energy generation in mitochondria
1/153
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
154 Terms
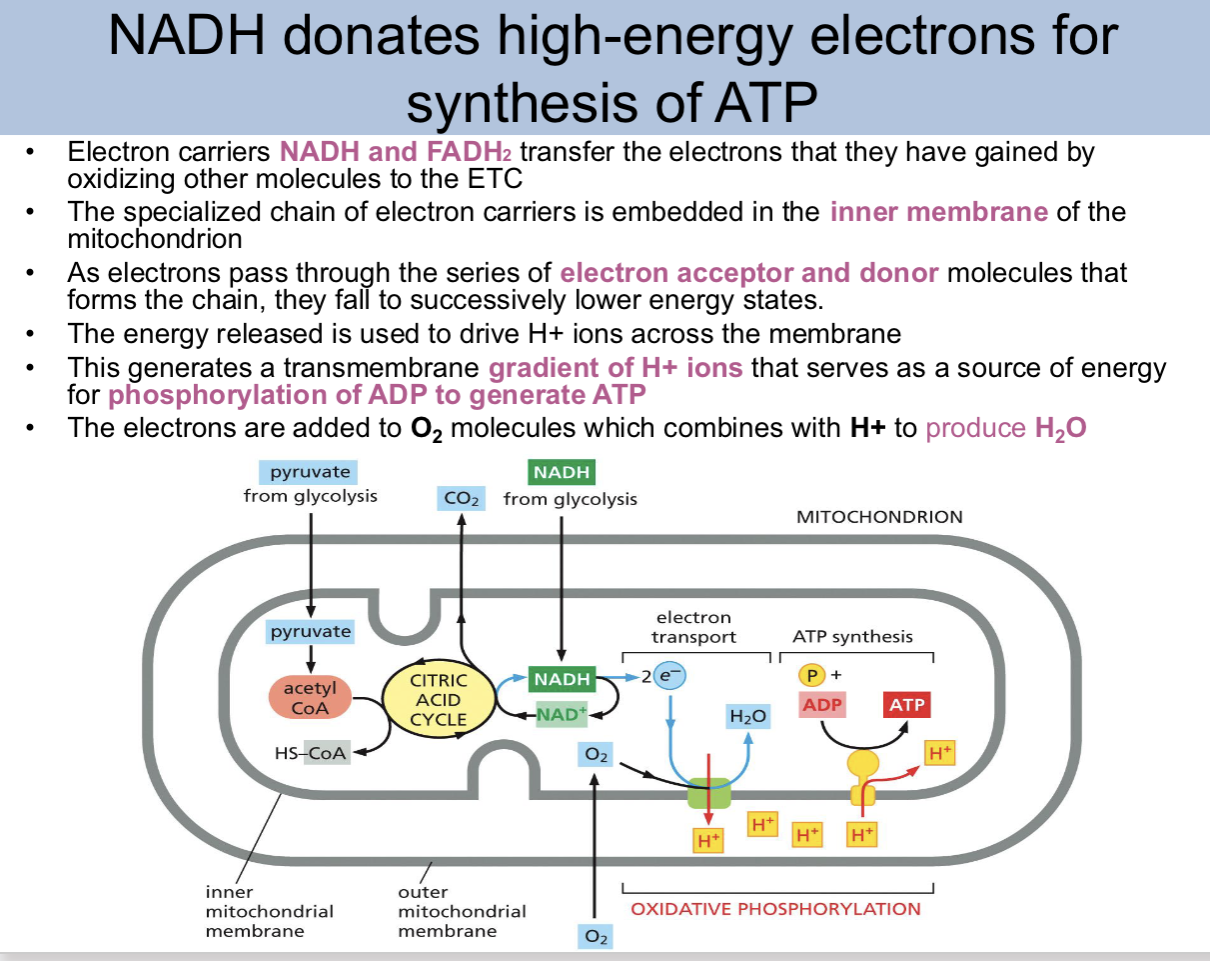
NADH donates _ for synthesis of ATP
high-energy electrons
electron carriers __ transfer electrons that they have gained by oxidizing other molecules to the ETC
NADH FADH2
the specialized chain of electron carriers is embedded where
the inner membrane of the mitochondrion
as electrons pass through the series of electron acceptor and donor molecules that forms the chain they _
fall to successively lower energy states
as electrons pass through the series of electron acceptor and donor molecules that forms the chain they fall to successively lower energy states
the energy released is used to drive
H+ ions across the membrane
as electrons pass through the series of electron acceptor and donor molecules that forms the chain they fall to successively lower energy states
the energy released is used to drive
This generates a
transmembrane gradient of H+ ions
as electrons pass through the series of electron acceptor and donor molecules that forms the chain they fall to successively lower energy states
the energy released is used to drive
This generates a transmembrane gradient of H+ ions that serves as a
source of energy for phosphorylation of ADP to generate ATP
as electrons pass through the series of electron acceptor and donor molecules that forms the chain they fall to successively lower energy states
the energy released is used to drive
This generates a transmembrane gradient of H+ ions that serves as a source of energy for phosphorylation of ADP to generate ATP
the electrons are added to — molecules which combines with — to produce _
O2, H+, H2O
energy generation in mitochondria slide
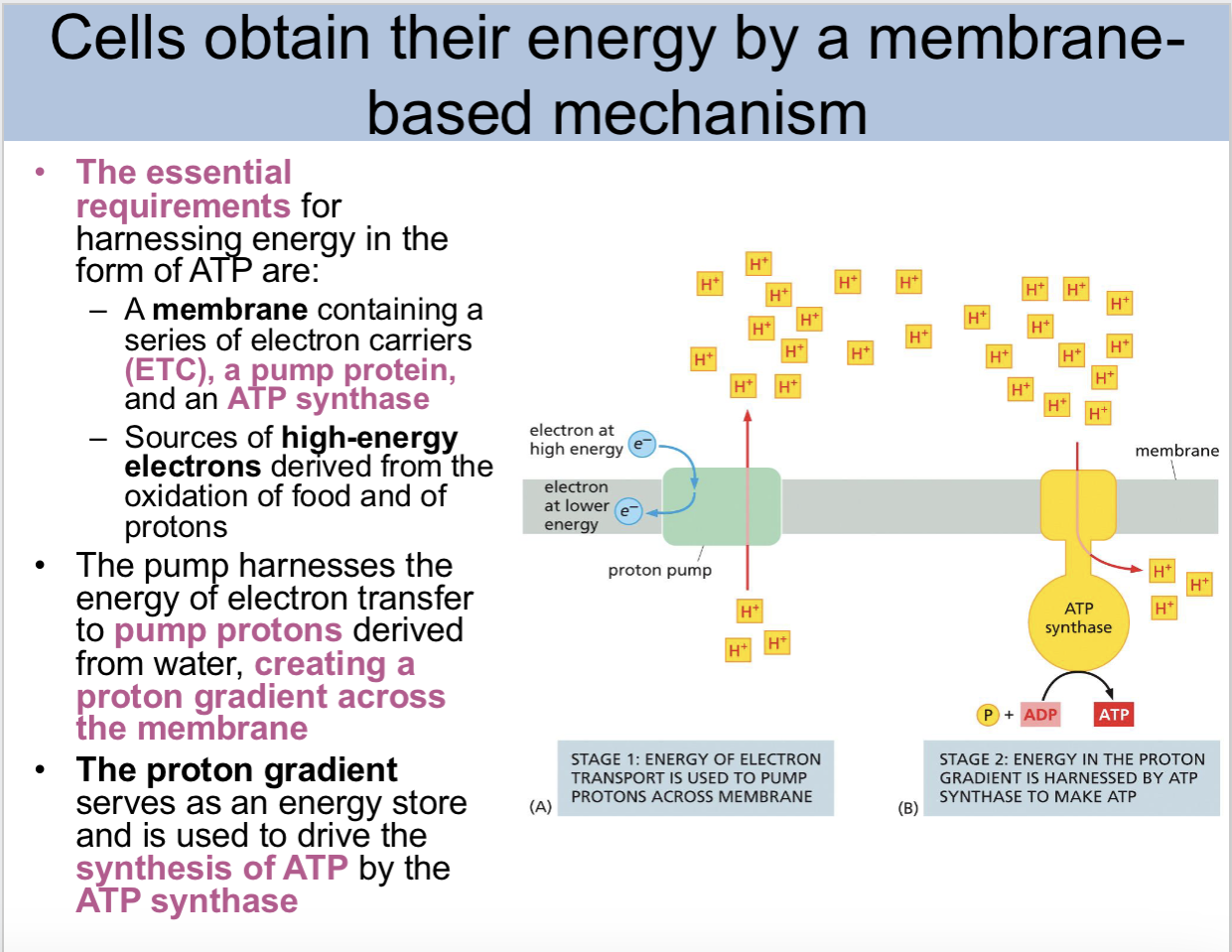
cells obtain their energy by a _base mechanism
membrane
The essential requirements for harnessing energy in the form of ATP are
A membrane containing a series of electron carriers (ETC), a pump protein, and an ATP synthase
Sources of high-energy electrons derived from the oxidation of food and of protons
the pump harnesses the energy of electron transfer to
pump protons derived from water creating a proton gradient across the membrane
the proton gradient serves as a
energy store and is used to drive the synthesis of ATP by the ATP synthase
stage one of harnessing energy from ATP
energy of electron transport is used to pump protons across the membrane
stage two of harnessing energy from ATP
energy in the proton gradient is harnessed by ATP synthase to make ATP
cells obtaining their energy by a membrane-based mechanism slide
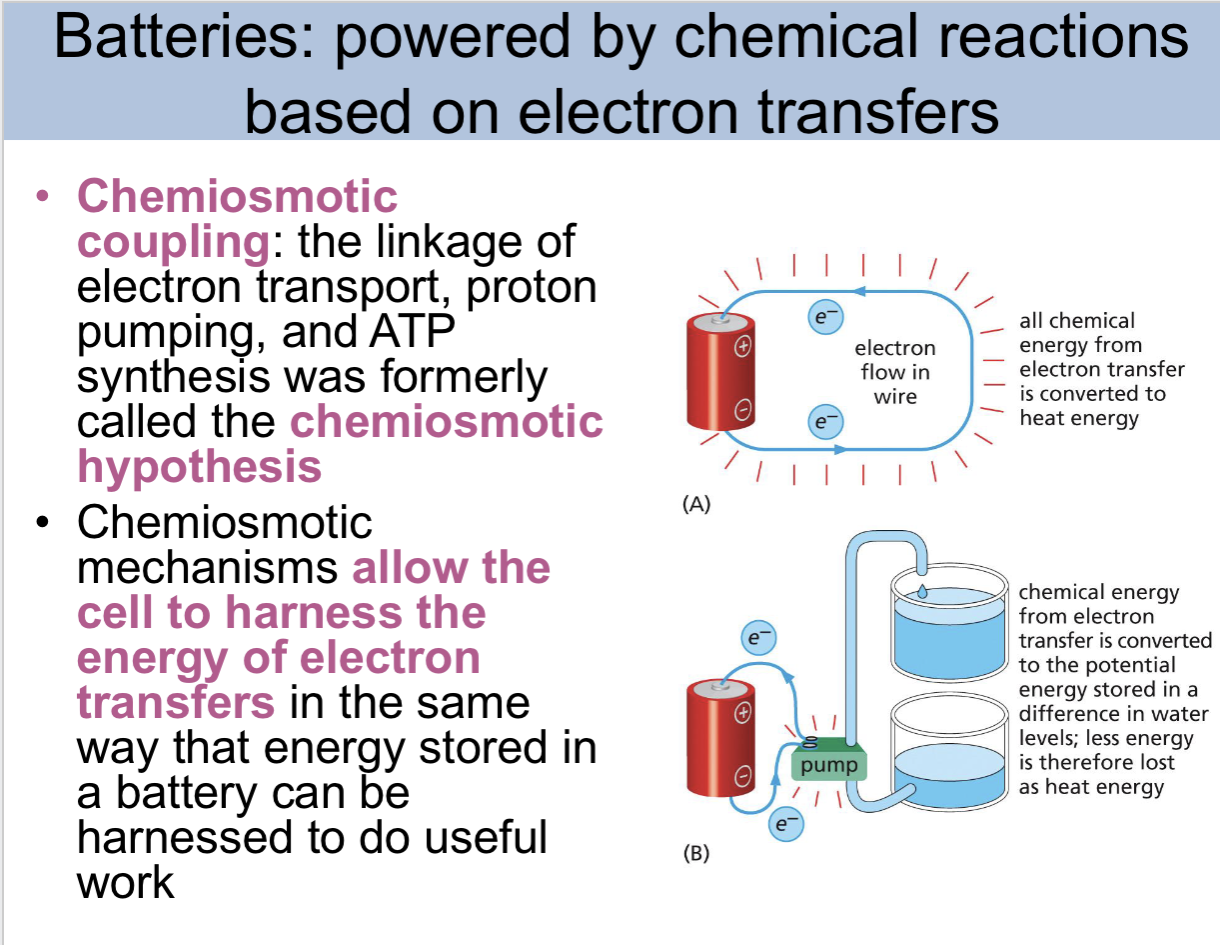
what are batteries powered by
chemical reactions based on electron transfers
chemiosmotic coupling
the linkage of electron transport, proton pumping, and ATP sythesi was formerly called the chemiosmotic hypotehsis
chemiosmotic mechanism allow the cell to
harness the energy of electron transfers in the same way that energy stored in a battery can be harnessed to do useful work
battery power slide
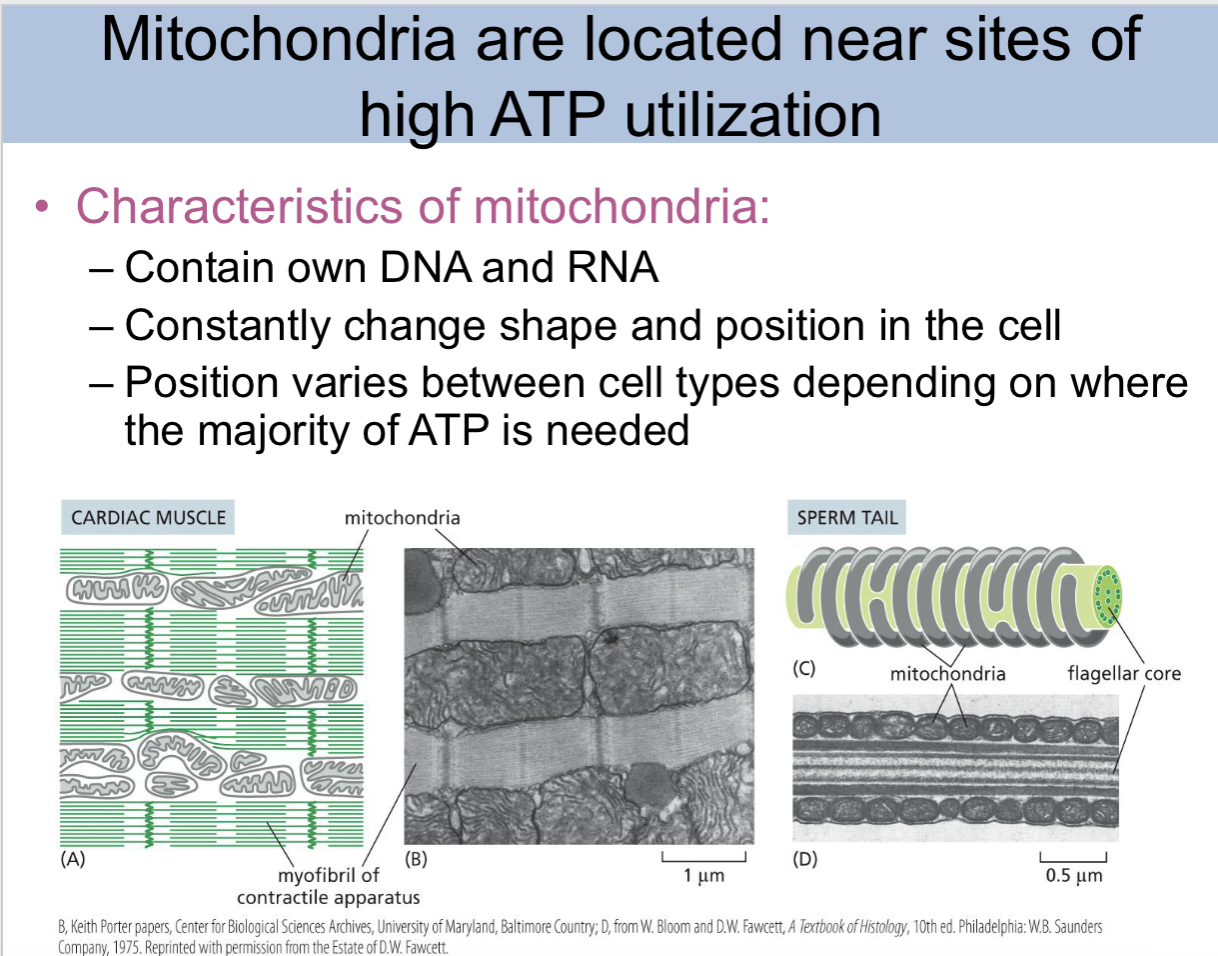
mitochondria are located near
sites of high ATP utilization
characteristics of mitochondria slide
contain own DNA and RNA, constantly change shape and position in the cell, position varies between cel types depending on where the majority of ATP is needed
characteristics of ATP slide
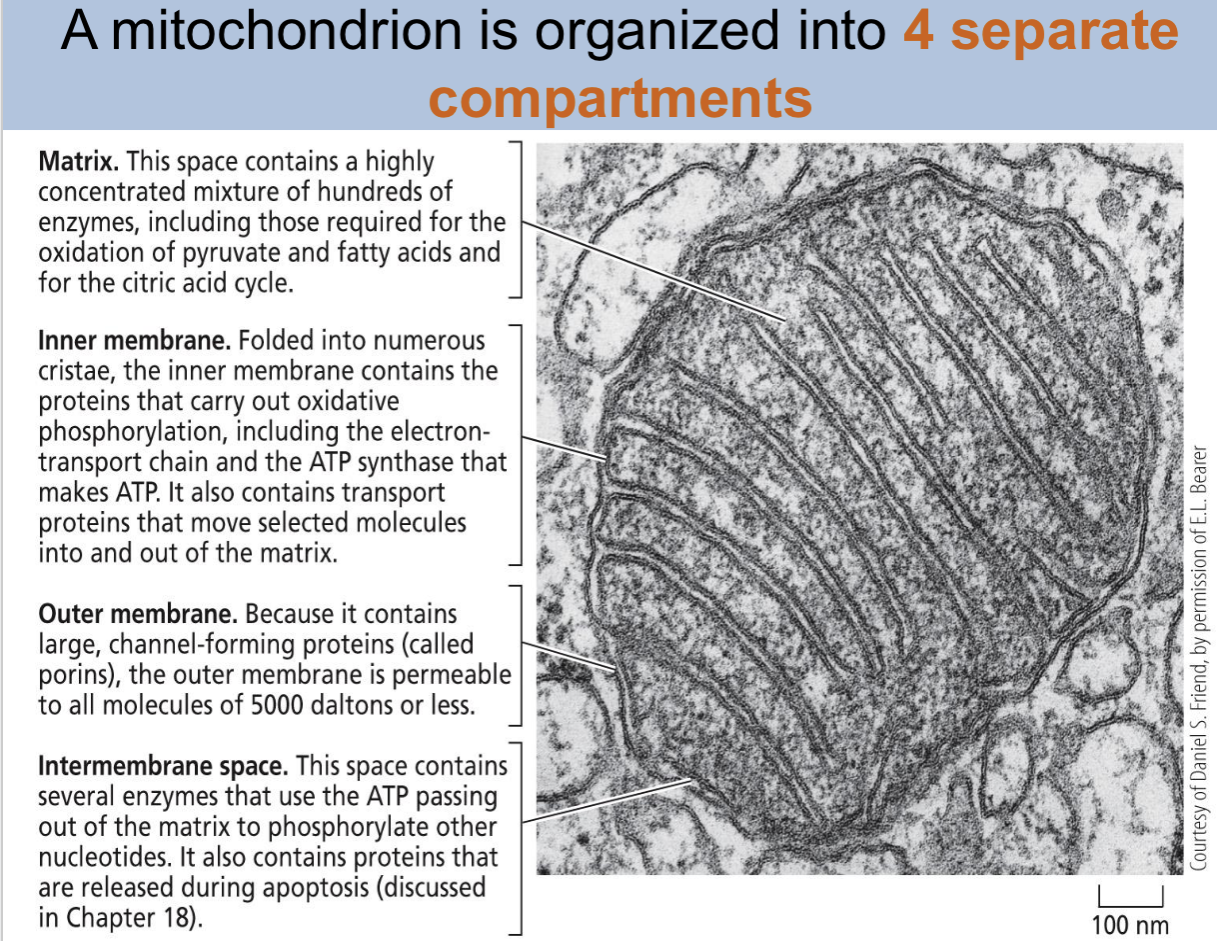
how many compartments is a mitochondria organized into
4
what are the 4 compartments of a mitochondria
matrix, inner membrane, outer membrane, intermembrane space
matrix of mitochondria
this space contains a highly concentrated mixture of hundreds of enzymes, including those required for the oxidation of pyruvate and fatty acids and for teh citric acid cycle
inner membrane mitochondria
folded into numerous cristae, the inner membrane contains proteins that carry oxidative phosphorylation, including the electron transport chain and ATP synthase that makes ATP. It also contains transport proteins that move selected molecules into and out of the matrix
out membrane mitochondria
because it contains large, channel-forming proteins(called porins), the outer membrane is permeable to all molecules of 5000 daltons or less
intermembrane space mitochondria
this space contains several enzymes that use the ATP passing out of the matrix to phosphorylate other nucleotides. It also contains proteins that are released during apoptosis.
how is mitochondria organized
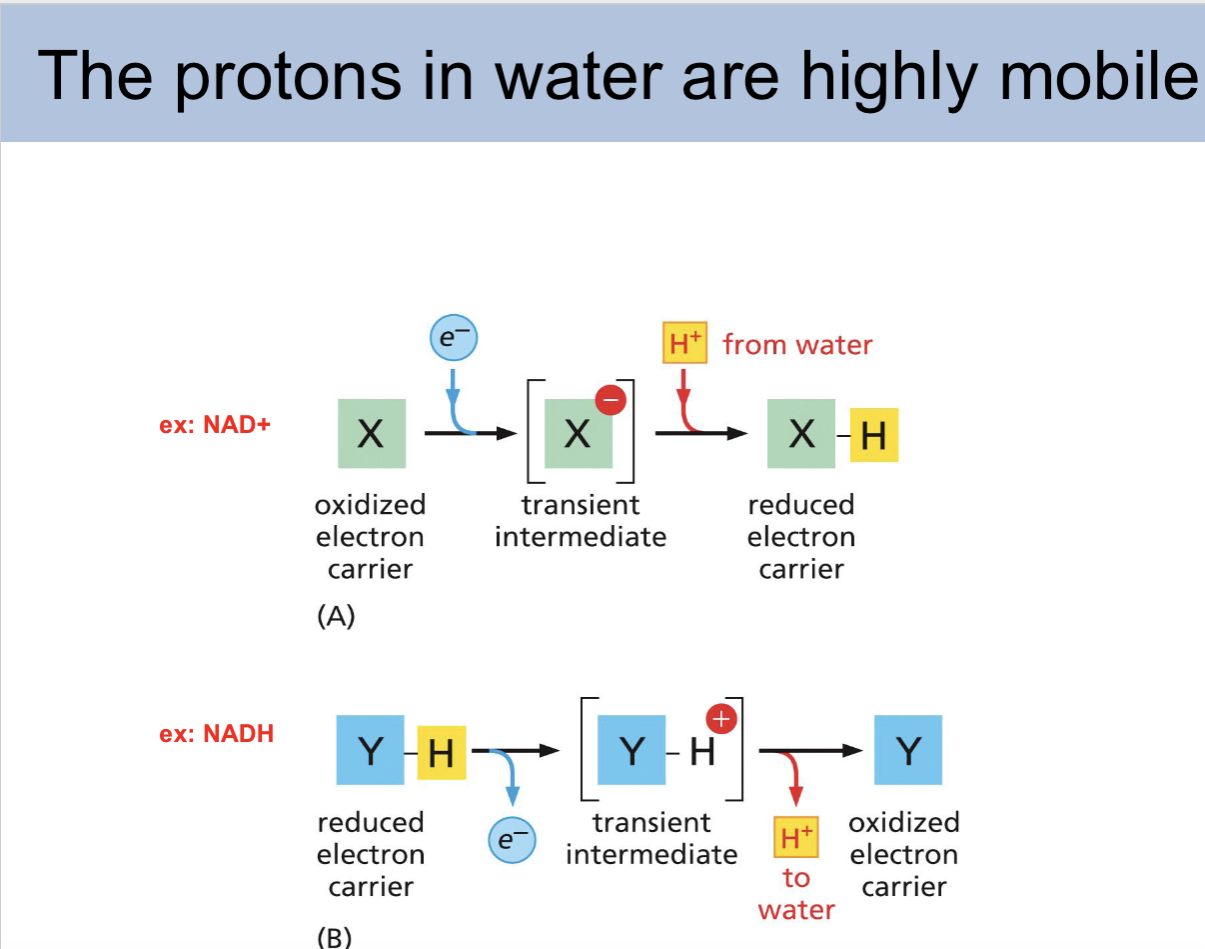
the protons in water are highly _
mobule
protons in water slide
NADH donates its electrons to the
ETC
NADH donating its electrons slide
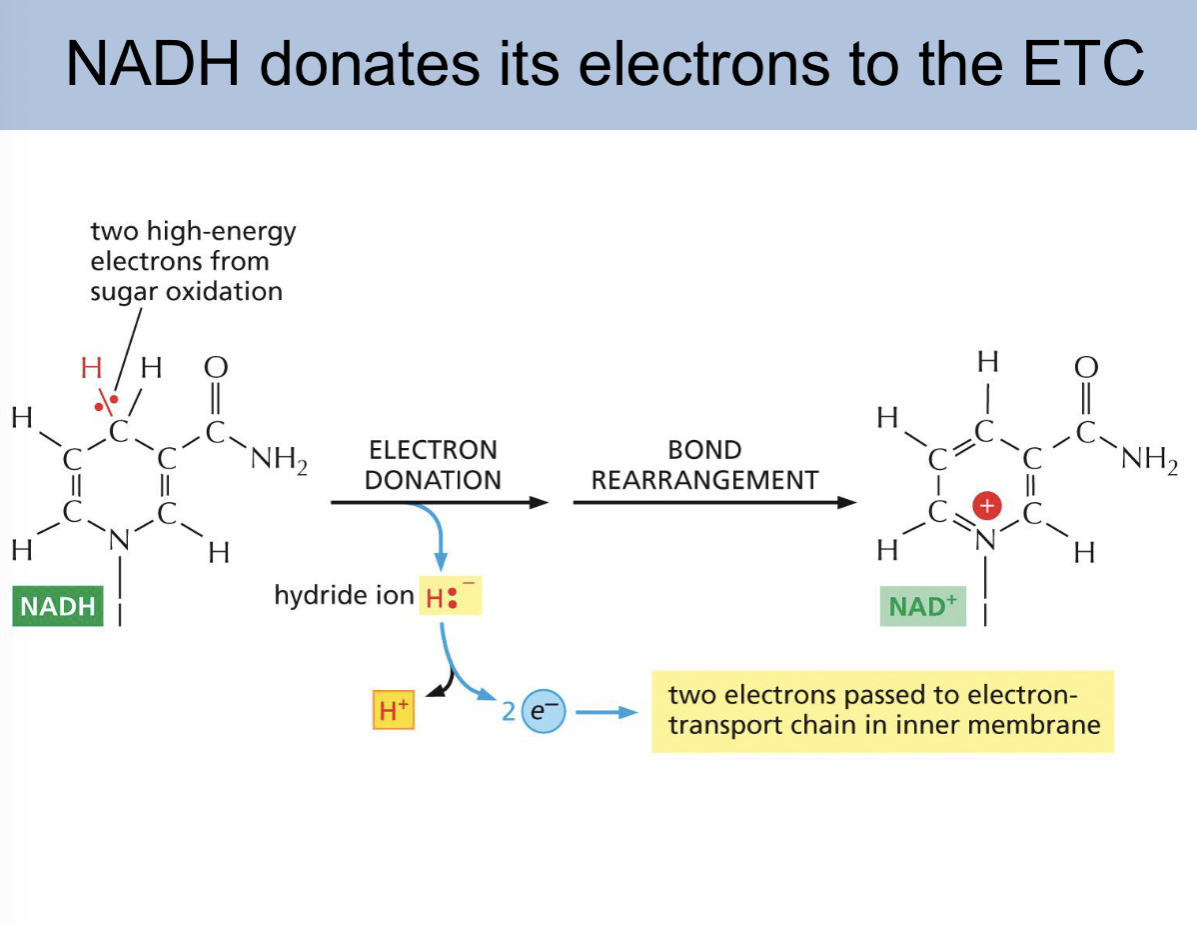
how many electrons does NADH donate to the ETC
2
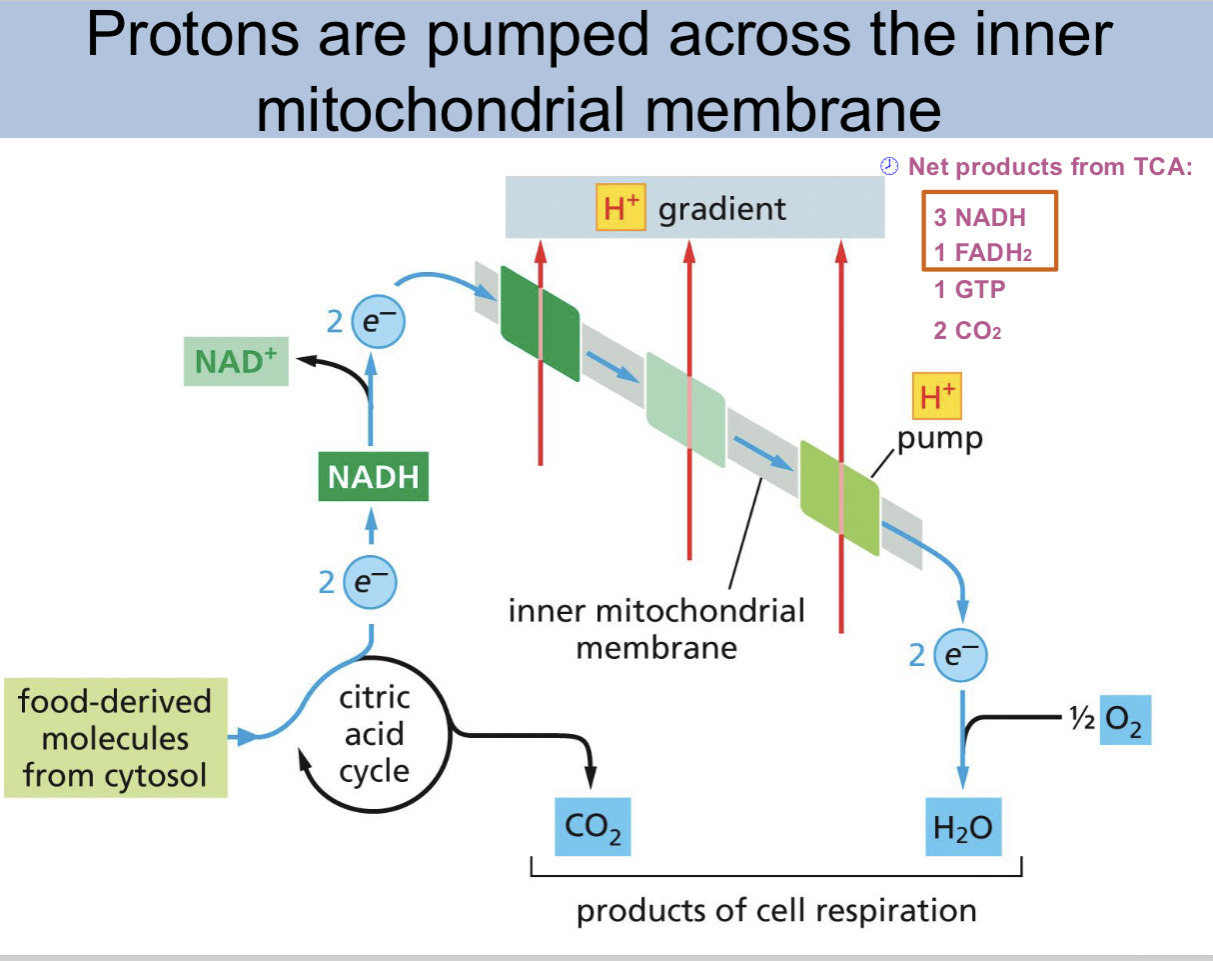
protons are pumped across the
inner mitochondrial membrane
protons are pumped across the inner mitochondrial membrane slide
electrons are transferred through __ in the ETC
3 respiratory enzyme complexes in the inner mitochondrial; membrane
what enzyme complexes make up the ETC
NADH dehydrogenase complex, cytochrome c reductase complex, cytochrome c oxidase complex
NADH dehydrogenase complex accepts _
electrons from NADH in the form of a hydride ion
enzymes in the NADH dehydrogenase complex catalyze what reaction
H- → 2e- + H+
teh three respirator enzymes of the ETC slide
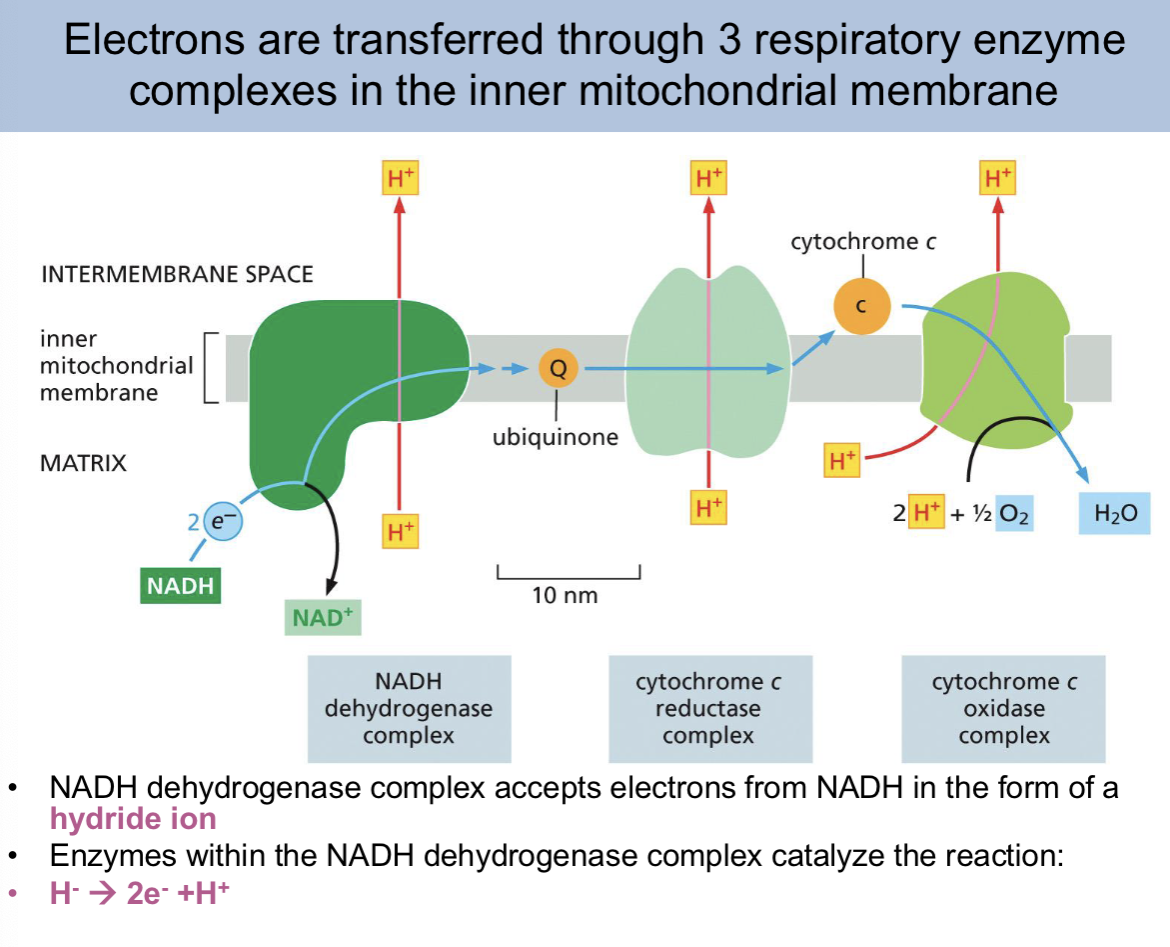
the __ of the electron carrier allows for the transfer to drive proton pump
orientation
as an electron passes along an electron transport chain, it can
bind and release a proton at each step
orientation of electron carrier slide
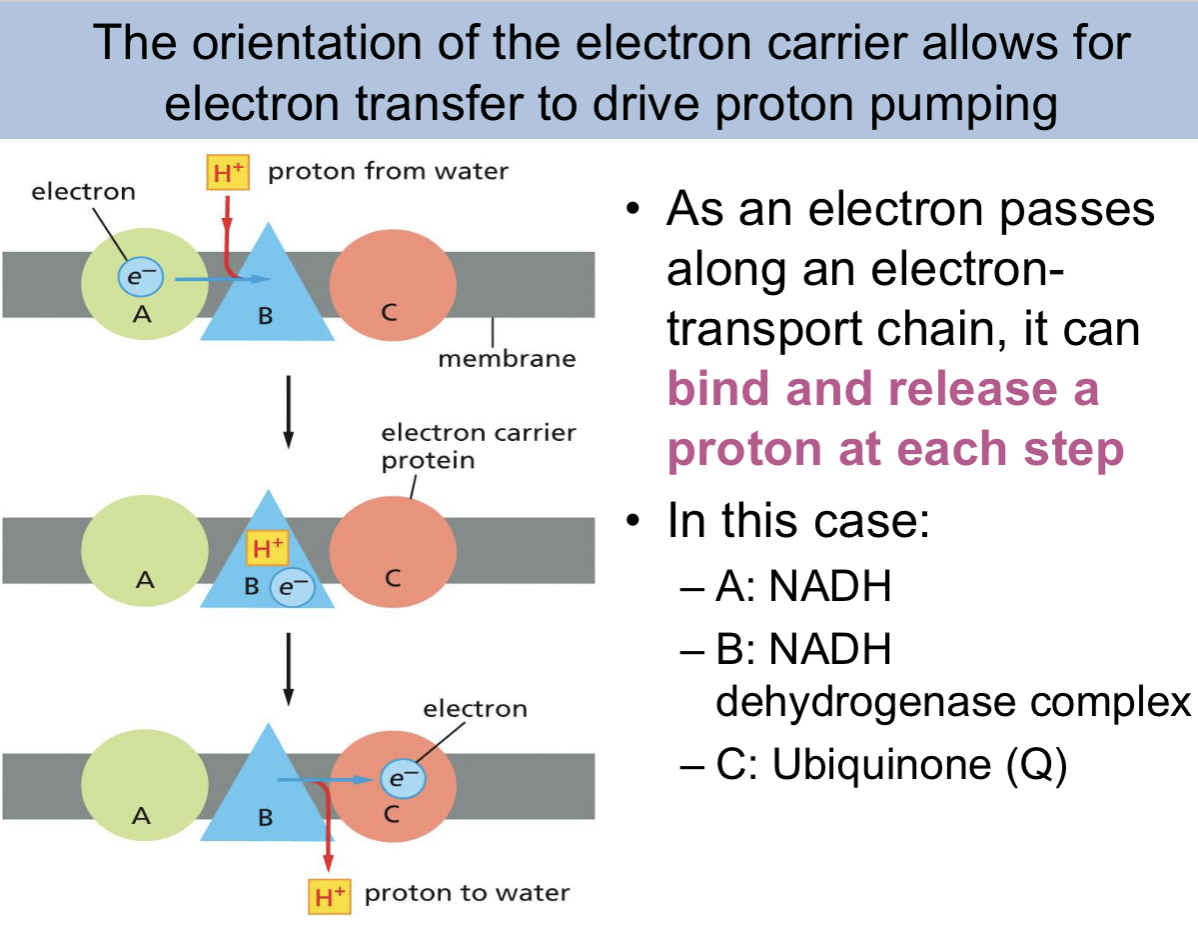
what do quinone do
carry electrons within the lipid bilayer
With each of the 3 respiratory enzyme complexes, electrons move mainly between
metal atoms that are tightly bound to the proteins
Electrons are carried between the different respiratory complexes by
molecules that diffuse along the lipid bilayer, picking up electrons from one complex and delivering them to another
Quinones are the only electron carriers in ETCs that can function without being
tightly bound to a protein
– Its long hydrophobic tail confines ubiquinone to the membrane
• Ubiquinone picks up electrons from NADH dehydrogenase complex or FADH2 and delivers
them to the cytochrome b-c1 complex
Quinones are the only electron carriers in ETCs that can function without being tightly
bound to a protein
– Its long hydrophobic tail ____
confines ubiquinone to the membrane
Ubiquinone picks up electrons from ___ or FADH2 and delivers
them to ——-
NADH dehydrogenase complex, the cytochrome b-c1 complex
quinone slide
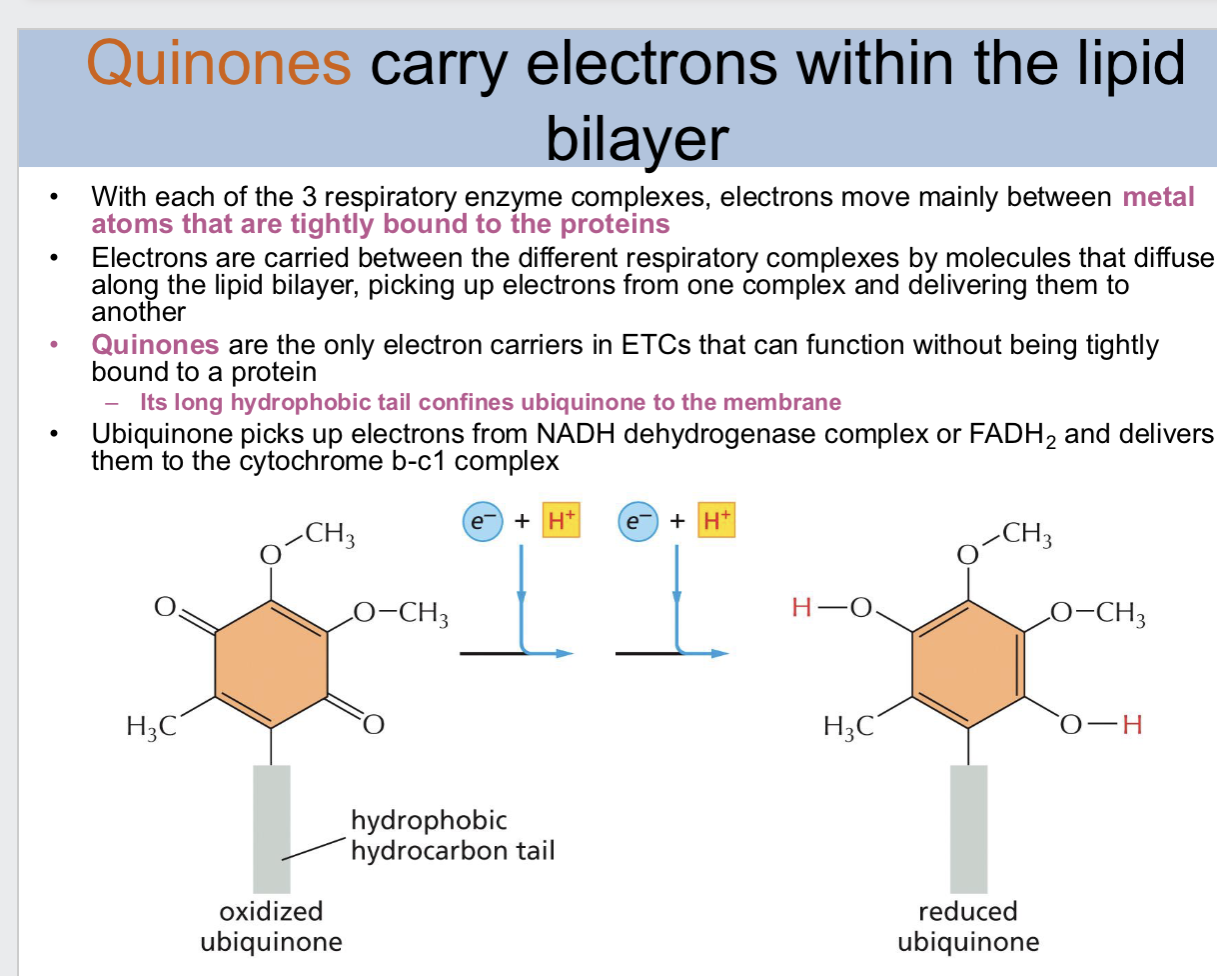
what is cytochrome c
an electron carrier in the ETC
cytochrome c is a _ protein
small
cytochrome c contains just over —— aa and is held ——
100, loosely on the outer face of the inner membrane by ionic interaction
what is bound to cytochrome c
heme
the _ atom in the bound heme can carry _
iron atom, a single electron
cytochrome c slide
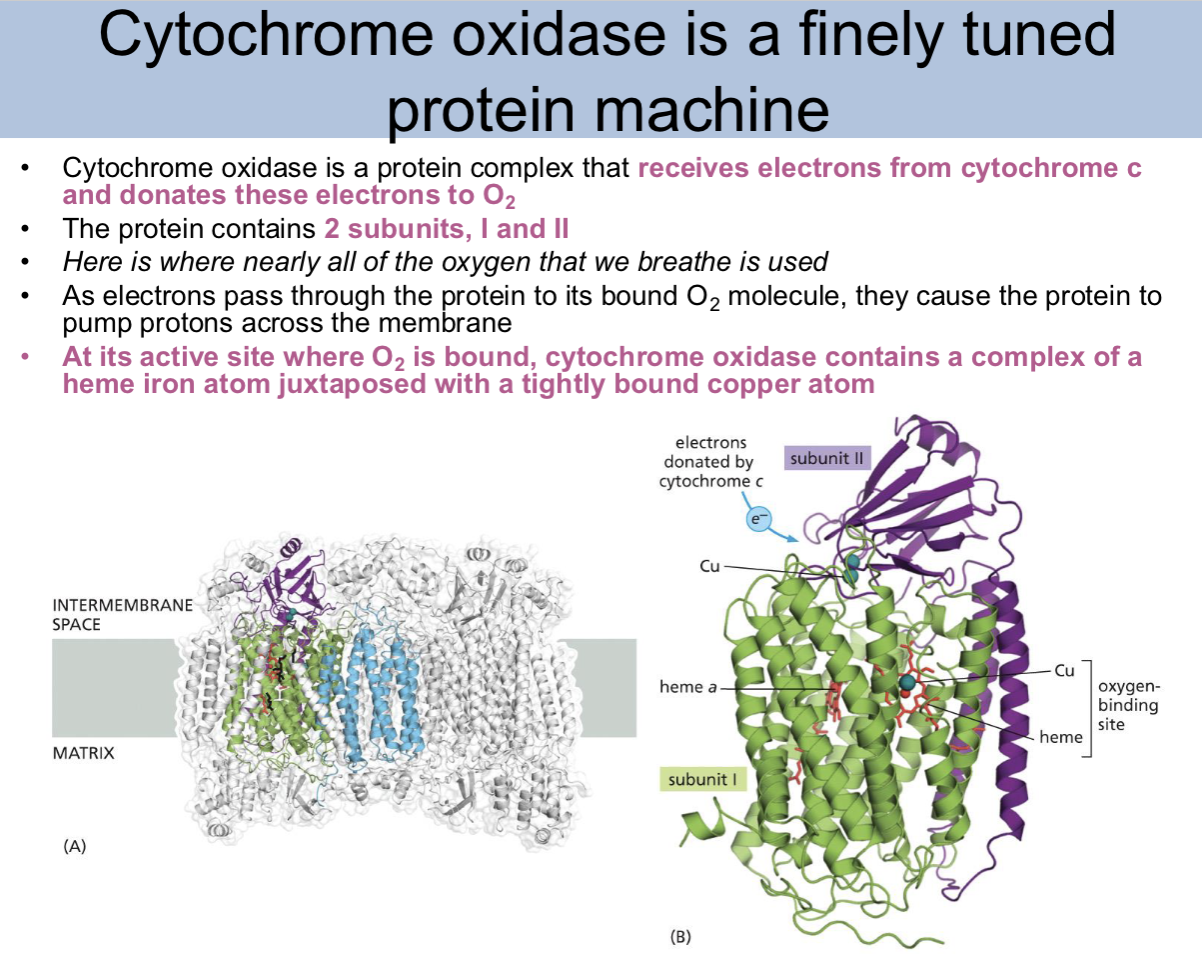
cytochrome oxidase is a
finely tuned protein machine (protein complex)
Cytochrome oxidase is
a protein complex that receives electrons from cytochrome c and donates these electrons to O2
how many subunits does cytochrome oxidase contain
2, I (one) and II (2)
where is nearly all the oxygen that we breathe used
cytochrome oxidase
As electrons pass through cytochrome oxidase to its bound O2 molecule, they cause
cytochrome oxidase to pump protons across the membrane
At its active site where O2 is bound, cytochrome oxidase contains
a complex of a heme iron atom juxtaposed with a tightly bound copper atom
cytochrome oxidase slide
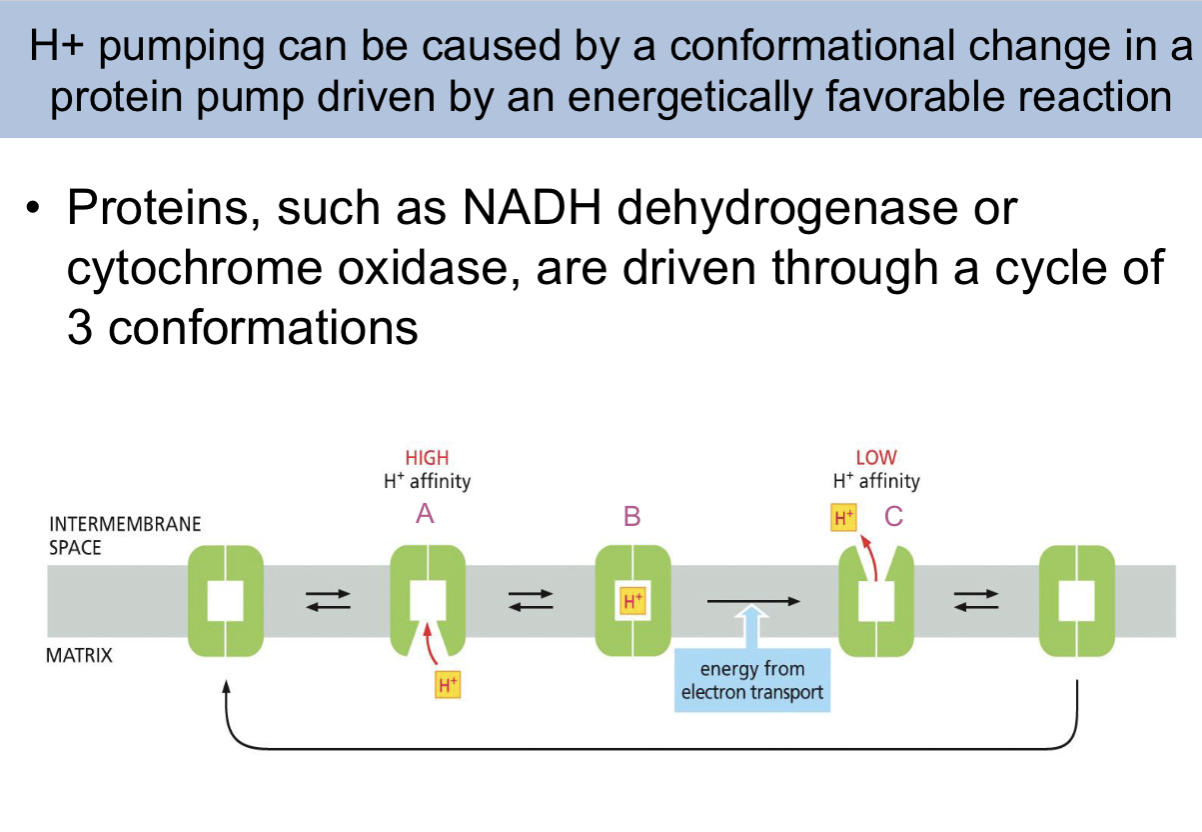
H+ pumping can be caused by a ___ in a protein pump driven by ——
conformational change, an energetically favorable reaction
Proteins, such as NADH dehydrogenase or cytochrome oxidase, are driven through a cycle of —
3 conformations
conformational change causing proton pumping slide
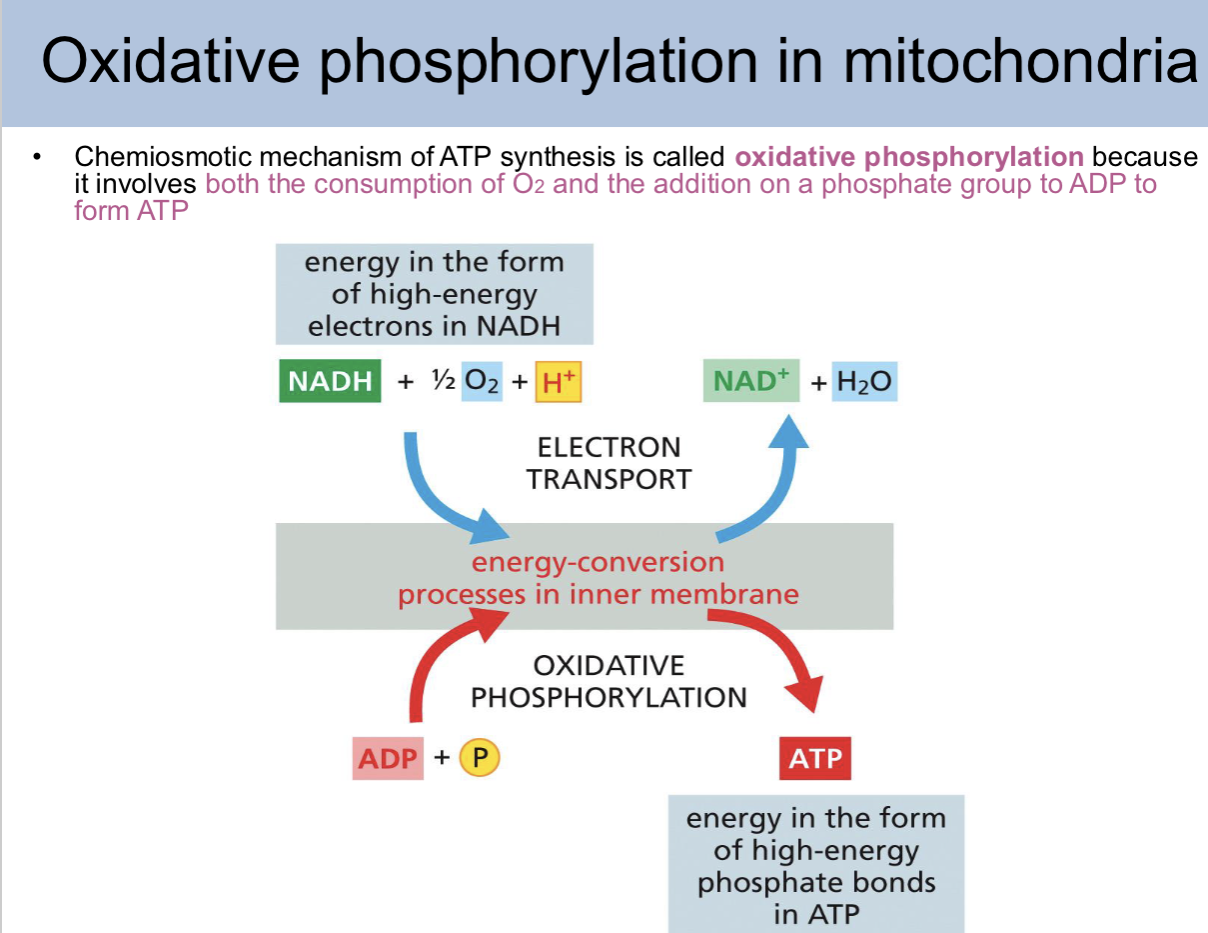
Chemiosmotic mechanism of ATP synthesis is called
oxidative phosphorylation
Chemiosmotic mechanism of ATP synthesis is called oxidative phosphorylation beacause
it involves both the consumption of O2 and the addition on a phosphate group to ADP to form ATP
oxidative phosphorylation in mitochondria
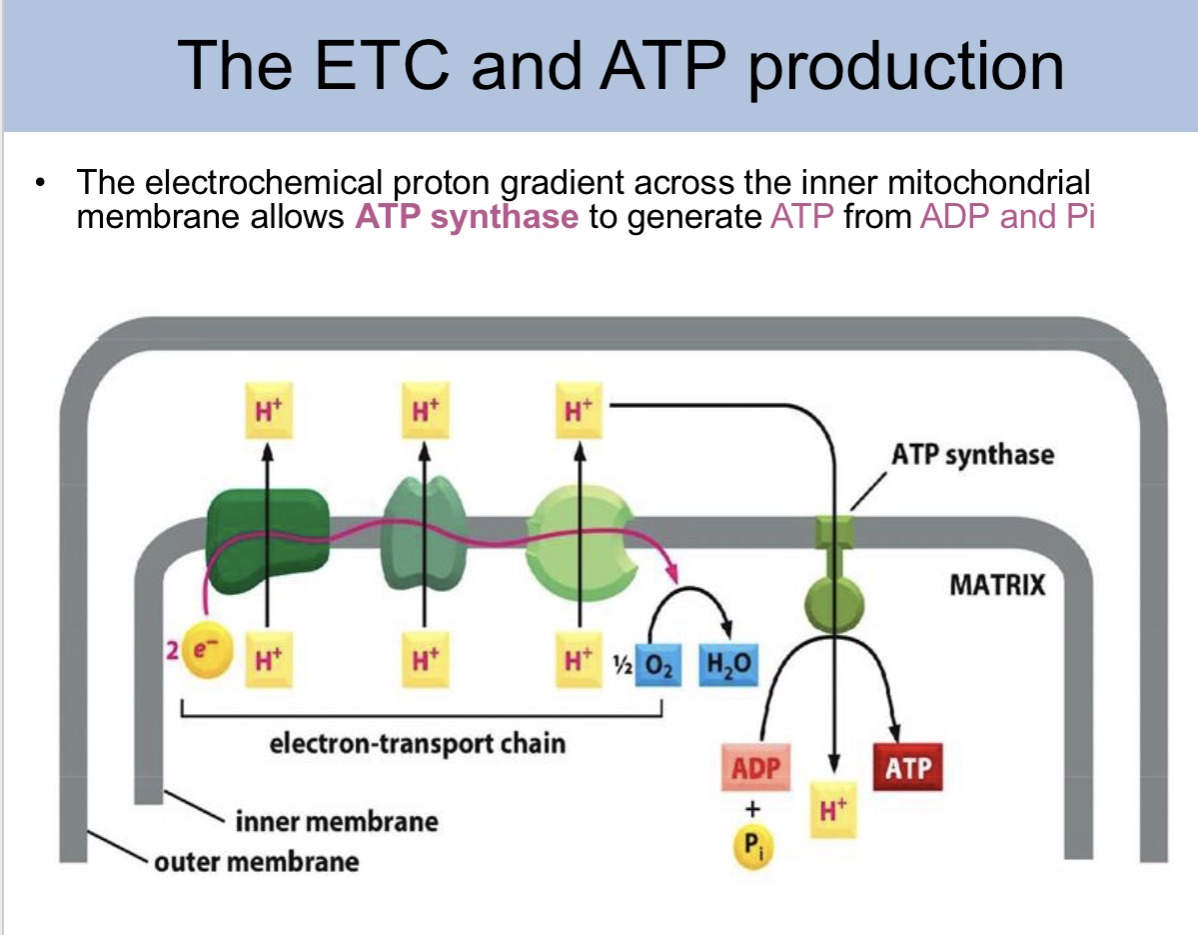
the electrochemical proton gradient across the inner mitochondrial membrane allows _ to generate ATP from ADP and Pi
ATP synthase
ETC and ATP production slide
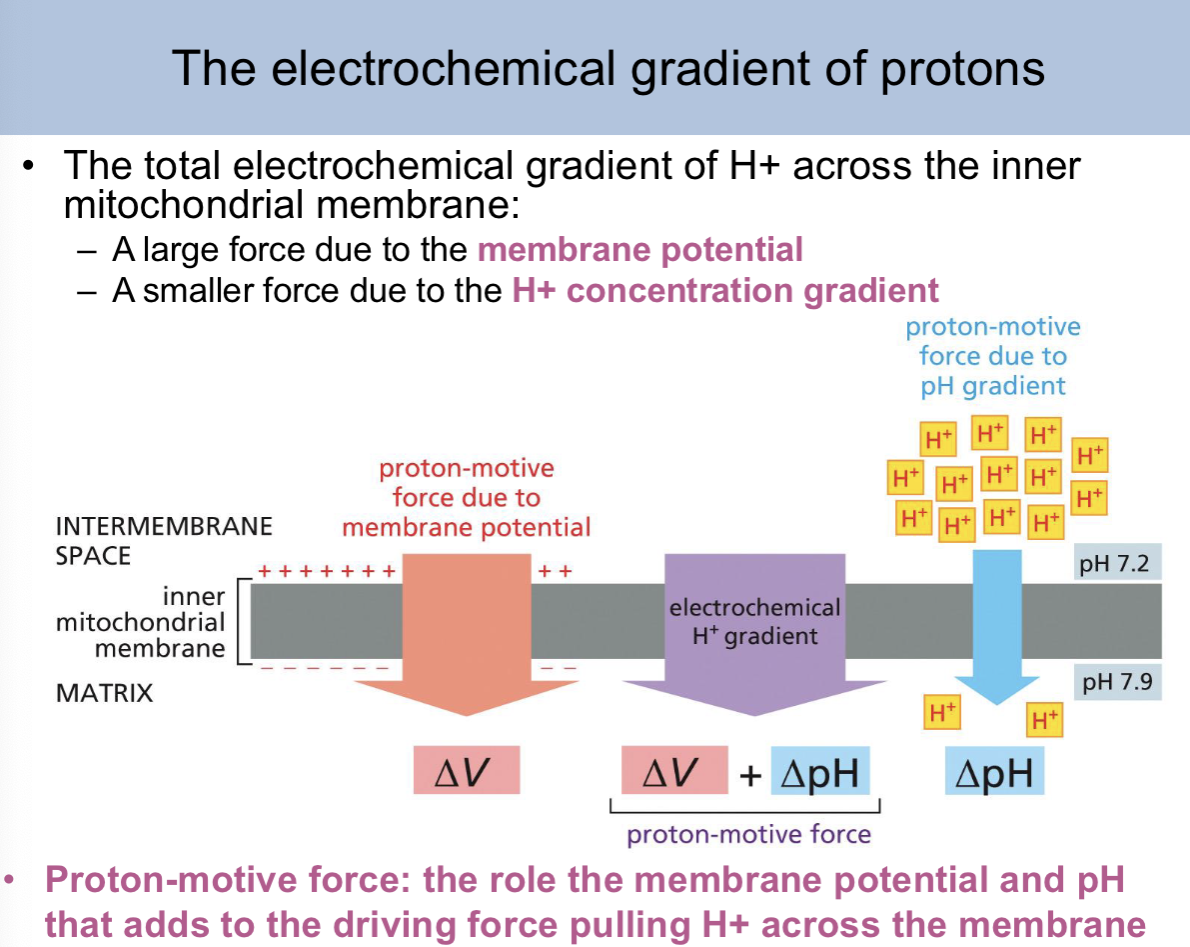
the total electrochemical gradient of H+ across the inner mitochondrial membrane is a combination of
a large force due to the membrane potential and a small force due to the H+ concentration graodent
proton motive force:
the role the membrane potential pH that adds to the driving force pulling H+ across the membrane
electrochemical gradient of protons slide
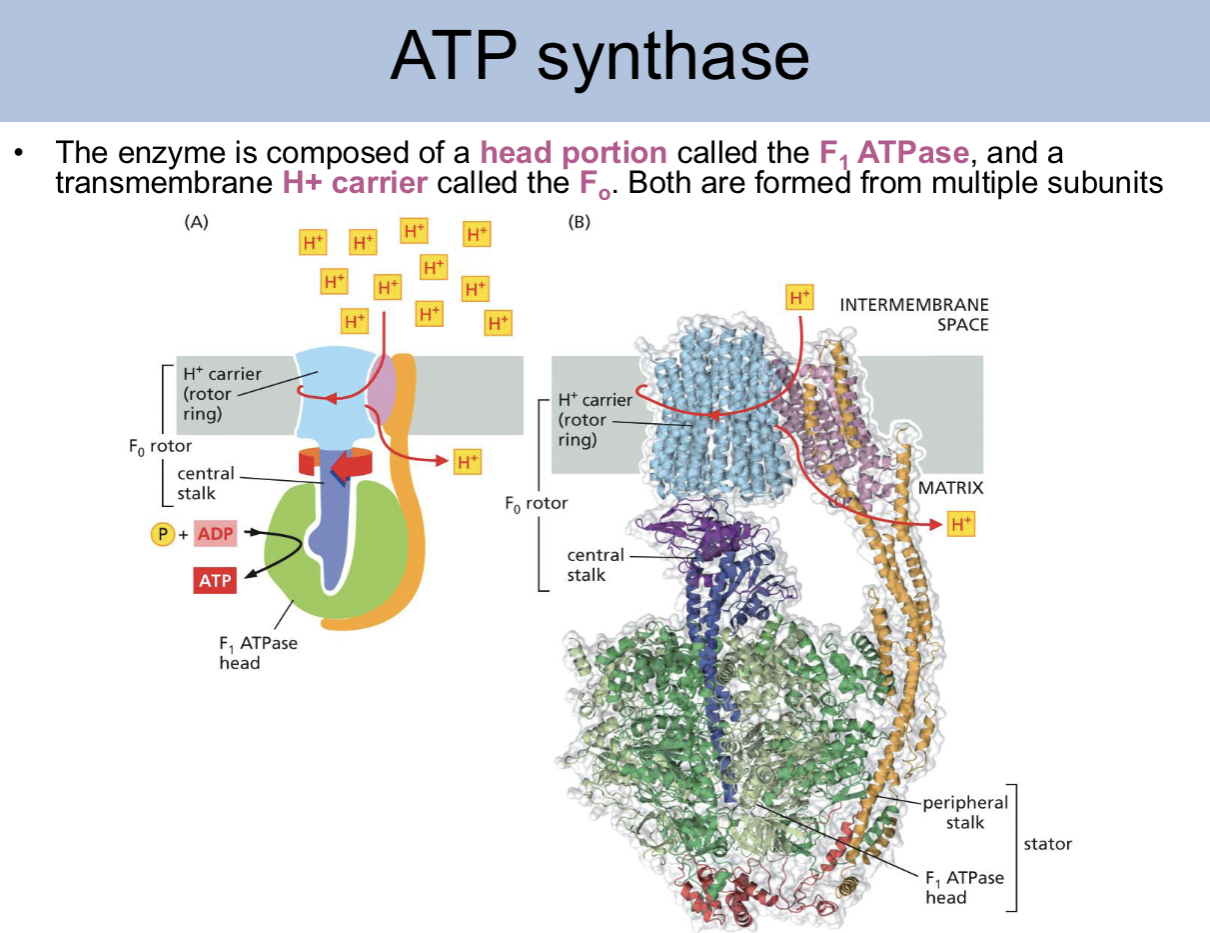
ATP synthase is composed of
a head portion called the F1 ATPase and a transmembrane H+ carrier called the F0 rotor. Both are formed from multile subunits
the F0 rotor is made up of what parts
the H+ carrier (rotor ring), and the central stalk
ATP synthase slide one
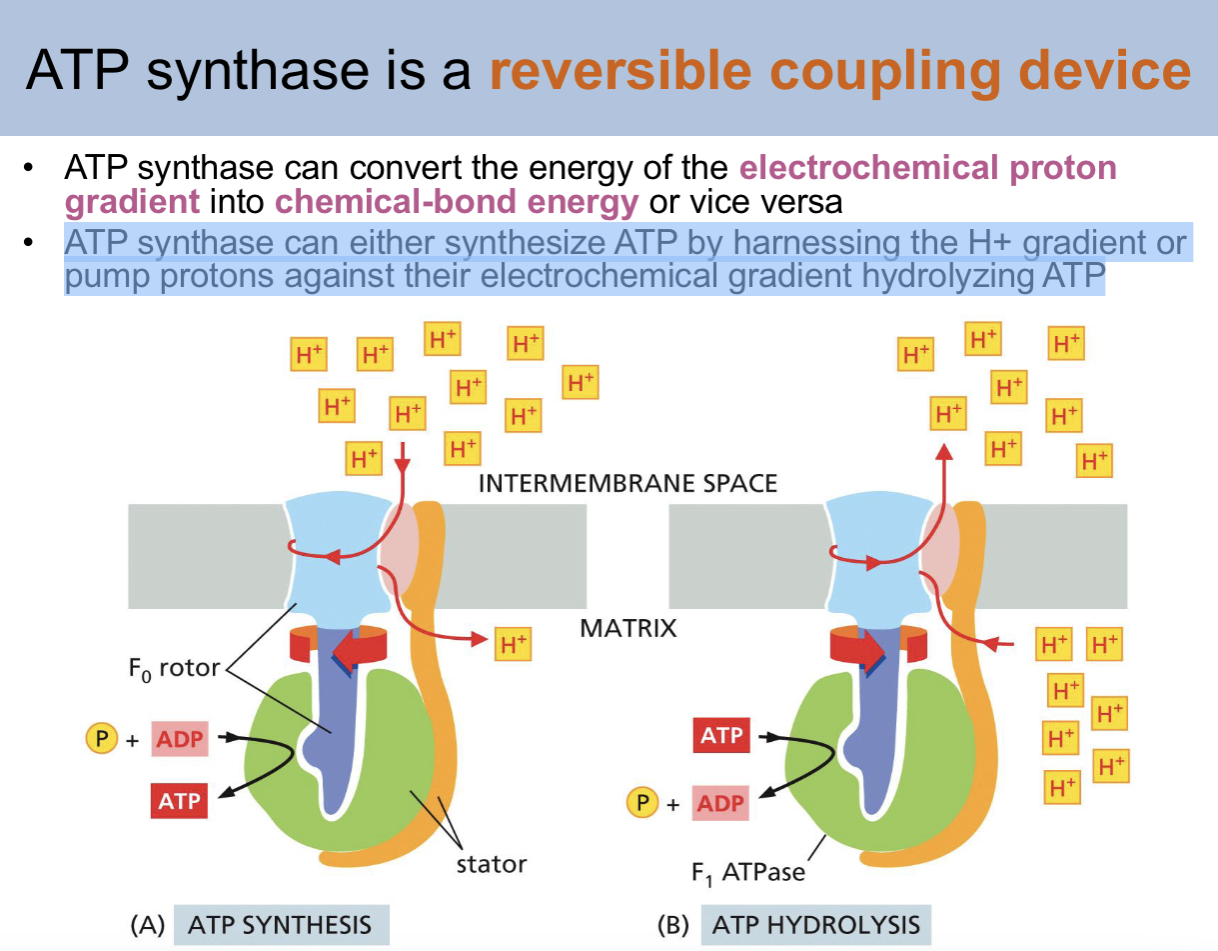
is ATP synthase a reversible coupling device
yes
ATP synthase can convert the energy of the _ into _
ATP synthase can convert the energy of the electrochemical proton gradient into chemical-bond energy or vice versa
ATP synthase can either—- or ——
synthesize ATP by harnessing the H+ gradient or pump protons against their electrochemical gradient hydrolyzing ATP
ATP synthase being reversible slide
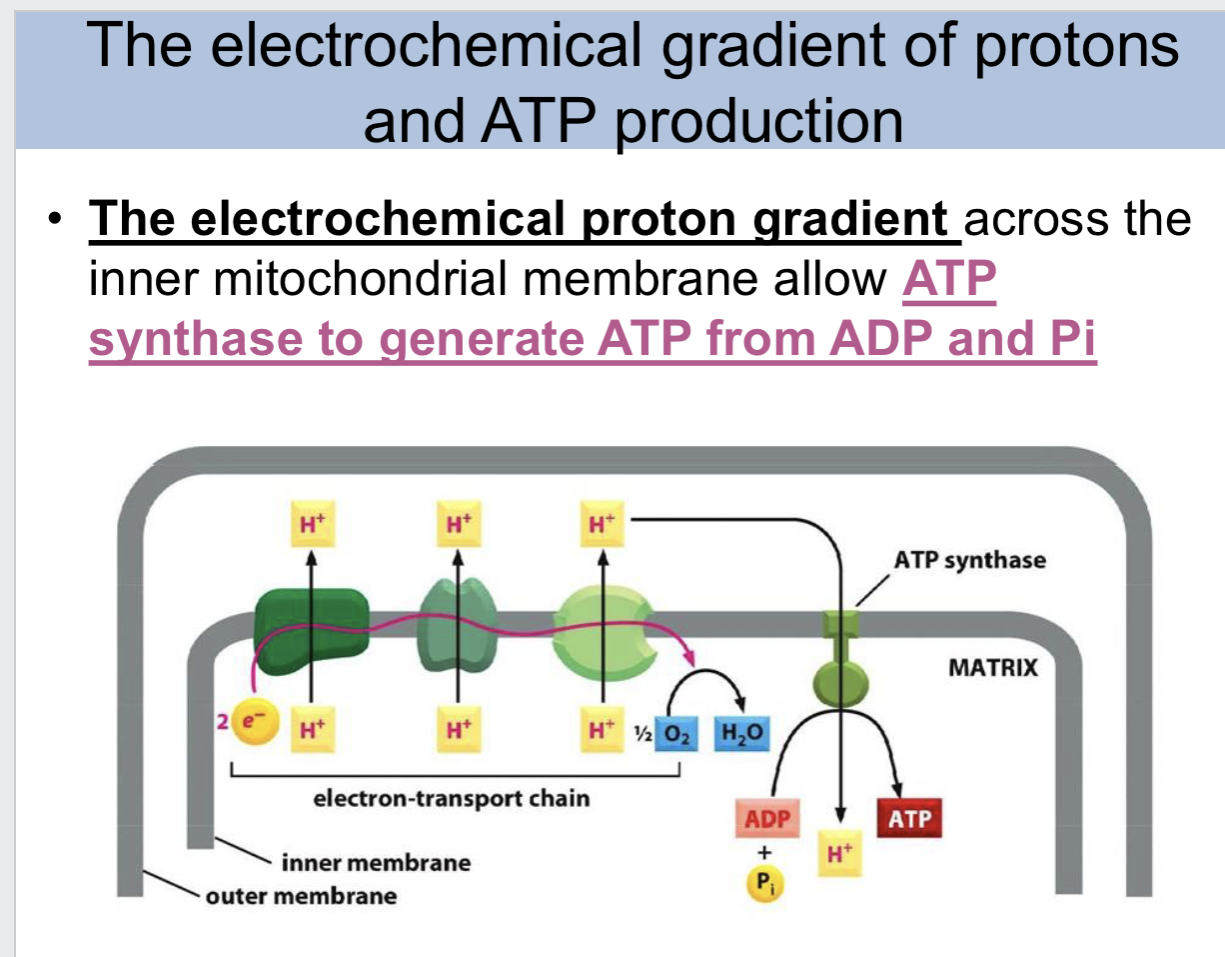
Overview: Mitochondria and oxidative phosphorylation slide

the electrochemical proton gradient across the inner mitochondrial membrane allow ATP synthase to
generate ATP from ADP and Pi
electrochemical gradient of protons and ATP production
mitochondria and systems within slide
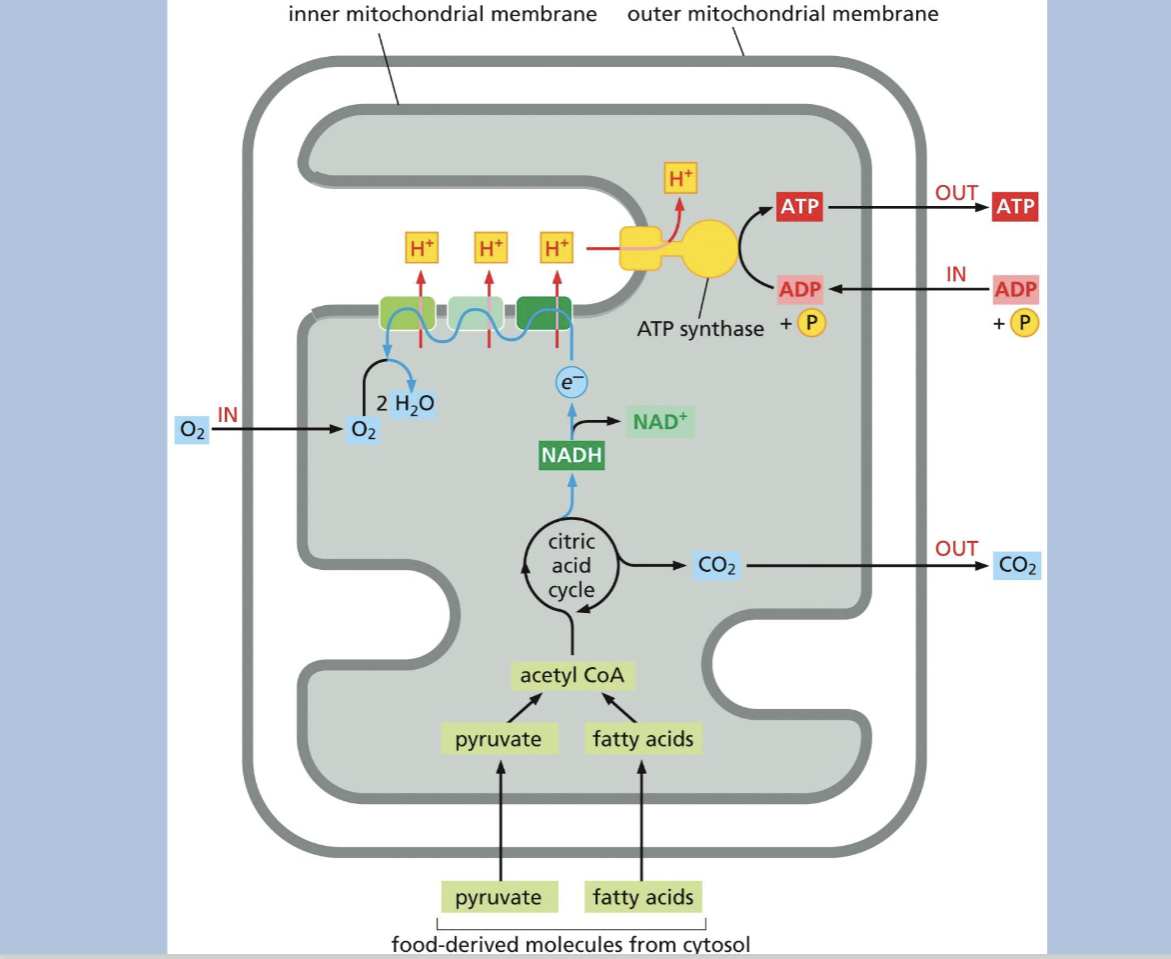
The electrochemical gradient of protons across the inner mitochondrial membrane is used to drive some
coupled transport processes
The electrochemical gradient of protons across the inner mitochondrial membrane is used to drive some coupled transport processes
symport:
antiport:
symport: electrochemical gradient of H+ drives the import of pyruvate and Pi
antiport: pump out ATP and in ADP (ADP-ATP exchange) that depends on a voltage gradient across the membrane (membrane potential), and move 1 negative charge out of the mitochondrion
symport and antiport slide
on glucose oxidation produces about how many ATP
30
in cytosol: 1 NADH →
1.5 ATP (transport of NADH across the inner membrane requires energy)
inside mitochondrial matrix: 1 NADH →
2.5 ATP
where does NADH pass their electrons to in the mitochondrial matrix
the 1st complex, the NADH dehydrogenase
inside mitochondrial matrix:
1 FADH2 →
1.5 ATP
where do FADH2 molecules pass their electrons in the mitochondrial matrix
to the second complex, cytochrome b-c1 complex
Mitochondria maintains a High —/— ratio in cells (— is — times higher than —)
ADP/ATP (ATP is 10 times higher than ADP)
ATP is used as an energy source to
drive energetically unfavorable reactions since ATP hydrolysis is energetically favorable
products slide

what are uncoupling agents
H+ carriers that can insert into the mitochondrial inner membrane