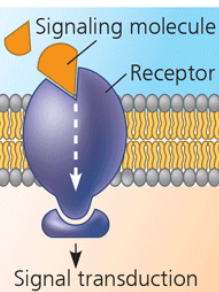
signal transduction
A membrane protein can have a binding site with a specific shape that fits the shape of a chemical messenger, like a hormone. The external messenger can cause the protein to change shape, causing it to send the message to the inside of the cell. This is usually done by binding to a cytoplasmic protein.
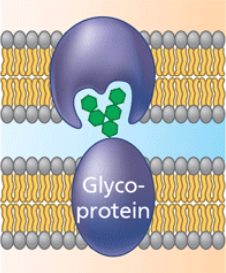
cell-cell recognition
some glycoproteins serve as identification tags that are specifically recognized by membrane proteins of other cells. This type of cell-cell binding is short lived, compared to intercellular joining.
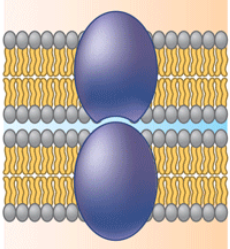
Intercellular joining
Membrane proteins of adjacent cells can hook together in various kinds of junctions.
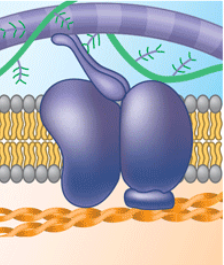
attachment to the cytoskeleton and extracellular matrix (ECM)
Microfilaments or other elements of the cytoskeleton may be non-covalently bound to membrane proteins. This helps maintain cell shape and stabilizes the location of certain membrane proteins. Proteins that can bind to ECM molecules can coordinate extracellular and intercellular changes.
many transmembrane proteins on the plasma membrane are…
glycoproteins (proteins that are attached to sugar).
membrane asymmetry
The side of the membrane facing the inside of the cell is different from the side facing the outside.
The inner membrane of the endoplasmic reticulum/vesicles/ Golgi is continuous with:
the outside of the cell and the outer layer of the membrane
selective permeability
a membrane allows some substances to cross more easily than others.
semi-permeable
permeability is based on size and polarity.
hydrophobic molecules are permeable.
small uncharged hydrophilic molecules are permeable.
Larger hydrophobic are usually impermeable.
Ions (charged substances) completely impermeable
Diffusion
the random thermal motion of particles of liquids, gases, or solids. In the presence of a concentration or electrochemical gradient, diffusion results in the net movement of a substance from a region where it was more concentrated to a region where it is less concentration.
(increases randomness)
solvent
something where other substances are diffused in
concentration gradient
the region along which the density of a chemical substance increases or decreases. Diffusion is a spontaneous process, needing no input of energy. Each substance diffuses down its own concentration gradient, unaffected by the concentration gradients of other substances.
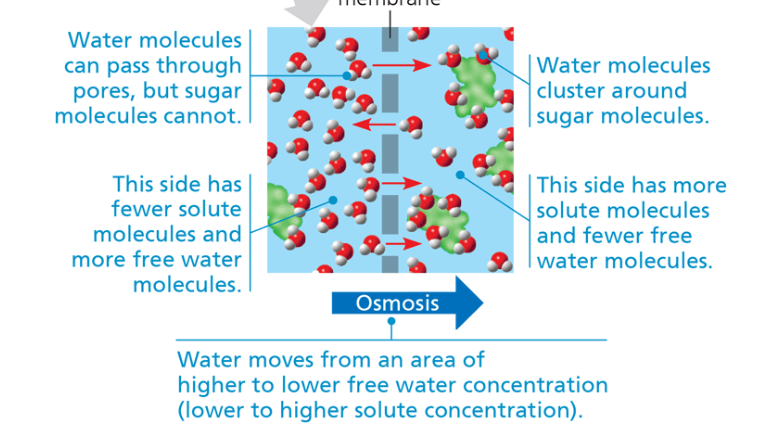
osmosis
diffusion of a solvent (water) across a selectively permeable membrane. The water moves from an area of higher to lower free water concentration. (lower to higher solute concentration)
passive transport
the diffusion of a substance across a biological membrane with no expenditure of energy
tonicity
the ability of a surrounding solution that causes a cell to gain or lose water. It depends in part on its concentration of solutes that cannot cross the membrane. If there’s a higher concentration of nonpenetrating solutes in the surrounding solution, water will tend to leave the cell and vice versa.
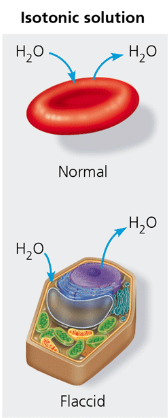
isotonic
referring to a solution that when surrounding a cell, causes no net movement of water into or out of the cell. (same concentration)
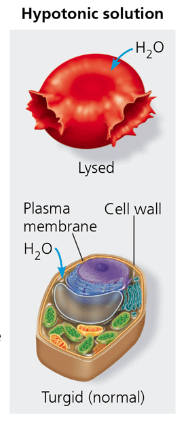
hypotonic
refers to a solution that when surrounding a cell, will cause the cell to take up water. (less solute concentration)
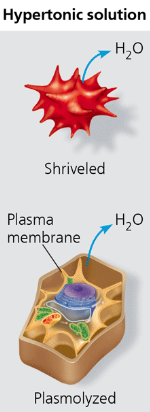
hypertonic
referring to a solution that, when surrounding a cell, will cause the cell to lose water. (more solute concentration)
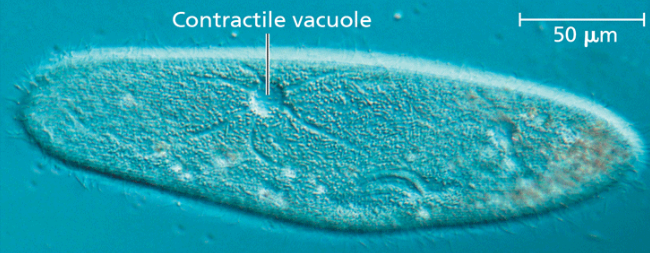
osmoregulation
the control of solute concentrations and water balance. example: protist paramecium caudatum. This protist lives in pond water, which is hypotonic to the cell. It has a plasma membrane that is less permeable to water than other cell membranes.
turgid
facilitated diffusion
the passage of molecules or ions down their electrochemical gradient across a biological membrane with the assistnace of specific transmembrane transport proteins, requiring no energy expenditure. (provides hydrophilici environment thoruhg the hydrophobic interior) (pores/channels
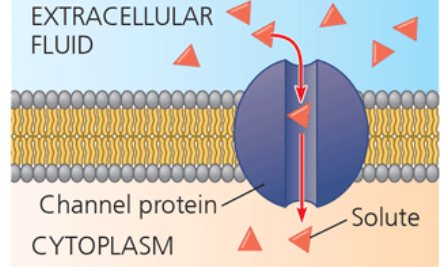
channel protein
has a channel through which water molecules or a specific solute can pass
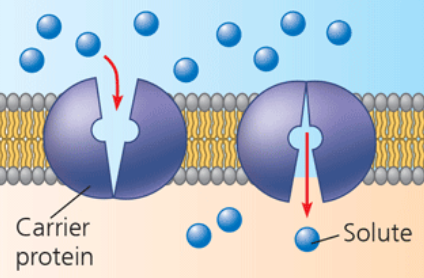
carrier protein
alternates between two shapes, moving a solute across the membrane during the shape change
active transport
moves solutes against their concentration gradients. requires inputs of energy in the form of ATP. active transporters are often referred to as “pumps”
sodium potassium pump
a transport protein in the plasma membrane of animal cells that actively transports sodium out of the cell and potassium into the cell
bulk transport
moves lots of things simultaneously
exocytosis
cellular secretion of biological molecules by fusion of vesicles ot plasma membrane—— AUUUUGHRAUUAAR
endocytosis
cellular uptake of substances (molecules, organisms, etc.) by formation of an endosome by plasma membrane.
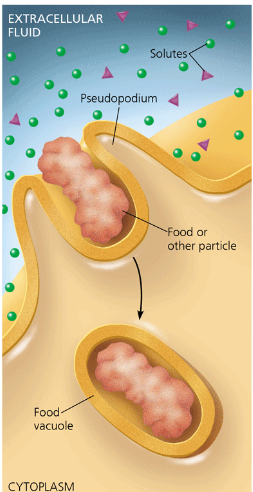
phagocytosis
a cell engulfs a particle by extending pseudopodia around it an packaging it within a membranous sac called a food vacuole. The particle will be digested after the food vacuole fuses with a lysosome containing hydrolytic enzymes.
also known as “cellular eating” S
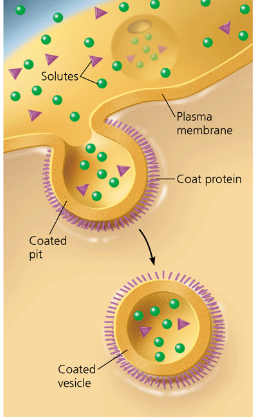
pinocytosis
a cell continually “gulps” droplets of extracellular fluid into tiny vesicles, formed by infoldings of the plasma membrane. The cell obtains molecules dissolved in the droplets.
also referred to as “cellular drinking.” It is constant and non-specific.
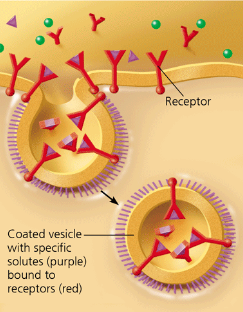
receptor mediated endocytosis
a type of pinocytosis that enables the cell to acquire bulk quantities of specific substances, even though those substances may not be very concentrated in the extracellular fluid. The plasma membrane has proteins with receptor sites that are exposed to the extracellular fluid. Specific solutes bind to the receptors. The receptor proteins cluster in coated pits, and each pit forms a vesicle containing the bound molecules. After the ingested material is liberated from the vesicle, the emptied receptors are recycled to the plasma membrane by the same vesicle.
metabolism
the totality of an organism’s chemical reactions, consisting of catabolic and anabolic pathways, which manage the material and energy resources of the organism. also known as: energy and matter transfer and transformation; the sum of all the chemical reactions in a system
metabolic pathway
a specific molecule is altered in a series of defined steps, resulting in a certain product. Each step is catalyzed by a specific enzyme, a macromolecule that speeds up a certain reaction.
catabolic pathways
a metabolic pathway that releases energy by breaking down complex molecules to simpler molecules. Usually results in a release of energy
exergonic
some energy is captured in “activated carriers of energy”
Anabolic pathways
a metabolic pathway that consumes energy to build complicated molecules from simpler ones.
(dn
sometimes called “biosynthetic pathways”
Biological organisms are open systems, meaning
they can transfer energy and matter with surroundings
thermodynamics
the study of energy transformation