CHEM LC 2
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
(LO 24) Which of the following is/are true?
Se has a smaller radius than Se2–
Se has a larger radius than Rb
Rb has a larger radius than Rb+
Rb+ has a smaller radius than Se2–
The effective nuclear charge of rubidium is about 1; the effective nuclear charge of selenium is about 4.
A, C, and D
|
B and D
(LO 27) Which of the following is/are true?
The first ionization energy of Cs is less than the first ionization energy of Ba.
The first ionization energy of Ba is greater than the first ionization energy of Ca.
The second ionization energy of Cs is greater than the second ionization energy of Ba.
The third ionization energy of Cs is greater than the third ionization energy of Ba.
A and C
LO 28) Which of the following is/are true?
Potassium has more metallic character than iron.
Electron affinities are generally negative (exothermic) while ionization energies are positive (endothermic).
Na has a more exothermic electron affinity than Cl.
Cl has a more exothermic electron affinity than Ar.
K has a more exothermic electron affinity than Na.
A, B, and D
LO 29) Which of the following is/are true?
Covalent bonding involves transferring electrons; ionic bonding involves sharing electrons.
Chemical bonds form to decrease the potential energy of the charged particles that compose atoms.
The empirical formula of C6H12O6 is C2H4O2.
More than one compound can have the same empirical formula.
The molecular formula cannot be the same as the empirical formula for a compound.
A covalent compound typically forms between two or more nonmetals; an ionic compound typically forms between a nonmetal and a metal.
B, D, and F
(LO 30) Which of the following is/are true?
The ionic compound formed from I and Sr is Sr2I.
The charge of Sn in SnO2 is 4+.
The ionic compound formed from Ba and Se is Ba2Se2.
The charge of Mn in Mn2O3 is 3+.
B and D
(LO 31) Which of the following is/are correct for both formula and name?
Ni2O3 is nickel(III) oxide
ZnBr2 is zinc bromide
Ag2S is silver(I) sulfide
CaF3 is calcium fluoride
Na3N is sodium nitrite
A and B
(LO 32) Which of the following is/are correct for both formula and name?
Mg(NO3)2 • 6H2O is magnesium nitrite hexahydrate
Fe(C2H3O2)2 • 4H2O is iron(II) acetate tetrahydrate
CaHPO4 is calcium hydrogen phosphate
BaBrO2 is barium hypobromite
Fe2(CrO4)3 is iron(III) dichromate.
Sr3N2 is strontium nitride
B, C, and F
What is the formal charge of the oxygen atom in this Lewis structure for N2O?
1
(LO 41) What is the formal charge of the oxygen atom in this Lewis structure of N2O?
-1
(LO 41) What is the formal charge of the oxygen atom for this Lewis structure of N2O?
0
(LO 41) Assign formal charges to the remaining atoms in each resonance structure of N2O. Which resonance structure (1, 2, or 3) is most favorable?
2
(LO 42) Draw the best resonance structure(s) for the bromate anion. Which of the following is/are true? Select all that apply.
There are three equally good resonance structures
In a best resonance structure, there are three single bonds
In a best resonance structure, there is a 0 formal charge on two oxygen atoms and a -1 formal charge on one oxygen atom
In a best resonance structure, there is a 0 formal charge on the bromine atom
In a best resonance structure, there are no lone pairs of electrons on bromine
a,c,d
LO 43) Draw the best Lewis structure for C2H4, C2H2, and C2H6; also consider the resonance structures for C6H6 (benzene, a 6-membered carbon ring shown below). Which of the following is/are true? Select all that apply.
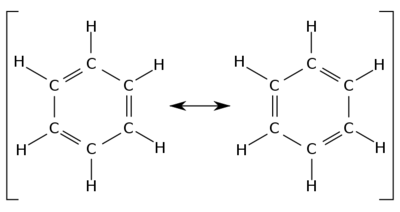
The bond between the two carbon atoms in C2H2 is shorter than the bond between the two carbon atoms in C2H4
The bond between the two carbon atoms in C2H6 is stronger than the bond between the two carbon atoms in C2H2
The bond between any two carbon atoms in C6H6 is longer than the bond between the two carbon atoms in C2H4
The bond between any two carbon atoms in C6H6 is weaker than the bond between the two carbon atoms in C2H6
Answer
a,c
(LO 44) Which of the following is/are true?
In VSEPR Theory, electron groups are generally positioned as far away from each other as geometry allows according to the number of electron groups on the atom
In VSEPR Theory, a lone pair of electrons counts as one electron group
In VSEPR Theory, a triple bond counts as three electron groups
In VSEPR Theory, a single bond takes up slightly more space around an atom than a lone pair of electrons.
If there are 3 bonding groups and 2 lone pairs, the molecular geometry will be T-shape
If there are 2 bonding groups and 2 lone pairs, the molecular geometry will be linear
Answer
A, B, and E
(LO 24) Which of the following is/are true? Select all that apply.
Ca has a smaller radius than Ca2+
S has a smaller radius than Ca
S2– has a smaller radius than Ca2+
The effective nuclear charge of Ca is about 2; the effective nuclear charge of S is about 6
Answer
B and D
(LO 27) Which of the following is/are true? Select all that apply.
The first ionization energy of Ba is greater than the first ionization energy of Ca.
The first ionization energy of K is less than the first ionization energy of Ca.
The second ionization energy of K is greater than the second ionization energy of Ca.
The third ionization energy of K is greater than the third ionization energy of Ca.
Answer
B and C
(LO 28) Which of the following is/are true? Select all that apply.
Calcium has more metallic character than nickel
K has a more exothermic electron affinity than Cs.
K has a more exothermic electron affinity than Br.
Kr has a more exothermic electron affinity than Br.
Answer
A and B
(LO 29, LO 30, and LO 33) Which of the following is/are true? Select all that apply.
Covalent bonding involves transferring electrons; ionic bonding involves sharing electrons.
Chemical bonds form to decrease the potential energy of the charged particles that compose atoms.
A covalent compound typically forms between two or more nonmetals; an ionic compound typically forms between a nonmetal and a metal.
The compound formed between Ca and Br has the formula Ca2Br
In Mn2O3, Mn has a 2+ charge
A double bond involves two adjacent atoms sharing 4 electrons
B, C, and F
(LO 31, LO 32, and LO 34) Which of the following is/are true? Select all that apply.
BaCl is barium chloride
ZnBr2 is zinc(II) bromide
Ag2Cr2O7 is silver dichromate
NiSO4 • 7H2O is nickel sulfate heptahydrate
Mn(C2H3O2)2 • 4H2O is manganese(II) acetate tetrahydrate
PF5 is monophosphorus pentafluoride
N2O4 is dinitrogen tetroxide
In2O3 is diindium trioxide
C, E, and G
(LO 35) Calculate the number of moles Ca(OH)2 in 946 dg Ca(OH)2.
1.28
(LO 35) How many molecules are contained in 25.0 ng N2O5?
Enter your answer in scientific notation using the following format: 1.00x1015 would be entered as 1.00E15
1.39E14
(LO 36) Calculate the percent by mass of oxygen in calcium carbonate hexahydrate. Enter a number without a % sign.
69.2
(LO 36) A certain compound is 14.5% F by mass. If the density of the compound is 4.11 g/mL, how many atoms of F are contained in 9.29 L of the compound?
Enter your answer in scientific notation using the following format: 1.00x1015 would be entered as 1.00E15
1.74E26
(LO 37) How many total atoms are contained in 56.0 g C12H24N4?
Enter your answer in scientific notation using the following format: 1.00x1015 would be entered as 1.00E15
Answer
6.01E24
LO 38) A sample from a compound contains 47.3707 g carbon, 5.96373 g hydrogen, and 23.6656 g oxygen. Its molar mass is about 625 g/mol. What is its molecular formula?
C24H36O9
C30H40O14
C34H56O10
C36H64O8
C28H32O16
C32H48O12
C21H24O12
Answer
f
(LO 38) When a 25.00 g sample of an organic compound containing only carbon, hydrogen, and oxygen undergoes combustion analysis, it produces 52.0530 g CO2 and 21.3085 g H2O. Find the empirical formula for the unknown compound.
C9H16O2
C4H8O
C5H8O
C10H18O3
C3H8O
C9H14O2
C9H18O4
Answer
G
(LO 39 and LO 46) Which of the following is/are true? Select all that apply.
In a nonpolar covalent bond, electrons are shared equally.
C—F bond should be more polar than a N—O bond.
C4H10 is polar
H2CO is polar
CH2F2 is polar
PF5 is polar
XeF4 is polar
SF4 is polar
A, B, D, E, and H
(LO 43) Which of the following is/are true? Select all that apply.
N2H2 has a shorter bond between the two nitrogen atoms than N2
N2H4 has a weaker bond between the two nitrogen atoms than N2H2
Nitrite has a longer bond between nitrogen and oxygen than CH3NO
Answer
B and C
|
A, D, and E
(LO 45) Complete the Lewis structure for NHCHCH2CHO by adding lone pairs of electrons. Which of the following is/are true?
The molecular geometry around the bolded atom is bent: NHCHCH2CHO
The molecular geometry around the bolded atom is trigonal pyramidal: NHCHCH2CHO
The molecular geometry around the bolded atom is tetrahedral: NHCHCH2CHO
The molecular geometry around the bolded atom is trigonal planar: NHCHCH2CHO
Answer
A, C, and D
(LO 40) Draw the Lewis structure for CH3OCH3. Which of the following is/are true?
There are no lone pairs in the Lewis structure.
There is one lone pair in the Lewis structure.
There are two lone pairs in the Lewis structure.
The oxygen atom is bonded to two carbon atoms.
The oxygen atom is bonded to one carbon atom.
The oxygen atom is bonded to one carbon atom and one hydrogen atom.
There are no double bonds.
There is one double bond.
There are two double bonds.
C, D, and G
(LO 40) Draw the Lewis structure for hypoiodite. Which of the following is/are true?
There are 2 lone pairs of electrons in the structure.
There is 4 lone pair of electrons in the structure.
There are 6 lone pairs of electrons in the structure.
There are 8 lone pairs of electrons in the structure.
There is a single bond in the structure.
There is a double bond in the structure.
There is a triple bond in the structure.
c and e
(LO 36) Calculate the percent hydrogen by mass in potassium hydrogen phosphate decahydrate. Enter a number without a % sign.
Answer
5.97